Abstract
Background
Pre-weaning diarrhea (PWD) is a syndrome affecting farm-raised neonatal mink kits. Apart from diarrhea it causes greasy skin exudation, dehydration, and distressed behavior and can ultimately lead to death. No specific causative agents have been identified and the syndrome is regarded as multifactorial. The aim of the present study was to investigate a possible inflammatory state in mink kits with PWD, as indicated by raised serum concentrations of the acute phase protein serum amyloid A (SAA) and by changes in intestinal pathomorphology and intestinal contents of bacteria. Samples collected from 20 diarrheic mink kits with PWD and 20 age-matched non-diarrheic control mink kits from two commercial Danish farms during the pre-weaning period (April–May) in 2016 were analyzed.
Results
Concentrations of SAA in serum samples from mink kits with PWD were significantly higher (up to 1000-fold) compared to non-diarrheic control mink kits. Significant features of enterocytic vacuolization, atrophy and fusion of villi in jejunum and mucosal atrophy of the colon of kits with PWD were found. Moreover, attachment of coccoid bacteria to enterocytes was more often found in kits suffering from PWD, while intra-cytoplasmic eosinophil bodies were more frequently observed in control kits. Cellular infiltrations with mononuclear and neutrophil leukocytes were not associated with disease status. Bacteria from the Staphylococcus intermedius group, such as Staphylococcus delphini, were more frequently cultivated from control mink kits, whereas Enterococcus spp. dominated in mink kits with PWD. Escherichia coli was cultivated from both control and mink kits with PWD, but with a higher frequency from mink kits with PWD.
Conclusion
A significant increase in circulating concentrations of SAA was found in PWD affected mink kits from 6 to 23 days old compared to controls. The histopathological changes in PWD mink kits suggest that the type of diarrhea is secretory. Attachment of coccoid bacteria, therefore, might be responsible for an enterotoxic effect causing a loss of balance in movements of ions and water leading to the vacuolization and swelling of the enterocytes. The slight to moderate infiltrations of neutrophils irrespectively of diarrheic status and the attachment of coccoid bacteria to enterocytes are comparable to observations found in piglets suffering from New Neonatal Porcine Diarrhea Syndrome. Mechanisms behind the correlation between increased SAA levels and the observed pathological intestinal features remain obscure.
Similar content being viewed by others
Background
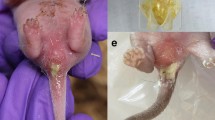
The pre-weaning diarrhea syndrome (PWD) in mink (Neovison vison) kits is a major cause of concern in the mink industry due to both economic losses and decreased animal welfare. It may affect more than 30% of the litters [1, 2] and has been observed in farm-raised neonatal mink for several decades worldwide [3]. Mink kits affected by PWD display diarrhea with concomitant excessive secretions from the cervical apocrine glands, and exudate on the skin surface, the tail, and the claws. Moreover, dehydration may ultimately lead to the death of the affected kits [4, 5]. The onset of clinical signs generally occurs in the whole litter during the pre-weaning period, at 5–20 days of age, with a morbidity rate varying from 0 to more than 30% of the litters, and a mortality of typically one or two kits per litter [6, own findings, 2017]. The PWD syndrome is considered multifactorial, and there is a lack of consistency in isolated bacteria and viruses in kits with PWD. Studies have aimed to define the causality of the syndrome and mink astrovirus (MiAstV) isolated from mink kits with PWD indicate involvement in the syndrome [7,8,9,10,11]. However, all of the proposed putative pathogens including also Campylobacter jejuni, rota-, calici-, and mink coronavirus, Escherichia coli, and Staphylococcus delphini have also been isolated from clinically healthy kits, so their role in PWD remains elusive [10,11,12,13]. Apart from having a multifactorial infectious origin, other factors including management factors have been associated with an increased risk of developing PWD. For example, litters from 1-year old females and in females with low energy supply in the late gestation period are at increased risk of being affected by PWD [2, 14]. Moreover, the presence of dogs on the farm area as well as the size of the farm (total number of females) have also been associated with high morbidity of PWD [2]. Regarding the intestinal pathomorphology accompanying PWD, only a few studies have been published [15, 16]. Moreover, intestinal lesions in mink kits suffering from PWD have not been classified, according to the standard pathomorphological paradigm, as either non-inflammatory/secretory, inflammatory or invasive [17]. A possible biomarker to assess infection or inflammation in mink kits suffering from PWD is serum amyloid A (SAA). SAA is an acute phase protein found in low concentrations in healthy animals and is released following inflammation, infection, or tissue injury in both mink [18,19,20] and many other species, including humans [21, 22]. It is synthesized predominantly by the liver in response to the cytokine interleukin 1, however, other organs such as the intestine, have also been shown to produce it [19]. The aim of the present study was to examine if the levels of SAA could be a biomarker for PWD in mink kits and to characterize and compare the intestinal pathomorphology, and the bacterial intestinal contents between healthy controls and mink kits suffering from PWD.
