Abstract
Background
The association between epicardial fat thickness and coronary artery disease (CAD) has been evaluated previously using echocardiography. Recently, multidetector computed tomography (MDCT), as a valuable tool in cardiovascular CT imaging, can improve characterization of CAD and give a more accurate volumetric quantitation of EF. The purpose of our study was to evaluate the relationship between the epicardial fat volume and CAD using multi-detector row CT.
Results
Out of the studied 120 patients, 22 patients were negative for CAD, while 98 patients had positive CAD. There was significant difference between both groups as regard epicardial fat volume (p < 0.001), and good relation was found between the amount of epicardial fat volume and coronary calcium score, number of affected vessel, plaque burden and degree of stenosis (p = < 0.001).
Conclusion
EAT volume was larger in the presence of obstructive CAD and atheromatous plaques. These data suggest that EAT is associated with the development of coronary atherosclerosis and potentially the most dangerous types of plaques.
Similar content being viewed by others
Background
Epicardial fat (EF) is the adipose tissue surrounding the heart constrained by the visceral pericardium and has the same origin as abdominal visceral fat. It encases coronary arteries without an intervening fascia and also has same innervation and blood supply as the coronary arterial wall. This unique anatomic relationship makes the EF being strongly related to the development of coronary artery disease (CAD) [1, 2].
The accumulation of EF is a rich source of free fatty acids (FFAs) and some inflammatory Cytokines and pro-atherogenic mediators that aggravate vessel wall inflammation and facilitate the progression of atherosclerosis and thus the development of CAD [3, 4].
Previously, the association between epicardial fat thickness and CAD has been evaluated using echocardiography. Nowadays, the introduction of multidetector computed tomography (MDCT) provides more objective results and a better quantitation of EF compared to the echocardiographic assessment due to its high acquisition speed, improved spatial resolution, intravenously contrast material bolus timing and reduced motion artifacts [5,6,7].
Multi-detector computed tomography (MDCT) allows for simultaneous assessment of coronary artery calcium (CAC), coronary artery stenosis and presence coronary plaque, plaque composition and plaque vulnerability as well as epicardial fat volume (EFV) [8].
Many studies have assessed the epicardial fat volume (EFV) and reported a good relationship between it and coronary artery calcium using MDCT, EFV increased in patients with significant coronary artery stenosis and in those with severe coronary artery calcification, and this increase in EFV was found to be associated with other biomarkers of disease severity, such as the Coronary Calcium Score and the Leaman Score [7, 9].
Therefore, the epicardial fat volume can be considered a novel biomarker useful for characterization of the severity of coronary artery disease and identification of patients at risk for CAD in addition to coronary angiography [8].
The aim of this study was to evaluate the relationship between the amount of epicardial fat and coronary artery disease using multi-detector row CT and to assess its added value to predict coronary artery disease latter on.
Methods
Study population
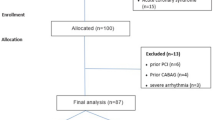
This prospective study was enrolled on 120 patients for multislice CT angiography of coronary arteries and measurement of epicardial fat volume. They were referred from cardiology department and private clinics with chest pain or dyspnea on effort to Radio-diagnosis department over a period from November 2018 and November 2020.
Approval of Research Ethics Committee (REC) and informed consent were obtained from all participants in this study after explanation of the benefits and risks of the procedure. Privacy and confidentiality of all patients' data were guaranteed. All data provision were monitored and used for scientific purpose only.
Any alert cooperative patient with symptoms suspecting coronary artery disease (as chest pain or dyspnea on effort) and referred for coronary CT angiography was included in this study. No gender predilection.
Exclusion criteria were patients refusing to participate in research, critically ill patients unable to perform breath hold required for scan, patients with severe uncontrollable cardiac arrhythmia or severe heart failure, patients with orthopnea, patients with allergy to the iodinated contrast material, patient with impaired renal function (creatinine level > 1.5 mg/dl), patients with left ventricular ejection fraction of less than 40%, patients with weight over 150 kg, and pregnant female patients.
All the included participants were subjected to the following
Data collection
-
Proper history taking, including personal history, history of the presenting symptom, and any medical diseases like diabetes mellitus or hypertension that may lead to progression of CAD.
-
Review of previous investigations, including echocardiography, recent stress ECG and stress echocardiography to detect any positive results for myocardial ischemia. We also reviewed the procedural data of previous PCI and CABG surgery if available.
Clinical examination
It included measuring patient’s height and weight to calculate body mass index and vital signs monitoring including heart rate and blood pressure measurement as a baseline for heart rate control during CT examination.
Laboratory investigations
-
Recent serum creatinine level.
-
Lipid profile, if available, including triglycerides and cholesterol levels.
Radiological examination:
The study was conducted using 320 row CT scanner (Aquilion one system, Toshiba Medical Systems, Tokyo, Japan). Multislice CT coronary angiography was done for all patients by these steps:
Patient preparation
-
Heart rate control:
-
Patients with heart rate above 65 bpm were given oral β-blockers one hour before scan.
