Summary
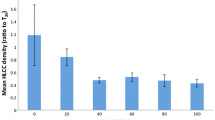
We describe procedures for isolating and culturing airway epithelial cells from chronically infected human lungs. Experience in our laboratory demonstrated the need to balance pathogen eradication against antibiotic toxicity to epithelial cells. To provide a logical basis for antibiotic selection and dose, we systematically analyzed the cytotoxicity of antibiotics useful against typical pathogens. Alone, colistin, ciprofloxacin, doxycycline, and tobramycin were moderately toxic at concentrations close to those used in cell culture, whereas amphotericin, ceftazidime, chloramphenicol, imipenem, meropenem, piperacillin, sulfamethoxazole/trimethoprim, and vancomycin were nontoxic even at concentrations many times the antimicrobial level. Epithelial cytotoxicity of combined antibiotics was additive, with no evidence of competition or synergism. Antibiotics had little effect on initial cell attachment and did not acutely lyse cells, but inhibited subsequent growth. Interestingly, cytotoxicity decreased markedly with increasing epithelial cell density. Cystic fibrosis (CF) and non-CF epithelial cells showed no differences in sensitivity to the antibiotics tested and initial exposure to antibiotics did not affect the electrophysiologic properties of resistance or short circuit current in well-differentiated cells. Tailored combinations of antibiotics at appropriate doses killed even multidrug-resistant bacteria. Thus, epithelial cells can usually be cultured from chronically infected CF airways.
Similar content being viewed by others
References
Bernacki, S. H.; Nelson, A. L.; Abdullah, L.; Sheehan, J. K.; Harris, A.; Davis, C. W.; Randell, S. H. Mucin gene expression during differentiation of human airway epithelia in vitro. Am. J. Respir. Cell. Mol. Biol. 20:595–604; 1999.
Costerton, J. W.; Cheng, K. J.; Geesey, G. G.; Ladd, T. I.; Nickel, J. C.; Dasgupta, M.; Marrie, T. J. Bacterial biofilms in nature and disease. Annu. Rev. Microbiol. 41:435–464; 1987.
Damour, O.; Hua, S. Z.; Lasne, F.; Villain, M.; Rousselle, P.; Collombel, C. Cytotoxicity evaluation of antiseptics and antibiotics on cultured human fibroblasts and keratinocytes. Burns 18:479–485; 1992.
Gilligan, P. H. Microbiology of cystic fibrosis lung disease. In: Yankaskas, J. R.; Knowles, M. R., ed. Cystic fibrosis in adults. Philadelphia, PA: Lippincott-Raven; 1999:93–114.
Gray, J. Prophylactic use of antibiotics in cells and tissues with a high risk of microbial contamination. In: Doyle, A.; Griffiths, J. B.; Newell, D. G., ed. Cell and tissue culture: laboratory procedures. Chichester: Wiley; 1998:4.1–4.10.
Guggino, W. B. Cystic fibrosis and the salt controversy. Cell 96:607–610; 1999.
Isberg, R. R.; Falkow, S. A single genetic locus encoded by Yersinia pseudotuberculosis permits invasion of cultured animal cells by Escherichia coli K-12. Nature 317:262–264; 1985.
Kaartinen, L.; Nettesheim, P.; Adler, K. B.; Randell, S. H. Rat tracheal epithelial cell differentiation in vitro. In Vitro Cell. Dev. Biol. 29A:481–492; 1993.
Lechner, J. F.; LaVeck, M. A. A serum-free method for culturing normal human bronchial epithelial cells at clonal density. J. Tissue Cult. Methods 9:43–48; 1985.
Martin, D. W.; Mohr, C. D. Invasion and intracellular survival of Burkholderia cepacia. Infect. Immun. 68:24–29; 2000.
National Committee for Clinical Laboratory Standards (NCCLS). Methods for dilution antimicrobial susceptibility tests for bacteria that grow aerobically. Approved Standard, M7-A4. 4th ed. Wayne, PA: National Committee for Clinical Laboratory Standards; 1997.
Nickel, J. C.; Ruseska, I.; Wright, J. B.; Costerton, J. W. Tobramycin resistance of Pseudomonas aeruginosa cells growing as a biofilm on urinary catheter material. Antimicrob. Agents Chemother. 27:619–624; 1985.
Raff, M. C. Social controls on cell survival and cell death. Nature 356:397–400; 1992.
Saiman, L.; Mehar, F.; Niu, W. W.; Neu, H. C.; Shaw, K. J.; Miller, G.; Prince, A. Antibiotic susceptibility of multiply resistant Pseudomonas aeruginosa isolated from patients with cystic fibrosis, including candidates for transplantation. Clin. Infect. Dis. 23:532–537; 1996.
Wei, L. Y.; Stutts, M. J.; Hoffman, M. M.; Roepe, P. D. Overexpression of the cystic fibrosis transmembrane conductance regulator in NIH 3T3 cells lowers membrane potential and intracellular pH and confers a multidrug resistance phenotype. Biophys. J. 69:883–895; 1995.
Wine, J. J. The genesis of cystic fibrosis lung disease. J. Clin. Invest. 103:309–312; 1999.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Randell, S.H., Walstad, D.L., Schwab, U.E. et al. Isolation and culture of airway epithelial cells from chronically infected human lungs. In Vitro Cell.Dev.Biol.-Animal 37, 480–489 (2001). https://doi.org/10.1290/1071-2690(2001)037<0480:IACOAE>2.0.CO;2
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1290/1071-2690(2001)037<0480:IACOAE>2.0.CO;2