Abstract
Background
The primary goal of this study was to determine overall survival (OS) in patients who underwent local treatment (metastasectomy or stereotactic body radiotherapy [SBRT]) or systemic therapy (chemotherapy or targeted therapy) for oligometastatic esophagogastric cancer. The secondary goal was to determine prognostic factors for OS.
Methods
Patients with synchronous or metachronous oligometastatic esophagogastric cancer who underwent local treatment or systemic therapy were included in this single-center, retrospective cohort study. Oligometastatic disease (OMD) included 1 organ or 1 extraregional lymph node station with ≤ 3 lesions. OS was determined after OMD detection. Treatment for OMD was categorized as (1) local treatment, (2) local plus systemic, (3) systemic therapy. The primary tumor was controlled after resection or definitive chemoradiotherapy.
Results
In total, 85 patients were included. Treatment for OMD was local treatment (58%), local plus systemic (14%), or systemic therapy (28%). The primary tumor was controlled in 68% of patients. Most patients were diagnosed with distal esophageal cancer (61%), with adenocarcinoma histology (76%), and presented with synchronous OMD (51%). OS after local treatment was 17 months (95% confidence interval [CI] 12–40), after local plus systemic therapy 35 months (95% CI 29–NA), and after systemic therapy 16 months (95% CI 11–NA). Better OS was independently associated with local plus systemic compared with local treatment (hazard ratio [HR] 2.11, 95% CI 1.05–5.07) or systemic therapy (HR 2.28, 95% CI 1.04–6.07).
Conclusions
Local plus systemic therapy for oligometastatic esophagogastric cancer was independently associated with improved OS and better OS compared with either systemic therapy or local treatment.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Gastric and esophageal cancer are the fifth and seventh most common cancer types worldwide with an estimated 1,033,701 and 572,034 new cases annually, respectively.1 Between 33 and 50% of esophagogastric cancer patients present with distant metastases at the time of initial diagnosis (i.e., synchronous metastases) and are usually treated with systemic therapy alone or best supportive care (BSC).2,3 In addition, between 20 and 50% of patients who have undergone multimodality treatment for locoregional disease develop distant metastases during follow-up (i.e., metachronous metastases).4,5,6,7,8
In a small portion of these metastatic patients, distant metastases are present in a limited number of lesions and organs only, so-called oligometastatic disease (OMD).9 The concept of OMD was first introduced in 1995 and described a clinical disease state of limited metastatic capacity.10 In 2020, a comprehensive classification system for OMD was proposed.11 “Induced OMD” was distinguished from “genuine OMD” (i.e., with vs. without previous history of polymetastatic disease, respectively). “Genuine OMD” was subdivided into “repeat OMD” and “de-novo OMD” (i.e., with vs. without a previous history of OMD, respectively).11 Finally, “de-novo OMD” was subdivided into “synchronous OMD” and “metachronous OMD.”11
Until now, no uniform criteria exist for the maximum number of lesions and organs to be considered OMD in esophagogastric cancer. However, most studies define OMD as a maximum of 3 lesions in 1 organ.12,13,14 Several, small, retrospective, nonrandomized studies suggest that local treatment (i.e., metastasectomy or stereotactic body radiotherapy (SBRT)) for oligometastatic esophagogastric cancer may improve overall survival (OS).15,16,17,18,19 However, because these studies have been focusing on patients who underwent local treatment and have not included patients who underwent systemic therapy alone, the general applicability of these studies remains unclear.
Therefore, the primary goal of this study was to determine the OS in patients who underwent local treatment and/or systemic therapy for synchronous or metachronous oligometastatic esophagogastric cancer. The secondary goal was to determine prognostic factors for OS and progression-free survival (PFS).
Material and methods
Ethical statement
This study was approved by the Institutional Review Board of the UMC Utrecht, and the need for informed consent was waived. This study was reported in accordance with the STROBE guidelines (Supplementary File 1), The Code of Ethics of the World Medical Association (Declaration of Helsinki) for experiments involving humans, and the Recommendations for the Conduct, Reporting, Editing, and Publication of Scholarly Work in Medical Journals.
