Abstract
Background
Obstructive Sleep Apnea (OSA) has been under-investigated in rural communities, particularly through a sex/gender lens. The purpose of this study was to examine the prevalence and correlates of OSA risk among rural-dwelling women and men in Saskatchewan, Canada.
Methods
Participants for this cross-sectional study were 2340 women and 2030 men living in rural Saskatchewan, Canada and were without a prior diagnosis of OSA. The dependent variable, OSA risk, was estimated from self-reported symptoms of OSA and percentage body fat. Independent variables included socio-demographic characteristics, health behaviors, comorbidities, and quality of life indicators. Multivariable logistic regression was the primary statistical technique employed, conducted separately for women and men.
Results
A greater proportion of men (30.1%) than women (19.4%) were at high risk of OSA. While many of the correlates of OSA risk were similar for women and men, sex differences emerged for marital status, educational attainment, financial strain, depression, asthma, and perceptions of community support.
Conclusion
A sizable minority of rural women and men may have possible undiagnosed OSA, which in turn, is associated with substantial comorbidity and reduced quality of life. Additional research with enhanced measurement and a longitudinal design is required to test the veracity of these findings and further clarify the role of sex/gender in relation to OSA risk in rural adults.
Similar content being viewed by others
Background
Obstructive Sleep Apnea (OSA) is characterized by loud snoring, excessive daytime sleepiness, and episodes of breathing cessation during sleep, causing the individuals to wake up choking and gasping (Laratta et al. 2017; Javaheri et al. 2017). Associated with numerous adverse physical, psychosocial, and economic consequences, OSA is a serious public health concern (Borsoi et al. 2022). While prevalence estimates of OSA vary considerably, depending on the population sampled and how OSA is defined and measured (Senaratna et al. 2017), several fairly consistent patterns have emerged, in that OSA: 1) may be increasing over time (Peppard et al. 2013), including in Canada (Pendharkar et al. 2023; Statistics Canada: Health Fact Sheet 2018); 2) is associated with older age (Senaratna et al. 2017) and obesity (Senaratna et al. 2017); and 3) at all ages, is identified more often in men than women (Bonsignore et al. 2019). Many people may have OSA but are undiagnosed (Fuhrman et al. 2012; Heinzer et al. 2015) and untreated OSA may increase risk of morbidity and mortality (Knauert et al. 2015; Fu et al. 2017). Two population subgroups that may be more prone to OSA under-recognition are women (Lindberg et al. 2017; Orbea et al. 2020) and rural dwellers (Spagnuolo et al. 2019).
The interaction of factors related to both sex (e.g., anatomical and physiological features of the upper airways) and gender (e.g., misattribution of OSA symptoms in women as depression) have been proposed to explain differences in the prevalence of OSA in women and men (Bonsignore et al. 2019; Bublitz et al. 2022). Common screening instruments may also be less sensitive for detecting OSA in women (Lindberg et al. 2017; Mou et al. 2019). In addition, OSA-related risk factors, comorbidities, and quality of life may differ for women and men (Thompson et al. 2022; Huang et al. 2018); however, sex-specific findings are rarely reported in the literature.
Rural dwellers are another segment of the population in which OSA may be more likely to go unrecognized, due, in part, to more limited access to primary and specialist health care (Spagnuolo et al. 2019). Rural adults may also be at greater risk of developing OSA than their urban counterparts, given older age and higher rates of obesity (Shields and Tjepkema 2006). To our knowledge, research investigating OSA risk and associated factors through a sex/gender lens has not been conducted in rural residents.
The objectives of this cross-sectional study of rural dwelling women and men in Saskatchewan, Canada were to determine: 1) the prevalence of high risk for OSA; and 2) the association of high risk OSA status with socio-demographic characteristics, health behaviors, comorbidities, and quality of life.
Methods
Design and sample
This research was set in Saskatchewan, a western Canadian province with a population of approximately 1 million people. The present study involved secondary analysis of cross-sectional data from the Saskatchewan Rural Health Study (SRHS) (Pahwa et al. 2012; Pahwa et al. 2017). The purpose of the SRHS was to enhance understanding of rural dwellers' respiratory health and determinants. The rural population for the SRHS was defined as ‘those living in towns and municipalities outside the commuting zone of larger urban centers with a population of 10,000 or more’ (Plessis et al. 2004).
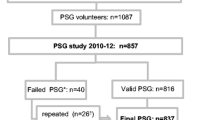
The SRHS was conducted in several phases, with detailed methods of phase 1 (2010) (Pahwa et al. 2012) and phase 2 (2014) (Pahwa et al. 2017) published previously. The study base consisted of tax-paying households located in rural municipalities (RMs) and small towns situated in one of four geographical quadrants (south-east, south-west, north-east and north-west) in the southern half of Saskatchewan. The local councils for the vast majority of these communities (32/36 RMs and 15/16 towns) provided mailing addresses. Dillman’s mail data collection methodology was followed to recruit adult study participants (Dillman 2000). A key informant in each household was asked to provide household-level information and individual information for each adult living in the household. Of the 11,004 eligible addresses in the phase 1 survey, responses were obtained from 4624 households, representing 8261 individuals. Various knowledge translations activities were employed to maintain engagement with participants for the next study phase.
Phase 2 of the SRHS provides the data for the present study, as additional screening questions for OSA were introduced into the survey. Questionnaires were mailed to all individuals who participated in the 2010 baseline survey. Initial mailing was done for 4454 households due to 170 households being lost to follow-up after baseline knowledge translation mailings were returned to sender. Questionnaires were returned from 2797 households, consisting of 4866 individuals. Participants were excluded (n = 415; 70% male) if they answered yes to the question “has a doctor ever said you had sleep apnea”, resulting in a sample size of 2340 women and 2030 men.
Measures
Dependent variable
OSA risk was estimated using a combination of self-reported symptoms of OSA and percentage body fat (%BF) (Thompson et al. 2022). The STOP questionnaire (Chung et al. 2008) was used to measure OSA symptoms and consisted of four questions measuring: (S) snoring (“Do you snore loudly? By ‘loudly’ I mean louder than talking or loud enough to be heard through closed doors”), (T) tiredness (“Do you often feel tired, fatigued, or sleepy during daytime?”), (O) observed apnea (“Has anyone ever observed you stop breathing in your sleep?”), and (P) high blood pressure (“Has a doctor ever told you that you have high blood pressure or hypertension?”). A score of one was applied each time participants answered “yes” to a question; scores for the STOP questionnaire ranged from 0–4 (Chung et al. 2008). Evidence of the predictive ability of the STOP tool has been reported with both clinical and general population samples (Chung et al. 2008; Patel et al. 2022).