-
Blood pressure and heart rate were monitored every 30 min till the desired heart rate (less than 65 bpm) was reached.
-
-
At scanner room:
-
The patients were supine on table with arms raised above their heads.
-
ECG electrodes were applied to chest wall, and ECG trace was monitored to ensure good amplitude of R wave that was used for scan trigger.
-
CT scan protocol
First, an anterior-posterior scout topogram was taken. Second, non-contrasted scan was taken for calcium scoring. The third step was the contrast medial injection; Non-ionic contrast media (Ultravist 370 mgI/ml) was injected through the peripherally inserted IV cannula using dual-head powered automatic injector (Stellant D, Medrad, Indianola, PA, USA) followed by 50 cc saline flushing. Contrast volume (in ml) = (scan time + 10) x injection flow rate. The injection flow rates were adapted according to Kv used; 100 kV: 4.0 ml/sec, 120 kV: 5.0 ml/sec and 135 kV: 6.0 ml/sec.
Image acquisition and reconstruction
-
Acquisition parameters: 0.35 s gantry rotation time, 100–135 kV, and variable mA according to patient body habitus.
-
Prospective ECG gating was used with volume scanning method.
-
Images were reconstructed at a slice thickness of 0.5 mm and 0.5 mm interval with smooth and sharp reconstruction kernels (FC03 and FC05 respectively) at 75% of R-R interval and at the best diastolic phase. The best systolic phase was also reconstructed in those whom scan window was set to include 30–80% of R-R interval.
Post-processing
The reconstructed images were transferred to workstation (Vitrea Fx, vital images, USA) to obtain multiplanar images in axial, sagittal and coronal planes as well as Maximum intensity projections, 3D Volume rendering technique and Curved Multiplanar Reconstruction images.
Image analysis
All CT images of studied patients were analyzed by two radiologists with 9 and 5 years of cardio-vascular CT experience, blinded to the clinical data in a standard clinical Picture Archiving and Diagnostic System workstation, and final decisions reached by consensus were reported. Image analysis included:
-
Assessment of image quality to exclude blooming or motion artifacts.
-
Evaluation of calcium score
Coronary calcium was defined as an area of at least three “face-connected” voxels in the axial plane in the course of a coronary artery, with an attenuation threshold value of 130 HU or greater. Calcium scores of each investigation were calculated and expressed as Agatston scores for standard of reference CT calcium score.
-
Evaluation of coronary arteries including
Assessment of coronary artery anatomy for possible coronary artery anomalies.
Characterization of coronary plaques and degree of stenosis; total plaque burden was calculated by adapting Leaman score which was calculated by multiplying the weighing factors regarding plaque composition, stenosis severity and location for a given segment.
-
Evaluation epicardial fat volume:
The visceral pericardium was traced manually from the root of the great vessels to the left ventricular apex, and all extra-pericardial tissue were excluded. These images were then segmented using an attenuation threshold varying between − 5 HU and − 250 HU in each slice. This effectively excluded myocardium, coronary arteries, coronary calcium, the aorta and blood pool. The epicardial fat at each level was then summed across slices and multiplied by the slice thickness and number of slices to determine “total epicardial fat volume.”
Statistical analysis
Data were fed to the computer and analyzed using IBM SPSS software package version 20.0. (Armonk, NY: IBM Corp). Qualitative data were described using number and percent. The Kolmogorov–Smirnov test was used to verify the normality of distribution. Quantitative data were described using range (minimum and maximum), mean, standard deviation, median and interquartile range (IQR). Significance of the obtained results was judged at the 5% level. Chi-square test was used for categorical variables, to compare between different groups, and Fisher’s Exact or Monte Carlo correction was used for correction for Chi-square when more than 20% of the cells have expected count less than 5. Student t-test was used or normally distributed quantitative variables, to compare between two studied groups, and F-test (ANOVA) for normally distributed quantitative variables, to compare between more than two groups, Pearson coefficient was used to correlate between two normally distributed quantitative variables, and Spearman coefficient was used to correlate between two distributed abnormally quantitative variables. Logistic Regression was used to detect the most affecting factor for affecting patients’ vessel stenosis ≥ 50%. Receiver operating characteristic curve (ROC) was tested to calculate the diagnostic ability of quantitative variable in prediction of categorical outcome.
Results
This study was conducted on 120 patients, twenty-two subjects were negative for coronary artery disease, and 98 patients were positive for coronary artery disease. Among the whole sample, 83 patients (69.2%) were males, while 37 of them (30.8%) were females. The age of the studied patients ranged from 28 to 73 years with mean age of 56.47 ± 9.15 years. Forty-seven studied patients (39.2%) were at the age group that ranged from 50 to > 60; 30 patients of them were males, and 17 patients were females.
