Abstract
In this paper we develop a stochastic mathematical model of cholera disease dynamics by considering direct contact transmission pathway. The model considers four compartments, namely susceptible humans, infectious humans, treated humans, and recovered humans. Firstly, we develop a deterministic mathematical model of cholera. Since the deterministic model does not consider the randomness process or environmental factors, we converted it to a stochastic model. Then, for both types of models, the qualitative behaviors, such as the invariant region, the existence of a positive invariant solution, the two equilibrium points (disease-free and endemic equilibrium), and their stabilities (local as well as global stability) of the model are studied. Moreover, the basic reproduction numbers are obtained for both models and compared. From the comparison, we obtained that the basic reproduction number of the stochastic model is much smaller than that of the deterministic one, which means that the stochastic approach is more realistic. Finally, we performed sensitivity analysis and numerical simulations. The numerical simulation results show that reducing contact rate, improving treatment rate, and environmental sanitation are the most crucial activities to eradicate cholera disease from the community.
Similar content being viewed by others
1 Introduction
According to WHO [15] the occurrence of an infectious disease causes death of thousands of individuals in a population. It is vital that tolerable attention is paid to prevent the spread of such an infectious disease using an effective controlling mechanism. Various infectious diseases are a result of poor hygienic conditions, contact between an infectious person and a susceptible person. Different infectious diseases, like common cold and tuberculosis, are air-borne, whereas some are water-borne like acute water diarrhea (AWD) or cholera (Ochoche [12]). From this, cholera is an infection of small intestine common in developing countries like Asia, Africa, and South and Central America due to poor sanitation and using uncleaned water (Beryl et al. [2]). Cholera bacteria shed in their stool for seven to fourteen days, they can infect others through contaminated food or water (Obeng [11]). Cholera is an acute diarrheal illness caused by the gram-negative bacteria Vibrio cholerae which lives in an aquatic environment (Cui et al. [5]).
The symptom of cholera includes extreme vomiting, dry mouth, irregular heartbeat, painless watery stools, little or no urine output, and low blood pleasure (Fatima et al. [7]). It can be transmitted either by direct and indirect transmission pathways (Fakai [6]). Human-to-human (direct) way of cholera transmission is from the infected individual to the other individuals (touching, biting, and sexual intercourse). Whereas indirect (environment-to-human) way of transmission of cholera is through ingesting vibrio cholera bacteria from contaminated foods and waters (Wang and Modnak [14]). In this paper we concentrate only on the direct transmission path way by incorporating the stochastic nature of the disease.
Mathematical modeling has been an important tool in analyzing the spread and control of infectious diseases and also in making decision as regards the intervention mechanisms for the control of disease (Adewale et al. [1]). Mathematical epidemiology contributed to the understanding of the behavior of infectious diseases, its impacts and possible future predictions about its spreading. Mathematical models are used in comparing, planning, implementing, evaluating, and optimizing various detection, prevention, therapy, and control programs (Bubniakova [3]). Several mathematical models on cholera were developed by different authors. The study done by Codeco [4] focused mainly on endemicity of cholera and suggested two controlling mechanisms. The other study done by Codeco [4] proposed an SIR-B deterministic model by adding an environmental component into the regular SIR model. The study done by Fatima et al. [7] in Nigeria also proposed a deterministic mathematical model for the control of cholera. Additionally, a lot of authors like Fakai [6], Adewale et al. [1], Beryl et al. [2], Javidi and Ahmad [8], and others developed mathematical model of cholera to explore the transmission dynamics and controlling strategies. But none of them consider some stochastic environmental factors that can cause cholera outbreaks like, water, rain fall, air temperature, etc. In this paper we consider environmental factors and develop a stochastic model of cholera dynamics.
The rest of the paper is organized as follows: In Sect. 2, the cholera model is described and formulated in deterministic as well as stochastic approach. In Sect. 3, qualitative analysis and sensitivity analysis of the model are discussed. In Sect. 4, we use MATLAB software to investigate numerical simulation results. Finally, our discussions and conclusions are presented in Sect. 5.
