Abstract
Background
Mozambique tilapia (Tilapia mossambica) are commonly used in the assessment of aquatic environment quality and also considered as useful bio-indicators during environmental pollution monitoring. Copper-oxide and dibutyltin are used as antifouling underwater hall paints.
Copper nanoparticles (Cu-NPs) are serious water pollutants but their impact in fish’s performance remains poorly understood.
Results
In the present study, we have exposed tilapia, a freshwater edible fish to two different doses (15 mg/L) of Cu-NPs and (2 mg/L) dibutyltin for 6 days. The doses selected were eco-relevant considering the contamination levels of certain water resources. The results indicated that the activity oxidative stress enzymes reduced glutathione (GSH), acetylcholinesterase (AChE), and glutathione-S-transferase (GST) were significantly decreased in the liver, brain, and gills of the treated groups when compared to control.
Conclusion
Taken together, the results suggest that short-term exposure of tilapia fish to Cu-NPs and dibutyltin causes oxidative stress and impart serious deleterious effects in the tissues which may affect fish growth and development and causes death. In the case of the gills, liver, and brain when exposed to both concentrations of CuO and dibutyltin (NPs), although the activity of these enzymes showed an inhibition in the liver when exposed to both concentrations of NPs. The present study investigated that CuO-NPs are more toxic than dibutyltin.
Similar content being viewed by others
Background
Antifouling paints containing tributyltin (TBT) began in the late 1980s. Although it still may be available in some parts of the world, antifouling paints containing TBT were ultimately banned in 2008. Copper began to become a concern in California in the 1990s. Copper has been used in antifouling paints for centuries because it is effective, available, and relatively inexpensive compared to other biocides. It is also easy to work with and can be used in many different formulations of antifouling paint. The most common forms of copper used today are cuprous oxide, copper thiocyanate, and copper flake.
In antifouling paints, copper compounds are used to control hard fouling, such as barnacles, mussels, and tube worms. Copper can also be effective against slime fouling, although it needs a much higher release rate to control slime than to control shell fouling.
The excessive contamination of aquatic ecosystems has evoked major environmental and health concerns worldwide (McNeil and Fredberg, 2011). The pollutants could increase the level of metals in natural water and seriously affect both fresh and marine habitats (El Nemr, 2012; El Nemr, Khaled, Moneer, and El Sikaily, 2012; Muhammad, Shah, and Khan, 2011; Yu et al. 2011)
Copper (Cu) in low amounts is considered as an essential micronutrient to all living organisms because it acts as a cofactor for various enzymes responsible for performing an essential metabolic activity. However, excessive concentrations of copper in aquatic ecosystems can exert adverse toxicological effects on freshwater organisms such as fish. In the last decade, several studies reported that waterborne exposure to soluble Cu can induce endocrine disruption and affect metabolic rates, oxidation stress, cell apoptosis, immune responses, swimming behavior, histopathology, growth parameters, digestive enzymes, and body composition. It has been suggested that the bioavailability of metal NPs by aquatic pollution and subsequent accumulation in fish constitute a substantial risk to human health and to the environment (Shaw and Handy, 2011). In this respect, it is important to study the effect of Cu-NPs using an edible fish as the animal model.
There is a growing concern that these products and their byproducts may discharge hazardous biochemical particles into aquatic habitats which in turn can affect their biota (Ghorade et al, 2014).
Dibutyltin (DBT) is closely related to our daily life and are widely used as polyvinyl chloride (PVC) plastic stabilizers, pesticides, marine antifouling coatings, and catalysts in rubber, polyurethane (PU), and the biodiesel industry (Brito et al. 2012; Harford, O’Halloran, and Wright, 2005; Xu, Zhou, and Zhao, 1990). Unfortunately, lots of studies have shown that DBTs are able to accumulate in and even contaminate the aquatic environment as well as cultivated land and remain unchanged for years.