Different risk factors for coronary artery disease were found in the studied patients including BMI > 25 in 116 patients (96.6%), dyslipidemia in 72 patients (60%), positive family history in 64 patients (35.3%), hypertension in 38 patients (31.7%), smoking in 30 patients (25%), and diabetes mellitus in 19 patients (15.8%).
In this study, 22 patients had negative CTA and 98 patients had positive CTA for CAD (Figs. 1, 2, 3 and 4). Among patients with positive CTA for CAD, 71 patients had no previous stent/surgery and 27 patients were post-stent/ CABG. After comparing three groups as regard age, sex, traditional risk factors, BMI and family history of cardiac diseases, there was significant difference between them as regard gender distribution (p = < 0.001), age (p = < 0.001) and hypertension (p = 0.001) which means strong correlation between these risk factors and coronary artery disease in selected sample. Comparison between the different studied groups according to different parameters is illustrated in Table 1.
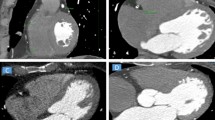
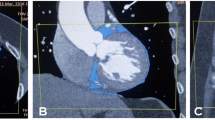
A–G images: A 65-year-old male patient (BMI = 33.6), dyslipidemic, non-hypertensive and non-diabetic, complaining of chest pain. Echocardiography revealed good systolic function (EF = 60%) and no regional wall motion abnormalities. A Two different views of 3D volume rendered images showing normal origin of coronary arteries with right dominant circulation, and mild ectasia at LAD proximal LAD segment. B Axial noncontrast curved planar reformatted image showing calcium score (17) at LAD and D1. C Axial curved planar reformatted image and D Two sagittal curved planar reformatted images showing atherosclerotic showing LAD, with mild proximal LAD ectasia (5.3 mm), a soft eccentric plaque is seen at proximal to mid-LAD segments exerting moderate stenosis (50%, length = 11 mm), with another small calcified plaque with positive remolding is seen at mid-LAD segment without significant stenosis, and a mixed eccentric plaque is seen at D1 ostium exerting severe stenosis (70%, length = 4.5 mm). E Two coronal curved planar reformatted image showing atherosclerotic LM, LCx, OM3, RCA, PDA and PLB without significant disease. F 3D volume rendered image and G 2D axial multiplanar reformatted image showing epicardial fat volume (223 cm3)
A–H images: 54-year-old male patient (BMI = 30.8), smoker, non-hypertensive and non-diabetic; and complaining of chest pain. Echocardiography revealed good systolic function (EF = 67%), and no regional wall motion abnormalities. A Two different views of 3D volume rendered images showing normal origin of coronary arteries with right dominant circulation. B 3D volume rendered image and C Axial maximum intensity projection image showing LV mid-inferoseptal divertiruclum. D Axial noncontrast curved planar reformatted image showing calcium score (1) only at LCx. E Two axial curved planar reformatted images showing atherosclerotic LM and LAD, with soft eccentric plaque with positive remolding is seen at LAD proximal segment exerting moderate stenosis (50%, length = 14 mm) displaying napkin ring sign (vulnerable plaque), and atherosclerotic LCx, OM1 and OM2, with no significant disease. Also, LAD mid-segment has superficial intra-myocardial course for about 13.4 mm being covered with 2.3 mm myocardial tissue without luminal compression. F Coronal curved planar reformatted image showing atherosclerotic RCA and PDA. A soft ulcerated concentric plaque is seen at RCA distal segment exerting severe stenosis (80%, length = 15 mm). G 3D volume rendered image and (H) 2D axial multiplanar reformatted image showing epicardial fat volume (197 cm3)
A–F images: A 67-year-old male patient (BMI = 31.1), smoker, dyslipidemic, hypertensive and non-diabetic, and complaining of chest pain and exertional dyspnea. Echocardiography revealed good systolic function (EF = 63%), dilated aortic root 4 cm, trivial mitral valve incompetent and no regional wall motion abnormalities. A Two different views of 3D volume rendered images, B Sagittal maximum intensity projection image, and C Coronal curved planar reformatted image showing sizable elongated tortuous abnormal arterial channel arising from the right superolateral wall of the LAD midsegment opposite D3 takeoff, arising superiorly crossing over RVOT being closely adherent to anterior wall of MPA then turning posteriorly and inferiorly between MPA and aortic root appearing to be ending blindly, and also, ectatic LM and LAD proximal and mid-segments with calcified eccentric plaques seen at LM ostium without significant stenosis and eccentric calcified plaque at LAD mid-segment exerting mild stenosis 30%. D Two axial curved planar reformatted image showing ectatic LM and mildly atherosclerotic LCx and OM branches with mild cardiac motion artifact at LCx mid- and distal segments with no evidence of significant coronary artery disease, and patent minimally atherosclerotic RCA and PLB with no significant disease. E 3D volume rendered image and F 2D axial multiplanar reformatted image showing epicardial fat volume (175 cm3)