Patient Inclusion
Between 2010 and 2021, consecutive patients diagnosed at the UMC Utrecht with synchronous or metachronous OMD from esophagogastric cancer were eligible for inclusion in this single-center, retrospective cohort study. OMD was limited to 1 organ or 1 extraregional lymph node station with ≤ 3 lesions.14 Patients who underwent local treatment and/or systemic therapy for oligometastases were included. Patients who underwent best supportive care were not included. Local treatment was defined as metastasectomy with the intention to perform a radical resection of all metastatic lesions or SBRT of all metastatic lesions using a SBRT scheme: ≥ 10 Gy per fraction with ≥ 1 fraction(s), ≥7 Gy per fraction with ≥ 5 fractions, ≥ 5 Gy per fraction with ≥ 12 fractions, or a total radiation dosage ≥ 50 Gy. Systemic therapy could include targeted therapy and/or chemotherapy.
Classification of Oligometastatic Lesions
Metachronous OMD was defined as OMD detected after completion of primary tumor treatment (resection of the primary tumor or definitive chemoradiotherapy). The location of OMD lesions was classified into an organ with hematogenous metastasis (i.e., brain, bone, liver, adrenal gland, lung, or soft tissue) or an extraregional lymph node station. OMD lesions were confirmed by pathological assessment or if pathological confirmation was not possible (e.g., because the lesion was not approachable for biopsy) with repeated follow-up imaging. The clinical and pathological stage was classified according to the TNM 8th edition of the International Union Against Cancer (UICC).20
Management
Management for OMD was categorized into (1) local treatment, (2) local plus systemic therapy, or (3) systemic therapy alone. Local treatment consisted of metastasectomy and/or SBRT for OMD. Local plus systemic therapy consisted of local treatment and systemic therapy for OMD. Systemic therapy consisted of systemic therapy alone for OMD. The primary tumor was controlled after resection or definitive chemoradiotherapy, without evidence of loco-regional recurrence.
Staging
Baseline staging for patients with esophageal cancer was with 18fluorodeoxyglucose positron emission tomography (18F-FDG PET) with integrated computed tomography (CT).21 Baseline staging for patients with gastric cancer was with CT and after 2016 for patients with advanced gastric cancer (i.e., ≥ cT3 or cN+) with PET/CT and diagnostic laparoscopy (according to Dutch national guidelines).22 Follow-up was done without standardized imaging and/or endoscopies as recommended in Dutch national guidelines and ESMO and NCCN guidelines).21,22,23,24 In patients with clinically suspected OMD, 18F-FDG PET/CT imaging was performed.
Outcomes
The primary outcome measure was OS. The secondary outcome measures were prognostic factors for OS and PFS. OS was defined as the time interval between the diagnosis of OMD and death or last follow-up. Prognostic factors for OS were analyzed using multivariable Cox proportional hazard models. PFS was defined as the time interval between the diagnosis of OMD and progression, last follow-up, or death.
Variables
Performance status was determined at the time of OMD diagnosis according to the World Health Organization (WHO) performance score.25 The disease-free interval was defined as the time interval between the detection of the primary tumor and OMD in patients with metachronous OMD.26 Recurrence of OMD was categorized into “local” when OMD was detected in the same treated OMD location and “systemic” when OMD was detected in another (nontreated) location. The primary tumor was not controlled in case of locoregional recurrence or no primary tumor treatment.
Statistical Analysis
Parametric data were presented as mean with standard deviation (SD) and non-parametric as median with interquartile range (IQR). Categorical data were presented as frequencies with proportions. Kaplan-Meier curves were constructed of PFS and OS. Univariable Cox proportional hazard models were used to identify prognostic factors associated with OS. For multivariable analyses, prognostic factors with a p-value of <0.25 in univariable analyses were entered in a model, and subsequent backward stepwise elimination was performed according to Akaike Information Criterion.27 Prognostic factors were expressed using hazard ratios (HRs) with 95% confidence intervals (CIs). The disease-free interval was dichotomized into ≤ 24 months or > 24 months.15 Data were analyzed using R for Windows, version 3.6.3.28 A p-value < 0.05 was considered statistically significant.
