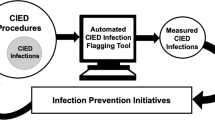
Abstract
Background
Cardiac implantable electronic device (CIED) surgical site infections (SSIs) have been outpacing the increases in implantation of these devices. While traditional surveillance of these SSIs by infection prevention and control would likely be the most accurate, this is not practical in many centers where resources are constrained. Therefore, we explored the validity of administrative data at identifying these SSIs.
Methods
We used a cohort of all patients with CIED implantation in Calgary, Alberta where traditional surveillance was done for infections from Jan 1, 2013 to December 31, 2019. We used this infection subgroup as our “gold standard” and then utilized various combinations of administrative data to determine which best optimized the sensitivity and specificity at identifying infection. We evaluated six approaches to identifying CIED infection using administrative data, which included four algorithms using International Classification of Diseases codes and/or Canadian Classification of Health Intervention codes, and two machine learning models. A secondary objective of our study was to assess if machine learning techniques with training of logistic regression models would outperform our pre-selected codes.
Results
We determined that all of the pre-selected algorithms performed well at identifying CIED infections but the machine learning model was able to produce the optimal method of identification with an area under the receiver operating characteristic curve (AUC) of 96.8%. The best performing pre-selected algorithm yielded an AUC of 94.6%.
Conclusions
Our findings suggest that administrative data can be used to effectively identify CIED infections. While machine learning performed the most optimally, in centers with limited analytic capabilities a simpler algorithm of pre-selected codes also has excellent yield. This can be valuable for centers without traditional surveillance to follow trends in SSIs over time and identify when rates of infection are increasing. This can lead to enhanced interventions for prevention of SSIs.
Similar content being viewed by others
Background
Surgical site infections (SSI) following cardiac implantable electronic device (CIED) implantation are hospital acquired infections (HAIs) that pose a substantial burden on our healthcare system [1, 2]. The annual rate of CIED implantations has increased over the past several decades due to the increasing need for pacemaker therapy in an aging population, and expanding indications for cardiac resynchronisation therapy (CRT) and prophylactic implantable cardioverter defibrillators (ICDs). Notably, the rate of CIED infections is outpacing the increase in implantation rate [3].
CIED infections can include local infections such as pocket infections, but can also include more severe systemic infections including bloodstream infections and infective endocarditis [4]. Randomized controlled trial data suggest relatively low baseline rates of CIED infection (i.e. approximately 1%) [5]. However, studies using large population-based observational data that better reflect everyday practice demonstrate CIED infection rates as high as 4% [6]. The consequence of CIED infection is increased patient morbidity and mortality. Additionally, CIED infections are associated with substantial medical resource use and costs due to repeat hospitalizations, multiple surgeries for device removal and possible re-implantation, and lengthy hospital stays for intravenous antibiotics [7, 8].
Given the impact of these infections, it is essential to perform surveillance of CIED infections within healthcare systems. Surveillance has been identified as a core component for any Infection Prevention and Control (IPC) program by international health organizations, which may include post-operative patient surveillance in the outpatient, emergency departments or inpatient setting, with detailed patient chart review, and tracking of microbiology results [9]. Unfortunately, comprehensive IPC surveillance programs for CIED infections can be costly and time-consuming, and require extensive human resources. Particularly in smaller centers without extensive funding for IPC programs this type of robust surveillance may not be practical.
An alternative method to identify CIED infections is the use of administrative data, which may be less expensive and resource intensive. However, the validity of such data for infection surveillance has been questioned and may be dependent on the type of HAI for identification [10]. A systematic review of administrative data for SSI surveillance found that across 34 studies, sensitivity and positive predictive values (PPV) only performed moderately, which may be due to pooling heterogeneous study populations undergoing very different surgical operations [11]. CIED infections were not one of the SSI included in that review.
Given the absence of studies assessing the use of administrative data for CIED infection surveillance, our study sought to validate administrative codes to robustly identify CIED infections compared to a gold standard of comprehensive IPC surveillance with chart review. A secondary objective was to determine the best administrative data approach for CIED infection identification using conventional selection of an unweighted set of preselected codes, or machine learning methods [12].