The STOP questionnaire can be combined with information on BMI (B), age (A), neck circumference (N), and sex/gender (G) to obtain a STOP-BANG score (Chung et al. 2008). The BANG component was not calculated for this study for several reasons. First, there was a large amount of missing data for neck circumference, especially among women, an issue that has been noted in prior research (Orbea et al. 2020). Second, BMI is dependent on age and sex (Gallagher et al. 1996); that is, even though women have more body fat than men (especially during certain life stages, such as menopause), as do older people compared to younger, the same BMI classification is applied. This awareness, combined with knowledge of our study population’s older age demographic (Pahwa et al. 2012; Pahwa et al. 2017) and the methodological approach adopted in a recent Canadian study of OSA prevalence and correlates (Thompson et al. 2022), resulted in our decision to use %BF to measure obesity. We estimated %BF using a validated prediction equation (Gallagher et al. 2000; Marin-Jimenez et al. 2022) which takes into account sex (coded: 0 female; 1 male) and age: 64.5 – 848 X (1/BMI) + 0.079 X age – 16.4 X sex + 0.05 X sex X age + 39.0 X sex X (1/BMI). Following recommended sex-specific guidelines for classification of obesity (WHO 1995), women with a %BF higher than 35 and men higher than 25 were considered obese and given a score of one.
STOP questionnaire and %BF scores were summed to obtain a STOP-Obesity score, with values ranging from 0 to 5; following the methods of a recent Canada-wide population based study of OSA risk using the STOP-Obesity tool (Thompson et al. 2022), participants with a score of three or greater were considered as being at high risk of OSA, and less than three, at low risk.
Independent variables
The independent variables were socio-demographic characteristics, health behaviors, comorbidities, and quality of life indicators. Socio-demographics included age (< 50yrs, 50–59, 60–69, 70–79, and 80 +), marital status (married, common law, widowed, separated/divorced, single/never married), home location (farm/town), and educational attainment (high school or less, post-secondary). In addition, income adequacy was a derived variable combining information on household income and number of residents with responses categorised into one of four groups: low income, medium income, high income, and missing. Current employment status was a categorical variable with four options for women (employed, retired, homemaker, and ‘other’) and three options for men (employed, retired, and ‘other’). The fewer number of categories for men were due to the small number indicating homemaker status (n = 2); this choice, therefore, was subsumed for men under 'other'. Financial strain was a categorical variable assessed on the basis of participants’ response to the question ‘At the end of the month, do you have some money left over: a) some money, b) just enough money, or c) not enough money’; responses were kept in their original form but with a ‘missing’ option incorporated.
For health behaviors, exercise was assessed with the question “Do you exercise?" (yes/no) and binge drinking was assessed with the question "How often in the past 12 months have you had five or more alcoholic drinks on one occasion?" (never, less than once/month, once/month, 2–3 times/month, once/week, more than once/week). Based on recommended operationalization of binge drinking (Bulloch et al. 2016), participants were categorized as having engaged in binge drinking if they indicated consuming five or more alcoholic drinks at least once a month. For women, it is recommended that the threshold be reduced to four drinks (Bulloch et al. 2016); therefore, it is likely that the prevalence of binge drinking in women was underestimated in this study. Three smoking statuses were also measured: current smoker (smoking in the past year), ex-smoker (no current smoking and a lifetime history of smoking at least 20 packs), or nonsmoker (all other categories).
Comorbidities were based on participants’ response to the question of whether they had ever been diagnosed (yes/no) with each of the following diseases: diabetes, high cholesterol, chronic obstructive pulmonary disease (COPD), asthma, and depression. In addition, injury was assessed by the question "During the past 12 months, were you seen by a doctor or other primary care giver for an injury?" (yes/no).
Quality of life was assessed with five indicators: self-rated physical health, self-rated mental health, change in health, life stress, and community support. Self-rated physical (mental) health was assessed with the question(s): "In general, would you say your physical (mental) health is excellent, very good, good, fair, poor"; responses were collapsed into two categories (excellent/very good/good and fair/poor). Change in health was assessed by the question "Compared to one year ago, how would you say your health is now" and grouped into three responses: much/somewhat better, about the same, much/somewhat worse. Life stress was measured by the question "Thinking about the amount of stress in your life, would you say most days are: a) not at all stressful, b) not very stressful, c) a bit stressful, d) quite stressful, or e) very stressful?", with responses grouped into three options (not at all/not very stressful, a bit stressful, quite/very stressful. Community social support was measured by the question "How well do you feel like you are generally supported (socially, emotionally, medically, etc.) by your community?" (Janzen et al. 2020). Participants were asked to mark their response along a 50 mm line, anchored between ‘no support’ and ‘extremely supported.’ Participants’ markings were measured, converted into numerical values (out of 50), and then categorized into tertiles: ‘low support’, ‘medium support,’ and ‘high support’.
Analyses
Initially, two sets of chi-square analyses were conducted to examine: 1) whether the distribution of study variables differed for women and men; and 2) the association of sleep apnea risk with each study variable, separately for women and men. Spearman rank correlations were conducted among all independent variables to assess multicollinearity. Following descriptive analyses, sex-specific multiple logistic regression analyses were conducted using a manual backward selection approach; only statistically significant variables were retained in the final models. Generalized estimating equations (GEE) were utilized to account for household clustering. The quasi-likelihood under the independence model criterion (QIC) goodness of fit statistic was used to identify the best fitting GEE model (Pan 2001). Statistical analyses were conducted using the Statistical Package for the Social Sciences v21 (IBM; http://www.spss.com).