Results
Patient Selection
Between 2010 and 2021, a total of 501 patients with synchronous or metachronous distant metastases from esophagogastric cancer were screened for “de-novo” OMD. Synchronous or metachronous OMD was identified in 106 patients (21%). Patients who underwent best supportive care for OMD were not included (n = 24). Patients who underwent best supportive care had a worse performance score and more OMD lesions (Supplementary Files 2 and 3). Consequently, a total of 85 “de-novo” OMD patients who underwent local treatment and/or systemic therapy were included. Figure 1 shows the patient inclusion.
Baseline Characteristics
Included patients had a mean age of 65 years (SD: 9.0), 77% were male, and 91% had a baseline WHO performance score of 0-1. Most patients were diagnosed with esophageal cancer of the distal third of the esophagus (61%) with adenocarcinoma histology (76%) and presented with synchronous OMD (51%). The mean disease-free interval was 24 months (SD: 20) in patients with metachronous OMD. The clinical disease stage was predominantly cT3 (75%) and cN1 (42%). The pathological stage was predominantly pT3 (58%) and pN1 (42%). Table 1 shows the baseline characteristics of included patients stratified for OMD treatment.
OMD Characteristics
The location of OMD lesions was the extraregional lymph nodes (28%), liver (21%), bone (21%), brain (15%), lung (8%), adrenal gland (8%), soft tissue (4%), or appendix (1%). The number of OMD lesions was 1 (74%), 2 (21%), or 3 (5%). OMD lesions were confirmed with pathological assessment in 76% or with repeated follow-up imaging in 24%. Most patients with bone, extraregional lymph node, lung, or adrenal gland oligometastases underwent local treatment (62%, 59%, 86%, 57%, respectively). Most patients with liver oligometastases underwent systemic therapy alone (72%). Table 2 demonstrates the location and number of OMD lesions stratified for OMD treatment.
Management for Primary Tumor and OMD
The primary tumor was controlled in 68% of patients. A controlled primary tumor was more common in patients who underwent local treatment or local plus systemic therapy compared with patients who underwent systemic therapy alone (86% and 75% vs. 29%). The primary tumor was controlled after neoadjuvant chemo(radio)therapy followed by resection (51%), chemoradiotherapy (14%), or upfront primary tumor resection (4%).
Treatment for OMD was either “local treatment” (58%), “local plus systemic therapy” (14%), or “systemic therapy” (28%). Local treatment consisted of patients who either underwent SBRT (35% of total), metastasectomy (16%), or metastasectomy and SBRT (6%). Local plus systemic therapy consisted of patients who either underwent systemic therapy plus SBRT (9%), systemic therapy plus metastasectomy (2%), or chemoradiotherapy (2%). Systemic therapy consisted of patients who underwent chemotherapy (21%) or chemotherapy and targeted therapy (7%) as a first-line treatment. Patients with synchronous OMD less often had a controlled primary tumor (42% vs. 76%) and more often underwent systemic therapy alone compared with metachronous OMD (50% vs. 16%). Supplementary Table 4 shows the treatment characteristics stratified for synchronous and metachronous OMD.
OS and Prognostic Factors
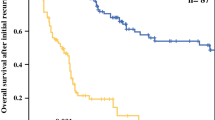
Median follow-up time was 17 months (range 1–119). Median OS across all included patients was 20 months (95% CI 15–35). OS after local treatment alone was 17 months (95% CI 12–40), local plus systemic therapy 35 months (95% CI 29–NA), and after systemic therapy alone 16 months (95% CI 11–NA). Figure 2 demonstrates the OS stratified for the treatment for OMD. In multivariable analysis, better OS was independently associated with local and systemic therapy as compared with local treatment alone or systemic therapy alone (HR 2.11, 95% CI 1.05–5.07 and HR 2.28, 95% CI 1.04–6.07). No other prognostic factors for OS were identified in multivariable analyses. Table 3 demonstrates the results of the univariable and multivariable Cox proportional hazard model analyses. OS in the best supportive care group was 6 months (95% CI 4–8, Supplementary File 5).
Progression-Free Survival
Median PFS across all included patients was 14 months (95% CI 11–21). PFS after local treatment alone was 10 months (95% CI 7–16), local plus systemic median PFS was not reached, and after systemic therapy 15 months (95% CI 11–NA; Fig. 3). A total of 51 patients (60%) developed OMD recurrence during follow-up, of whom 44 patients (51%) developed systemic OMD recurrence and 7 patients (9%) locoregional OMD recurrence (i.e., OMD recurrence in the same treated OMD location).