Methods
The study protocol was developed based on the modified Standards for Reporting of Diagnostic Accuracy (STARD) criteria, and recommendations on the design and reporting of administrative data validation studies [13, 14].
Study design and cohort
We identified a cohort of adults patients (i.e., age ≥ 18 years) in Calgary, Alberta who underwent de novo CIED implantation (including pacemaker (PM), ICD, or CRT) or generator replacement between January 1, 2013 and December 31, 2019. Among these patients undergoing CIED surgery, those developing a CIED infection within one year of surgery were identified through manual chart review enabled through a formal IPC Surveillance program. CIED infections were adjudicated using the Centers for Disease Control and Prevention/National Healthcare Safety Network (NHSN) standardized definitions for complex SSIs (i.e., deep or organ space) [15]. Superficial infections without involvement of the device or leads were not included as these types of infections are not an indication for CIED system removal and lead extraction [16]. This study focused on complex SSIs following CIED implantation given the substantial burden to health system (I.e., hospitalization, prolonged antibiotics, re-operation for system removal and reimplantation).These “gold standard” CIED infections were then used to validate infections identified through administrative coding data from the International Classification of Diseases-10th revision in Canada (ICD-10-CA) and Canadian Classification of Health Intervention (CCI) administrative codes (ref https://www.cihi.ca/en/cci-coding-structure).
Data sources
Paceart™
CIED implantations were identified using the PaceArt™ database, which is a repository of all device-related clinical encounters for any patient followed within the province of Alberta, Canada. As comprehensive IPC surveillance for CIED infections is not readily available across the province, we limited the base cohort to patients with CIED procedures performed within the Calgary zone. Calgary is an urban city with a population of approximately 1.5 million. Paceart™ contains information regarding indications for device implantation, type of device, date of operation and basic demographic information. Repeat procedures were censored in the two-year period from index surgical date to avoid double counting patient encounters that may be attributed to a single CIED infection (e.g. for a patient who underwent de novo pacemaker implantation requiring a lead revision 2 months later, only the initial implant was counted as an index procedural date).
Infection prevention and control
Complex SSI cases were obtained via collaboration with Alberta Health Services (AHS) Calgary Zone Infection Prevention & Control (IPC) (one IPC department completes surveillance for Calgary zone). Superficial infections were not collected. All patients with CIED implantation are actively followed for one year to determine if they develop infection using various sources including hospital admissions, chart review, and microbiology data. This surveillance is performed by a trained Infection Control Professional with AHS who has access to patient charts. Case identification follows NHSN definitions for eligibility and complex infection criteria. Infections that are identified are brought forth to a committee for case adjudication including the Infection Control Professional, Cardiac Electrophysiologists and Infectious Diseases physicians. Surveillance results are fed back to clinicians, and quality improvement strategies are implemented if CIED infection rates increase beyond acceptable levels. The importance of surveillance is recognized as an intervention which can lead to decreases in post-surgical infections. This IPC data set formed our reference (gold standard) CIED infection target variable, which was used to validate the administrative codes.
AHS analytics
AHS is a single health system that services the entire province of Alberta, Canada. We utilized AHS analytics data which provides healthcare information on all Alberta residents with an Alberta Health Care Insurance Plan (> 99% of provincial coverage) in order to obtain information from the Discharge Abstract Database (DAD) for our patients in Calgary. This data was utilized to provide information on all hospital admissions and their associated ICD-10-CA and CCI codes (Additional file 1) for our baseline cohort, for one year after their initial CIED implantation or generator replacement. We assumed that all patients with a complex CIED infection would present to the Emergency Department or be admitted to hospital for management and that essentially all patients would require removal of their device.
Statistical analyses
We identified the CCI codes published by Canadian Institute for Health Information (CIHI), which could potentially be used to identify CIED procedures (Additional file 1). These administrative codes were used to identify potential CIED infection cases through AHS analytics data. All patients in the base cohort were followed for one year from their initial CIED implantation or generator replacement to determine if they had the “infection” codes in subsequent hospital admissions.
