Abstract
Background
This study sought to examine the impact of magnesium supplementation on clinical outcomes and biochemical factors among hospitalized patients with COVID-19.
Methods
This double-blind, randomized clinical trial was conducted at Razi Hospital, Ahvaz, Iran, between September 2021 and March 2022. Participants aged 18–70 years with moderate disease severity were enrolled. Magnesium supplementation (300 mg daily) was administered to the intervention group, while the control group received a placebo. Clinical outcomes, including the need for oxygen therapy, oxygen saturation, respiratory rate, fever, hs-CRP and TNF-α levels, as well as quality of life and mental health, were assessed. Blood samples were collected to measure biochemical variables.
Results
The main result was the count of individuals requiring oxygen therapy. Additional outcomes comprised of oxygen saturation, respiratory rate, fever, hs-CRP and TNF-α levels, as well as quality of life and mental health. Out of 64 participants, 60 completed the study. The results showed that magnesium supplementation significantly reduced the number of patients requiring oxygen therapy (9 vs. 14; P < 0.001). Moreover, the magnesium group demonstrated improved oxygen saturation compared to the control group (4.55 ± 2.35 vs. 1.8 ± 1.67; P < 0.001). Furthermore, we observed a noteworthy enhancement in the quality of life and depression score in the magnesium group. No significant differences were observed in respiratory rate, fever, hs-CRP, and TNF-α levels (P > 0.05).
Conclusion
The findings suggest that magnesium supplementation may have beneficial effects on clinical outcomes and arterial oxygen saturation in COVID-19 patients. More investigation is necessary to delve into its potential mechanisms and long-term effects on patient outcomes.
Trial registration
This study is registered on Iranian Registry of Clinical Trials (IRCT) under identifier IRCT20210413050957N1. (The registration date: May 1, 2021).
Similar content being viewed by others
Introduction
The global outbreak of COVID-19 caused by the SARS-CoV-2 virus has presented an unprecedented challenge to public health worldwide, prompting an urgent search for effective treatment strategies [1]. As of March 19, 2023, the worldwide consequences of the COVID-19 pandemic triggered by the SARS-CoV-2 virus led to in 761,402,282 confirmed cases and 6,887,000 fatalities worldwide [2]. Since 2019, this disease has become the biggest health-related challenge in the whole world and has imposed a lot of financial problems on healthcare systems [3].
The human body relies on its immune system as the primary defense mechanism against viral infections and diseases [4, 5]. Upon pathogen invasion, various immune response pathways are activated to combat the threat [6, 7]. Nutrition plays a pivotal role in enhancing these immune response mechanisms, thereby bolstering the body’s ability to defend against pathogens [8,9,10]. Studies have demonstrated a notable increase in the risk of mortality and adverse events during hospitalization among adult COVID-19 patients with nutritional deficiencies or malnutrition [11,12,13]. In COVID-19 patients, malnutrition or susceptibility to malnutrition correlates with heightened inflammation and compromised immune function, potentially exacerbating disease progression and complicating its outcomes [14, 15]. Hence, nutrients with the potential to enhance immunity against the coronavirus (COVID-19) and mitigate the risk of infection or progression of associated factors play a crucial role in both preventing the disease and treating afflicted individuals [16, 17].
Among the various micronutrients under investigation, magnesium has gained attention due to its essential role in immune function, pulmonary health, and cellular metabolism [10, 18]. Magnesium (Mg) is the second most plentiful positively charged ion found within cells, following potassium. About 99% of the total magnesium in the body is concentrated within the intracellular area, while approximately 1% is distributed in the bloodstream and other fluids outside the cells [19,20,21]. Low levels of magnesium in the blood, which are quite prevalent in some countries, are commonly observed among elderly individuals due to inadequate dietary intake, certain health conditions (such as diabetes), and the use of multiple medications [12, 22, 23].
Mg partakes in various metabolic and biochemical processes and plays a crucial role in numerous essential functions in the body. These include bone development, neuromuscular activity, signal transmission, energy production, as well as the metabolism of glucose, lipids, and proteins [24, 25]. Additionally, Mg contributes to the stability of DNA and RNA, as well as the regulation of cell growth and specialization [26]. It’s important to mention that magnesium (Mg) plays a role in controlling the functions of both the innate and adaptive immune systems [27]. This can potentially provide protective benefits against COVID-19. For example, Mg helps stabilize mastocyte membranes, regulates the activity of neutrophils and macrophages, and hinders the Toll-like receptor a/nuclear factor-κB (NF-κB) pathway [28, 29]. Moreover, magnesium (Mg) adjusts the cytotoxic activities of natural killer (NK) cells and CD8 + T lymphocytes [30].
Given the significance of magnesium in immune modulation and its prevalent deficiency among patients, this study aimed to investigate the impact of magnesium supplementation on clinical outcomes and biochemical factors in hospitalized COVID-19 patients. By addressing these critical gaps in the literature and providing novel insights into the potential benefits of magnesium supplementation, this study contributes to the ongoing efforts to optimize treatment strategies for COVID-19.
