Abstract
Background
Melanoma and the immune system are intimately related. However, the association of immunosuppressive medications (ISMs) with survival in melanoma is not well understood. The study evaluated this at a population level.
Methods
A cohort of patients with a diagnosis of invasive cutaneous melanoma (2007–2015) was identified from the Ontario Cancer Registry and linked to identify demographics, stage at diagnosis, prescription of immunosuppressive medications (both before and after diagnosis), and outcomes. The demographics of patients with and without prescriptions for ISM were compared. Patients eligible for Ontario’s Drug Benefit Plan were included to ensure accurate prescription data. The primary outcome was overall survival. Cox Proportional Hazards Regression models identified factors associated with mortality, including use of ISM as a time-varying covariate.
Results
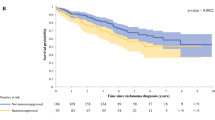
Of the 4954 patients with a diagnosis of cutaneous melanoma, 1601 had a prescription for ISM. The median age of the patients was 74 years. Overall, 58.4% of the patients were men (60.5% of those without ISM and 54% of those using ISM; p < 0.001). The use of oral immunosuppression was associated with an increased hazard of death (hazard ratio, 5.84; 95% confidence interval, 5.11–6.67; p < 0.0001) when control was used for age, disease stage at diagnosis, anatomic site, comorbidity, and treatment. Other factors associated with death were increasing age, male sex, increased disease stage, truncal location of primary melanoma, and inadequate treatment. In sensitivity analysis with steroid-only ISM use excluded, survival did not differ significantly (p = 0.355).
Conclusions
The use of immunosuppressive steroids for melanoma is associated with worse overall survival. Use of steroids should be limited when possible.




Similar content being viewed by others
References
Džambová M, Sečníková Z, Jiráková A, et al. Malignant melanoma in organ transplant recipients: incidence, outcomes, and management strategies: a review of literature. Dermatol Ther. 2016;29:64–8. https://doi.org/10.1111/dth.12276.
Zattra E, Fortina AB, Bordignon M, Piaserico S, Alaibac M. Immunosuppression and melanocyte proliferation. Melanoma Res. 2009;19:63–8. https://doi.org/10.1097/cmr.0b013e328322fc20.
Barker JN, MacDonald DM. Eruptive dysplastic naevi following renal transplantation. Clin Exp Dermatol. 1988;13:123–5. https://doi.org/10.1111/j.1365-2230.1988.tb00676.x.
Leonardi GC, Falzone L, Salemi R, et al. Cutaneous melanoma: from pathogenesis to therapy (review). Int J Oncol. 2018;52:1071–80. https://doi.org/10.3892/ijo.2018.4287.
Kubica AW, Brewer JD. Melanoma in immunosuppressed patients. Mayo Clin Proc. 2012;87:991–1003. https://doi.org/10.1016/j.mayocp.2012.04.018.
McCarter MD, Baumgartner J, Escobar GA, et al. Immunosuppressive dendritic and regulatory T cells are upregulated in melanoma patients. Ann Surg Oncol. 2007;14:2854–60. https://doi.org/10.1245/s10434-007-9488-3.
Mahmoud F, Shields B, Makhoul I, et al. Immune surveillance in melanoma: from immune attack to melanoma escape and even counterattack. Cancer Biol Ther. 2017;18:451–69. https://doi.org/10.1080/15384047.2017.1323596.
Eggermont AMM, Blank CU, Mandala M, et al. Adjuvant pembrolizumab versus placebo in resected stage III melanoma. N Engl J Med. 2018;378:1789–801. https://doi.org/10.1056/nejmoa1802357.
Schachter J, Ribas A, Long GV, et al. Pembrolizumab versus ipilimumab for advanced melanoma: final overall survival results of a multicentre, randomised, open-label phase 3 study (KEYNOTE-006). Lancet. 2017;390:1853–62. https://doi.org/10.1016/s0140-6736(17)31601-x.
Robert C, Schachter J, Long GV, et al. pembrolizumab versus ipilimumab in advanced melanoma. clinical trial, phase III comparative study, multicenter study, randomized controlled trial, research support, non-U.S. gov’t. N Engl J Med. 2015;372:2521–32. http://dx.doi.org/10.1056/NEJMoa1503093.