2 Model formulation and description
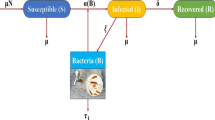
The model considers a total human population size (\(P(t)\)) and is divided into four compartments, namely susceptible represented by \(S(t)\), infected (\(I(t)\)), treated (\(T(t)\)), and recovered (\(R(t)\)) classes. Susceptible individuals (\(S(t)\)) are those individuals that cannot be infected, but can get infection sometime in the future, infected individuals (\(I(t)\)) are individuals who have developed the symptom of cholera and are able to transmit the disease, treatment class (\(T(t)\)) includes those individuals that get treatment at a time t for \(t > 0\) after they have been infected with cholera, and recovered compartment (\(R(t)\)) includes those individuals that have recovered from cholera disease and got temporary immunity.
Population in the susceptible compartment will be increased from the recovered compartment with a rate of δ by losing temporary immunity and also with a recruitment rate of π. However, its number decreases by the natural causing death rate of μ and also moving to the infected compartment with the rate of α. Population in the infected compartment will be increased by the contact rate of α, and also its number decreases by the natural causing death rate of μ, cholera causing death rate τ, and moving to the treatment compartment with the treatment rate of σ. Population in the treatment compartment increases from the infected compartment with the treatment rate of σ and decreases with the recovery rate of γ and the natural causing death rate of μ. Population in the recovered compartment also increases by the recovery rate of γ, but its number will decrease by the natural causing death rate of μ and by losing their immunity at the rate of δ.
The model is guided by the following assumptions: the size of homan population is constant, the birth rate and death rate are not equal, all parameters are nonnegative, all individuals are susceptible in the community, therapeutic treatment is applied to the infectious individuals, the treated individuals (individuals that are on treatment) do not transmit cholera disease to the susceptible human population, on recovery there is temporary immunity and there is the disease induced death (disease causing death). By the above descriptions and assumptions, our model is expressed diagrammatically in Fig. 1.
From the description and flow diagram of Fig. 1, the following deterministic model equation is obtained in equation (2):
with the initial condition \(S(0)= S_{0}\), \(I(0)= I_{0}\), \(T(0)=T_{0}\), \(R(0)=R_{0}\).
Since the above deterministic model in equation (2) does not consider stochastic environmental factors and lacks realistic conditions, we extended it to a stochastic model.
To extend, we introduce Brownian motion (\(B_{i}(t)\)) and the intensity of stochastic environmental factors (\(\beta _{i}\)) on equation (1) and multiply it by dt. Then we obtain the following stochastic model of cholera dynamics in equation (3):
where \(\beta _{1}, \beta _{2}, \beta _{3}, \beta _{4} \geq 0\) denotes the intensity of Brownian motion and \(B_{1}\), \(B_{2}\), \(B_{3}\), \(B_{4}\) are independent Brownian motions.
3 Qualitative analysis
In this section, some basic qualitative behaviors of the model, including the invariant region, positivity of the solution, disease-free equilibrium point, basic reproduction numbers, local and global stabilities of disease-free equilibrium, endemic equilibrium point, stability of endemic equilibrium, and sensitivity analysis, are discussed.
3.1 Invariant region
In this subsection we obtain invariant regions of model equations (1) and (2).
3.1.1 Invariant region for deterministic model
To get invariant region, we consider the total population at a time t given by \(P(t) = S(t) + I(t) + T(t) + R(t)\). Then differentiating \(P(t)\) with respect to time t on both sides, we get
Substituting \(\frac{dS(t)}{dt}\), \(\frac{dI(t)}{dt}\), \(\frac{dT(t)}{dt}\), and \(\frac{dR(t)}{dt}\) from equation (1) into equation (3), we get
If there is no infectious individual due to cholera disease, which means (\(\tau =0\), \(g=0\)), then equation (4) becomes
By the separation of variables of differential in equality (5), we obtain
where C and D are constant.