Methods
Animals
In total, 20 mink (Neovison vison) kits with PWD and 20 age-matched healthy kits (controls), between 6 and 23 days old, were obtained during the pre-weaning period (April–May) from two (A and B) commercial Danish mink farms with outbreaks of PWD. Sick mink kits were selected based on the liquid consistency of the feces and the following clinical manifestations: sticky cutaneous exudation, red swollen anus and perineal soiling (own findings, 2017). Mink female and their kits were housed in separate cages with conventional nest boxes, females were fed conventional feed and had unlimited access to water. Mink kits were euthanized with CO or CO2, bled, and the intestine was removed aseptically immediately thereafter.
Serum amyloid A ELISA
Un-stabilized blood samples from the kits were obtained at the time of euthanasia. Blood was allowed to clot and serum was obtained as the supernatant after centrifugation at 4000g for 15 min at 4 °C. Serum was stored at − 20 °C until analysis. A commercially available multispecies sandwich ELISA (Phase SAA assay, Tridelta Development Ltd., Kildare, Ireland, #TP 802) was used to quantify SAA concentrations in the serum samples. This assay is a quantitative sandwich ELISA using rat anti–human monoclonal antibodies [23]. Mink kit serum samples were diluted 500 times according to the manufacturer’s instructions for canine SAA. The lower limit of quantification was 6.25 μg/mL (canine SAA units) as given by the lowest concentration of the standard dilution series included on each plate. Readings below the lower limit of quantification were assigned the value 6.3 µg/mL. Data was transferred to GraphPad Prism version 7 (GraphPad Software, San Diego, California, USA, https://www.graphpad.com) for graphic representation and for statistical analysis. Significant difference in SAA concentrations in circulation between PWD kits and control kits were identified using the Mann–Whitney test in Prism. P values < 0.05 were considered significant.
Histology
Duodenal, mid-intestinal (jejunum) and aboral colon sections from each kit were fixed with needles on a piece of styrene foam and placed in 10% neutral buffered formalin in a CellStor Pot (CellPath, Newtown, Powys, UK). Two pieces from each of the three gut sections were embedded in paraffin and sections of 4–5 µm were prepared and stained with hematoxylin and eosin (HE). Selected sections were stained with Periodic acid–Schiff (PAS), Gram, and Warthin–Starry (WS). Each of the three sections was histologically examined and graded blindly according to the following manifestations (Table 1).
For each of the included animals a score for duodenum + jejunum and colon sections was established, respectively. Thus, for the dichotomous variables the summary score was “absent” (−), if absent in all gut sections and “present” (+), if present in any of the gut sections. For the assessment of vacuolization a cumulated score was established and categorized as absent (−) if the cumulated score was 0, or present (+) if the cumulated score was > 0. For the assessment of infiltration with neutrophils in the villi a cumulated score was established, and categorized based on the mean score as high or low i.e. ≥ 2/< 2 for duodenum + jejunum sections. Associations between disease status of the kit and each variable was tested with the SAS Enterprise Guide 7.1 using Chi square and where relevant Fisher’s exact test (expected < 5). A cumulated number of mitosis in enterocytes of duodenum and jejunum from PWD and control mink kits were tested with Students t-test. P values < 0.05 were considered significant.