Discussion
This cohort study reports on patients with synchronous or metachronous oligometastatic esophagogastric cancer who underwent local treatment (i.e., metastasectomy or SBRT) and/or systemic therapy for OMD. Local treatment combined with systemic therapy for OMD was associated with a favorable OS (median OS 35 months) and independently associated with improved OS compared with patients who underwent local treatment alone (median OS 17 months) or systemic therapy alone (median OS 16 months) for OMD.
This improved OS after local and systemic therapy for OMD compared with local treatment alone might in part be explained by improved PFS (median PFS not reached vs. 10 months) suggesting improved systemic control by adding systemic therapy to local treatment. In addition, this improved OS also might be explained by the selection of patients who responded well to systemic therapy who were offered subsequent local treatment for OMD. Previous studies have administered chemotherapy first as a tool for selecting patients with the highest likelihood to benefit from additional local treatment based on their response to treatment.29
OS of patients who underwent local plus systemic therapy was comparable with a phase II, nonrandomized trial that included patients with oligometastatic gastric or gastroesophageal junction cancer patients who underwent systemic therapy and resection of the primary tumor and metastases (35 months versus 31.3 months, respectively).29
Moreover, OS patients who underwent local plus systemic therapy for OMD was better than a phase II trial, including patients with esophageal squamous cell carcinoma who underwent SBRT and 50% additional systemic therapy (median OS 24.6).30 The lower OS in this trial might be explained by limited use of systemic therapy in this trial (50%). Accordingly, median OS of patients who underwent local treatment alone for OMD without systemic therapy in our study was worse as compared with this study (16.0 versus 24.6 months).30 Besides the omission of systemic therapy, the lower OS in this cohort might be explained by the inclusion of 12 patients (14% of included patients) with brain OMD in our study (of whom 92% received local treatment), while these patients were excluded from previous trials.29,30 Brain OMD are associated with lower OS as compared with extracranial OMD.31
Future prospective studies are warranted to determine which patients benefit the most from local treatment for OMD. In our study, OMD was defined as distant metastases in 1 organ or 1 extraregional lymph node station with ≤ 3 lesions. This definition was comparable with recent literature on OMD in esophagogastric cancer.14 However, perhaps this definition of OMD should not be applicable to the brain as OMD in this organ is associated with worse OS.31 A universal multidisciplinary consensus statement is warranted to initiate future trials in this field. The OligoMetastatic Esophagogastric Cancer (OMEC) consortium was designed to develop a multidisciplinary consensus statement for the definition and treatment for oligometastatic esophagogastric cancer.14,32 The OMEC consortium consists of 46 esophagogastric cancer experts centers in Europe and is endorsed by the European Organisation for Research and Treatment of Cancer (EORTC), European Society for Radiotherapy and Oncology (ESTRO), European Society of Medical Oncology (ESMO), European Society of Surgical Oncology (ESSO), European Society for Diseases of the Esophagus (ESDE), the European chapter of the International Gastric Cancer Association (IGCA), and the Dutch Upper GI Cancer Group (DUCG).
There are certain limitations to this study that warrant caution for the interpretation of results. First, selection bias may have resulted in a potential overestimation of OS. Second, the applicability and generalizability of these results remain challenging because of a lack of a uniform definition of OMD in esophagogastric cancer. Strengths of this study include the homogenous study cohort, because only patients with de-novo OMD were included (according to the most recent OMD consensus classification).11 Other strengths include the applicability of our results because also patients were included who underwent systemic therapy alone for OMD. Therefore, this study provides a contemporaneous comparator into selection and outcomes after different approaches to treatment for OMD.
Conclusions
This study included patients with oligometastatic esophagogastric cancer limited to 1 organ with ≤ 3 lesions or 1 extra-regional lymph node station with ≤ 3 lesions who underwent local treatment (metastasectomy or SBRT) and/or systemic therapy for oligometastases. Local treatment combined with systemic therapy was associated with a median OS of 35 months compared with 17 months after local treatment alone and 16 months after systemic therapy alone. Future prospective studies are warranted to confirm these results.