After solving and evaluating equation (6) as t goes to ∞, we have \(\lim_{t}\to \infty P(t)=\frac{\pi }{\mu }\), which implies that \(0 \leq P(t) \leq \frac{\pi }{\mu }\).
Therefore, the model is biologically meaningful and bounded in the domain
3.1.2 Invariant region for stochastic model
Theorem 1
The region Ω is almost surely positive invariant of our stochastic model equation (2).
Proof
Let us take \(K_{0}\) as a large integer so that if \((S_{0}, I_{0}, T_{0}, R_{0}) \in R^{4}_{+}\), then every component of \((S_{0}, I_{0}, T_{0}, R_{0})\) lies in the interval \([\frac{1}{k_{0}}, 1]\). For each integer \(K \geq k_{0}\), we can define stopping time:
We want to prove \(P(\tau = \infty )\), which is \(P (\tau < M) = 0\) for \(M > 0\), so that it allows us to show \(\lim_{t \to \infty }\sup P(\tau _{k} < 0) = 0\).
Now consider a Lyapunov function V technically:
Then, by using Ito’s formula, for \(M > 0\), \(t \in [0, M \wedge \tau _{k} ]\) to \(X(t) = (S(t), I(t), T(t), R(t))\), we obtain that
Hence, we have
Let \(a = \pi + \delta R(t) -\alpha S(t) I(t) - \mu S(t)\), \(b = \beta _{1} S(t)\), \(d = \alpha S(t) I(t) - (\mu + d + \sigma )I(t)\), and \(f = \beta S(t) I(t)\), then equation (7) becomes
Now, rearranging equation (8), we get
Let \(LV = [- \frac{ \pi }{S(t)} - \frac{\delta R(t)}{S(t)} - \sigma \frac{I(t)}{T(t)} - \gamma \frac{T(t)}{R(t)} + \alpha I(t) - \alpha S(t) + 4\mu + \tau + \sigma + \delta + \gamma + g + \beta _{1}^{2} + \beta _{2}^{2} + \beta _{3}^{2} + \beta _{4}^{2} ]\).
Then \(dV(t) = LVdt - [\beta _{1} \,dB_{1}(t) + \beta _{2} \,dB_{2}(t) + \beta _{3} \,dB_{3}(t) + \beta _{4} \,dB_{4}(t) ] \).
We have \(LV = \alpha I(t) + 4\mu + \tau + \sigma + \delta + \gamma + g + \beta _{1}^{2} + \beta _{2}^{2} + \beta _{3}^{2} + \beta _{4}^{2} = C \), and we get \(dV(t) \leq C \,dt - [\beta _{1} \,dB_{1}(t) + \beta _{2} \,dB_{2}(t) + \beta _{3} \,dB_{3}(t) + \beta _{4} \,dB_{4}(t) ] \).
Then, by integrating both sides from \(0 \to \tau _{k} \wedge M \) implies that
where \(\tau _{k} \wedge M = \min \{\tau _{n}, t\} \). Taking expectation of the above inequalities yields
Since \(V(X(\tau _{k} \wedge M)) > 0\), then
Now, for \(\tau _{k}\), there are some components of \(X(\tau _{k})\), say \(S(\tau _{k})\), such that \(0 < S(\tau _{k}) \leq \frac{1}{k} < 1\).
Therefore, \(V(X(\tau _{k})) \geq - \ln (\frac{1}{k}) \), this comes from
Thus, from equation (13) and the above equation, we get
From (12)–(14) it follows that
Letting \(k \to \infty \) and by taking limit sup to equation (16), for all \(M > 0\), we obtain that
Therefore, \(\lim_{t \to \infty } \sup P(\tau _{k} < M) = 0 \), this completes the proof of the theorem. □
3.2 Positivity of the solutions
In this subsection, we obtain nonnegative solutions for future time with their respective initial conditions.