Bacteriology
Post mortem, the intestine was removed from the abdomen as previously described [24]. The aboral part of the colon was aseptically opened with a sterile scalpel and swabbed for bacterial cultivation (Transport Swabs, VWR, Radnor, PA, USA). The total of 17 pooled swab control samples were generated by pooling swabs from two healthy kits from the same litter, on the same day, from 17 litters age-matched to the kits affected by PWD. The age-matching of the mink kits unfortunately resulted in some of the kits being pooled into the same tube. This left only 17 pooled samples, instead of 20, from kits without PWD. From the diarrheic kits, the samples were cultured individually. The swabs were stored at 5 °C until analysis by blinded cultivation. The swabs were inoculated on blood agar plates enriched with 5% bovine blood (blood agar base, Oxoid, Thermo Scientific, Watham, MA, USA) and incubated at 37 °C aerobically for 1–2 days. The two most frequently occurring bacterial colonies were then sub-cultivated before they were identified by Matrix-Assisted Laser Desorption Ionization Time-Of-Flight mass spectrometry (MALDI–TOF–MS) using a VITEK MS MALDI-TOF (BioMérieux, Marcy-l’Etoile, France) as previously described [25]. If two bacterial species were equally frequent next to a single more frequent species both were sub-cultivated, and hence three isolates were identified.
Results
Serum SAA concentrations
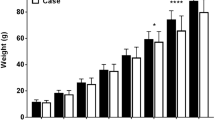
The difference in serum SAA concentration between PWD kits and control kits was highly statistically significant (Mann–Whitney test, P < 0.0001) (Fig. 1). Only 2/20 (10%) control mink kits had SAA concentrations higher than the lower limit of quantification of 6.3 µg/mL, while 17/20 (85%) kits with PWD had SAA concentrations higher than 6.3 µg/mL.
Serum concentration of serum amyloid A (SAA) protein in PWD and healthy control mink kits as determined by SAA ELISA. Median of the serum SAA concentrations with 95% confidence interval is depicted (n = 20). The Mann–Whitney test showed a statistically significant higher concentration of serum SAA in PWD kits than in control kits (****P < 0.0001)
Histology
The results of the histologic evaluations are presented in Table 2. Compared to normal kits (Fig. 2a) PWD mink kits expressed significant features of enterocytic hydropic vacuolization (PAS negative) in the jejunum and colon (Fig. 2b, c). Atrophy and fusion of villi in the small intestine and atrophy of the mucosa lining in the colon were also associated with presence of the disease, and were most prominent in jejunum and colon (Fig. 2d, e). The association between attachment of coccoid bacteria to enterocytes and disease status was highly significant in duodenum + jejunum and colon (P < 0.0001 and P = 0.001, respectively), and was equally present in all parts of the intestine (Fig. 2b). Selected Gram stained sections revealed that the coccoid bacteria were Gram positive. Rod-shaped bacteria were also significantly observed more frequently in intestines from PWD kits, but the attachment to villi was sporadic and in combination with coccoid bacteria. Intra-cytoplasmic eosinophilic bodies (PAS positively stained vacuoles) were significantly more frequent in control kits compared to PWD kits, and most often in jejunum (Fig. 2f). Infiltrations with mononuclear and neutrophil leucocytes were not associated with disease status. Moreover, the mitotic activity of enterocytes in the small intestine were significantly higher in PWD kits compared to control kits (P = 0.003).
Photomicrographs of representative examples of intestines from mink kits. a Jejunum from healthy mink kit. b Vacuolization on the tip of the villi (arrows) and pronounced attachment of coccoid bacteria (arrow heads) to the enterocytes in jejunum of a mink kit with PWD. c Severe vacuolization and hypertrophied enterocytes in the colon of a mink kits with PWD. d Colon from a healthy mink kit. e Atrophy of the mucosa of the colon from a mink kit with PWD. Inset: higher magnification of the mucosal atrophy. f Eosinophilic bodies in the enterocytes from jejunum in a healthy mink kit (arrows)
Bacteriology
The number of bacterial isolates obtained from healthy mink kits and kits with PWD is shown in Table 3. The dominating species cultivated from healthy control mink kits was from the Staphylococcus intermedius group, whereas Enterococcus spp. was the main species found in mink kits with PWD. On Farm B, E. coli was cultivated more frequently from mink kits with PWD (58.8% of the isolates) than from control kits (41.7% of isolates). On farm A, E. coli was not isolated from any of the control kits, but accounted for 28.6% of the isolates from the mink kits suffering from PWD.