A–G images: 72-year-old male patient (BMI = 28), dyslipidemic, hypertensive and non-diabetic, and complaining of exertional dyspnea and palpitation. Echocardiography revealed good systolic function (EF = 58%), mild mitral incompetence with sclerotic aortic valve without obstruction, mild tricuspid incompetence and segmental wall motion abnormalities at rest in the form of basal inferior and posterior hypokinesia. A Two different views of 3D volume rendered images, and B Sagittal curved planar reformatted image showing patent LIMA to distal LAD and SVG to OM1 with good opacification of distal LAD and OM1 segments beyond the anastomotic sites, and diffusely diseased RCA with occluded proximal LAD segment. C Two axial curved planar reformatted images showing patent SVG to OM1 without focal stenosis with good filling of the OM artery beyond the anastomotic site, and occluded distal LM proximal LAD with soft plaque. D Two coronal curved planar reformatted images showing occluded distal LM proximal LAD with soft plaque, the distal LAD beyond the anastomotic site is patent and free of significant disease, and occluded distal LM proximal LCx with soft plaque. E Sagittal curved planar reformatted image showing diffusely disease atherosclerotic RCA with patent ostium and totally occluded proximal, mid- and distal segments by soft and mixed plaques. F 3D volume rendered image and G 2D axial multiplanar reformatted image showing epicardial fat volume (247 cm3)
As regard coronary circulation dominance, 91 patients (75.8%) were with right dominant circulation as shown in (Fig. 1A), 20 patients (16.7%) were with left dominant circulation and 9 patients (7.5%) were with co-dominant circulation. As regard coronary arteries origin, 104 patients (86.7%) were with normal coronary origin (86.7%), two separate ostia of LAD and LCx was found at 6 patients (5%), high LM takeoff was found at 6 patients (5%) and anomalous origin was found at 4 patients (3.3%) (Fig. 5).
A–F images: A 48-year-old female patient (BMI = 35.7), dyslipidemic, non-diabetic and non-hypertensive, complaining of chest pain and exertional dyspnea. Echocardiography revealed good systolic function (EF = 66%), no regional wall motion abnormalities. A Two different views of 3D volume rendered images, B Axial Maximum Intensity Projection (MIP) image, and C Sagittal curved planar reformatted image showing mildly ectatic proximal segment of LAD with anomalous origin of the RCA from mid-LAD just opposite to D2 takeoff and right dominant circulation. Another small coronary artery arises from right coronary cusp supplying SA node branch and becomes very diminutive in caliber distally. D Axial curved planar reformatted image of LM and Ramus intermedius. E 3D volume rendered image and F 2D axial multiplanar reformatted image showing epicardial fat volume (212 cm3)
Calcium score was estimated in the studied patients represented by Agatston units that ranged from zero to 516 with median (IQR) 17.50 (0.0–83.50), stratified by Zero Agatston units in 36 patients (30%), minimal (1–10) in 13 patients (10.8%), mild (11–100) in 43 patients (35.8%), moderate (101–400) in 27 patients (22.5%) and more than 400 at only one patient (0.8%). Additionally, adapted Leaman score was estimated for patients with positive CTA for CAD with its value ranged from 0.46 to 17.25 with median (IQR) 4.61 (3.23–6.77).
Regarding degree of stenosis, 38 patients (13.6%) had lesion/s less than 50% stenosis and 60 patients (50%) had lesion/s ≥ 50% stenosis (Fig. 4). As regard number of vessel affection, 34 patients (28.3%) had one vessel disease, 33 patients (27.5%) had two vessel disease (Fig. 2E, F), 23 patients (19.25%) had three vessel disease and 8 patients (6.7%) had multi-vessel disease.
Soft plaques were detected at 53 patients (44.2%), mixed plaques were detected at 70 patients (58.3%), and calcified plaques were detected at 46 patients (38.3%). As regard high-risk plaques, napkin ring sign was detected at only 10 patients (8.3%) as shown at (Fig. 2E).
The positive CTA 98 patients were subdivided according to segmental distribution into:
-
LM disease in 17 patients (14.2%); 12 patients of them (16.9%) without previous intervention/surgery.
-
Ostial and ostio-proximal segmental affection; 64 patients (53.3%) had ostial and ostio-proximal LAD affection, 18 patients (15%) had ostial and ostio-proximal LCx affection, and 23 patients (19.2%) had ostial and ostio-proximal RCA affection.
-
Mid-segments affection; 35 patients (53.3%) had mid-LAD segment affection, 4 patients (3.3%) had mid-LCx segment affection, and 14 patients (11.7%) had mid-RCA segment affection.
-
Distal segments affection; 3 patients (2.5%) had distal LAD segment affection, and 13 patients (10.8%) had distal RCA segment affection.
-
Branches affection; 6 patients (5%) had ramus intermedius lesions, 15 patients (12.5%) had D1 lesions (Fig. 1C, D), 4 patients (3.3%) had D2 lesions without previous intervention/surgery, 19 patients (15.8%) had OM1 lesions, 2 patients (1.7%) had PDA lesions, and 3 patients (2.5%) had PLB lesions.
