Abstract
Background
We compared the results of hereditary cancer multigene panel testing among patients ≤ 45 years of age diagnosed with ductal carcinoma in situ (DCIS) versus invasive breast cancer (IBC) in a large integrated health care system.
Methods
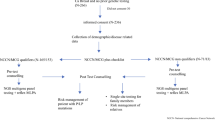
A retrospective cohort study of hereditary cancer gene testing among women ≤ 45 years of age diagnosed with DCIS or IBC at Kaiser Permanente Northern California between September 2019 and August 2020 was performed. During the study period, institutional guidelines recommended the above population be referred to genetic counselors for pretesting counseling and testing.
Results
A total of 61 DCIS and 485 IBC patients were identified. Genetic counselors met with 95% of both groups, and 86.4% of DCIS patients and 93.9% of IBC patients (p = 0.0339) underwent gene testing. Testing differed by race/ethnicity (p = 0.0372). Among those tested, 11.76% (n = 6) of DCIS patients and 16.71% (n = 72) of IBC patients had a pathogenic variant (PV) or likely pathogenic variant (LPV) based on the 36-gene panel (p = 0.3650). Similar trends were seen in 13 breast cancer (BC)-related genes (p = 0.0553). Family history of cancer was significantly associated with both BC-related and non-BC-related PVs in IBC, but not DCIS.
Conclusion
In our study, 95% of patients were seen by a genetic counselor when age was used as an eligibility criterion for referral. While larger studies are needed to further compare the prevalence of PVs/LPVs among DCIS and IBC patients, our data suggest that even in younger patients, the prevalence of PVs/LPVs in BC-related genes is lower in DCIS patients.

Similar content being viewed by others
References
NCCN clinical practice guidelines in oncology (NCCN guidelines®) for invasive breast cancer V.4.2022©. National Comprehensive Cancer Network, Inc.; 2023. All rights reserved. Accessed 1 Jan 2023.
Buys SS, Sandbach JF, Gammon A, et al. A study of over 35,000 women with breast cancer tested with a 25-gene panel of hereditary cancer genes. Cancer. 2017;123(10):1721–30. https://doi.org/10.1002/cncr.30498.
Tung N, Lin NU, Kidd J, et al. Frequency of germline mutations in 25 cancer susceptibility genes in a sequential series of patients with breast cancer. J Clin Oncol. 2016;34(13):1460–8. https://doi.org/10.1200/JCO.2015.65.0747.
van der Groep P, van Diest PJ, Menko FH, Bart J, de Vries EG, van der Wall E. Molecular profile of ductal carcinoma in situ of the breast in BRCA1 and BRCA2 germline mutation carriers. J Clin Pathol. 2009;62(10):926–30. https://doi.org/10.1136/jcp.2009.065524.
Petridis C, Arora I, Shah V, et al. Frequency of pathogenic germline variants in BRCA1, BRCA2, PALB2, CHEK2 and TP53 in ductal carcinoma in situ diagnosed in women under the age of 50 years. Breast Cancer Res. 2019;21(1):58. https://doi.org/10.1186/s13058-019-1143-y.
Smith KL, Adank M, Kauff N, et al. BRCA mutations in women with ductal carcinoma in situ. Clin Cancer Res. 2007;13(14):4306–10. https://doi.org/10.1158/1078-0432.CCR-07-0146.
Hall MJ, Reid JE, Wenstrup RJ. Prevalence of BRCA1 and BRCA2 mutations in women with breast carcinoma In Situ and referred for genetic testing. Cancer Prev Res (Phila). 2010;3(12):1579–85. https://doi.org/10.1158/1940-6207.CAPR-09-0218.
Bayraktar S, Elsayegh N, Gutierrez Barrera AM, et al. Predictive factors for BRCA1/BRCA2 mutations in women with ductal carcinoma in situ [published erratum appears in Cancer. 2014;120(6):927]. Cancer. 2012;118(6):1515-22. https://doi.org/10.1002/cncr.26428.
US Census Bureau. QuickFacts California. Available at https://www.census.gov/quickfacts/CA (2022)
US Census Bureau. QuickFacts United States. Available at: https://www.census.gov/quickfacts/fact/table/US/PST045221 (2021)
Samadder NJ, Riegert-Johnson D, Boardman L, et al. Comparison of universal genetic testing vs guideline-directed targeted testing for patients with hereditary cancer syndrome [published erratum appears in JAMA Oncol. 2021;7(2):312]. JAMA Oncol. 2021;7(2):230–7. https://doi.org/10.1001/jamaoncol.2020.6252.
Ow SGW, Ong PY, Lee SC. Discoveries beyond BRCA1/2: multigene testing in an Asian multi-ethnic cohort suspected of hereditary breast cancer syndrome in the real world. PLoS ONE. 2019;14(3):e0213746. https://doi.org/10.1371/journal.pone.0213746.
Hwang ES, McLennan JL, Moore DH, Crawford BB, Esserman LJ, Ziegler JL. Ductal carcinoma in situ in BRCA mutation carriers. J Clin Oncol. 2007;25(6):642–7. https://doi.org/10.1200/JCO.2005.04.0345.
Claus EB, Petruzella S, Matloff E, Carter D. Prevalence of BRCA1 and BRCA2 mutations in women diagnosed with ductal carcinoma in situ. JAMA. 2005;293(8):964–9. https://doi.org/10.1001/jama.293.8.964.