Discussion
We report for the first time that PWD in mink kits between 6 and 23 days old is associated with a significant increase in circulating concentrations of SAA. Two of the control kits showed elevated concentrations of SAA in serum, however only one of them (1315 µg/mL; Fig. 1) showed signs of cholangitis, which could explain the elevated concentration of SAA (data not shown). SAA have been shown by Bruun et al. [20] to be elevated in mink after subcutaneous injection with lipopolysaccharide from E. coli. The gastrointestinal tract of mink is very short and the passage of feed through the entire gastrointestinal tract takes approximately 2–3 h [26]. Previous studies have shown the difficulty in culturing bacteria from the small intestine in mink [27] and therefore we decided to use swabs from the colon, where the density of bacteria is relatively high. Furthermore, the decision to only cultivate the bacteria aerobically was based on the fact that only aerobic bacteria have so far been isolated from PWD-affected mink, whereas anaerobic bacteria have not [11, 28, 29]. We isolated E. coli most frequently from kits suffering from PWD, which corresponds well with previous findings that E. coli is commonly isolated from PWD mink kits [11, 16, 30, 31]. A quantitative study of healthy mink kits showed that the intestinal counts of E. coli were highest when kits were around 4 weeks of age [30], but in the present study E. coli was isolated from younger kits suffering from PWD. However, E. coli has also been identified as part of the normal mink kit intestinal microflora [11, 16, 30, 32]. Moreover, analysis of virulence factors of E. coli has revealed that the population of E. coli in mink consists of several serogroups with no apparent association to outbreaks of PWD [11, 32]. In general, enterococci are not regarded as pathogenic in mink, but Enterococcus hirae has been associated with diarrhea in 4–7 week old mink kits submitted for diagnostic testing (Chriél, pers. comm., 2017), as well as in other neonatal animals such as suckling rats, kittens and piglets [33,34,35]. Gut sections from PWD mink kits showed clear disease associated changes, including vacuolization of enterocytes and pronounced attachment of coccoid bacteria, both of which have been observed in other studies [7, 15, 16]. The intracytoplasmic eosinophilic bodies (PAS positive vacuoles) within the enterocytes that most frequently were identified in the healthy mink kits have also been described previously [15, 16]. Although their role has not been clarified in mink, their staining properties and localization is comparable to the absorptive, intra-cytoplasmic vacuoles found in neonates of pigs and ruminants [36]. Therefore, the presence of these vacuoles in enterocytes of healthy mink kits is not surprising and may be related to the intestinal uptake of maternal milk antibodies [37], taking place up to 4–5 weeks after birth in mink kits [37]. Although more pronounced changes of the gut architecture like atrophy and fusion of villi were present in the PWD kits, no significant difference in the degree of neutrophil and mononuclear leucocyte infiltration were observed between controls and PWD mink kits. This lack of histopathological signs of inflammation indicates that PWD in the mink kits represents a secretory type of diarrhea. The observed attachment of coccoid bacteria may be responsible for an enterotoxic effect causing a loss of balance of movements of ions and water leading to the vacuolization and swelling of the enterocytes. Interestingly, the attachment of enterococci and E. coli to enterocytes and the slight to moderate infiltrations of neutrophils irrespective of diarrheic status has recently been found in piglets suffering from New Neonatal Porcine Diarrhea Syndrome (NNPDS) [38,39,40], suggesting similarities in mechanisms between diarrhea in the pre-weaning period of mink kits and piglets. Elevated levels of SAA and adhesion of bacteria to the intestinal wall has been seen for segmented filamentous bacteria (SFB), which adhere to the enterocytes, inducing epithelial SAA production [41, 42]. Atarashi et al. [43] colonized rats and germ-free mice with SFB from 20 strains of bacteria isolated from feces from patients suffering from ulcerating colitis and from E. coli and found that they all promote the induction of Th17 cells, which in turn could lead to an increased SAA production in enterocytes. Research on local expression of SAA in the intestine is needed to elucidate if circulating SAA levels are increased as a consequence of local production of SAA by epithelial intestinal cells in mink kits affected by PWD. In rodent models, a local induction of SAA in enteric epithelial cells in response to an altered microbiota in the absence of inflammation has indeed been demonstrated [43], however the impact on circulating SAA concentrations has not been reported. Although more pronounced changes of the gut architecture, like atrophy and fusion of villi were present in the PWD kits, no significant difference in the degree of neutrophil and mononuclear leucocyte infiltration were observed between controls and PWD mink kits. Thus, the possibility of an association between increased SAA levels and an unidentified inflammatory state in mink kits suffering from PWD should be investigated in more detail.