There was significant difference between negative CTA patients and positive CTA patients as regard epicardial fat volume (t test = 5.467, p < 0.001). By Receiver operator characteristic curve (ROC curve), epicardial fat volume > 124 cm3 is identified as optimal cut off value (sensitivity 78.57%, Specificity 72.73%) to detect positive CTA patients (AUC = 0.833, p = < 0.001). These findings are illustrated in Table 2.
As regard traditional risk factors, there was significant correlation between epicardial fat volume and gender distribution (t test = 2.402, p = 0.018), dyslipidemia (t test = 4.310, p = < 0.001) and hypertension (t test = 2.258, p = 0.026). There was also significant correlation between epicardial fat volume and BMI (F test = 27.988, p = < 0.001), mean value of epicardial fat volume increased from 61.0 ± 7.07 to 223.6 ± 46.04 at the range from 18.5–24.9 to > 35 and median value increased from 60.50 to 246.0 at the same range.
A significant correlation between epicardial fat volume and coronary artery disease was found represented by multiple variables. Firstly, there was strong significant correlation between epicardial fat volume and calcium score Agatston units; the mean value of cumulative number of the epicardial fat volume increased from 127.2 cm3 ± 39.27 to 177.7 cm3 ± 32.89 at the range from 1–10 to (101–400)/ > 400 and median value increased from 124 cm3 to 168.5 cm3 at the same range (F = 15.048, p = < 0.001) as shown in Table 3. By Pearson coefficient, there was a strong positive correlation between epicardial fat volume and calcium score (r = 0.713, p = < 0.001) as illustrated at Fig. 6.
There was also significant correlation between epicardial fat volume and degree of stenosis (f = 12.975, p = < 0.001); the mean value of cumulative number of the epicardial fat volume was significantly higher at patients with lesions exerting diameter reduction ≥ 50% (F = 15.048, p = < 0.001). By spearman coefficient, there was strong positive correlation between epicardial fat volume and number of vessel affection (r = 0.782, p = < 0.001) as shown in Table 4.
Also, a significant correlation between epicardial fat volume and all plaque types (soft, mixed and calcified plaques) was detected with no specific predilection to specific plaque type. The mean value of cumulative number of the epicardial fat volume was significantly higher at patients with soft, mixed and calcified plaque lesions (p = 0.01, p = 0.001 and p = < 0.001 for soft, mixed and calcified plaque respectively). Controversy, there was no significant difference between mean value of cumulative number of the epicardial fat volume for patients had high-risk plaque criteria (NRS) and patients with no detectable high-risk plaque criteria (t = 1.374, p = 0.172) as shown in Table 5.
As regard segment involvement stenosis, the mean value of the cumulative number of the epicardial fat volume was significantly higher at patients with ostial and ostio-proximal LAD lesions, mid-segment LAD lesions, and mid- segment LCx lesions (p = 0.005, p = 0.003 and p = 0.048 respectively) as illustrated in Table 6.
In patients with obstructive coronary artery disease (≥ 50% stenosis), age, sex, dyslipidemia, hypertension, epicardial fat volume at cutoff value > 124 cm3 & calcium score ≥ 101 were univariate significant risk factors/ predictors for obstructive coronary artery disease. In multivariate logistic regression analysis, four models were proposed; model A retained all candidates risk factors. Calcium score (≥ 101) and epicardial fat volume > 124 cm3 were added resulting in exclusion of dyslipidemia and hypertension as significant risk factors. In model B, calcium score was a major risk factor/predictor. In model C, epicardial fat volume > 124 cm3 was a major risk factor/predictor. In model D, epicardial fat volume > 124 cm3 was the most powerful risk factor/predictor, calcium score was adjusted, but still significant risk factor/ predictor. By Hosmer and lemeshow test for goodness of fit model, model D has the best performance. These findings are illustrated in Tables 7 and 8.
On comparing the proposed models using receiver-operator characteristic curves, the area under curve was higher for model D (0.984, 95% CI 0.959–1.009) than those for model A (0.683, 95% CI 0.545–0.864), model B (0.704, 95% CI 0.545–0.864) and model C (0.885, 95% CI 0.74–1.03) as illustrated in Table 9.
Discussion
Considering its noninvasive technique and recent advances in temporal and spatial resolution, the measurement of epicardial fat volume by MDCT might be helpful in the prevention and treatment of coronary artery disease as it can early detect the risk factors and the sequences of these risk factors. Multidetector computed tomography (MDCT) allows simultaneous assessment of coronary artery calcium (CAC), coronary artery stenosis, coronary plaques and epicardial fat volume (EFV) without increased radiation exposure or cost [10].