Tilapia is the most common freshwater fish that is frequently used in toxicological studies as a biological indicator because of its high growth rate, significant tolerance to environmental stress, ease of reproduction, and high market demand (El-Sayed, 2006), and used to evaluate the health of aquatic ecosystems, because pollutants are building up in the food chain (Farkas, Farkas, and Hyde, 2004). Fish are sensitive to many variables in their environment; hence, they play a significant role in the assessment of water quality (AlGhais, 2013). So, the determination of metal levels in fish is tremendously important for the health of human beings (Uysal, Emre, and Ko¨se, 2008).
Therefore, our study purposed to further investigate the effects of copper-oxide nanoparticles (CuO-NPs) and dibutyltin on tilapia fish to compare CuO-NPs and dibutyltin bioaccumulation in the gills, liver, and brain. The present study aims to provide a comparative study between CuO and dibutyltin (NPs) at a concentration to declare their deleterious effects on oxidative biomarkers of fresh water tilapia and the measurement of oxidative stress.
Methods
Experimental fish maintenance and treatment
Experimental fish in the present study were tilapia. They were taken from an unpolluted fish farm of EPDA located in Ras Al Khaimah, UAE. The initial body length and weight of fish were 10–11.5 cm and 14.4–25.8 g, respectively. All tilapia were transported in plastic containers with continuous aeration to the lab. All fish (four fish/aquarium) were maintained for 2 weeks in glass aquaria with 50 L aerated, dechlorinated tap water. Water temperature was maintained at 25 °C, while salinity and pH were 1.12–1.002 and 8.7–7.6, respectively. Photoperiod was 12 h light /12 h dark. During the acclimatization period, fish were fed once daily with commercial pellet food (20% crude protein, 4% crude fat, 5% crude fiber, 12% crude ash, and 10% crude moisture). Dead fish as well as any fish showing any unusual performances were excluded.
Treatment with CuO and dibutyltin
After the acclimatization period (1 week), each of the four fishes were transferred to small glass aquaria for lethal concentration determination. Nominal concentrations used for CuO was 15 mg/L and for dibutyltin was 2 mg/L. The exposure period was 96 h; with the same temperature, dissolved oxygen and pH as in the acclimatization period. The dead fish was recorded in each concentration. A control was handled identically but without exposure to CuO and dibutyltin particles. The conditions of the experiments were as those of the acclimatization period and water was constantly (every day) checked for pH, temperature, salinity, and dissolved oxygen. Fish were fed once every day.
Sample collection
At the end of the treatments, the weights of whole fish as well as of the brain, liver, and gill were measured. Tissues were dissected out and used for analysis. Samples were obtained from all fishes (control and treated) in replicates similar procedure was followed. Tissues were used for biochemical and protein analysis. Tissues from fishes were pooled to obtain biological samples, and a total sample was used for all experimentations.
Measurement of biomarkers
For evaluation of oxidative damage, the liver, brain, and gills were homogenized in cold buffer (pH 7.4) per gram tissue using a homogenizer. Then the homogenates were centrifuged at 4000 rpm for 15 min, and the supernatants were stored in the refrigerator until used. Oxidative stress was detected in the supernatant of the tissue homogenate (GST, GSH, AChE, protein).
GST assay
Jakoby, Habig, and Jakoby (1980) protocol was followed for measuring glutathione-S-transferase (GST) activity. GST catalyzes the conjugation of GSH to CDNB through the thiol group of the glutathione and making CDNB-GSH adduct and this CDNB-GSH adduct was used to measure GST activity.
In brief, 1% sample solutions (liver) and 5% sample solutions (gills) prepared from 10% solutions prepared for GSH assay determination, mixed with 0.1 M Tris HCl buffer (pH 7.4) using tissue homogenizer (Tissue homogenizer, REMI RQ-127A, India), solutions kept for incubation up to 6 h in refrigerator. Centrifuged (cold centrifuge, Sigma, USA) at 9000 cps at 4 °C and for 20 min. 0.1 ml of supernatant was used for determination of GST (glutathione-S-transferase) content. Add 0.3 ml of 0.1 M acetate buffer (pH 8.0) followed by addition of 2.25 ml of water, and 0.3 ml 0.02 M glutathione reduced (glutathione reduced, CAS no.:,1.01090.005, Merck, Germany) the absorbance was measured at 340 nm after the immediate addition of 0.05 ml 0.1 M CDNB (1-chloro 2,4 dinitrobenzene, CAS no.: 97-00-7, Acros Organics, India). Time reaction was monitored at 0 min,5 min,10 min, and 15 min.