Results
Based on STOP-Obesity scores, 19.4% of women and 30.1% of men were classified as high risk of OSA. Table 1 shows the distribution of all other study variables by sex. No age difference was present, but a greater proportion of men than women were married, employed, living on a farm, had a high school education or less, and were in the highest income adequacy grouping. Women and men did not differ in financial strain. A higher percentage of women reported exercising and being a non-smoker, whereas men were more likely to binge drink. A greater proportion of men than women reported the presence of diabetes, heart disease, and high cholesterol, with depression showing the opposite pattern. While no sex difference emerged in the prevalence of asthma, COPD, self-rated mental health, or life stress, a higher proportion of women than men indicated positive self-rated physical health, better health compared to the previous year, and high social support.
Table 2 shows the relationship between OSA risk and each study variable, separately for women and men. With the exception of binge drinking and injury, all variables were significantly associated with OSA risk, and for most variables, the nature of association was similar for women and men. That is, high OSA risk was associated with older age, town rather than farm residence, lower income, lower education, being a current or ex-smoker, not exercising, and the presence of a chronic condition (ie., heart disease, diabetes, high cholesterol, COPD, and depression). Elevated OSA risk was also associated with lower quality of life, in the form of fair/poor self-rated physical and mental health, worse health compared to the previous year, and high life stress. Regarding sex differences, OSA risk was highest for married men and divorced women, and asthma was associated with high OSA risk in women but not in men. Among the employment categories, retired women and “other” status men had the highest prevalence of high OSA risk. For financial strain, high OSA risk in women was associated with not having enough money at the end of the month, whereas in men, high OSA risk was most common among those with missing data. Finally, low community support was related to high OSA risk in men but unrelated in women.
Table 3 displays the final adjusted models predicting high OSA risk for women and men. For both women and men, high risk of OSA was associated with older age, town residence (compared to farm), not exercising, heart disease, diabetes, high cholesterol, fair/poor self-rated physical health, and greater life stress. For women only, the presence of asthma, depression, and financial strain were also related to an elevated odds of OSA risk. Conversely, for men only, high risk of OSA was associated with being married/partnered, lower educational attainment, and lower community support.
Discussion
In this sample of rural adults, based on their STOP-Obesity score, 19.4% of women and 30.1% of men were at risk of OSA. Many of the correlates of OSA risk were similar for women and men, with high OSA risk associated with older age, town residence, not exercising, the presence of chronic conditions such as heart disease, diabetes, and high cholesterol, fair/poor self-rated physical health, and life stress. Sex differences were observed in the relationship between OSA risk and marital status, education, financial strain, asthma, depression, and community support.
Prevalence
A greater proportion of men (30%) than women (19%) were considered at high risk of OSA, findings consistent with a vast amount of previous research (Senaratna et al. 2017; Bonsignore et al. 2019; Fuhrman et al. 2012; Heinzer et al. 2015; Thompson et al. 2022; Huang et al. 2018). The reasons underlying this pattern are no doubt complex and likely related to the interaction of exposures linked to both sex and gender (Bonsignore et al. 2019; Bublitz et al. 2022). The prevalence of OSA risk observed here is not inconsistent with the 9%-38% range reported in a recent systematic review of objectively measured OSA in community samples (Senaratna et al. 2017). Our estimates are higher, however, than those reported in a recent Canada-wide study also applying STOP-Obesity scores, whereby 13.1% of women and 21.9% of men were classified as being at high risk of OSA (Thompson et al. 2022). Our higher estimates could have been due, in part, to a greater prevalence of obesity in our sample; that is, people living in rural areas of Canada tend to have an elevated risk of obesity when compared to the national average (Shields and Tjepkema 2006), and this elevation may be even more pronounced in several provinces, including Saskatchewan (Chen et al. 2009). Also, while Thompson objectively measured %BF (Thompson et al. 2022), our estimate was based on a prediction equation, which may have resulted in an overestimation, thus inflating OSA risk prevalence (Gallagher et al. 2000). It is important to note, however, that Thompson et al. acknowledge that their estimates were considerably lower than those obtained in prior research with similar sample demographics (Thompson et al. 2022). Generally speaking, meaningful comparisons of OSA prevalence between studies are extremely challenging, given the wide variability in population characteristics and study methodologies (Senaratna et al. 2017).
Socio-demographics
Previous research suggests that OSA risk increases with age (Senaratna et al. 2017), and is consistent with the findings of the present study. The areas of the hypothalamus undergo reductions in volume with age, contributing to fragmentation in critical stages of sleep and homeostatic dysregulation, disrupting the dopaminergic system and increasing the risk of sleep disorders (Sosso and Matos 2021). For women, OSA risk increases after menopause due to lower progesterone levels, reducing the responsiveness of hypoxia chemoreceptors to oxygen reduction or depletion (Bonsignore et al. 2019; Sosso and Matos 2021; Mallampalli and Carter 2014; Jeler and Mihaltan 2016), often leading to smaller sex differences in prevalence during this life stage. Although menopausal status was not assessed in this study, the ratio of male to female OSA risk was largest among those less than 60 years of age and smallest in those between 60 and 69 years, the latter of which corresponds to an age range in which the majority of women would likely be post-menopausal (Costanian et al. 2018). The highest prevalence of OSA in this study was observed in 70–79 year old women (26.6%) and men (37.9%), which is not inconsistent with the results of a recent systematic review of OSA in the general population in which observed rates among older adults were described by authors as “strikingly high” (Senaratna et al. 2017).
In this rural sample, we found town residents, both women and men, were more likely to be at high risk of OSA than farm residents. This finding is difficult to explain given that factors which may be associated with town residence and a greater risk of OSA, such as older age and the presence of chronic conditions, were adjusted for in the multivariable analysis. Additional research is needed to identify those determinants of OSA risk that may be more common in town than farm dwellers that were unmeasured in this study.
Sex differences in relation to OSA risk emerged for several demographic factors in this study. Regarding marital status, being partnered was associated with a high risk of OSA for men but was unrelated for women. Some previous research has similarly reported a higher prevalence of OSA in those married or cohabiting (Fuhrman et al. 2012; Charokopos et al. 2007); being partnered increases the likelihood of another person sleeping nearby and possibly alerting a partner to their OSA symptoms and encouraging health professional contact. Although the focus of the present study was not on diagnosis, several questions on our screening tool involved snoring and observed sleep apnea, both of which might become more apparent in the presence of a partner. But why was a similar relationship not observed for partnered women in this study? Some research suggests that men may be less likely than women to notice sleep-related abnormalities in their partners (Breugelmans et al. 2004), possibly emanating from traditional gender role expectations whereby girls and women are raised to be attentive to the needs of others, and men, more individualistic and self-focused. It is important to note, however, we did not have information on the sex/gender of partners in this study. Also, some research has failed to find any association between marital status and OSA diagnosis or symptomology (Krüger et al. 2023; Spuy et al. 2018). Additional research is clearly needed to clarify this relationship between gender, OSA, and marital status.