This study was conducted on 120 patients divided into three groups; (22 patients) with no coronary artery disease, (71 patients) with coronary artery disease without previous stent/operation, and (27 patients) with post-stent/CABG coronary artery disease.
Coronary artery disease (CAD) is a global burden and mortality leader in 31% of all deaths worldwide. Many risk factors can predispose to CAD including obesity, hypertension, diabetes mellitus, smoking and high cholesterol. Atherosclerosis is considered the main cause for developing Coronary artery disease [11].
The most common risk factor for coronary artery disease in the current study was BMI > 25 in 96.6%, followed by dyslipidemia in 60% of the patients with significant difference between three groups as regard gender distribution (p = < 0.001), age (p = < 0.001) and hypertension (p = 0.001) which means strong correlation between these risk factors and coronary artery disease in selected sample.
Our results were supported by study of Khurana et al. [12] which revealed that the prevalence of either of the traditional risk factors (smoking, hypertension, diabetes mellitus, family history of CAD, elevated serum cholesterol, elevated LDL, low serum HDL, elevated serum triglycerides) was significantly higher in the “CAD present” group as compared with the “CAD absent” group. No significant difference was seen in the distribution of weight and body mass index (BMI) between the two groups. However, Shehata, et al. [13] reported no statistically significant difference concerning age or sex distribution characteristics among the studied groups (p value: 0.17 and 0.09, respectively).
The epicardial adipose tissue (EAT) covers 80% of the cardiac surface, mostly in the grooved segments along the paths of the coronary arteries making them embedded in this fat, and this creates a perfect environment for a local interaction between a metabolically active EAT and the coronary vessels. As the EF volume virtually increases, it becomes hypoxic and dysfunctional with increased lipolysis and resultant inflammation causing metabolic shift, and alteration of the homeostasis, and thus progression of atherosclerotic plaques [14, 15].
Early identification and treatment of atherosclerotic plaques before narrowing of coronary artery lumen can prevent and possibly even reverse the progression of CAD, returning the plaques to a stable form [16].
Calcium score can reflect the overall burden of coronary atherosclerosis and may predict the risk of future CAD events [15]. It was estimated in this study represented by Agatston units and ranged from zero to 516 with median (IQR) 17.50 (0.0–83.50). Additionally, adapted Leaman score was estimated for patients with positive CTA with its value ranged from 0.46 to 17.25 with median (IQR) 4.61 (3.23–6.77).
Bettencourt et al. [10] observed that EFV was positively related to the CAC score. This is proven in our study as increased CAC score was associated with increased EAT volume with a strong positive relationship between EFV and CAC score.
The present study showed that there was significant difference between negative CTA patients and positive CTA patients for CAD as regard epicardial fat volume (p < 0.001), epicardial fat volume > 124 cm3 is identified as optimal cut off value (sensitivity 78.57%, Specificity 72.73%) to detect positive CTA patients (p = < 0.001).
Kamal et al. [17] found that EFT of 5.5 mm is the cut-off value at which we can expect the presence of CAD with a high sensitivity and specificity. Faghihi et al. [18] found an EFT cut-off value of 2.95 mm in their study with a sensitivity of 83% and specificity of 75%; Eroglu et al. [19] found a cut-off point of 5.2 mm with 85% sensitivity and 81% specificity. The difference between them may be attributed to the difference in measurement of EFT either at end-systole or end-diastolic frames.
The current study showed a significant correlation between epicardial fat volume and gender distribution (p = 0.018), dyslipidemia (p = < 0.001), hypertension (p = 0.026), and BMI (p = < 0.001), strong positive correlation between epicardial fat volume and calcium score (p = < 0.001), significant correlation between epicardial fat volume and degree of stenosis, and significant correlation between epicardial fat volume and all plaque types (soft, mixed and calcified plaques) with no specific predilection to specific plaque type. As regard segment involvement stenosis, the mean value of the cumulative number of the epicardial fat volume was significantly higher at patients with ostial and ostio-proximal LAD lesions, mid-segment LAD lesions, and mid- segment LCx lesions.
Our results were supported by study of Alexopoulos et al. [20] as they reported that EAT volume correlated with age, height, body mass index (BMI), and CAC score. EAT volume increased significantly with the severity of luminal stenosis (p < 0.001), and in patients with no plaques, calcified, mixed, and non-calcified plaques (p < 0.001). The EAT volume was significantly larger in patients with mixed or non-calcified plaques compared to patients with calcified plaques or no plaques (all p < 0.01 or smaller).
In the study of Bastarrika et al. [21] performed using automatic volumetry of epicardial fat by MSCT, patients with significant coronary artery stenosis had significantly greater EFV than those without significant CAD and this was found in our study.
Hirata et al. [22] tested the hypothesis that echocardiographic adipose thickness especially adipose thickness in anterior interventricular groove, where the left descending coronary artery runs, can be a marker for the presence and severity of CAD. There was agreement between this study and affected coronaries in our study as there was significant relationship between EFV and proximal LAD and D1 lesions (p = 0.020). Rosito et al. [23] in a substudy of the Framingham Heart Study found that epicardial fat was correlated with multiple measures of adiposity and cardiovascular disease risk factors; they also found that epicardial fat was associated with coronary artery calcification.