Theorem 2
If our initial values are \(S(0) \geq 0, I(0) \geq 0, T(0) \geq 0, R(0) \geq 0 \) then the solutions of \((S, I, T, R)\) are positive for all \(t \geq 0 \).
Proof
To prove Theorem 2, let us take the first equation of model (1):
Then, after solving equation (17) by using the separation of variables and applying the initial conditions, we get
After some steps, we get \(S(t) \geq S(0) e^{-(\alpha I + \mu ) t}\).
Therefore, \(S(t) \geq S(0) e^{-(\alpha I + \mu ) t} \geq 0\).
Next, let us take the second equation of model (1):
Then, solving equation (19) by using the separation of variables and applying the initial condition, we obtain
As \(t \to 0\), then \(e^{-(\mu + d + \theta + \tau )t} \to 1\), this implies \(I(0) = D\).
Therefore, \(I(t) \geq I(0) e^{-(\mu + d + \theta + \tau )t} \geq 0\).
Similarly, we took the third and fourth equations of model (1).
Now, taking similar steps to the above gives
This completes the proof of the theorem. □
Therefore, our model solutions of equations (1) and (2) are positive for future time.
3.3 Disease-free equilibrium point
To obtain a disease-free equilibrium point, set model equation (1) to zero and there are no infectious individuals in the population, which means \(I = 0\), \(T = 0\), \(R = 0\).
Then the system of equations (1) and (2) is simplified, which gives
Since \(I = T = R = 0\), then \(\pi - \mu S = 0\), which implies \(S = \frac{\pi }{\mu }\).
Therefore, our disease-free equilibrium point is \(E_{0} = (\frac{\pi }{\mu }, 0, 0, 0 )\).
3.4 Basic reproduction number
To determine the basic reproduction number \((R_{0})\) of equations (1) and (2), we use the next generation matrix method by Beryl et al. [2]. We have two basic reproduction numbers (deterministic and stochastic).
3.4.1 Basic reproduction number for deterministic model
In view of that, first let us take the newly infectious class
Now, by the principle of next generation matrix approach, we obtain
The next step is obtaining the Jacobian matrix of f and v with respect to I at \(E_{0}= (\frac{\pi }{\mu }, 0, 0, 0)\). Let F and V be the Jacobian matrix of f and v, respectively, then
Then, evaluating F and V at a disease-free equilibrium point \(( E_{0} = (\frac{\pi }{\mu }, 0, 0, 0 ))\), we obtain
Then \(FV^{-1}\) becomes
The eigenvalue of \(FV^{-1}\) can be obtained by
Here, by the next generation matrix principle, the largest eigenvalue is the basic reproduction number.
Therefore, our basic reproduction number for the deterministic model is
3.4.2 Basic reproduction number for stochastic model
By taking the infected class of (2), we obtain the basic reproductive number of stochastic approach:
Using twice differentiable function of Ito’s formula, we can derive our stochastic basic reproduction number. Let us take \(f(t, I(t)) = \ln (I(t))\), then its Taylor series expression becomes
where \(\frac{\partial f}{\partial t} = 0\), \(\frac{\partial f}{\partial I(t)} = \frac{1}{I(t)}\), \(\frac{\partial ^{2} f}{\partial I^{2}(t)} = -\frac{1}{I^{2}(t)}\), \(\frac{\partial ^{2} f}{\partial t \partial I} = 0\), \(\frac{\partial ^{2} f}{\partial t^{2}} = 0\).
Then equation (26) becomes
Let \(a = \alpha S(t) I(t) - (\mu + \tau + \sigma )I(t)\) and \(b = \beta _{2} I(t)\), then
By applying the chain rule, we get
Then
By using the next generation matrix, let
Now f and v at DFEP become
Then \(fv^{-1} = \frac{\alpha \frac{\pi }{\mu } - \frac{1}{2}\beta _{2}{^{2}}}{(\mu + \tau + \sigma )}\)
where \(R_{0}^{D} = \frac{\alpha \pi }{\mu (\mu + \tau + \sigma )}\).