Ribas A, Kefford R, Marshall MA, et al. Phase III randomized clinical trial comparing tremelimumab with standard-of-care chemotherapy in patients with advanced melanoma. clinical trial, phase III comparative study, randomized controlled trial, research support, non-U.S. gov’t. J Clin Oncol. 2013;31:616–22. http://dx.doi.org/10.1200/JCO.2012.44.6112.
Larkin J, Chiarion-Sileni V, Gonzalez R, et al. Combined nivolumab and ipilimumab or monotherapy in untreated melanoma. clinical trial, phase iii, comparative study, randomized controlled trial, research support, non-U.S. gov’t. N Engl J Med. 2015;373:23–34. http://dx.doi.org/10.1056/NEJMoa1504030.
DePry JL, Reed KB, Cook-Norris RH, Brewer JD. Iatrogenic immunosuppression and cutaneous malignancy. Clin Dermatol. 2011;29:602–13. https://doi.org/10.1016/j.clindermatol.2011.08.009.
Collins L, Quinn A, Stasko T. Skin cancer and immunosuppression. Dermatol Clin. 2019;37:83–94. https://doi.org/10.1016/j.det.2018.07.009.
Dahlke E, Murray CA, Kitchen J, Chan AW. Systematic review of melanoma incidence and prognosis in solid organ transplant recipients. Transplant Res. 2014;3:10. https://doi.org/10.1186/2047-1440-3-10.
Miao Y, Everly JJ, Gross TG, et al. De novo cancers arising in organ transplant recipients are associated with adverse outcomes compared with the general population. Transplantation. 2009;87:1347–59. https://doi.org/10.1097/tp.0b013e3181a238f6.
Matin RN, Mesher D, Proby CM, et al. Melanoma in organ transplant recipients: clinicopathological features and outcome in 100 cases. Am J Transplant. 2008;8:1891–900. https://doi.org/10.1111/j.1600-6143.2008.02326.x.
Greenberg JA, Hohmann SF, Hall JB, Kress JP, David MZ. Validation of a method to identify immunocompromised patients with severe sepsis in administrative databases. Ann Am Thorac Soc. 2016;13:253–8. https://doi.org/10.1513/annalsats.201507-415bc.
Cortegiani A, Madotto F, Gregoretti C, et al. Immunocompromised patients with acute respiratory distress syndrome: secondary analysis of the LUNG SAFE database. Crit Care. 2018;22:157. https://doi.org/10.1186/s13054-018-2079-9.
Frankenthaler A, Sullivan RJ, Wang W, et al. Impact of concomitant immunosuppression on the presentation and prognosis of patients with melanoma. Melanoma Res. 2010;20:496–500. https://doi.org/10.1097/cmr.0b013e32833e9f5b.
Benchimol EI, Guttmann A, Mack DR, et al. Validation of international algorithms to identify adults with inflammatory bowel disease in health administrative data from Ontario, Canada. J Clin Epidemiol. 2014;67:887–96. https://doi.org/10.1016/j.jclinepi.2014.02.019.
Widdifield J, Bombardier C, Bernatsky S, et al. An administrative data validation study of the accuracy of algorithms for identifying rheumatoid arthritis: the influence of the reference standard on algorithm performance. BMC Musculoskelet Disord. 2014;15:216. https://doi.org/10.1186/1471-2474-15-216.
Gershon AS, Wang C, Guan J, Vasilevska-Ristovska J, Cicutto L, To T. Identifying individuals with physcian diagnosed COPD in health administrative databases. COPD. 2009;6:388–94. https://doi.org/10.1080/15412550903140865.
Gershon AS, Wang C, Guan J, Vasilevska-Ristovska J, Cicutto L, To T. Identifying patients with physician-diagnosed asthma in health administrative databases. Can Respir J. 2009 2009;16:183–8. https://doi.org/10.1155/2009/963098.
Canada Go. Canada Vigilance adverse reaction online database. Retrieved xxxx at https://www.canada.ca/en/health-canada/services/drugs-health-products/medeffect-canada/adverse-reaction-database.html.
Board TPMPR. Use of the World Health Organization defined daily dose in Canadian drug utilization and cost analyses. In: System TNPDUI, editor. Government of Canada Website2010.
Look Hong NJ. Melanoma patterns of care in Ontario: a call for a strategic alignment of multidisciplinary care: response to letter. J Surg Oncol. 2018;117:1613. https://doi.org/10.1002/jso.25006.
Health Services Research & Development Center JHBSoPH. The Johns Hopkins University ACG System: State of the Art Technology and a Tradition of Excellence in One Integrated Solution. 2012. Retrieved xxxx at https://www.johnshopkinssolutions.com/wp-content/uploads/2014/04/ACG-White-Paper-Technical-Dec-2012-rev.pdf.