GST catalyzes the conjugation of GSH to CDNB through the thiol group of the glutathione and making CDNB-GSH adduct (Fig. 1). The enzyme activity was calculated based on units/protein at different time reaction 0 min, 5 min, 10 min, and 15 min.
GSH
According to Beutler, Duron, and Kelly (1963), this reaction mechanism involves oxidation of glutathione reduced (GSH) (glutathione, CAS no.: 1.04090.005, Merck) by 0.01 Mole DTNB (5,5 dithio bis-2-dinitro benzoic acid, CAS no.: 422592J, VWR, UK) to form glutathione disulfide and yellow derivative of 5,thio 2-nitrobenzoic acid and its measured at 412 nm by spectrophotometer.
There is amount of GSH (glutathione, CAS no.: 1.04090.005, Merck) present in the solution (5%) is followed by oxidation with DTNB (5,5 dithio bis-2-dinitro benzoic acid, CAS no.: 422592 J,VWR, UK) produce Glutathione disulfide, and concentration depends on the amount of DTNB oxidized with glutathione (Fig. 2).
Ten percent sample solutions (liver, gills) were prepared by using tissue homogenizer (tissue homogenizer, REMI RQ-127A, India) mixed with 0.1 M Tris HCl buffer with EDTA (pH 7.4). Pipetted 0.75 ml of gills solution (10%) and 0.5 ml of liver solution (10%) made final volume of both sample up to 1.0 ml with water. 0.25 ml of 20% v/v trichloacetic acid was added and sample were kept for incubation in refrigerator about 45 min. 0.8 ml of water was added after incubation and was centrifuged at 2000 rpm for 20 min at room temperature. In the supernatant, 0.3 ml of 2 M tris base and 0.1 ml of 0.01 M DTNB (5,5 dithio bis 2-dinitrobenzoic acid, CAS no.: 422592J, VWR, UK) were added. Absorbance measured at 412 nm after 10 min by spectrophotometer (UV spectrophotometer, GENWAY 7315, UK).
AChE
There is a rate of production of thiocholine (acetylcholine iodide, CAS no.: 1866-15-5,VWR, UK), and this is measured by continuous reaction of hydrolysis of thiocholine with DTNB (0.01 M) (5,5 dithio bis-2-dinitro benzoic acid, CAS no.: 422592J, VWR, UK) to produce yellow color compound 5-thio-2-nitrobenzene ion. The rate of color production of the reaction is measured at 412 nm by spectrophotometry (Fig. 3).
1% sample solutions (liver, gills) was prepared from 10% solutions of GSH Assay determination, mixed with 0.1 M Tris HCl buffer (pH 7.4), and 1% brain sample solutions was prepared from 5% brain stock solution by using tissue homogenizer (tissue homogenizer, REMI RQ-127A, India) mixed with 0.1 M Tris HCl buffer with EDTA (pH 7.4), centrifuged (cold centrifuge, Sigma, USA) at 9000 cps at 4 °C and for 20 min. 0.1 ml of supernatant was used for determination of acetylcholine esterase (ACHe) content. 0.3 ml of 0.1 M Tris HCl buffer (pH 8.0) was added, followed by the addition of 2.4 ml of water, and the absorbance was measured at 412 nm after the immediate addition of 0.01 M DTNB (5,5 dithio bis 2-dinitro benzoic acid, CAS no.: 422592J, VWR, UK) time reaction was monitored at 0 min, 5 min,10 min, and 15 min.
Protein estimation
Protein was estimated by the method of Lowry, Rosebrough, Farr, and Randall, 1951. The liver, brain, and gill samples of fish muscle was taken out, washed with ice-cold normal saline, dried and weighed (Bhardwaj and Garg, 2014: Al Ghais and Bhardwaj, 2018).