Therefore, our basic reproduction number for stochastic model is
From equation (28) we see that \(R_{0}^{S} < R_{0}^{D}\), which means the stochastic approach is more realistic than the deterministic approach.
3.5 Local stability of disease-free equilibrium
In this subsection we show the local stability of disease-free equilibrium in the case of deterministic as well as stochastic models.
3.5.1 Local stability of disease-free equilibrium in the case of deterministic model
Theorem 3
The disease-free equilibrium point of system (1) is locally asymptotically stable if and only if \(R_{0}^{D} < 1\), unstable if \(R_{0}^{D} > 1\).
Proof
To prove Theorem 3, first we construct a Jacobian matrix of model equation (1).
Then the Jacobian matrix is obtained by
where
Then the Jacobian matrix is given by equation (30).
Then the Jacobian matrix evaluated at \(E_{0}\) becomes
From equation (31) the eigenvalues are evaluated as follows:
The characteristic polynomial of equation (32) becomes
By factorizing the characteristic polynomial equation, eigenvalues are as follows:
Since \(\lambda _{1} < 0\), \(\lambda _{2} < 0\), and \(\lambda _{3} < 0\), but we do not know the sign of \(\lambda _{4}\). However, to be stable all the eigenvalues have to be negative and \(\lambda _{4}\) has to be less than zero. This means
We know that from equation (24) we have \(R_{0}^{D} = \frac{\alpha \pi }{\mu (\mu + \tau + \sigma )}\).
Hence, equation (34) becomes
Therefore, our disease-free equilibrium point is locally asymptotically stable if and only if \(R_{0}^{D} < 1\). □
3.5.2 Local stability of disease-free equilibrium in the case of stochastic model
Theorem 4
If \(R_{0}^{S} < 1\), then for any initial values of \((S_{0}, I_{0}, T_{0}, R_{0})\in R_{+}^{3}\), I(t) obeys \(\lim_{t \to \infty }\sup \frac{1}{t}\ln I(t) \leq (\mu + \tau + \sigma )(R_{0}^{S} - 1)< 0\).
Proof
Let us take \(F(t, I(t)) = \ln I(t)\), by Ito’s formula
Integrating equation (35) on both sides, we have
Then evaluating equation (36) at \(E_{0}\), we obtain
where a martingale \(G(t) = \int _{0}^{t}\beta _{2} \,dB(t)\).
By the strong law of large numbers of martingale, we have \(\lim_{t \to \infty } \sup \frac{G(t)}{t}=0\) almost surely.
Then divide both sides of equation (37) by t. By letting \(t \to \infty \), we get
Taking \(\lim_{t \to \infty } \sup \) on both sides of equation (38), we obtain
Obviously \((\mu + \tau + \sigma )> 0\), therefore \(R_{0}^{S} - 1 \) less than zero:
Therefore, our disease-free equilibrium point is locally asymptotically stable if and only if \(R_{0}^{S} < 1\). □
3.6 Global stability of disease-free equilibrium
Theorem 5
If \(R_{0}^{D} < 1\), then \(E_{0}\) is globally asymptotically stable in Ω.
Proof
Now let us construct a Lyapunov function technically as follows:
Differentiating equation (41), we obtain
Evaluating equation (42) at \(S = S_{0} = \frac{\pi }{\mu }\), we obtain
So that \(\frac{dL}{dt} \leq 0\) if \(R_{0}^{D} - 1 < 0\).
Obviously, we know that \([\pi + \alpha \sigma + \frac{\pi }{\mu } ](\mu + \tau + \sigma ) > 0\),which means to be \(\frac{dL}{dt} \leq 0\), must be \(R_{0}^{D} - 1 < 0\).
Therefore, by LaSalle’s invariance principle, every solution of model equation (1) with a given initial conditions in Ω approaches to \(E_{0}\) at t and goes to infinity whenever \(R_{0}^{D} \leq 0\).