Regarding socioeconomic status (SES), lower education and greater financial strain were associated with high risk of OSA for men only and women only, respectively, whereas household income was unrelated to OSA for both sexes. The broader literature to a certain extent mirrors the findings of the present study, with mixed results reported, often dependent on the particular SES indicator, sex/gender, and the measure of OSA (Spuy et al. 2018; Cunningham et al. 2021; Tufik et al. 2010; Li et al. 2008; Adams et al. 2012). Although results have been variable, including some studies which suggest no association between SES and OSA (Spuy et al. 2018), research to date leans towards the presence of an increased prevalence of OSA among those lower in the social hierarchy (Park et al. 2022), which is generally consistent with the findings of this study. Compared to other chronic conditions, research examining the relationship between SES and OSA is somewhat in the beginning stages, and it is currently unclear as to whether the increased occurrence observed among those less advantaged is linked to OSA risk, severity, access to medical care, and/or reverse causality (Park et al. 2022).
Health behaviors
Exercise in the present study was associated with reduced odds of OSA risk for both women and men, which is consistent with previous research (Huang et al. 2018; Spuy et al. 2018; Sunwoo et al. 2018; Hall et al. 2020), including the results of a recent longitudinal study (Liu et al. 2022). Various mechanisms have been proposed to explain how physical inactivity or a sedentary lifestyle may increase OSA risk such as systemic inflammation, insulin resistance, and lower extremity oedema (Dempsey et al. 2010; Huang et al. 2021; Yumino et al. 2010; Duan et al. 2022). Conversely, it is also possible that OSA-related difficulties may deter individuals from engaging in physical activities due to time constraints or diminished physical capabilities (Hiestand et al. 2006).
Binge drinking in the present study was unrelated to OSA risk, a finding in contrast with the results of several meta-analyses reporting alcohol use to be associated with an increased likelihood of OSA (Simou et al. 2018; Taveira et al. 2018). Alcohol may increase OSA risk by reducing the body’s sensitivity to blood-oxygen reduction during sleep and binge drinking can increase this stress (Simou et al. 2018; Urbanik et al. 2020). Alcohol may also aggravate OSA severity by increasing levels of apnea, episodes of breathing cessation (Scanlan et al. 2000). However, varied measurement and a lack of longitudinal research precludes any definitive statements about the precise nature of the relationship between alcohol and OSA (Qian et al. 2023). In this cross-sectional study, we used a threshold measure of 5 or more drinks on a single occasion at least once a month to determine binge drinking and failed to find any association for women or men; this measure, although standard (Bulloch et al. 2016), combines individuals who binge drink once a month with those binging more than once a week. In supplementary exploratory analysis (data not shown) we recoded the binge drinking variable from dichotomous to ordinal, with results suggesting an association between binge drinking and increased OSA risk, but only among men in the most extreme binge drinking category (i.e., more than once a week). Clearly, additional research is needed to clarify the relationship between alcohol use (e.g., quantity and pattern) and OSA risk and whether associations vary by sex/gender.
Comorbidities
Previous research has reported associations between OSA and an increased occurrence of heart disease (Thompson et al. 2022; Kim et al. 2021), diabetes (Heinzer et al. 2015; Thompson et al. 2022; Huang et al. 2018; Hall et al. 2020; Kim et al. 2021), and high cholesterol (Hall et al. 2020), which was similarly observed for both women and men in the present study. Systemic inflammation is caused by visceral fats, the excess fats surrounding abdominal organs, which secrete inflammatory substances and reduce lung volumes (Drager et al. 2013; McArdle et al. 2007). People with OSA have resistance to leptin, a substance that inhibits hunger (Kim et al. 2021), and resistance to insulin, implicated in narrowing the upper airway by causing inflammation and increasing muscle fatigue (McArdle et al. 2007; Edwards et al. 2020). These metabolic dysregulations with insulin resistance show why diabetes is a common comorbidity of OSA (Drager et al. 2013). The intermittent hypoxia from breathing cessation in sleep among people with OSA also influences higher cholesterol production, inflammation, and lower clearance of fats (McArdle et al. 2007), which accounts for the prevalence of high cholesterol among the participants at high OSA risk. While some research suggests a stronger association between OSA and some cardiometabolic outcomes in women than men, such as diabetes (Cunningham et al. 2021), others have failed to report such differences (Bonsignore et al. 2019). The relationship between OSA risk and cardiometabolic comorbidities are no doubt complex and likely operate in a bi-directional fashion which we are unable to clarify further in this cross-sectional analysis.
In the present study, high OSA risk related to increased odds of depression among women but not men. An association between OSA and greater depression has been reported in a large body of research (Edwards et al. 2020; Garbarino et al. 2020). The higher rates of depression, alongside OSA, can be explained by the increased SNS activity due to fragmented sleep and systemic inflammation (Gupta and Knapp 2014; Canessa et al. 2011). In a different sense, the use of serotonin to affect upper airway muscles contribute to further sleep fragmentations and neurotransmitter imbalances, serving as another link to depression (Canessa et al. 2011). Consistent with our findings, considerable research has reported a relationship between OSA and depression only in women (Heinzer et al. 2018; Mokhlesi et al. 2016; Davies et al. 2019), whereas other studies have observed increased odds in men as well (Garbarino et al. 2020; Gupta and Knapp 2014). Interestingly, in our univariate results, men at high risk of OSA also had an elevated prevalence of depression compared to those at low OSA risk, but this association became nonsignificant in multivariable analysis.