Muscle homogenate (5% w/v) was prepared in ice-cold distilled water with the help of homogenizer. 0.2 ml of tissue homogenate was mixed with 1.3 ml of distilled water and 0.5 ml of 20% trichloroacetic acid to precipitates proteins. The tubes were allowed to stand at 4 °C for 30 min and centrifuge at 2500 rpm to sediments protein precipitates.
The sediments were dissolved in 0.1 N sodium hydroxide solution. A suitable aliquot of protein solution thus obtained was taken out in another tube and made up to 0.5 ml with reagent A. Then, 2.5 ml of reagent C was added and shaken. After 10 min, 0.25 ml of Folin and Ciocalteus Phenol reagent was added. After 30 min, the blue-colored solution was appeared and measured at 660 nm by a spectrophotometer.
Statistical analysis
Data are expressed as mean. Pairwise comparisons were performed. Experimental error was determined for triplicate assays and expressed as standard deviation (SD).
Results
Effect of Cu-NPs and dibutyltin on fish and tissue weight
At the end of the experiment, the weight of the whole fish and tissues (liver, gills, brain) were noted for each group (Table 1) and changes in relation to control were found. However, the treated group showed a more pronounced effect and a significant increase of tissue (liver, gills, brain) and body weight were observed when compared to the control (Fig 4).
Effect of CuO-NPs and dibutyltin on enzymes (GSH, AChE, GST) and protein
The levels of various enzymes were analyzed in the liver, brain, and gills of control and treated groups and exposure to Cu-NPs was found to modify the enzyme performance more.
GSH
For GSH, Cu-NPs effected more gills than in dibutyltin as compared to control. The level of toxicity of Cu was more in case of gills (Fig. 5). In the case of Cu-treated liver, the GSH reduced to half the original concentration as compared to control.
AChE
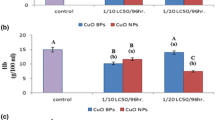
For AChE, the brain was the most affected in case of Cu-treated than dibutyltin compared to control but in case of the liver the Cu-treated showed 50% reduction as compared to control. In the case of gills, toxicity with dibutyltin was more than Cu-NPs as compared to control (Fig. 6).
GST assay
For GST, the toxicity with Cu-NPs was more in case of liver and gills than in dibutyltin as compared to control (Fig. 7).
Protein
The amount of protein was reduced to 99% in the case of gills treated with Cu-NPs. But in case of brain and liver, fish treated with Cu-NPs and dibutyltin showed 50% reduction (Fig. 8).
Discussion
Oxidative stress is a state of abundance of reactive oxygen species (ROS), which interferes with biological processes by disturbing or damaging homeostasis. In this study, the activity of oxidative stress enzyme AChE, GSH, and GST indicates the alteration of normal homeostasis. Cu-NPs are causative molecules for generating oxidative stress and responsible for cell death (Fahmy and Cormier, 2009). In addition, Cu-NPs disturb the normal mitochondrial homeostasis, which further causes oxidative stress to the cell (Federici, Shaw, and Handy, 2007). In fish, the gills are the first line of defense for any pollutant by secreting mucous (Handy and Maunder, 2009). Subsequently, gills respond through the generation of edema with the lifting of gill lamellar epithelium by binding to Naþ/Kþ-ATPase and inhibiting toxin entry (Stagg and Shuttleworth, 1982). Cu is a well-known inhibitor of gill respiration and ionoregulation (Grosell, Blanchard, Brix, and Gerdes, 2007; Handy, 2003).
The present study also analyzed the liver as a central compartment for Cu metabolism (Grosell, Boetius, Hansen, and Rosenkilde, 1996; Handy, Sims, Giles, Campbell, and Musonda, 1999; Kamunde, Grosell, Higgs, and Wood, 2002). Previous reports documented that fish exposed to Cu-NPs displayed blood accumulation and increase in sinusoid space, which is an indication of liver damage (Arellano, Storch, and Sarasquete, 1999; Shaw and Handy, 2011.