We used three different pre-defined algorithms to track infection. First, we searched a combination of ICD-10-CA codes for infection including: infection following a procedure (T814), infection of an implantable cardiovascular or other device (T827, T857) or surgical procedure as the cause of an abnormal reaction (Y83), with the CCI codes listed in Additional file 1 which represent CIED procedures (Algorithm 1). Second, we assessed an approach from a recent publication using non-validated ICD-10-CA codes to track infection: Infection of an implantable cardiovascular or other device (T827, T857), infective endocarditis (I330, I339, I38, I398), or cellulitis of the chest wall or other unspecified site (L0330, L0339, L038, L039) (Algorithm 2) [8]. Third, we used an approach of only CCI codes related to implantation/removal of CIEDs as follows 1.YY.54.^, 1.YY.54.LA-NJ, 1.HZ.55.^, 1.HD.53.^, 1.HD.53.GR-JA, 1.HD.54.^, 1.HD.54.GR-JA, 1.HD.55.^, 1.HD.55.GP-JB, or 1.HD.55.GR-JA (Algorithm 3) [17]. For all three approaches above, we used standard epidemiological methods to calculate sensitivity, specificity, PPV, negative predictive value (NPV) and area under the receiver operating characteristic (ROC) curve (AUC) metrics for the administrative code classification predictions.
An additional analysis was conducted where we trained a machine learning model to determine the most appropriate codes to identify infection rather than the traditional pre-identification of codes as outlined above. Logistic regression models were trained using Python and the scikit-learn library [17]. Due to the high class imbalance of the dataset (133:1 occurrence of infection) the class weights were scaled to be inversely proportional to frequency when training the model. L1 regularization was used on the model coefficients during training for its feature selection capability and to avoid overfitting the model [18]. Due to the limited number of infection examples a stratified cross validation strategy was applied to evaluate the models. A test set was not used because the primary aim was a comparison of the different approaches. The outer cross validation loop consisted of five random stratified folds of the full dataset. The same strategy was used to evaluate the performance of the traditional approaches. For the logistic regression models a nested inner training loop consisted of a grid search for regularization strength using three stratified folds of the training data split.
Ultimately, we evaluated two logistic regression models: a model trained on ICD-10-CA and CCI codes with regularization C = 0.6 (ICD & CCI Model), as well as a model trained on only ICD-10-CA codes with a regularization C = 0.4 (ICD Model). Finally, we simply looked for the ICD-10-CA code T827 given its definition of “infection of an implantable cardiovascular device”.
Following validation of administrative codes, we compared temporal trends between the CIED infections identified via gold standard IPC surveillance and those identified using the optimal administrative approach. Analysis was completed using Python version 3.9.12 and Scikit-learn version 1.1.1. Ethics for this study was obtained from the University of Calgary Health Research Ethics Board (REB20-2186).
Results
Study cohort
Between January 1 2013 and December 31 2019, there were 3536 CIED procedures performed, and a total of 5631 hospitalizations. Among these 3536 procedures, there were 42 infections (1.2%) identified through comprehensive IPC surveillance.
Baseline characteristics of the overall cohort and those with SSI’s can be found in Table 1. The most common procedure was PM insertion (72%). However, SSIs following PM insertion only accounted for 45% overall. ICDs which accounted for 20% of all device implantations accounted for 36% of SSIs.