Hence, disease-free equilibrium is globally asymptotically stable in the region Ω. □
3.7 Endemic equilibrium point
In this subsection, we obtain the equilibrium point at which no disease is present in the population.
The endemic equilibrium point of our model is denoted by \(E_{1} = (S^{*}, I^{*}, T^{*} R^{*})\). The endemic equilibrium can be obtained by equating all the expressions of model equation (1) to zero:
Let us take the second equation of system (44), we get
Then, solving for \(S^{*}\), we have
From the third equation in (44), we solve for \(I^{*}\):
Now, solving for \(R^{*}\) and \(T^{*}\) by substituting equation (45) and (46) into the first equation of (33), we obtain
Then, by taking simultaneously the fourth equation of (44) and equation (47), we get
Finally, from equation (48), we obtain
Then substituting equation (49) into the second equation of (48), we have
Now substituting equation (50) into equations (46) and (49), we obtain
Therefore, endemic equilibrium point of the model is
3.8 Stability of endemic equilibrium point
In this subsection, we discuss the stability of endemic equilibrium \(E_{0}\) by considering a Lyapunov function L technically as follows:
Now, differentiating both sides of equation (51) with respect to t, we get
Then, simplifying equation (52), we obtain the following equation:
Now, by simplifying and rearranging (the positive part on one side and the negative part on the other side) equation (53), we have
Now, replacing U for the positive terms and V for the negative terms of equation (54), we obtain
and
Then, equation (54) is replaced by U and V, and we have
If \(U < V\), then \(\frac{dL}{dt} \leq 0\) and \(\frac{dL}{dt} = 0\) if and only if our equilibrium points are \((S = S^{*}, I = I^{*}, T=T^{*}, R=R^{*})\).
From this, we observe that \(E_{0}\) is the largest set of compact invariant singletons in \(\{(S = S^{*}, I = I^{*}, T=T^{*}, R=R^{*}) \in \Omega : \frac{dL}{dt} = 0\}\).
Therefore, \(E_{0}\) (endemic equilibrium) is globally asymptotically stable in Ω if \(U < V\).
3.9 Sensitivity analysis
In this subsection, we obtain sensitivity analysis of the model to determine the effect of each parameter on basic reproduction number \((R_{0} )\). To perform the sensitivity analysis of \((R_{0} )\), we use the following formula:
where \(n_{i}\) is the parameters of \(R_{0}\) (Tilahun [13]).
For \(n = \pi \):
For \(n = \alpha \):
For \(n = \mu \):
For \(n = \tau \):
Finally, for \(n = \sigma \):
3.10 Interpretation of sensitivity analysis
The interpretation of the sensitivity indices given in Table 1 is as follows. Those parameters that have positive sensitivity indices \((\pi , \alpha )\) have a big contribution to the expansion of cholera disease in the human population if their values are increased by keeping the rest of parameters constant. And those parameters that have negative sensitivity indices \((\mu , \tau, \sigma )\) show a great effect in bringing down the disease from the population if their values are decreased by keeping the rest of parameters constant. Due to the reason that \(R_{0}\) (basic reproductive number) increases as its parameter value increases, the average number of secondary infection increases in the population; and \(R_{0}\) decreases as its parameter value decreases, which means that the average number of secondary infection decreases in the human population.
4 Numerical results and discussion
In this section, some numerical results are presented to demonstrate how change in parameters of the model influences various performance measures of the system. Models (1) and (2) were simulated using MAT LAB software to obtain the following graphs. Additionally, we used \(S(0)=12\), \(I(0)=8\), \(T(0)=5\), and \(R(0) = 3\) as initial values, where the initial values are estimated. In Table 2, the listed parameter values are used for numerical simulation purposes.