Frank TS, Deffenbaugh AM, Reid JE, et al. Clinical characteristics of individuals with germline mutations in BRCA1 and BRCA2: analysis of 10,000 individuals. J Clin Oncol. 2002;20(6):1480–90. https://doi.org/10.1200/JCO.2002.20.6.1480.
Powell CB, Laurent C, Garcia C, et al. Factors influencing genetic counseling and testing for hereditary breast and ovarian cancer syndrome in a large US health care system. Clin Genet. 2022;101(3):324–34. https://doi.org/10.1111/cge.14100.
Childers CP, Childers KK, Maggard-Gibbons M, Macinko J. National estimates of genetic testing in women with a history of breast or ovarian cancer [published erratum appears in J Clin Oncol. 2018;36(4):432]. J Clin Oncol. 2017;35(34):3800-3806. https://doi.org/10.1200/JCO.2017.73.6314.
Katz SJ, Ward KC, Hamilton AS, et al. Gaps in receipt of clinically indicated genetic counseling after diagnosis of breast cancer. J Clin Oncol. 2018;36(12):1218–24. https://doi.org/10.1200/JCO.2017.76.2369.
Turza L, Lovejoy LA, Turner CE, Shriver CD, Ellsworth RE. Eligibility, uptake and response to germline genetic testing in women with DCIS. Front Oncol. 2022;12:918757. https://doi.org/10.3389/fonc.2022.918757.
Hafertepen L, Pastorino A, Morman N, et al. Barriers to genetic testing in newly diagnosed breast cancer patients: do surgeons limit testing? Am J Surg. 2017;214(1):105–10. https://doi.org/10.1016/j.amjsurg.2016.08.012.
Delikurt T, Williamson GR, Anastasiadou V, Skirton H. A systematic review of factors that act as barriers to patient referral to genetic services. Eur J Hum Genet. 2015;23(6):739–45. https://doi.org/10.1038/ejhg.2014.180.
Chapman-Davis E, Zhou ZN, Fields JC, et al. Racial and ethnic disparities in genetic testing at a hereditary breast and ovarian cancer center. J Gen Intern Med. 2021;36(1):35–42. https://doi.org/10.1007/s11606-020-06064-x.
Kurian AW, Ward KC, Howlader N, et al. Genetic testing and results in a population-based cohort of breast cancer patients and ovarian cancer patients. J Clin Oncol. 2019;37(15):1305–15. https://doi.org/10.1200/JCO.18.01854.
Cragun D, Weidner A, Lewis C, et al. Racial disparities in BRCA testing and cancer risk management across a population-based sample of young breast cancer survivors. Cancer. 2017;123(13):2497–505. https://doi.org/10.1002/cncr.30621.
Sutton AL, Hurtado-de-Mendoza A, Quillin J, et al. Reducing disparities in receipt of genetic counseling for underserved women at risk of hereditary breast and ovarian cancer. J Womens Health (Larchmt). 2020;29(8):1131–5. https://doi.org/10.1089/jwh.2019.7984.
Reid S, Cadiz S, Pal T. Disparities in genetic testing and care among black women with hereditary breast cancer. Curr Breast Cancer Rep. 2020;12(3):125–31. https://doi.org/10.1007/s12609-020-00364-1.
Pagán JA, Su D, Li L, Armstrong K, Asch DA. Racial and ethnic disparities in awareness of genetic testing for cancer risk. Am J Prev Med. 2009;37(6):524–30. https://doi.org/10.1016/j.amepre.2009.07.021.
Singer E, Antonucci T, Van Hoewyk J. Racial and ethnic variations in knowledge and attitudes about genetic testing. Genet Test. 2004;8(1):31–43. https://doi.org/10.1089/109065704323016012.
Canedo JR, Miller ST, Myers HF, Sanderson M. Racial and ethnic differences in knowledge and attitudes about genetic testing in the US: systematic review. J Genet Couns. 2019;28(3):587–601. https://doi.org/10.1002/jgc4.1078.
Catz DS, Green NS, Tobin JN, et al. Attitudes about genetics in underserved, culturally diverse populations. Community Genet. 2005;8(3):161–72. https://doi.org/10.1159/000086759.
Suther S, Kiros GE. Barriers to the use of genetic testing: a study of racial and ethnic disparities. Genet Med. 2009;11(9):655–62. https://doi.org/10.1097/GIM.0b013e3181ab22aa.
Modell SM, Allen CG, Ponte A, Marcus G. Cancer genetic testing in marginalized groups during an era of evolving healthcare reform. J Cancer Policy. 2021;28:100275. https://doi.org/10.1016/j.jcpo.2021.100275.
Nikolaidis C, Duquette D, Mendelsohn-Victor KE, et al. Disparities in genetic services utilization in a random sample of young breast cancer survivors. Genet Med. 2019;21(6):1363–70. https://doi.org/10.1038/s41436-018-0349-1.
Acknowledgment
This research was supported by the Kaiser Permanente Graduate Medical Education and a DARE (Delivery Science and Applied Research) grant.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosure
Diana S. Hsu, Sheng-Fang Jiang, Laurel A. Habel, Elizabeth Hoodfar, Audrey Karlea, Leslie Manace-Brenman, Jessica M. Dzubnar, and Veronica C. Shim have no conflicts of interest to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hsu, D.S., Jiang, SF., Habel, L.A. et al. Germline Genetic Testing Among Women ≤ 45 Years of Age with Ductal Carcinoma In Situ Versus Invasive Breast Cancer in a Large Integrated Health Care System. Ann Surg Oncol 30, 6454–6461 (2023). https://doi.org/10.1245/s10434-023-13745-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-023-13745-4