Administrative data algorithms for CIED infection identification
All of the algorithms to identify infections utilizing administrative codes performed well. Sensitivity, specificity, PPV, NPV and AUC for each approach can be found in Table 2. Ranked based on AUC, the highest performing approach was the ICD/CCI code based logistic regression model with an average AUC of 96.8%. The ICD code only model followed closely behind with an average AUC of 96.1%. A visual representation of each AUC can be found in Fig. 1. Trends in SSIs over time identified by administrative data using all included approaches as well as the “gold standard” cohort of SSIs identified by IPC can be seen in Fig. 2. All approaches overpredict the number of SSIs, as seen in the PPV metric. The improved proximity of the trendline profile and values to the gold standard is evident for the top performing models/algorithms. While sensitivity and specificity are excellent, the PPV is low. This is due to the fact that PPV is affected by the prevalence of the “disease,” in this case the CIED infection. When prevalence is low the PPV will also be low irrespective of the sensitivity and specificity. However, the sensitivity and specificity reflect the ability to discriminate between a true positive and a true negative result. These characteristics are what is required to ensure a test or algorithm is useful for diagnosis [19].
The best performing machine learning model was comprised of 48 non-zero ICD and CCI codes, which are shown along with their weights in Table 3. Training with a high regularization strength of 0.1 reduces this list to the top 5 most predictive codes in order of importance: T827 (Infection and inflammatory reaction due to other cardiac and vascular device, implants and grafts), Y831 (Surgical operation with implant of artificial internal device as the cause of abnormal reaction of the patient, or of later complication, without mention of misadventure at the time of the procedure), 1IS53 and 1IS53GR (Implantation of internal device, vena cava (superior and inferior), percutaneous transluminal approach), 1HZ53 (Implantation of an internal device, heart). The ROC curve for this model can be visualized in Fig. 3. All trained logistic regression models prioritized T827 as the most important indicator of SSI regardless of regularization.
Discussion
We found that between January 1, 2013 and December 31, 2019 there were a total of 3,536 CIED implantations including generator replacements in Calgary zone. Of these, 42 (1.2%) developed a complex SSI. While PM insertions were the most common CIED procedure, ICD and CRT implantation accounted for a disproportionately greater amount of SSIs compared to their implantation rate, which is consistent with previous literature [8]. We found that administrative data was able to perform very well (AUC > 90%) at identifying SSIs post CIED implantation, including both pre-selected ICD/CCI code algorithms and a machine learning approach. Trends in SSIs were similar between the traditionally collected data, and the best performing administrative data approaches.
Our findings are consistent with previous recent work done on validating administrative data for SSIs. Prior work done on SSIs following hip and knee replacement found that pre-selected ICD code algorithms using administrative data were able to achieve sensitivity > 80% and specificity close to 100% compared to a gold standard of SSI collection [20, 21]. Previous studies exploring HAIs in general have examined the use of administrative data to identify these infections. The results have been inconsistent with some studies reporting that administrative data using ICD codes was of limited benefit in identifying HAIs [22]. The previously mentioned systematic review [11] which encompassed 57 studies, including 34 looking specifically at SSIs, found a wide range of sensitivity for using ICD codes to identify HAI ranging from 10 to 100% and PPV from 11 to 95% suggesting that administrative data was extremely variable at identifying infections [11]. However, it is important to note that many of those studies were done using ICD-9 revision codes as opposed to the more granular ICD-10 codes [23] which likely improve the yield of administrative data at identifying infections. Our current findings in corroboration with the aforementioned recent studies on SSIs do suggest that administrative data can be an effective tool at identifying SSIs.
There is very little literature available validating the use of administrative data for identification of SSIs following CIED implantation and comparing the use of pre-selected ICD/CCI codes and machine learning. A recent study by Mull et al. did use a unique method of combining structured and text data in Veterans Health Facilities electronic medical records in order to identify SSIs following CIED implantation [24]. In their validation logistic regression model they were able to achieve an AUC of 90%. However, this type of work would rely on the availability of data via an electronic medical record system which is not always available at centers. Furthermore, the results have limited generalizability as collected data fields may vary depending on the electronic medical system deployed in a particular healthcare system.