GSH plays an important role in non-enzymatic antioxidant system, since it acts as a reductant in conjugation with xenobiotics (Kanak, Dogan, Eroglu, Atli, and Canli, 2014). In the present study, GSH contents in the liver and gill tissues of tilapia exposed to CuO-NPs showed a significant decrease, except those exposed to dibutyltin in the case of gill tissues, when compared with the control groups after 4 days. These results are similar to Xiong, Fang, Yu, Sima, and Zhu (2011), who stated that ZnO NPs and a bulk ZnO suspension caused a decrease in GSH content in the liver tissue of zebrafish compared to controls.
Results indicated that, CuO (NPs) have a more toxic effect than dibutyltin in the liver and gill tissues in most oxidative stress parameters. Therefore, CuO potential toxicity should not be ignored (Blinova, Ivask, Heinlaan, Mortimer, and Kahru, 2010; Buffet et al. 2011; Saison et al. 2010). Also, the results indicate the difference in effects between the selected concentrations of CuO-NPs and dibutyltin and these may be due to the aggregation of particles in water.
Conclusion
Exposure of tilapia fish to CuO-NPs and dibutyltin increased the activity of oxidative stress enzymes that might lead to the excessive production of free radicals and disturbance of internal homeostasis indicating that this compound has a profound adverse effect on fish health. Based on our analysis, we suggest that short-term exposure of Cu-NPs and dibutyltin even at a low dose can cause oxidative stress, and this may lead to developmental disarray in the tilapia. Cu-NPs could cause more toxic effects than dibutyltin as antifouling agent. Also, this study is helpful to understand and make comparisons between Cu-NPs and dibutyltin toxicity to aquatic organisms but those in the laboratory, so further studies are required to assess the current environmental burden of NPs in aquatic ecosystems to determine, monitor, and/or regulate the use and release of Cu-NPs.
Change history
08 July 2019
Following publication of the original article (Ghais et al., 2019), it was noticed that Figures 2 and 3 were incomplete.
Abbreviations
- AChE:
-
Acetylcholinesterase
- CDNB:
-
1-Chloro 2,4 dinitrobenzene
- CF1:
-
Control fish
- Cu:
-
Copper
- Cu-NPs:
-
Copper nanoparticles
- DBT:
-
Dibutyltin
- GSH:
-
Reduced glutathione
- GST:
-
Glutathione-S-transferase
- NPs:
-
Nanoparticles
- TBF1:
-
Treated dibutyltin fish
- TBT:
-
Tributyltin
- TCF1:
-
Treated copper fish
References
Al Ghais, S., & Bhardwaj, V. (2018). Nannochloropsis as potential fish feed. International Journal of Science and Research, 7(12), 278–282 ISSN: 2319-7064.
AlGhais, M. S. (2013). Acetylcholinesterase, glutathione and hepatosomatic index as potential biomarkers of sewage pollution and depuration in fish. Marine Pollution Bulletin, 74, 183–186.
Arellano, J. M., Storch, V., & Sarasquete, C. (1999). Histological changes and copper accumulation in liver and gills of the senegales sole, Solea senegalensis. Ecotoxicology and Environmental Safety, 44, 62–72.
Beutler, E., Duron, O., & Kelly, M. B. (1963). Improved method for the determination of blood glutathione. The Journal of Laboratory and Clinical Medicine, 61, 882–888.
Bhardwaj, V., & Garg, N. (2014). Pectinase production by Delftia acidovorans isolated from fruit waste under submerged fermentation. International Journal of Science and Research, 3, 261–265.
Blinova, I., Ivask, A., Heinlaan, M., Mortimer, M., & Kahru, A. (2010). Ecotoxicity of nanoparticles of CuO and ZnO in natural water. Environmental Pollution, 158, 41–47.