The most frequent bacterial isolate from control mink kits belonged to the S. intermedius group, which in mink has been shown to be S. delphini [44]. This finding is in line with previous studies of the normal intestinal microflora of mink kits [12, 30]. Although mink are natural hosts of S. delphini this bacterial species was suggested to be responsible for an outbreak of PWD through enterotoxin production, which again leads to the question if S. delphini could be an opportunistic bacterium after a primary infection during an episode of PWD [29]. In contrast, MiAstV, mink coronavirus, mink enteritis virus, and rotavirus A were not found by PCR [45] in any of the kits analyzed (data not shown), suggesting a low presence of virus.
Conclusions
We identified a significant increase in circulating concentrations of SAA and attachment of coccoid bacteria in kits affected by PWD. The slight to moderate infiltrations of neutrophils irrespectively of diarrheic status and the attachment of coccoid bacteria to enterocytes show similarities with observations found in piglets suffering from NNPDS, and suggest that PWD in mink is a secretory type of diarrhea.
References
Chriél M. Greasy kits-an efficient effort limits losses. Dansk Pelsdyravl. 1994;4:180–1 (in Danish).
Birch JM, Agger JF, Dahlin C, Jensen VF, Hammer AS, Struve T, et al. Risk factors associated with diarrhea in Danish commercial mink (Neovison vison) during the pre-weaning period. Acta Vet Scand. 2017;59:43.
Svennekjær NC. Mink diseases and current problems in mink farming. Dansk Pelsdyravl. 1954;12:384–404 (in Danish).
Hyldgaard-Jensen C. “Greasy kits” in mink. Observations over a three year period. Dansk Veterinærtidskrift. 1989;72:566–71 (in Danish).
Clausen TN, Dietz HH. Greasy kits. Annual Report, Kopenhagen Research. Kopenhagen Fur. 2004; p. 209–14 (in Danish).
Olesen CR, Hansen M, Clausen TN. Sticky kits-risk factors and causality. Dansk Pelsdyravl. 1992;4:170–2 (in Danish).
Englund L, Chriél M, Dietz HH, Hedlund KO. Astrovirus epidemiologically linked to pre-weaning diarrhoea in mink. Vet Microbiol. 2002;85:1–11.
Ullman K, Hammer AS, Hedlund KO, Thorén P, Czifra G. Prevalence of mink astrovirus (MiAstV) in faecal samples in mink kits during the pre-weaning period. Annual Report, Kopenhagen Research. Kopenhagen Fur. 2007. p. 159–64 (in Danish).
Hammer AS, Ullman K, Czifra G. Astrovirus infections in mink kits-virological and pathological findings in mink kits with pre-weaning diarrhea submitted for diagnostic investigation. Annual Report, Kopenhagen Research. Kopenhagen Fur. 2007. p. 165–70 (in Danish).
Hansen S, Krarup LJ, Agger JF, Ullman K, Hedlund KO, Klingström J, et al. Investigation of parvo-, corona-, and astrovirus as causative agents in outbreaks of diarrhea in Danish mink kits in the nursing and growth period 2013—Preliminary results. Annual Report, Kopenhagen Research. Kopenhagen Fur. 2014. p. 131–8 (In Danish).
Jørgensen M, Scheutz F, Strandbygaard B. Escherichia Coli and virus isolated from “Sticky Kits”. Acta Vet Scand. 1996;37:163–9.
Guardabassi L, Schmidt KR, Petersen TS, Espinosa-Gongora C, Moodley A, Agersø Y, Olsen JE. Mustelidae are natural hosts of Staphylococcus delphini group A. Vet Microbiol. 2012;159:351–3.
Bell JA, Manning DD. Prevalence of Campylobacter jejuni in ranch mink at pelting: cultural, serological, and histological evidence of infection. Can Vet J. 1990;31:367–71.
Møller SH, Chriél M. Health effects of feeding strategies in the pre mating and gestation periods of mink. Scientifur. 2000;24:37–41.
Järplid B, Mejerland T. Villous atrophy in the small intestine of mink kits with diarrhea. Scientifur. 1998;22:157–60.
Dietz HH, Rattenborg E. Histopatological and bacteriological investigation of healthy and greasy mink kits. Annual Report, Kopenhagen Research. Kopenhagen Fur. 1996. p. 135–8 (in Danish).