Machine learning is becoming increasingly recognized as an important tool for infection identification and control. A review of 52 studies looked at the ability of machine learning technique to detect or predict sepsis, HAIs (including SSIs) and other infections. However, they found that understanding and interpretability of the models was rarely addressed [25]. Our study provides a clear application and interpretation of a machine learning approach for identifying SSIs following CIED implantation contributing to the literature. Our study also demonstrated that one unique code T827 still had a very high AUC (> 90%), and therefore in situations where it may not be practical to compute a weighted total of 48 codes this could be a simpler alternative. Nevertheless, validation of administrative codes to identify CIED infection lays the foundation to adapt these algorithms in clinical practice. Potential applications include fully automated or semi-automated surveillance supplemented by traditional IPC methods [26]; however, implementation will depend on local health system needs, acceptability and resources.
Our study has several additional strengths. First, our results are generalizable as the current study validates the use of widely available administrative data—ICD-10 and CCI codes, for identification of SSIs following CIED implantation. In settings that do not use CCI codes for procedural reporting, our study provides several algorithms based on ICD-10 codes alone. Second, our work compares different methods of more traditional pre-selected data codes and machine learning approaches to logistic regression in order to identify the most optimal way to use administrative data for infection detection. Several algorithms had comparable test characteristics, which provides options for implementation depending on the needs and resources of regional health systems. For example, the algorithms using only ICD-10 codes would be applicable outside Canada, where CCI codes are not available. Finally, our study is facilitated by the availability of comprehensive IPC surveillance for CIED infections s which provides a “gold standard” to use for validation of the administrative data.
The limitations of this work must also be considered. The number of identified infections was very small given that the availability of the gold standard infection data was restricted to one city. However, this population-based study included a cohort of all CIED implantations from a large regional health zone comprised of urban, community and rural hospitals, which improves the external validity of our study. Another limitation was that IPC surveillance identified infections within one year post procedure. While most infections would occur during this timeframe any infections that occurred very late (beyond one year) would have been missed.
Conclusions
While traditional IPC surveillance is the current gold standard for identifying CIED infections, this comprehensive approach requires substantial time and resources, which may not be feasible, especially in smaller hospital centers. The current study validates a method using administrative data for accurate identification of complex SSIs following CIED implantation. While the best performance came from the machine learning models, pre-selected ICD codes also performed very well (AUCs > 90%). At centers with limited data analytic capacity where machine learning applications and validation would not be feasible, the more traditional approach of pre-selected ICD-10 codes could be used to track SSIs and trends over time. Our findings set the foundation for more accessible surveillance of CIED infections and identification of outlier infection rates compared to historic trends, which can lead to targeted interventions for prevention of SSIs.
Availability of data and materials
The data that support the findings of this study are available from Alberta Health Services but restrictions apply to the availability of these data, which were used under license for the current study, and so are not publicly available. Data are however available from the authors upon reasonable request and with permission of Alberta Health Services and appropriate ethics.
Abbreviations
- CIED:
-
Cardiac implantable electronic devices
- PM:
-
Pacemaker
- ICD:
-
Implantable cardioverter defibrillator
- CRT:
-
Cardiac resynchronization therapy
- IPC:
-
Infection prevention and control
- SSI:
-
Surgical site infection
- HAI:
-
Hospital acquired infection
- AHS:
-
Alberta Health Services
- DAD:
-
Discharge Abstract Database
- ICD:
-
International Classification of Diseases
- CCI:
-
Canadian Classification of Health Interventions
- AUC:
-
Area under the receiver operating characteristic Curve
- ROC:
-
Receiver operating characteristic
- PPV:
-
Positive predictive value
- NPV:
-
Negative predictive value
- NHSH:
-
National Healthcare Safety Network
- CIHI:
-
Canadian Institute for Health Information
References
Pakyz AL, Wang H, Ozcan YA, Edmond MB, Vogus TJ. Leapfrog hospital safety score, magnet designation, and healthcare-associated infections in United States Hospitals. J Patient Saf. 2017. https://doi.org/10.1097/pts.0000000000000378.
Portable ultraviolet light surface-disinfecting devices for prevention of hospital-acquired infections: a health technology assessment. Ont Health Technol Assess Ser. 2018;18(1):1–73.