Another sex-specific association in this study emerged regarding asthma, with elevated odds reported among high OSA risk women but not men. A recent systematic review (Davies et al. 2019) concluded that between 19 and 60% of people with asthma also have OSA, and that their co-occurrence predicts poorer health outcomes. Systemic inflammation is considered a key pathophysiologic mechanism linking OSA and asthma (Ragnoli et al. 2021). Consistent with our study, some previous research has reported a more pronounced association between OSA and asthma in women than men (Bonsignore et al. 2018 Dec; Krishnan et al. 2022; Chua et al. 2023), whereas some studies have indicated a similar, elevated occurrence of asthma for both (Thompson et al. 2022).
Quality of life
A growing body of research suggests that quality of life (QOL) may be severely compromised in individuals with OSA (Chua et al. 2023; Moyer et al. 2001; Appleton et al. 2018). We incorporated several indicators of QOF into the present study and found that women and men at high risk of OSA were significantly more likely than their low risk counterparts to report poor self-rated physical health and high stress levels, associations which have been similarly reported in prior research (Ward et al. 2022; Wong et al. 2021; Pauletto et al. 2021). The adverse impact of OSA symptoms on daily life can be considerable (Moyer et al. 2001) and may be more pronounced if left untreated or undertreated (Pauletto et al. 2021). In addition, high OSA risk men in this study, but not women, reported feeling less supported by their community. This result is somewhat challenging to interpret, given the broad nature of the question [How well do you feel like you are generally supported (socially, emotionally, medically, etc.) by your community?]. While we could not find any prior research using a similar variable, this association is broadly consistent with the lower QOL generally reported by those with OSA (Moyer et al. 2001; Appleton et al. 2018). It is possible that we have identified a group of vulnerable rural men at high risk of OSA who experience social isolation and who are medically underserved. Additional research is required to understand this sex-specific association, and whether such a finding can be linked with traditionally masculine attributes of stoicism and a lower likelihood of help-seeking behavior.
Study strengths and limitations
In addition to our positioning of sex/gender as an important consideration when examining OSA risk in a rural setting, strengths of this study include a large sample size and the inclusion of a broad array of risk factors. There are also several limitations in the current study. The gold standard for assessment of OSA, polysomnography, was not available in this study. However, the fact that the prevalence of high risk for OSA in this study was comparable to that from previous studies in general population samples lends some support regarding the validity of our screening tool. It is important to note, however, that the psychometric properties of the OSA tool used in this study are less established than those of other standard screening measures, such as STOP-BANG; further research is required to provide evidence of the validity of the STOP-Obesity tool. Our independent variables were also self-reported which may have resulted in some misclassification, as might the binary nature of many of these variables. In addition, the design of our study was cross-sectional thus preventing us from inferring causal associations.
Conclusion
A sizable minority of rural women and men may have possible undiagnosed OSA, which in turn, is associated with substantial comorbidity and reduced quality of life. While many of the correlates of OSA risk were similar for women and men, several sex differences also emerged. Additional research with enhanced measurement and a longitudinal design is required to test the veracity of these findings and further clarify the role of sex/gender in relation to OSA risk in rural adults.
Availability of data and materials
Original microdata files for the Saskatchewan Rural Health Study used to support the findings of this study are available from Drs. Pahwa and Dosman at the Canadian Centre for Rural and Agricultural Health, University of Saskatchewan, upon reasonable request.
Abbreviations
- OSA:
-
Obstructive sleep apnea
- SRHS:
-
Saskatchewan Rural Health Study
- SES:
-
Socioeconomic status
- COPD:
-
Chronic obstructive pulmonary disease
- BMI:
-
Body mass index
References
Adams RJ, Piantadosi C, Appleton SL, Hill CL, Visvanathan R, Wilson DH, McEvoy RD. Investigating obstructive sleep apnoea: will the health system have the capacity to cope? A Population Study. Aust Health Rev. 2012;36(4):424–9.
Appleton S, Gill T, Taylor A, McEvoy D, Shi Z, Hill C, Reynolds A, Adams R. Influence of gender on associations of obstructive sleep apnea symptoms with chronic conditions and quality of life. Int J Environ Res Public Health. 2018;15(5):930.
Bonsignore MR, Pepin JL, Anttalainen U, Schiza SE, Basoglu OK, Pataka A, Steiropoulos P, Dogas Z, Grote L, Hedner J, McNicholas WT. Clinical presentation of patients with suspected obstructive sleep apnea and self-reported physician-diagnosed asthma in the ESADA cohort. J Sleep Res. 2018;27(6):e12729.
Bonsignore MR, Saaresranta T, Riha RL. Sex differences in obstructive sleep apnoea. Eur Respir Rev. 2019;28(154):190030.
Borsoi L, Armeni P, Donin G, Costa F, Ferini-Strambi L. The invisible costs of obstructive sleep apnea (OSA): systematic review and cost-of-illness analysis. PLoS ONE. 2022;17(5):e0268677.
Breugelmans JG, Ford DE, Smith PL, et al. Differences in patient and bed partner-assessed quality of life in sleep-disordered breathing. Am J Respir Crit Care Med. 2004;170:547–52.
Bublitz M, Adra N, Hijazi L, Shaib F, Attarian H, Bourjeily G. A narrative review of sex and gender differences in sleep disordered breathing: gaps and opportunities. Life (Basel). 2022;12(12):2003.
Bulloch AG, Williams JV, Lavorato DH, Patten SB. Trends in binge drinking in Canada from 1996 to 2013: a repeated cross-sectional analysis. CMAJ Open. 2016;4(4):E599-604.
Canessa N, Castronovo V, Cappa SF, Aloia MS, Marelli S, Falini A, et al. Obstructive sleep apnea: brain structural changes and neurocognitive function before and after treatment. Am J Respir Crit Care Med. 2011;183(10):1419–26.
Charokopos N, Leotsinidis M, Tsiamita M, Karkoulias K, Spiropoulos K. Sleep apnea syndrome in a referral population in Greece: influence of social factors. Lung. 2007;185:235–40.
Chen Y, Rennie D, Dosman JA. Changing prevalence of obesity in a rural community between 1977 and 2003: a multiple cross-sectional study. Public Health. 2009;123:15–9.
Chua AP, Soh ZY, Rahman SA, Luo N, Shorey S. Lived experience of patients with sleep apnea: a systematic synthesis of qualitative evidence. Qual Life Res. 2023;32(5):1447–67.
