Abstract
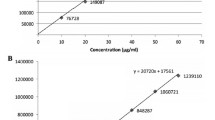
A simple high-performance liquid chromatographic method for simultaneous determination of ethylenediaminetetraacetic acid (EDTA), sorbic acid, and diclofenac sodium was developed and validated. Separation was achieved on a C18 column (10 cm × 4.6 mm) using gradient elution. The mobile phase consisted of acetonitrile–ammonium dihydrogen phosphate buffer solution (0.01 M, pH = 2.5, containing 0.8% tetra-n-butyl ammonium hydroxide). The detector wavelength was set at 254 nm. Under these conditions, separation of three compounds was achieved in less than 10 min. The effect of two metal salts and metal concentration on peak area of EDTA was investigated. The pH effect on retention of EDTA and sorbic acid was studied. The method showed linearity for EDTA, sorbic acid, and diclofenac in the ranges of 2.5–100.0, 5.0–200.0, and 20.0–120.0 μg/mL, respectively. The within- and between-day relative standard deviations ranged from 0.52 to 1.94%, 0.50 to 1.34%, and 0.78 to 1.67% for EDTA, sorbic acid, and diclofenac, respectively. The recovery of EDTA, sorbic acid, and diclofenac from pharmaceutical preparation ranged from 96.0–102.0%, 99.7–101.5%, to 97.0–102.5%, respectively. To the best of our knowledge, this is the first report about simultaneous determination of EDTA, sorbic acid, and diclofenac.





Similar content being viewed by others
REFERENCES
Rowe RC, Sheskey PJ, Weller PJ. Handbook of pharmaceutical excipients. 4th ed. London: Science and Practice; 2003. p. 225.
Charvalos E, Tzatzarakis M, Tsatsakis A, Petrikkos G. Controlled release of water-soluble polymeric complexes of sorbic acid with antifungal activities. Appl Microbiol Biotechnol. 2001;57:770–5.
Christianah MA, Li PK, Florey K. Analytical profiles of drug substances, vol. 19. NY: Academic; 1998. p. 123–40.
Dale M, Parfit K. The complete drug reference. 32nd ed. UK: Pharmaceutical Press; 1999. p. 32.
Sillanpää M, Sihvonen ML. Analysis of EDTA and DTPA. Talanta. 1997;44:1487–97.
Laine P, Matilainen R. Simultaneous determination of DTPA, EDTA, and NTA by UV–visible spectrometry and HPLC. Anal Bioanal Chem. 2005;382:1601–9.
Nowack B, Kari FG, Hilger SU, Sigg L. Determination of dissolved and adsorbed EDTA species in water and sediments by HPLC. Anal Chem. 1996;68:561–6.
Laamanen PL, Blanco E, Cela R, Matilainen R. Improving sensitivity in simultaneous determination of copper carboxylates by nonaqueous capillary electrophoresis. J Chromatogr A. 2006;1110:261–7.
Laamanen PL, Mali A, Matilainen R. Simultaneous determination of DTPA, EDTA, and NTA by capillary electrophoresis after complexation with copper. Anal Bioanal Chem. 2005;381:1264–71.
Fung YS, Luk SF. Polarographic determination of sorbic acid in fruit juices and soft drinks. Analyst. 1990;115:1219–21.
García I, Ortiz MC, Sarabia L, Vilches C, Gredilla E. Advances in methodology for the validation of methods according to the International Organization for Standardization: application to the determination of benzoic and sorbic acids in soft drinks by high-performance liquid chromatography. J Chromatogr A. 2003;992:11–27.
Saad B, Bari MF, Saleh MI, Ahmad K, Talib MKM. Simultaneous determination of preservatives (benzoic acid, sorbic acid, methylparaben and propylparaben) in foodstuffs using high-performance liquid chromatography. J Chromatogr A. 2005;1073:393–7.
Tang Y, Wu M. The simultaneous separation and determination of five organic acids in food by capillary electrophoresis. Food Chem. 2007;103:243–8.
Lau OW, Luk SF. Iodimetric method for the determination of sorbic acid in soft drinks. Analyst. 1987;112:1269–72.
de Micalizzi YC, Pappano NB, Debattista NB. First and second order derivative spectrophotometric determination of benzyl alcohol and diclofenac in pharmaceutical forms. Talanta. 1998;47:525–30.
de Cόrdova MLF, Barrales PO, Daíz AM. Sensitive and selective determination of diclofenac sodium in pharmaceutical preparations by solid phase ultraviolet absorptiometry. Anal Chim Acta. 1998;369:263–8.