Gelberg HB. Alimentary system and the peritoneum, omentum, mesentery and peritoneal cavity. In: Zachary JF, editor. Pathol basis Vet Dis. St. Louis: Elsevier; 2016. p. 324–411.
Jespersen A, Jensen HE, Agger JF, Heegaard PMH, Damborg P, Aalbæk B, et al. The effect of color type on early wound healing in farmed mink (Neovison vison). BMC Vet Res. 2017;13:135.
Rygg M, Nordstoga K, Husby G, Marhaug G. Expression of serum amyloid A genes in mink during induction of inflammation and amyloidosis. Biochim Biophys Acta. 1993;1216:402–8.
Bruun CF, Rygg M, Nordstoga K, Sletten K, Marhaug G. Serum amyloid A protein in mink during endotoxin induced inflammation and amyloidogenesis. Scand J Immunol. 1994;40:337–44.
Petersen HH, Nielsen JP, Heegaard PMH. Application of acute phase protein measurements in veterinary clinical chemistry. Vet Res. 2004;35:164–87.
De Buck M, Gouwy M, Wang JM, Van Snick J, Opdenakker G, Struyf S, et al. Structure and expression of different serum amyloid A (SAA) variants and their concentration-dependent functions during host insults. Curr Med Chem. 2016;23:1725–55.
McDonald TL, Weber A, Smith JW. A monoclonal antibody sandwich immunoassay for serum amyloid A (SAA) protein. J Immunol Methods. 1991;144:149–55.
Hammer ASV, Dietz HH. Necropsy of the mink. In: Jensen HE, editor. Necropsy A Handb Atlas. 1st ed. Frederiksberg C: Biofolia; 2011. p. 231–50.
Fang H, Ohlsson AK, Ullberg M, Özenci V. Evaluation of species-specific PCR, Bruker MS, VITEK MS and the VITEK 2 system for the identification of clinical Enterococcus isolates. Eur J Clin Microbiol Infect Dis. 2012;31:3073–7.
Gugołek A, Zalewski D, Strychalski J, Konstantynowicz M. Food transit time, nutrient digestibility and nitrogen retention in farmed and feral American mink (Neovison vison) - a comparative analysis. J Anim Physiol Anim Nutr. 2013;97:1030–5.
Pedersen K, Jørgensen M. Lactic acid bacteria for mink. Colonization and persistence of Enterococcus faecium Cernelle 68 in the digestive tract of mink. Acta Vet Scand. 1992;33:95–103.
Jørgensen M. E. coli-bacteria frequent guests in wet kits. Dansk Pelsdyravl. 1994;12:546–51 (in Danish).
Sledge DG, Danieu PK, Bolin CA, Bolin SR, Lim A, Anderson BC, et al. Outbreak of neonatal diarrhea in farmed mink kits (Mustella vison) associated with enterotoxigenic Staphylococcus delphini. Vet Pathol. 2010;47:751–7.
Vulfson L, Pedersen K, Chriél M, Andersen TH, Dietz HH. Assessment of the aerobic faecal microflora in mink (Mustela vison Schreiber) with emphasis on Escherichia coli and Staphylococcus intermedius. Vet Microbiol. 2003;93:235–45.
Hammer AS, Rasmussen LB, Hansen S, Krarup L, Damgaard PP, Aalbek B. Bacterial etiology of diarrhea in mink kits. In: NJF Semin Proc; 2014. p. 170–2.
Vulfson L, Pedersen K, Chriél M, Frydendahl K, Andersen TH, Madsen M, et al. Serogroups and antimicrobial susceptibility among Escherichia coli isolated from farmed mink (Mustela vison Schreiber) in Denmark. Vet Microbiol. 2001;79:143–53.
Etheridge ME, Yolken RH, Vonderfecht SL. Enterococcus hirae implicated as a cause of diarrhea in suckling rats. J Clin Microbiol. 1988;26:1741–4.
Nicklas JL, Moisan P, Stone MR, Gookin JL. In situ molecular diagnosis and histopathological characterization of enteroadherent Enterococcus hirae infection in pre-weaning-age kittens. J Clin Microbiol. 2010;48:2814–20.
Larsson J, Lindberg R, Aspán A, Grandon R, Westergren E, Jacobson M. Neonatal piglet diarrhoea associated with enteroadherent Enterococcus hirae. J Comp Pathol. 2014;151:137–47.