Chung F, Yegneswaran B, Liao P, Chung SA, Vairavanathan S, Islam S, Khajehdehi A, Shapiro CM. STOP questionnaire: a tool to screen patients for obstructive sleep apnea. Anesthesiology. 2008;108(5):812–21.
Costanian C, McCague H, Tamim H. Age at natural menopause and its associated factors in Canada: cross-sectional analyses from the Canadian Longitudinal Study on Aging. Menopause. 2018;25(3):265–72.
Cunningham J, Hunter M, Budgeon C, Murray K, Knuiman M, Hui J, Hillman D, Singh B, James A. The prevalence and comorbidities of obstructive sleep apnea in middle-aged men and women: the Busselton Healthy Ageing Study. J Clin Sleep Med. 2021;17(10):2029–39.
Davies SE, Bishopp A, Wharton S, Turner AM, Mansur AH. The association between asthma and obstructive sleep apnea (OSA): a systematic review. J Asthma. 2019;56(2):118–29.
Dempsey JA, Veasey SC, Morgan BJ, et al. Pathophysiology of sleep apnea. Physiol Rev. 2010;90:47–112.
Dillman DA. Mail and internet surveys: the tailored design method. 2nd ed. New York, NY: Wiley & Sons Inc; 2000.
Drager LF, Togeiro SM, Polotsky VY, Lorenzi-Filho G. Obstructive Sleep Apnea: A Cardiometabolic Risk in Obesity and the Metabolic Syndrome. J Am Coll Cardiol. 2013;59(6):1027–33.
Duan X, Zheng M, Zhao W, Huang J, Lao L, Li H, Lu J, Chen W, Liu X, Deng H. Associations of depression, anxiety, and life events with the risk of obstructive sleep apnea evaluated by berlin questionnaire. Front Med. 2022;9:1–10.
Edwards C, Almeida OP, Ford AH. Obstructive sleep apnea and depression: A systematic review and meta-analysis. Maturitas. 2020;142:45–54.
Fu Y, Xia Y, Yi H, Xu H, Guan J, Yin S. Meta-analysis of all-cause and cardiovascular mortality in obstructive sleep apnea with or without continuous positive airway pressure treatment. Sleep Breath. 2017;21:181–9.
Fuhrman C, Fleury B, Nguyên XL, Delmas MC. Symptoms of sleep apnea syndrome: high prevalence and underdiagnosis in the French population. Sleep Med. 2012;13(7):852–8.
Gallagher D, Visser M, Sepulveda D, Pierson RN, Harris T, Heymsfield SB. How useful is body mass index for comparison of body fatness across age, sex, and ethnic groups? Am J Epidemiol. 1996;143(3):228–39.
Gallagher D, Heymsfield SB, Heo M, Jebb SA, Murgatroyd PR, Sakamoto Y. Healthy percentage body fat ranges: an approach for developing guidelines based on body mass index. Am J Clin Nutr. 2000;72(3):694–701.
Garbarino S, Bardwell WA, Guglielmi O, Chiorri C, Bonanni E, Magnavita N. Association of anxiety and depression in obstructive sleep apnea patients: a systematic review and meta-analysis. Behav Sleep Med. 2020;18(1):35–57.
Gupta MA, Knapp K. Cardiovascular and psychiatric morbidity in obstructive sleep apnea (OSA) with insomnia (sleep apnea plus) versus obstructive sleep apnea without insomnia: a case-control study from a nationally representative US sample. PLoS ONE. 2014;9(3):e90021.
Hall KA, Singh M, Mukherjee S, Palmer LJ. Physical activity is associated with reduced prevalence of self-reported obstructive sleep apnea in a large, general population cohort study. J Clin Sleep Med. 2020;16(7):1179–87.
Heinzer R, Vat S, Marques-Vidal P, Marti-Soler H, Andries D, Tobback N, Mooser V, Preisig M, Malhotra A, Waeber G, Vollenweider P. Prevalence of sleep-disordered breathing in the general population: the HypnoLaus study. Lancet Respir Med. 2015;3(4):310–8.
Heinzer R, Marti-Soler H, Marques-Vidal P, Tobback N, Andries D, Waeber G, Preisig M, Vollenweider P, Haba-Rubio J. Impact of sex and menopausal status on the prevalence, clinical presentation, and comorbidities of sleep-disordered breathing. Sleep Med. 2018;51:29–36.
Hiestand DM, Britz P, Goldman M, Phillips B. Prevalence of symptoms and risk of sleep apnea in the US population. Chest. 2006;130(3):780–6.
Huang T, Goodman M, Li X, et al. C-reactive protein and risk of obstructive sleep apnea in four US cohorts. Chest. 2021;159:2439–48.
Huang T, Lin BM, Markt SC, Stampfer MJ, Laden F, Hu FB, et al. Sex differences in the associations of obstructive sleep apnoea with epidemiological factors. Eur Respir J. 2018;51(3):1702421.
Janzen B, Karunanayak C, Rennie D, Lawson J, Dosman JA, Pahwa P. Depression and binge drinking in farm and non-farm rural adults in Saskatchewan. Can Rural Remote Health. 2020;20(1):174–85.
Javaheri S, Barbe F, Campos-Rodriguez F, Dempsey JA, Khayat R, Javaheri S, Malhotra A, Martinez-Garcia M, Mehra R, Pack AI, Polotsky VY, Redline S, Somers VK. Sleep apnea: types, mechanisms, and clinical cardiovascular consequences. J Am Coll Cardiol. 2017;69(7):841–58.
Jeler EC, Mihaltan FD. Obstructive sleep apnea and risk factors. Ro J Neurol. 2016;14(4):155–9.
Kim DH, Kim B, Han K, Kim SW. The relationship between metabolic syndrome and obstructive sleep apnea syndrome: a nationwide population-based study. Sci Rep. 2021;11(1):8751.
Knauert M, Naik S, Gillespie MB, Kryger M. Clinical consequences and economic costs of untreated obstructive sleep apnea syndrome. World J Otorhinolaryngol Head Neck Surg. 2015;1(1):17–27.
Krishnan S, Chai-Coetzer CL, Grivell N, Lovato N, Mukherjee S, Vakulin A, Adams RJ, Appleton SL. Comorbidities and quality of life in Australian men and women with diagnosed and undiagnosed high-risk obstructive sleep apnea. J Clinical Sleep Med. 2022;18(7):1757–67.