In this study, Logistic regression analysis for the effect of different parameters on obstructive coronary artery disease (≥ 50% stenosis) was performed and revealed that age, sex, dyslipidemia, hypertension, epicardial fat volume at cutoff value > 124cm3 & calcium score ≥ 101 were univariate significant risk factors/ predictors for obstructive coronary artery disease. Our results were supported by study of Samy et al. [24] as they found a significant relation between the thickness of both EF and pericoronary fat as well as the Ca score and the severity of CAD, with higher values in group 3 with obstructive CAD compared with groups 1 and 2. Several studies showed results consistent with our findings. Some of these studies used also EF thickness; Demircelik et al. [25] evaluated the relationship between epicardial adipose tissue and pericoronary fat thickness (PCFT) measured with 64-MDCT. They concluded that epicardial adipose tissue and PCFT scores were higher in patients with obstructive CADs. Other studies used pericardial fat volume.
Iwasaki et al. [26] studied 197 patients with suspected CAD who underwent 64-MDCT and coronary angiography. Cross-sectional tomographic cardiac slices (3.0 mm thick) from base to apex (30–40 slices per heart) were traced semiautomatically, and epicardial fat volume (EFV) was measured by assigning Hounsfield units ranging from − 30 to − 250. EFV was associated with coronary atherosclerosis, and EFV increased steeply in patients with significant coronary artery stenosis and in those with severe coronary artery calcification. Quantitation of EF may be useful, in addition to coronary artery Ca score and coronary angiography, to identify patients at risk for CAD.
The main limitation in our study was the time consuming procedure used in volumetric assessment of EAT by MDCT. Also, the assessed amount of EAT may be of less benefit in the pathophysiology of CAD than functional characteristics of fat surrounding the coronary arteries. Furthermore, there were no available follow-up data to evaluate the outcome in these patients to reach the prognostic value of EFV.
Conclusion
Epicardial fat volume is an independent risk factor for obstructive coronary artery disease suggesting the necessity for its integration in coronary artery disease risk stratification.
We recommend widespread developing automatic software to facilitate EAT quantification for being a clinically useful parameter in patients undergoing MSCT for coronary angiography.
Availability of data and materials
The author's confirm that all data supporting the finding of the study are available within the article, and the raw data and data supporting the findings were generated and available at the corresponding author on request.
Abbreviations
- BMI:
-
Body mass index
- CABG:
-
Coronary artery bypass grafting
- CAC:
-
Coronary artery calcium
- CAD:
-
Coronary artery disease
- cMPR:
-
Curved multiplanar reformations
- EAT:
-
Epicardial adipose tissue
- EF:
-
Epicardial fat
- EFV:
-
Epicardial fat volume
- FFAs:
-
Free fatty acids
- HU:
-
Hounsfield unit
- LAD:
-
Left anterior descending
- LCx:
-
Left circumflex
- LCA:
-
Left coronary artery
- LM:
-
Left main
- MDCT:
-
Multi-detector computed tomography
- NRS:
-
Napkin ring sign
- PCI:
-
Percutaneous coronary intervention
- PDA:
-
Posterior descending artery
- PCFT:
-
Pericoronary fat thickness
- PLB:
-
Postero-lateral branch
- RCA:
-
Right coronary artery
- ROI:
-
Region of interest
References
Bertaso AG, Bertol D, Duncan BB et al (2013) Epicardial fat: definition, measurements and systematic review of main outcomes. Arq Bras Cardiol 101(1):e18-28
Mazurek T, Kiliszek M, Kobylecka M et al (2014) Relation of proinflammatory activity of epicardial adipose tissue to the occurrence of atrial fibrillation. Am J Cardiol 113(9):1505–1508
Ridker PM, Everett BM, Thuren T et al (2017) Antiinflammatory therapy with canakinumab for atherosclerotic disease. N Engl J Med 377(12):1119–1131
McKenney ML, Schultz KA, Boyd JH et al (2014) Epicardial adipose excision slows the progression of porcine coronary atherosclerosis. J Cardiothorac Surg 9(1):1–11
Raggi P, Zona S, Scaglioni R et al (2015) Epicardial adipose tissue and coronary artery calcium predict incident myocardial infarction and death in HIV-infected patients. J Cardiovasc Comput Tomogr 9(6):553–558
You S, Sun JS, Park SY et al (2016) Relationship between indexed epicardial fat volume and coronary plaque volume assessed by cardiac multidetector CT. Medicine 95(27):e4164
Iwasaki K, Urabe N, Kitagawa A et al (2018) The association of epicardial fat volume with coronary characteristics and clinical outcome. Int J Cardiovasc Imaging 34(2):301–309
Hodaş R, Pop S, Opincariu D et al (2016) Correlations between severity of coronary lesions and epicardial fat volume in patients with coronary artery disease–a multislice CT-based study. J Interdiscip Med 1(1):71–78