Uzal FA, Plattner BL, Hostetter JM. Intestine. Normal form and function. In: Maxie MG, editor. Pathol Domest Anim. 6th ed. Philadelphia: Saunders Elsevier; 2016. p. 60–3.
Coe JE, Race RE. Ontogeny of Mink IgG, IgA, and IgM (40039). Proc Soc Exp Biol Med. 1978;157:289–92.
Kongsted H, Jonach B, Haugegaard S, Angen Ø, Jorsal S, Kokotovic B, et al. Microbiological, pathological and histological findings in four Danish pig herds affected by a new neonatal diarrhoea syndrome. BMC Vet Res. 2013;9:206.
Jonach B, Boye M, Stockmarr A, Jensen TK. Fluorescence in situ hybridization investigation of potentially pathogenic bacteria involved in neonatal porcine diarrhea. BMC Vet Res. 2014;10:68.
Hedegaard CJ, Strube ML, Hansen MB, Lindved BK, Lihme A, Boye M, et al. Natural pig plasma immunoglobulins have anti-bacterial effects: potential for use as feed supplement for treatment of intestinal infections in pigs. PLoS ONE. 2016;11:1–14.
Sano T, Huang W, Hall JA, Yang Y, Chen A, Gavzy SJ, et al. An IL-23R/IL-22 Circuit regulates epithelial serum amyloid A to promote local effector Th17 responses. Cell. 2015;163:381–93.
Bel S, Hooper LV. A bacterial nudge to T-cell function. Nature. 2015;526:328–30.
Atarashi K, Tanoue T, Ando M, Kamada N, Nagano Y, Narushima S, et al. Th17 Cell induction by adhesion of microbes to intestinal epithelial cells. Cell. 2015;163:367–80.
Larsen C. Hemolytic Staphylococci in mink. Veterinary Master Thesis. University of Copenhagen; 2014 (in Danish).
Hartby CM, Andersen MR, Kvistgaard LK, Chriél M, Larsen LE, Hjulsager CK. Pooling of faecal samples for quantitative virus diagnostics by real-time PCR. In: Mäki-Tanila A, Valaja J, Mononen J, Sironen T, Vapalahti O, editors. Proceenings XIth Int Sci Congr Fur Anim Prod. Helsinki: Libris Oy; 2016. p. 27–30.
Authors’ contributions
RM and JMB conceived the study. RM and JMB planned the sampling, performed the experiments and drafted the manuscript with contributions from PMHH, HEJ, MC, JFA and TS. RM, JMB and TS did the necropsies. RM analyzed the SAA concentration. JMB and HEJ evaluated all histological sections and described the histopathological lesions. JMB did the bacteriology. TS participated in the collection of blood and samples. All authors read and approved the final manuscript.
Acknowledgements
Farmers K. J. Hansen and J. Jensen who helped with the sampling for the project are greatly acknowledged. The technical assistance and invaluable help from Henriette Vorsholt, Marion Baltzer Petersen, Mette Sif Hansen, Dennis Brok, and Annie Ravn Pedersen are also greatly acknowledged.
Competing interests
The authors declare that they have no competing interests.
Availability of data and assay
The datasets used and/or analyzed during the current study are available from the corresponding author and JMB on reasonable request.
Consent for publication
Animal owners were explained the study purposes and procedures and agreed by written consent to participate.
Ethics approval and consent to participate
The study was approved by the Danish Animal Experiments Inspectorate, (license 2016-15-0201-00906, https://www.foedevarestyrelsen.dk/english/Animal/AnimalWelfare/Pages/The-Animal-Experiments-Inspectorate.aspx).
Funding
The project was co-funded by Pelsdyrafgiftsfonden (The Danish production levy fund for mink), Pelsdyrenes Forskningsfond, the Innovation Fund Denmark, and the Technical University of Denmark.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated.
About this article
Cite this article
Mathiesen, R., Birch, J.M., Chriél, M. et al. Mink (Neovison vison) kits with pre-weaning diarrhea have elevated serum amyloid A levels and intestinal pathomorphological similarities with New Neonatal Porcine Diarrhea Syndrome. Acta Vet Scand 60, 48 (2018). https://doi.org/10.1186/s13028-018-0403-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s13028-018-0403-7