Brito, Y. C., Ferreira, D. A. C., Fragoso, D. M. D. A., Mendes, P. R., de Oliveira, C. M. J., Meneghetti, M. R., & Meneghetti, S. M. P. (2012). Simultaneous conversion of triacylglycerides and fatty acids into fatty acid methyl esters using organometallic tin (IV) compounds as catalysts. Applied Catalysis A: General, 443–444, 202–206.
Buffet, P. E., Tankoua, O. F., Jin-Fen Pan, J. F., Berhanu, D., Herrenknecht, C., Poirier, L., … Mouneyrac, C. (2011). Behavioural and biochemical responses of two marine invertebrates Scrobicularia plana and Hediste diversicolor to copper oxide nanoparticles. Chemosphere, 84, 166–174.
El Nemr, A. (2012). Environmental Pollution and its Relation to Climate Change, (p. 694). Hauppauge, New York: Nova Science Publishers Inc. 13: 978-1-61761-794-2.
El Nemr, A., Khaled, A., Moneer, A. A., & El Sikaily, A. (2012). Risk probability due to heavy metals in bivalve from Egyptian Mediterranean coast. Egyptian Journal of Aquatic Research, 28, 67–75.
El-Sayed, A. F. M. (2006). Tilapia Culture. Oxfordshire: CABI publishing, CABI International Wallingford.
Fahmy, B., & Cormier, S. A. (2009). Copper oxide nanoparticles induce oxidative stress and cytotoxicity in airway epithelial cells. Toxicology In Vitro, 23, 1365–1371.
Farkas, J., Farkas, P., & Hyde, D. (2004). Liver and gastroenterology tests. In M. Lee 3rd (Ed.), Basic Skills in Interpreting Laboratory Data, (pp. 330–336). Bethesda: American Society of Health-System Pharmacists.
Federici, G., Shaw, B. J., & Handy, R. D. (2007). Toxicity of titanium dioxide nanoparticles to rainbow trout (Oncorhynchus mykiss): Gill injury, oxidative stress, and other physiological effects. Aquatic Toxicology, 84, 415–430.
Ghorade, I. B., Lamture, S. V., & Patil, S. S. (2014). Assessment of heavy metal content in Godavari river water. IMPACT: International Journal of Research in Applied, Natural and Social Sciences, 2, 23–26.
Grosell, M., Blanchard, J., Brix, K. V., & Gerdes, R. (2007). Physiology is pivotal for interactions between salinity and acute copper toxicity to fish and invertebrates. Aquatic Toxicology, 84, 162–172.
Grosell, M., Boetius, I., Hansen, H. J. M., & Rosenkilde, P. (1996). Influence of preexposure to sublethal levels of copper on 64Cu uptake and distribution among tissues of the european eel (Anguilla anguilla). Comparative Biochemistry and Physiology: Comparative Pharmacology and Toxicology, 114, 229–235.
Handy, R. D. (2003). Chronic effects of copper exposure versus endocrine toxicity: Two sides of the same toxicological process? Comparative Biochemistry and Physiology: Molecular & Integrative Physiology, 135, 25–38.
Handy, R. D., & Maunder, R. J. (2009). Osmoregulation and ion transport: Integrating physiological, molecular and environmental aspects. London: Essential Reviews in Experimental Biology, Society for Experimental Biology Press.
Handy, R. D., Sims, D. W., Giles, A., Campbell, H. A., & Musonda, M. M. (1999). Metabolic trade-off between locomotion and detoxification for maintenance of blood chemistry and growth parameters by rainbow trout (Oncorhynchus mykiss) during chronic dietary exposure to copper. Aquatic Toxicology, 47, 23–41.
Harford, A. J., O’Halloran, K., & Wright, P. F. (2005). The effects of in vitro pesticide exposures on the phagocytic function of four native Australian freshwater fish. Aquatic Toxicology, 75, 330–342.
Jakoby, W. B., Habig, W. H., & Jakoby, W. B. (Eds.) (1980). Enzymatic basis of detoxication pp. 63–94. New York: Academic Press.