4.1 Comparison of deterministic and stochastic trends of the model
In Fig. 2, the numerical results of the comparison of deterministic and stochastic trends of the model in the community are displayed by keeping all parameters unchanged. By taking the whole compartments of the model for deterministic figure and by adding a white noise to a deterministic equation, we illustrate a stochastic figure. Now, from this figure, we see that running the simulation results for the stochastic model is slower than for the deterministic model, this is due to environmental factors. The amount of infectious population decreases and the number of individuals who get treatment increases after a certain point of time due to treating the infected people in the community with the rate \((\sigma )\) in the stochastic case as well as in the deterministic one. Moreover, the stochastic behavior of the curves shows the real life behavior compared to the deterministic approach. We conclude from this figure that the stochastic solution is closer to the real solution of the cholera model than the deterministic approach. So, using the stochastic model is better than the deterministic one because the stochastic model considers a white noise or stochastic environmental factors.
4.2 Effect of contact rate on cholera infected population
In this subsection, we obtain the numerical simulation results of the impact of contact rate (α) on the number of infectious individuals \(I(t)\). The simulation results that are displayed in Fig. 3 are obtained by different values of contact rate (α) from \(\alpha = 0.001\) to \(\alpha = 0.017\) and keeping the rest of parameters constant. From Fig. 3, we see that as the contact rate increases the number of infectious individuals in the community increases, which means if the susceptible individuals contact with infected individuals either by shaking hands or eating foods with the same materials this would increase the infectious population. In addition, we observe that the stochastic curves show a sound wave property due to environmental factors or random behavior of the disease, but in the deterministic model the curve looks like a smooth line, which implies it does not consider any factors in the environment for cholera disease. Hence, the disease persists in the community when there is an increase in the contact rate, even though the rest of the parameters are kept constant. Due to this, healthy workers must control this parameter.
4.3 Effect of treatment rate on cholera infected population
We investigated the effects of treatment rate on the number of infectious population. In Fig. 4, the experimental results are obtained by taking different values of treatment rate σ and by keeping the rest of the parameters constant. The simulation results in Fig. 4 reveal that increasing the values of treatment rate leads to increase in the infectious individuals. We also observe in Fig. 4 that in the case of deterministic approach as well as stochastic approach the decrements of infectious population are obtained as the treatment rate in the population increases. Therefore, increasing the treatment rate σ plays a vital role in the reduction of cholera disease dynamics in the community.
4.4 Effects of recovery rate on recovered individuals
In this subsection, we use the impact of recovery rate γ on the size of recovered individuals to display Fig. 5 by varying γ (recovery rate) from \(\gamma = 0.03\) to \(\gamma = 0.7\) and keeping the rest of the parameter values fixed. In the case of deterministic approach, Fig. 5 shows that the graph goes up smoothly as the recovery rate γ increases, which means that if the infectious individuals get treatment and recover more with the recovery rate γ, the number of recovered individuals increases in the community. Also, in the stochastic case, it displays that the number of recovered individuals increases as the value of recovery rate increases, with the graph going up and down. These ups and downs show the random behavior of the model. From this, we conclude that the recovered population becomes bigger by increasing the recovery rate (γ), and the stochastic approach is more advisable than the deterministic one because the stochastic approach considers environmental white noise.
5 Discussions and conclusions
In Sect. 2, we proposed and described briefly an SITRS cholera disease dynamics model with two approaches: deterministic and stochastic. In Sect. 3, we analyzed the qualitative behavior of the model by obtaining the invariant region, existence of a positive invariant solution set, equilibrium points, the basic reproduction number for deterministic as well as stochastic model; local stability of disease-free equilibrium for both models was checked; the global stability, which is obtained by a Lyapunov function, sensitivity analysis, and their interpretation were studied. In Sect. 3, the two reproduction numbers were obtained by using the next generation matrix method and by using twice differentiable Ito’s formula for stochastic reproduction number. Out of these two reproduction numbers the stochastic reproduction number is much smaller than the deterministic one. This implies that the stochastic approach is more realistic (close to the accurate solution) than the deterministic approach, because the stochastic model considers stochastic environmental factors or takes the randomness process. In Sect. 4, the numerical simulation results were discussed and analyzed by using MATLAB software by comparing the deterministic approach with the stochastic one. From our simulation results in Sect. 4, we conclude that increasing cholera treatment rate has a big contribution to eliminating cholera disease from the community, and increasing the recovery rate contributes to the reduction of the infection. Other results that were obtained in this section are as follows: decreasing the contact rate has a big influence on controlling cholera disease dynamics in the community. Therefore, for healthy workers as well as policy makers we recommend improvement of the treatment: by decreasing the contact rate and by increasing the recovery rate we can eradicate the disease from the community.