Data sharing statement
All data generated and analyzed during this study are included in this published article (and its supplementary information files).
References
Sung H, Ferlay J, Siegel RL, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2021;71(3):209–49. https://doi.org/10.3322/caac.21660
Bernards N, Creemers GJ, Nieuwenhuijzen GAPP, Bosscha K, Pruijt JFMM, Lemmens VEPPPP. No improvement in median survival for patients with metastatic gastric cancer despite increased use of chemotherapy. Ann Oncol. 2013;24:3056–60. https://doi.org/10.1093/annonc/mdt401
Wu S-GG, Zhang W-WW, Sun J-YY, Li F-YY, Lin Q, He Z-YY. Patterns of distant metastasis between histological types in esophageal cancer. Front Oncol. 2018. https://doi.org/10.3389/fonc.2018.00302
Hulscher JBF, Van Sandick JW, Tijssen JGP, Obertop H, Van Lanschot JJB. The recurrence pattern of esophageal carcinoma after transhiatal resection. J Am Coll Surg. 2000;191(2):143–8. https://doi.org/10.1016/S1072-7515(00)00349-5.
Nakagawa S, Kanda T, Kosugi SI, Ohashi M, Suzuki T, Hatakeyama K. Recurrence pattern of squamous cell carcinoma of the thoracic esophagus after extended radical esophagectomy with three-field lymphadenectomy. J Am Coll Surg. 2004;198(2):205–11. https://doi.org/10.1016/j.jamcollsurg.2003.10.005.
Parry K, Visser E, van Rossum PSNN, Mohammad NH, Ruurda JP, van Hillegersberg R. Prognosis and treatment after diagnosis of recurrent esophageal carcinoma following esophagectomy with curative intent. Ann Surg Oncol. 2015;22 Suppl 3(S3):1292–300. https://doi.org/10.1245/s10434-015-4840-5
Dresner SM, Griffin SM. Pattern of recurrence following radical oesophagectomy with two-field lymphadenectomy. Br J Surg. 2000;87(10):1426–33. https://doi.org/10.1046/j.1365-2168.2000.01541.x.
Cunningham D, Allum WH, Stenning SP, et al. Perioperative chemotherapy versus surgery alone for resectable gastroesophageal cancer. N Engl J Med. 2006;355(1):11–20. https://doi.org/10.1056/NEJMoa055531.
Reyes DK, Pienta KJ. The biology and treatment of oligometastatic cancer. Oncotarget. 2015. https://doi.org/10.18632/oncotarget.3455.
Hellman S, Weichselbaum RR. Oligometastases. J Clin Oncol. 1995;13(1):8–10. https://doi.org/10.1200/JCO.1995.13.1.8.
Guckenberger M, Lievens Y, Bouma AB, et al. Characterisation and classification of oligometastatic disease: a European Society for Radiotherapy and Oncology and European Organisation for Research and Treatment of Cancer consensus recommendation. Lancet Oncol. 2020;21(1):e18-28. https://doi.org/10.1016/S1470-2045(19)30718-1.
Chen Y, Cheng X, Song H, et al. Outcomes of concurrent chemoradiotherapy versus chemotherapy alone for esophageal squamous cell cancer patients presenting with oligometastases. J Thorac Dis. 2019;11(4):1536–45. https://doi.org/10.21037/jtd.2019.03.10.
Yamashita H, Ogita M, Aoki S, Abe O, Nakagawa K. Linear accelerator-based stereotactic body radiation therapy in the treatment of oligometastatic disease. Mol Clin Oncol. 2020;13(2):109–14. https://doi.org/10.3892/mco.2020.2065.
Kroese TE, van Laarhoven HWM, Nilsson M, et al. Definition of oligometastatic esophagogastric cancer and impact of local oligometastasis-directed treatment: A systematic review and meta-analysis. Eur J Cancer. 2022;166:254–69. https://doi.org/10.1016/j.ejca.2022.02.018.
Seesing MFJ, Van Der Veen A, Brenkman HJF, et al. Resection of hepatic and pulmonary metastasis from metastatic esophageal and gastric cancer: a nationwide study. Dis Esophagus. 2020;32(12):1–8. https://doi.org/10.1093/dote/doz034.