Kamunde, C., Grosell, M., Higgs, D., & Wood, C. M. (2002). Copper metabolism in actively growing rainbow trout (Oncorhynchus mykiss): Interactions between dietary and waterborne copper uptake. Journal of Experimental Biology, 205, 279–290.
Kanak, E. G., Dogan, Z., Eroglu, A., Atli, G., & Canli, M. (2014). Effects of fish size on the response of antioxidant systems of Oreochromis niloticus following metal exposures. Fish Physiology and Biochemistry, 40, 1083–1091.
Lowry, O. H., Rosebrough, N. J., Farr, A. L., & Randall, R. J. (1951). Protein measurement with the Folin phenol reagent J. Biol. Chem, 193, 265–275.
McNeil, D. G., & Fredberg, J. (2011). Environmental water requirements of native fishes in the Middle River catchment, Kangaroo Island, South Australia, A Report to the SA Department for water (p. 50). Adelaide. SARDI Publication No. f2011/000060-1. SARDI Research Report Series No. 528: South Australian Research and Development Institute (Aquatic sciences).
Muhammad, S., Shah, M. T., & Khan, S. (2011). Health risk assessment of heavy metals and their source apportionment in drinking water of Kohistan region, northern Pakistan. Microchemical Journal, 98, 334–343.
Saison, C., Perreault, F., Daigle, J. C., Fortin, C., Claverie, J., Morin, M., & Popovic, R. (2010). Effect of core-shell copper oxide nanoparticles on cell culture morphology and photosynthesis (photosystem II energy distribution) in the green alga, Chlamydomonas reinhardtii. Aquatic Toxicology, 96, 109–114.
Shaw, B. J., & Handy, R. D. (2011). Physiological effects of nanoparticles on fish: A comparison of nanometals versus metal ions. Environment International, 37, 1083–1097.
Stagg, R. M., & Shuttleworth, T. J. (1982). The effects of copper on ionic regulation by the gills of the seawater-adapted flounder (Platichthys flesus L.). Journal of Comparative Physiology, 149, 83–90.
Uysal, K., Emre, Y., & Ko¨se, E. (2008). The determination of heavy metal accumulation ratios in muscle, skin and gills of some migratory fish species by inductively coupled plasma-optical emission spectrometry (Icp-Oes) in Beymelek Lagoon (Antalya/Turkey). Microchemical Journal, 90, 67–70.
Xiong, D., Fang, T., Yu, L., Sima, X., & Zhu, W. (2011). Effects of nano-scale TiO2, ZnO and their bulk counterparts on zebra fish: Acute toxicity, oxidative stress and oxidative damage. The Science of the Total Environment, 409, 1444–1452.
Xu, R., Zhou, D., & Zhao, D. (1990). Effect of organotin stabiliser on the thermal stabilisation of PVC: Part I—The influence of dibutyltin dilaurate on polyene sequences. Polymer Degradation and Stability, 27, 203–210.
Yu, G. B., Liu, Y., Yu, S., Wu, S. C., Leung, A. O. W., Luo, X. S., … Wong, M. H. (2011). Inconsistency and comprehensiveness of risk assessments for heavy metals in urban surface sediments. Chemosphere, 85, 1080–1087.
Acknowledgements
There is no acknowledgment.
Funding
Not applicable.
Availability of data and materials
The relevant data and materials are available in the present study.
Author information
Authors and Affiliations
Contributions
I would like to thank the fishery department of EPDA. SAG supervised the entire project. VB and PK performed all the experiments. The supervision of the laboratory work was performed by VB. PK and OAS helped in fish dissection. VB analyzed the data and wrote the manuscript. I would also like to thank Mr. Jamsheed in kindly providing fishes for the experiment. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests. All procedures followed were in accordance with the ethical standards (institutional and national). All institutional and national guidelines for the care and use of laboratory animals were followed.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Al Ghais, S., Bhardwaj, V., Kumbhar, P. et al. Effect of copper nanoparticles and organometallic compounds (dibutyltin) on tilapia fish. JoBAZ 80, 32 (2019). https://doi.org/10.1186/s41936-019-0101-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s41936-019-0101-7