Availability of data and materials
The data we used for this research is from respective published articles that are cited.
References
Adewal, S.O., Adeniran, G.A., Olopade, I.A., Ajao, S.O., Mohammed, I.T.: Mathematical and sensitivity analysis of the dynamical spread of cholera. Int. J. Innov. Appl. Stud. 19(1), 46–54 (2017)
Beryl, M.O., George, L.O., Fredrick, N.O.: Mathematical analysis of a cholera transmission model incorporating media coverage. Int. J. Pure Appl. Math. 111(2), 219–231 (2016)
Bubniakova, L.: The mathematics of infectious diseases (2007)
Codeco, C.T.: Endemic and epidemic dynamics of cholera: the role of the aquatic reservoir. BMC Infect. Dis. 1(1), 1 (2001)
Cui, J.A., Wu, Z., Zhou, X.: Mathematical analysis of a cholera model with vaccination. J. Appl. Math. 2014, Article ID 324767 (2014)
Fakai, S.A., Ibrahim, M.O., Siddiqui, A.M.: A deterministic mathematical model on cholera dynamics and some control strategies. Int. J. Sci. Eng. Technol. 8(3), 1115–1118 (2014)
Fatima, S., Krishnarajah, I., Jaffar, M.Z.A.M., Adam, M.B.: A mathematical model for the control of cholera in Nigeria. Res. J. Environ. Earth Sci. 6(6), 321–325 (2014)
Javidi, M., Ahmad, B.: A study of a fractional-order cholera model. Appl. Math. Inf. Sci. 8(5), 2195–2206 (2014)
Kamuhanda, E.A., Shaibu, O., Mary, W.: Mathematical modelling and analysis of the dynamics of cholera. Glob. J. Pure Appl. Math. 14(9), 1259–1275 (2018)
Lemos-Paiao, A.P., Silva, C.J., Torres, D.F.M.: A cholera mathematical model with vaccination and the biggest outbreak of world’s history. arXiv preprint (2013). arXiv:1810.05823
Obeng, C.: Deterministic SIRB cholera model with logistic approach in Dormaa-Ahenkro, Ghana. Doctoral dissertation (2015)
Ochoche, J.M.: A mathematical model for the transmission dynamics of cholera with control strategy. Int. J. Sci. Technol. 2(11), 212–217 (2013)
Tilahun, G.T.: Mathematical Model for Co-Infection of Pneumonia and Typhoid Fever Disease with Optimal Control. Doctoral dissertation, JKUAT (2018)
Wang, J., Modnak, C.: Modeling cholera dynamics with controls. Can. Appl. Math. Q. 19(3), 255–273 (2011)
WHO: Vaccine-preventable diseases surveillance standards (2019)
Acknowledgements
The authors would like to express their deepest appreciation to Haramaya University for the financial support. WAW also acknowledges the financial support from the DST/NRF SARChI Chair in Mathematical Models and Methods in Biosciences and Bioengineering at the University of Pretoria, grant No. 82770. Moreover, all the authors are grateful to the anonymous reviewers.
Funding
All costs are covered by Haramaya University, Ethiopia.
Author information
Authors and Affiliations
Contributions
All authors contributed equally starting from model development to the analysis of the results. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing interests.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Tilahun, G.T., Woldegerima, W.A. & Wondifraw, A. Stochastic and deterministic mathematical model of cholera disease dynamics with direct transmission. Adv Differ Equ 2020, 670 (2020). https://doi.org/10.1186/s13662-020-03130-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s13662-020-03130-w