Buajatti E, Chiodo S, Watson T, et al. Ontario Atlas of Adult Mortality 1992–2015, Version 2.0: Trends in Public Health Units. Population Health Analytics Lab, Toronto, ON, 2018.
Gatov E, Kurdyak P, Sinyor M, Holder L, Schaffer A. Comparison of vital statistics definitions of suicide against a coroner reference standard: a population-based linkage study. Can J Psychiatry. 2018;63:152–60. https://doi.org/10.1177/0706743717737033.
Burchell AN, Raboud J, Donelle J, et al. Cause-specific mortality among HIV-infected people in Ontario, 1995–2014: a population-based retrospective cohort study. CMAJ Open. 2019;7:E1–7. https://doi.org/10.9778/cmajo.20180159.
Widdifield J, Paterson JM, Huang A, Bernatsky S. Causes of death in rheumatoid arthritis: how do they compare to the general population? Arthritis Care Res Hoboken. 2018;70:1748–55. https://doi.org/10.1002/acr.23548.
Abdel-Qadir H, Austin PC, Lee DS, et al. A population-based study of cardiovascular mortality following early-stage breast cancer. JAMA Cardiol. 2017;2:88–93. https://doi.org/10.1001/jamacardio.2016.3841.
Assi H, Wilson KS. Immune toxicities and long remission duration after ipilimumab therapy for metastatic melanoma: two illustrative cases. Curr Oncol. 2013;20:e165–9. https://doi.org/10.3747/co.20.1265.
Harsch IA. Hypothesis: does adrenalitis caused by immune checkpoint-inhibitors put melanoma patients at an elevated risk for recurrence? J Immunother Cancer. 2019;7:166. https://doi.org/10.1186/s40425-019-0651-8.
Weber JS, Hodi FS, Wolchok JD, et al. Safety profile of nivolumab monotherapy: a pooled analysis of patients with advanced melanoma. J Clin Oncol. 2017;35:785–92. https://doi.org/10.1200/jco.2015.66.1389.
Eggermont AMM, Kicinski M, Blank CU, et al. Association between immune-related adverse events and recurrence-free survival among patients with stage III melanoma randomized to receive pembrolizumab or placebo: a secondary analysis of a randomized clinical trial. JAMA Oncol. 2020. https://doi.org/10.1001/jamaoncol.2019.5570.
Sciascia S, Radin M, Roccatello D, Sanna G, Bertolaccini ML. Recent advances in the management of systemic lupus erythematosus. F1000Res. 2018;7. https://doi.org/10.12688/f1000research.13941.1.
Smolen JS, Landewé R, Breedveld FC, et al. EULAR recommendations for the management of rheumatoid arthritis with synthetic and biological disease-modifying antirheumatic drugs: 2013 update. Ann Rheum Dis. 2014;73:492–509. https://doi.org/10.1136/annrheumdis-2013-204573.
Matsuoka K, Kobayashi T, Ueno F, et al. Evidence-based clinical practice guidelines for inflammatory bowel disease. J Gastroenterol. 2018;53:305–53. https://doi.org/10.1007/s00535-018-1439-1.
Acknowledgements
This study was supported by the ICES, which is funded by an annual grant from the Ontario Ministry of Health and Long-Term Care (MOHLTC). The opinions, results, and conclusions reported in this paper are those of the authors and are independent from the funding sources. No endorsement by ICES or the Ontario MOHLTC is intended or should be inferred. Parts of this material are based on data and information compiled and provided by CIHI. Parts of this material are based on data and information provided by Cancer Care Ontario (CCO). The opinions, results, view, and conclusions reported in this paper are those of the authors and do not necessarily reflect those of CCO or CIHI. The authors thank IMS Brogan Inc. for use of their Drug Information Database. The authors also acknowledge Service Ontario for use of the ORG-D data. This study was funded by internal philanthropic funding (Doug and Adrianne Mahaffy).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This paper was accepted as a poster to the annual Society of Surgical Oncology meeting, and was virtually presented in August 2020.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Bogach, J., Wright, F.C., Austin, J. et al. Medical Immunosuppression and Outcomes in Cutaneous Melanoma: A Population-Based Cohort Study. Ann Surg Oncol 28, 3302–3311 (2021). https://doi.org/10.1245/s10434-020-09224-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-020-09224-9