Kitagawa T, Yamamoto H, Sentani K et al (2015) The relationship between inflammation and neoangiogenesis of epicardial adipose tissue and coronary atherosclerosis based on computed tomography analysis. Atherosclerosis 243(1):293–299
Bettencourt N, Toschke AM, Leite D et al (2012) Epicardial adipose tissue is an independent predictor of coronary atherosclerotic burden. Int J Cardiol 158(1):26–32
Okada K, Ohshima S, Isobe S et al (2014) Epicardial fat volume correlates with severity of coronary artery disease in nonobese patients. J Cardiovasc Med 15(5):384–390
Khurana R, Yadav A, Buxi TB et al (2018) Correlation of epicardial fat quantification with severity of coronary artery disease: a study in Indian population. Indian Heart J 70:S140–S145
Shehata SM, Zaiton FM, Warda MA (2018) Role of MDCT in evaluation of epicardial fat volume as an independent risk factor for coronary atherosclerosis. Egypt J Radiol Nuclear Med 49(2):329–337
Djaberi R, Schuijf JD, van Werkhoven JM et al (2008) Relation of epicardial adipose tissue to coronary atherosclerosis. Am J Cardiol 102(12):1602–1607
Iacobellis G, Willens HJ (2009) Echocardiographic epicardial fat: a review of research and clinical applications. J Am Soc Echocardiogr 22(12):1311–1319
Verhagen SN, Visseren FL (2011) Perivascular adipose tissue as a cause of atherosclerosis. Atherosclerosis 214(1):3–10
Kamal D, Abd ElMoteleb AM, Samir R et al (2018) Epicardial fat thickness can predict severity and multivessel distribution in Egyptian patients with atherosclerotic coronary artery stenosis. Egypt Heart J 70(4):323–327
Faghihi S, Vasheghani-Farahani A, Parsaee M et al (2015) Association between epicardial fat thickness and premature coronary artery disease: a case control study. Res Cardiovasc Med 4(2):e25679
Eroglu S, Sade LE, Yildirir A et al (2009) Epicardial adipose tissue thickness by echocardiography is a marker for the presence and severity of coronary artery disease. Nutr Metab Cardiovasc Dis 19(3):211–217
Alexopoulos N, McLean DS, Janik M et al (2010) Epicardial adipose tissue and coronary artery plaque characteristics. Atherosclerosis 210(1):150–154
Bastarrika G, Broncano J, Schoepf UJ et al (2020) Relationship between coronary artery disease and epicardial adipose tissue quantification at cardiac CT: comparison between automatic volumetric measurement and manual bidimensional estimation. Acad Radiol 17(6):727–734
Hirata Y, Kurobe H, Akaike M et al (2011) Enhanced inflammation in epicardial fat in patients with coronary artery disease. Int Heart J 52(3):139–142
Rosito GA, Massaro JM, Hoffmann U et al (2008) Pericardial fat, visceral abdominal fat, cardiovascular disease risk factors, and vascular calcification in a community-based sample: the Framingham Heart Study. Circulation 117(5):605–613
Samy NI, Abdalzez WF, Alnaggar MF (2020) Epicardial fat measured by multidetector computed tomography and coronary artery disease. Menoufia Med J 33(1):303
Demircelik MB, Yilmaz OC, Gurel OM et al (2014) Epicardial adipose tissue and pericoronary fat thickness measured with 64-multidetector computed tomography: potential predictors of the severity of coronary artery disease. Clinics 69(6):388–392
Iwasaki K, Matsumoto T, Aono H et al (2011) Relationship between epicardial fat measured by 64-multidetector computed tomography and coronary artery disease. Clin Cardiol 34(3):166–171
Acknowledgements
To all the participants for their cooperation and patience.
Funding
No funding. Not applicable for this section.
Author information
Authors and Affiliations
Contributions
KA suggested the research idea; RA ensured the original figures and data in the work, minimized the obstacles to the team of work, correlated the study concept and design and had the major role in analysis; DA collected data in all stages of manuscript, performed data analysis. HA supervised the study with significant contribution to design the methodology, manuscript revision and preparation. MF correlated the clinical data of patient and matched it with the findings, drafted and revised the work. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Informed written consents were taken from the patients, and the study was approved by ethical committee of Tanta university hospital, faculty of medicine. Committee’s reference number: 32542/9/18.
Consent for publication
All participants included in the research gave written informed consent to publish the data included within this study.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Taha, D.A., El Shafey, R.A.A., Hamesa, M.F. et al. Relationship between epicardial fat volume measured by multi-detector computed tomography and coronary artery disease. Egypt J Radiol Nucl Med 52, 235 (2021). https://doi.org/10.1186/s43055-021-00615-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s43055-021-00615-w