Krüger M, Obst A, Bernhardt O, Ewert R, Penzel T, Stubbe B, Fietze I, Ivanovska T, Biffar R, Daboul A. Socioeconomic factors do not predict sleep apnea in a population sample from Mecklenburg-Western Pomerania. Germany Sleep Breath. 2023;27(2):459–67.
Laratta CR, Ayas NT, Povitz M, Pendharkar SR. Diagnosis and treatment of obstructive sleep apnea in adults. CMAJ. 2017;189(48):1481–8.
Li X, Sundquist K, Sundquist J. Socioeconomic status and occupation as risk factors for obstructive sleep apnea in Sweden: a population-based study. Sleep Med. 2008;9(2):129–36.
Lindberg E, Benediktsdottir B, Franklin KA, Holm M, Johannessen A, Jögi R, Gislason T, Real FG, Schlünssen V, Janson C. Women with symptoms of sleep-disordered breathing are less likely to be diagnosed and treated for sleep apnea than men. Sleep Med. 2017;35:17–22.
Liu Y, Yang L, Stampfer MJ, et al. Physical activity, sedentary behaviour and incidence of obstructive sleep apnoea in three prospective US cohorts. Eur Respir J. 2022;59(2):2100606.
Mallampalli MP, Carter CL. Exploring sex and gender differences in sleep health: a Society for Women’s Health Research Report. J Womens Health (larchmt). 2014;23(7):553–62.
Marin-Jimenez N, Cruz-Leon C, Sanchez-Oliva D, Jimenez-Iglesias J, Caraballo I, Padilla-Moledo C, Cadenas-Sanchez C, Cuenca-Garcia M, Castro-Piñero J. Criterion-related validity of field-based methods and equations for body composition estimation in adults: a systematic review. Curr Obes Rep. 2022;11(4):336–49.
McArdle N, Hillman D, Beilin L, Watts G. Metabolic risk factors for vascular disease in obstructive sleep apnea: a matched controlled study. Am J Respir Crit Care Med. 2007;175(2):190–5.
Mokhlesi B, Ham SA, Gozal D. The effect of sex and age on the comorbidity burden of OSA: an observational analysis from a large nationwide US health claims database. Eur Respir J. 2016;47(4):1162–9.
Mou J, Pflugeisen BM, Crick BA, Amoroso PJ, Harmon KT, Tarnoczy SF, Shirley Ho S, Mebust KA. The discriminative power of STOP-Bang as a screening tool for suspected obstructive sleep apnea in clinically referred patients: considering gender differences. Sleep Breath. 2019;23:65–75.
Moyer CA, Sonnad SS, Garetz SL, Helman JI, Chervin RD. Quality of life in obstructive sleep apnea: a systematic review of the literature. Sleep Med. 2001;2(6):477–91.
Orbea CA, Lloyd RM, Faubion SS, Miller VM, Mara KC, Kapoor E. Predictive ability and reliability of the STOP-BANG questionnaire in screening for obstructive sleep apnea in midlife women. Maturitas. 2020;135:1–5.
Pahwa P, Karunanayake CP, Hagel L, Janzen B, Pickett W, Rennie D, Senthilselvan A, Lawson J, Kirychuk S, Dosman J. The Saskatchewan rural health study: an application of a population health framework to understand respiratory health outcomes. BMC Res Notes. 2012;5:1–3.
Pahwa P, Rana M, Pickett W, Karunanayake CP, Amin K, Rennie D, Lawson J, Kirychuk S, Janzen B, Koehncke N, Dosman J. Cohort profile: the Saskatchewan rural health study—adult component. BMC Res Notes. 2017;10:1–7.
Pan W. Akaike’s information criterion in generalized estimating equations. Biometrics. 2001;57(1):120–5.
Park JW, Hamoda MM, Almeida FR, Wang Z, Wensley D, Alalola B, Alsaloum M, Tanaka Y, Huynh NT, Conklin AI. Socioeconomic inequalities in pediatric obstructive sleep apnea. J Clin Sleep Med. 2022;18(2):637–45.
Patel D, Tsang J, Saripella A, Nagappa M, Islam S, Englesakis M, Chung F. Validation of the STOP questionnaire as a screening tool for OSA among different populations: a systematic review and meta-regression analysis. J Clin Sleep Med. 2022;18(5):1441–53.
Pauletto P, Réus JC, Bolan M, Massignan C, Flores-Mir C, Maia I, Gozal D, Hallal AL, Porporatti AL, Canto GD. Association between obstructive sleep apnea and health-related quality of life in untreated adults: a systematic review. Sleep Breath. 2021;25(4):1773–89.
Pendharkar SR, Sharpe H, Rosychuk RJ, Laratta CR, Fong A, Duan QM, Ronksley PE, MacLean JE. Temporal and Regional Trends in Obstructive Sleep Apnea Using Administrative Health Data in Alberta. Canada Ann Am Thorac Soc. 2023;20(3):433–9.
Peppard PE, Young T, Barnet JH, et al. Increased prevalence of sleep-disordered breathing in adults. Am J Epidemiol. 2013;177(9):1006–14.
du Plessis V, Beshiri R, Bollman RD, Clemenson H. Definitions of “Rural”. Agriculture and Rural Working Paper Series, Working Paper No. 61. Catalogue no. 21–601-MIE- No. 061. Ottawa: Agricultural Division, Statistics Canada; 2004.
Qian Y, Dharmage SC, Hamilton GS, Lodge CJ, Lowe AJ, Zhang J, Bowatte G, Perret JL, Senaratna CV. Longitudinal risk factors for obstructive sleep apnea: a systematic review. Sleep Med Rev. 2023;71:101838.
Ragnoli B, Pochetti P, Raie A, Malerba M. Interrelationship between obstructive sleep apnea syndrome and severe asthma: from endo-phenotype to clinical aspects. Front Med. 2021;8:1–8.
Scanlan M, Roebuck T, Little PJ, Naughton MT. Effect of moderate alcohol upon obstructive sleep apnoea. Eur Respir J. 2000;16(5):909–13.