Sena MM, Chaudhry ZF, Collins CH, Poppi RJ. Direct determination of diclofenac in pharmaceutical formulations containing B vitamins by using UV spectrophotometry and partial least squares regression. J Pharm Biomed Anal. 2004;36:743–9.
Castellano PM, Vignaduzzo SE, Maggio RM, Kaufman TS. Application of a chemometric method for simultaneous determination of acetaminophen and diclofenac in content-uniformity and drug-dissolution studies. Anal Bioanal Chem. 2005;382:1711–4.
Arancibia JA, Boldrini MA, Escandar GM. Spectrofluorimetric determination of diclofenac in the presence of α-cyclodextrin. Talanta. 2000;52:261–8.
Patil ST, Sundaresan M, Bhoir IC, Bhagwat AM. Packed column supercritical fluid chromatographic separation and estimation of acetaminophen, diclofenac sodium and methocarbamol in pharmaceutical dosage forms. Talanta. 1998;47:3–10.
González L, Yuln G, Volonté MG. Determination of cyanocobalamin, betamethasone, and diclofenac sodium in pharmaceutical formulations, by high performance liquid chromatography. J Pharm Biomed Anal. 1999;20:487–92.
Abdel-Hamid ME, Novotny L, Hamza H. Determination of diclofenac sodium, flufenamic acid, indomethacin and ketoprofen by LC-APCI-MS. J Pharm Biomed Anal. 2001;24:587–94.
Mayer BX, Namiranian K, Dehghanyar P, Stroh R, Mascher H, Muller M. Comparison of UV and tandem mass spectrometric detection for the high-performance liquid chromatographic determination of diclofenac in microdialysis samples. J Pharm Biomed Anal. 2003;33:745–54.
Chmielewska A, Konieczna L, Plenis A, Lamparczyk H. Sensitive quantification of chosen drugs by reversed-phase chromatography with electrochemical detection at a glassy carbon electrode. J Chromatogr B. 2006;839:102–11.
Nebot C, Gibb SW, Boyd KG. Quantification of human pharmaceuticals in water samples by high performance liquid chromatography–tandem mass spectrometry. Anal Chim Acta. 2007;598:87–94.
Panusa A, Multari G, Incarnato G, Gagliardi L. High-performance liquid chromatography analysis of anti-inflammatory pharmaceuticals with ultraviolet and electrospray-mass spectrometry detection in suspected counterfeit homeopathic medicinal products. J Pharm Biomed Anal. 2007;43:1221–7.
Cakirer O, Kilic E, Atakol O, Kenar A. The non-aqueous titrimetric assay of the selected anti-inflammatory agents using tetra-n-butylammonium hydroxide as titrant. J Pharm Biomed Anal. 1999;20:19–26.
Pimenta AM, Araújo AN, Montenegro MCBSM. Simultaneous potentiometric and fluorimetric determination of diclofenac in a sequential injection analysis system. Anal Chim Acta. 2002;470:185–94.
Shamsipur M, Jalali F, Ershad S. Preparation of a diclofenac potentiometric sensor and its application to pharmaceutical analysis and to drug recovery from biological fluids. J Pharm Biomed Anal. 2005;37:943–7.
Hassan SSM, Mahmoud WH, Elmosallamy MAF, Almarzooqui MH. Iron (II)-phthalocyanine as a novel recognition sensor for selective potentiometric determination of diclofenac and warfarin drugs. J Pharm Biomed Anal. 2005;39:315–21.
Mazurek S, Szostak R. Quantitative determination of diclofenac sodium and aminophylline in injection solutions by FT-Raman spectroscopy. J Pharm Biomed Anal. 2006;40:1235–42.
Cagnasso CE, López LB, Rodríguez VG, Valencia ME. Development and validation of a method for the determination of EDTA in non-alcoholic drinks by HPLC. J Food Comp Anal. 2007;20:248–51.
ACKNOWLEDGMENTS
This research work was supported by Bakhtar Bioshimi Pharmaceutical Company and Iran National Science Foundation (INSF). The author would like to thank Mr. Bahman Barazesh for the excellent and open collaboration.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Heydari, R., Shamsipur, M. & Naleini, N. Simultaneous Determination of EDTA, Sorbic Acid, and Diclofenac Sodium in Pharmaceutical Preparations Using High-Performance Liquid Chromatography. AAPS PharmSciTech 14, 764–769 (2013). https://doi.org/10.1208/s12249-013-9962-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1208/s12249-013-9962-0