Kondo H, Okumura T, Ohde Y, Nakagawa K. Surgical treatment for metastatic malignancies. Pulmonary metastasis: Indications and outcomes. Int J Clin Oncol. 2005;10(2):81–5. https://doi.org/10.1007/s10147-004-0472-7
Kaifi JT, Gusani NJ, Deshaies I, et al. Indications and approach to surgical resection of lung metastases. J Surg Oncol. 2010;102(2):187–95. https://doi.org/10.1002/jso.21596.
Riquet M, Foucault C, Cazes A, et al. Pulmonary resection for metastases of colorectal adenocarcinoma. Ann Thorac Surg. 2010;89(2):375–80. https://doi.org/10.1016/j.athoracsur.2009.10.005.
Weichselbaum RR, Hellman S. Oligometastases revisited. Nat Rev Clin Oncol. 2011;8(6):378–82. https://doi.org/10.1038/nrclinonc.2011.44.
Rice TW, Patil DT, Blackstone EH. 8th edition AJCC/UICC staging of cancers of the esophagus and esophagogastric junction: Application to clinical practice. Ann Cardiothorac Surg. 2017. https://doi.org/10.21037/acs.2017.03.14.
Netherlands Cancer Registry. Richtlijn Oesofaguscarcinoom. 2015.
Netherlands Cancer Registry. Richtlijn Maagcarcinoom.
Ajani JA, D’Amico TA, Bentrem DJ, et al. Esophageal and Esophagogastric Junction Cancers, Version 2.2019, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Cancer Netw. 2019.https://doi.org/10.6004/jnccn.2019.0033
Lordick F, Mariette C, Haustermans K, et al. Oesophageal cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2016;27:v50–7. https://doi.org/10.1093/annonc/mdw329.
Oken MM, Creech RH, Davis TE. Toxicology and response criteria of the Eastern Cooperative Oncology Group. Am J Clin Oncol Cancer Clin Trials. 1982;5(6):649–55. https://doi.org/10.1097/00000421-198212000-00014.
Lievens Y, Guckenberger M, Gomez D, et al. Defining oligometastatic disease from a radiation oncology perspective: An ESTRO-ASTRO consensus document. Radiother Oncol. 2020;148:157–66. https://doi.org/10.1016/j.radonc.2020.04.003.
deLeeuw J. Introduction to Akaike (1973) Information theory and an extension of the maximum likelihood principle. In: Springer, New York, NY; 1992:599–609. https://doi.org/10.1007/978-1-4612-0919-5_37
R: The R Project for Statistical Computing.
Al-Batran S-EE, Homann N, Pauligk C, et al. Effect of neoadjuvant chemotherapy followed by surgical resection on survival in patients with limited metastatic gastric or gastroesophageal junction cancer: The AIO-FLOT3 trial. JAMA Oncol. 2017;3(9):1237–44. https://doi.org/10.1001/jamaoncol.2017.0515
Liu Q, Zhao K, Chen Y, Lu S. Stereotactic body radiotherapy for patients with oligometastatic esophageal squamous cell carcinoma: preliminary results of a Phase 2 Clinical Trial. Int J Radiat Oncol. 2019;105(1):S129. https://doi.org/10.1016/j.ijrobp.2019.06.112.
Nobel TB, Sihag S, Xing XX, et al. Oligometastases after curative esophagectomy are not one-size-fits-all. Ann Thorac Surg. 2021. https://doi.org/10.1016/j.athoracsur.2021.03.002.
Kroese TE, van Hillegersberg R, Schoppmann S, et al. Definitions and treatment of oligometastatic oesophagogastric cancer according to multidisciplinary tumour boards in Europe. Eur J Cancer. 164:18–29. https://doi.org/10.1016/j.ejca.2021.11.032.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosures
All authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Tiuri E. Kroese and George S. Buijs are shared first authors.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kroese, T.E., Buijs, G.S., Burger, M.D.L. et al. Metastasectomy or Stereotactic Body Radiation Therapy With or Without Systemic Therapy for Oligometastatic Esophagogastric Cancer. Ann Surg Oncol 29, 4848–4857 (2022). https://doi.org/10.1245/s10434-022-11541-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-022-11541-0