Senaratna CV, Perret JL, Lodge CJ, Lowe AJ, Campbell BE, Matheson MC, Hamilton GS, Dharmage SC. Prevalence of obstructive sleep apnea in the general population: a systematic review. Sleep Med Rev. 2017;34:70–81.
Shields M, Tjepkema M. Regional differences in obesity. Health Rep. 2006;17(3):61–7.
Simou E, Britton J, Leonardi-Bee J. Alcohol and the risk of sleep apnoea: a systematic review and meta-analysis. Sleep Med. 2018;42:38–46.
Sosso FE, Matos E. Socioeconomic disparities in obstructive sleep apnea: a systematic review of empirical research. Sleep Breath. 2021;25(4):1729–39.
Spagnuolo CM, McIsaac M, Dosman J, Karunanayake C, Pahwa P, Pickett W. Distance to specialist medical care and diagnosis of obstructive sleep apnea in rural Saskatchewan. Can Respir J. 2019;14(2019):1683124.
Statistics Canada: Health Fact Sheet. Sleep apnea in Canada, 2016 and 2017. 2018. Retrieved from https://www150.statcan.gc.ca/n1/pub/82-625-x/2018001/article/54979-eng.htm.
Sunwoo JS, Hwangbo Y, Kim WJ, Chu MK, Yun CH, Yang KI. Prevalence, sleep characteristics, and comorbidities in a population at high risk for obstructive sleep apnea: a nationwide questionnaire study in South Korea. PLoS ONE. 2018;13(2):e0193549.
Taveira KV, Kuntze MM, Berretta F, de Souza BD, Godolfim LR, Demathe T, De Luca CG, Porporatti AL. Association between obstructive sleep apnea and alcohol, caffeine and tobacco: a meta-analysis. J Oral Rehab. 2018;45(11):890–902.
Thompson C, Legault J, Moullec G, Baltzan M, Cross N, Dang-Vu TT, Martineau-Dussault MÈ, Hanly P, Ayas N, Lorrain D, Einstein G. A portrait of obstructive sleep apnea risk factors in 27,210 middle-aged and older adults in the Canadian Longitudinal Study on Aging. Sci Rep. 2022;12(1):5127.
Tufik S, Santos-Silva R, Taddei JA, Bittencourt LR. Obstructive sleep apnea syndrome in the Sao Paulo epidemiologic sleep study. Sleep Med. 2010;11(5):441–6.
Urbanik D, Martynowicz H, Mazur G, Poręba R, Gać P. Environmental factors as modulators of the relationship between obstructive sleep apnea and lesions in the circulatory system. J Clinical Med. 2020;9(3):836.
van der Spuy I, Zhao G, Karunanayake C, Pahwa P. Predictors of sleep apnea in the Canadian population. Can Respir J. 2018;28:6349790.
Ward SA, Storey E, Gasevic D, Naughton MT, Hamilton GS, Trevaks RE, Wolfe R, O’Donoghue FJ, Stocks N, Abhayaratna WP, Fitzgerald S. Sleep-disordered breathing was associated with lower health-related quality of life and cognitive function in a cross-sectional study of older adults. Respirology. 2022;27(9):767–75.
WHO. Physical status: the use and interpretation of anthropometry Report of a WHO expert committee. World Health Organization Technical Report Series. 1995;854:1–452.
Wong JL, Martinez F, Aguila AP, Pal A, Aysola RS, Henderson LA, Macey PM. Stress in obstructive sleep apnea. Sci Rep. 2021;11(1):1–9.
Yumino D, Redolfi S, Ruttanaumpawan P, et al. Nocturnal rostral fluid shift: a unifying concept for the pathogenesis of obstructive and central sleep apnea in men with heart failure. Circulation. 2010;121:1598–605.
Acknowledgements
We would like to thank the reviewers for their helpful comments and suggestions to enhance the quality of this manuscript.
The Saskatchewan Rural Health Study Team consists of: James Dosman, MD (Designated Principal Investigator, University of Saskatchewan (UofS), Saskatoon, SK Canada); Punam Pahwa, PhD (Co-Principal Investigator, UofS, Saskatoon SK Canada); John Gordon, PhD (Co-Principal Investigator, UofS, Saskatoon SK Canada); Yue Chen, PhD (University of Ottawa, Ottawa Canada); Roland Dyck, MD (UofS, Saskatoon SK Canada); Louise Hagel (Project Manager, UofS, Saskatoon SK Canada); Bonnie Janzen, PhD (UofS, Saskatoon SK Canada); Chandima Karunanayake, PhD (UofS, Saskatoon SK Canada); Shelley Kirychuk, PhD (UofS, Saskatoon SK Canada); Niels Koehncke, MD (UofS, Saskatoon SK Canada); Joshua Lawson, PhD, (UofS, Saskatoon SK Canada); William Pickett, PhD (Queen's University, Kingston ON Canada); Roger Pitblado, PhD (Professor Emeritus, Laurentian University, Sudbury ON Canada); Donna Rennie, RN, PhD, (UofS, Saskatoon SK Canada); Ambikaipakan Senthilselvan, PhD (University of Alberta, Edmonton, AB, Canada). We are grateful for the contributions of all the participants who donated their time to complete and return the survey.
Informed consent
Informed consent was obtained from all subjects involved in the study.
Funding
Data used in this study were from the Saskatchewan Rural Health Study, funded by a grant from the Canadian Institutes of Health Research MOP-187209-POP-CCAA-11829.
Author information
Authors and Affiliations
Contributions
This research was undertaken towards partial fulfillment of I.Z.’s B.A. & Sc. (Hons) degree in Health Studies under the supervision of B.J. P.P. and J.D. acquired the data used in this manuscript. I.Z and B.J. conceptualized the present study, analyzed the data, and wrote the initial manuscript draft. P.P., C.K., and J.D. reviewed the manuscript and provided feedback. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
The study was approved by the Biomedical Research Ethics Board of the University of Saskatchewan (PSY-REC: #2021–2022-HLST-005).
Consent for publication
Not applicable.
Competing interests
The authors declare no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zaman, I.B., Janzen, B., Karunanayake, C. et al. Sex-specific prevalence and correlates of possible undiagnosed obstructive sleep apnea in rural Canada. Sleep Science Practice 8, 5 (2024). https://doi.org/10.1186/s41606-024-00097-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s41606-024-00097-5