Greenspon AJ, Patel JD, Lau E, et al. 16-Year Trends in the Infection Burden for Pacemakers and Implantable Cardioverter-Defibrillators in the United States: 1993 to 2008. J Am Coll Cardiol. 2011;58(10):1001–6.
Joy PS, Kumar G, Poole JE, London B, Olshansky B. Cardiac implantable electronic device infections: Who is at greatest risk? (1556–3871 (Electronic))
Tarakji KG, Mittal S, Kennergren C, et al. Worldwide Randomized Antibiotic EnveloPe Infection PrevenTion Trial (WRAP-IT). (1097–6744 (Electronic))
Rennert-May E, Chew D, Lu S, Chu A, Kuriachan V, Somayaji R. Epidemiology of cardiac implantable electronic device infections in the United States: a population-based cohort study. Heart Rhythm. 2020. https://doi.org/10.1016/j.hrthm.2020.02.012.
Blomström-Lundqvist C, Traykov V, Erba PA, et al. European Heart Rhythm Association (EHRA) international consensus document on how to prevent, diagnose, and treat cardiac implantable electronic device infections-endorsed by the Heart Rhythm Society (HRS), the Asia Pacific Heart Rhythm Society (APHRS), the Latin American Heart Rhythm Society (LAHRS), International Society for Cardiovascular Infectious Diseases (ISCVID) and the European Society of Clinical Microbiology and Infectious Diseases (ESCMID) in collaboration with the European Association for Cardio-Thoracic Surgery (EACTS). Europace. 22(4):515–549. https://doi.org/10.1093/europace/euz246
Daneman N, Homenauth E, Saskin R, Ng R, Ha A, Wijeysundera HC. The predictors and economic burden of early-, mid- and late-onset cardiac implantable electronic device infections: a retrospective cohort study in Ontario, Canada. (1469–0691 (Electronic))
Storr J, Twyman A, Zingg W, et al. Core components for effective infection prevention and control programmes: new WHO evidence-based recommendations. Antimicrob Resist Infect Control. 2017;6(1):6. https://doi.org/10.1186/s13756-016-0149-9.
Moehring RW, Staheli R, Miller BA, Chen LF, Sexton DJ, Anderson DJ. Central line-associated infections as defined by the centers for medicare and medicaid services’ hospital-acquired condition versus standard infection control surveillance why hospital compare seems conflicted. Infect Control Hosp Epidemiol. 2013;34(3):238–44. https://doi.org/10.1086/669527.
van Mourik MSM, van Duijn PJ, Moons KGM, Bonten MJM, Lee GM. Accuracy of administrative data for surveillance of healthcare-associated infections: a systematic review. BMJ Open. 2015;5(8): e008424. https://doi.org/10.1136/bmjopen-2015-008424.
Petrosyan Y, Thavorn K, Smith G, et al. Predicting postoperative surgical site infection with administrative data: a random forests algorithm. BMC Med Res Methodol. 2021;21(1):179. https://doi.org/10.1186/s12874-021-01369-9.
Benchimol EI, Guttmann A, Mack DR, et al. Validation of international algorithms to identify adults with inflammatory bowel disease in health administrative data from Ontario, Canada. J Clin Epidemiol. 2014;67(8):887–96. https://doi.org/10.1016/j.jclinepi.2014.02.019.
Cohen JF, Korevaar DA, Altman DG, et al. STARD 2015 guidelines for reporting diagnostic accuracy studies: explanation and elaboration. BMJ Open. 2016;6(11):e012799. https://doi.org/10.1136/bmjopen-2016-012799.
Horan TC, Andrus M, Dudeck MA. CDC/NHSN surveillance definition of health care-associated infection and criteria for specific types of infections in the acute care setting. Am J Infect Control. 2008;36(5):309–32. https://doi.org/10.1016/j.ajic.2008.03.002.
Kusumoto FM, Schoenfeld MH, Wilkoff BL, et al. 2017 HRS expert consensus statement on cardiovascular implantable electronic device lead management and extraction. Heart Rhythm. 2017;14(12):e503–51. https://doi.org/10.1016/j.hrthm.2017.09.001.
Parkash R, Sapp J, Gardner M, Gray C, Abdelwahab A, Cox J. Use of administrative data to monitor cardiac implantable electronic device complications. Can J Cardiol. 2019;35(1):100–3. https://doi.org/10.1016/j.cjca.2018.10.018.
Demir-Kavuk O, Kamada M, Akutsu T, Knapp EW. Prediction using step-wise L1, L2 regularization and feature selection for small data sets with large number of features. BMC Bioinform. 2011;12:412. https://doi.org/10.1186/1471-2105-12-412.
Bewick V, Cheek L, Ball J. Statistics review 13: receiver operating characteristic curves. Crit Care. 2004;8(6):508–12. https://doi.org/10.1186/cc3000.
Rennert-May E, Manns B, Smith S, et al. Validity of administrative data in identifying complex surgical site infections from a population-based cohort after primary hip and knee arthroplasty in Alberta, Canada. Am J Infect Control. 2018;46(10):1123–6. https://doi.org/10.1016/j.ajic.2018.03.018.
Kandel CE, Jenkinson R, Widdifield J, et al. Identification of prosthetic hip and knee joint infections using administrative databases-a validation study. Infect Control Hosp Epidemiol. 2021;42(3):325–30. https://doi.org/10.1017/ice.2020.449.
Stevenson KB, Khan Y, Dickman J, et al. Administrative coding data, compared with CDC/NHSN criteria, are poor indicators of health care-associated infections. Am J Infect Control. 2008;36(3):155–64. https://doi.org/10.1016/j.ajic.2008.01.004.
Jetté N, Quan H, Hemmelgarn B, et al. The development, evolution, and modifications of ICD-10: challenges to the international comparability of morbidity data. Med Care. 2010;48(12):1105–10. https://doi.org/10.1097/MLR.0b013e3181ef9d3e.
Mull HJ, Stolzmann KL, Shin MH, Kalver E, Schweizer ML, Branch-Elliman W. Novel method to flag cardiac implantable device infections by integrating text mining with structured data in the Veterans Health Administration’s Electronic Medical Record. JAMA Netw Open. 2020;3(9):e2012264. https://doi.org/10.1001/jamanetworkopen.2020.12264.
Luz CF, Vollmer M, Decruyenaere J, Nijsten MW, Glasner C, Sinha B. Machine learning in infection management using routine electronic health records: tools, techniques, and reporting of future technologies. Clin Microbiol Infect. 2020;26(10):1291–9. https://doi.org/10.1016/j.cmi.2020.02.003.
Verberk JDM, Aghdassi SJS, Abbas M, et al. Automated surveillance systems for healthcare-associated infections: results from a European survey and experiences from real-life utilization. J Hosp Infect. 2022;122:35–43. https://doi.org/10.1016/j.jhin.2021.12.021.
Acknowledgements
We would like to acknowledge Alberta Health Services Analytics for their support in providing data, and IPC Calgary zone.
Funding
The authors received third party funding to support this work from the MSI Foundation. The MSI Foundation had no role in the design of the study or any of the work done for this study.
Author information
Authors and Affiliations
Contributions
The original project was conceived by authors ER, DC and JL. Authors KC, DC, and ER completed data collection and validation. All statistical analyses were completed by author MM. ER and MM completed the manuscript with input and revision from authors DC, SS, DE, OL, JL, KC and KB. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
This study was approved by the University of Calgary Health Research Ethics Board (REB20-2186).
Consent for Publication
Not applicable.
Competing Interests
The authors declare they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Additional file 1
. CCI Codes representing CIED implantation.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Rennert-May, E., Leal, J., MacDonald, M.K. et al. Validating administrative data to identify complex surgical site infections following cardiac implantable electronic device implantation: a comparison of traditional methods and machine learning. Antimicrob Resist Infect Control 11, 138 (2022). https://doi.org/10.1186/s13756-022-01174-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s13756-022-01174-z