Methods
Research design and participant enrollment
This research conducted at Razi Hospital, Ahvaz, Iran, between September 2021 and March 2022, involved a double-blind, randomized clinical trial on COVID-19-infected patients who were hospitalized in the infectious ward. In order to qualify for involvement, people were required to fulfill the following conditions: be aged between 18 and 70 years, have received a verified COVID-19 diagnosis via a positive RT-PCR test, nasopharyngeal swab, experiencing respiratory symptoms (such as breathlessness, chest discomfort, and sensations of pressure) with or without an accompanying fever (equal or more than 38 °C) and Spo2 between 90% and 93%. Patients with moderate disease severity were included in this study. Patients who were critically ill and were hospitalized in the intensive care unit (ICU) or had a mild illness and did not need to be hospitalized were not included in the study.
Other inclusion criteria include absence of pregnancy and breastfeeding, without history of diabetes, high blood pressure, heart disease, kidney dysfunction, and psychological disorders like depression, Body mass index less than 35 and more than 18, Vitamin D level above 30 ng/ml, absence of hypomagnesemia that refers to a deficiency in magnesium levels (below 1.7 mg/dL) and hypermagnesemia signifies an excess of magnesium levels (above 2.6 mg/dL), and absence of alcohol and drug abuse. Exclusion criteria included unwillingness to continue participating in the study, hospitalization of the patient in the ICU, Taking antioxidant and anti-inflammatory food supplements such as vitamin E, omega 3, vitamin D, etc. and contraindication, intolerance or allergy to magnesium supplement.
The sample size calculation was based on a previous study by Tan et al. [31] which reported a 60% reduction in the proportion of cases requiring oxygen therapy in the intervention group compared to the control group. With a power of 80%, alpha level of 0.05, and estimated proportions of 61.5% in the control group and 24% in the intervention group, the sample size was determined to be 25 individuals per group. Considering a 20% attrition rate, the final sample size comprised 30 individuals in each group.
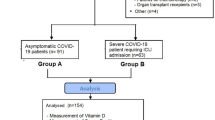
The general scheme of the intervention is shown in Fig. 1. The method of assigning patients to the intervention and control groups was the approach of employing randomized permutation blocks with a block size of 4 (referring to the table associated with random permutations). The participants were randomly allocated in a ratio of 1:1 to the intervention and control group. A computer-generated list was created using random permuted blocks to ensure that the allocation to subjects and investigators remained concealed. The intervention and placebo were kept hidden from researchers, patients, infectious disease specialist and clients. Magnesium supplements and placebo were completely similar in terms of appearance, color, fragrance, and packaging, so that the blinding process could be fully implemented.
At the study’s inception, the researchers thoroughly briefed the patients on the process of implementing the study and all participants provided written informed consent to partake in the study. Patients had the option to exit the study at any point if they were unwilling to cooperate. The study was conducted in compliance with the Declaration of Helsinki and the protocol of the research was approved by the ethics committee of Ahvaz Jundishapur University of Medical Sciences (Ethical code: IR.AJUMS.REC.1400.0255) and This study is registered on Iranian Registry of Clinical Trials (IRCT) under identifier IRCT20210413050957N1. (The registration date: May 1, 2021).
Intervention
Participants within the intervention group were provided with 300 mg of oral magnesium supplement in the form of magnesium citrate capsules daily, and subjects in the control group received a placebo containing starch. Magnesium citrate powder was obtained from Behan Sar Pharmaceutical Company, and capsules containing magnesium and placebo were prepared by an expert at the Faculty of Pharmacy of Jundishapur University and provided to the study. The placebo capsules contained starch and were similar in appearance to magnesium supplements. The duration of intervention in patients was from the time of admission to the time of discharge from the hospital. In case of changes in the diet and medication protocol of the patients, the changes were reviewed by the research team, and in case of interference with the study objectives, the patient was excluded from the study.
Data about the physical dimensions of the body and past medical records
Information pertaining to age, weight, height, past medical conditions, prescribed medications, blood pressure, serum lipids, random blood glucose, and respiratory condition was obtained by examining medical records.
Dietary intake
Nutritional intake of patients was done by recording of 24-hour dietary recall at both the start and conclusion of the research. The N4 software was utilized to analyze data on dietary intakes, with national food composition tables serving as a reference for the analysis of food intake.
Primary and secondary outcome
The confirmatory result of this study was the number of patients who needed oxygen therapy. Secondary outcomes included SaO2, respiratory rate, fever, hs-CRP and TNF-α levels, the well-being and psychological state of individuals’ lives and mental health.
Measurements
To measure biochemical variables, 7 cc of blood were taken from the patients at the beginning of the study and on the day of discharge from the hospital. Diaclone Research’s ELISA kits from Besançon, France were employed to evaluate the levels of hs-CRP and TNF-α concentration. To assess serum magnesium concentration, the measurements were conducted using an auto-analyzer utilizing a photometric method (BT-3500, Biotecnica Instruments, Rome, Italy) using Biosystem Kits (Barcelona, Spain). Depression status of patients was measured by Beck Depression inventory-II (BDI-II). The research conducted by Hamidi and colleagues, Persian version of the questionnaire was utilized, demonstrating satisfactory validity (alpha = 0.92) and reliability (r = 0.64) [32]. Individuals participating in the study filled out a self-report inventory consisting of 21 items. The items were rated on a scale of 0–3, where a higher score indicates more severe symptoms. The total score of the questionnaire is then classified into the following categories [33]: for scores ranging from 14 to 19, it indicates a mild level of depression. Scores between 20 and 28 suggest a moderate level of depression, while scores between 29 and 63 indicate a severe level of depression. The patients’ anxiety was evaluated by Spielberger State-Trait Anxiety Inventory (STAI). In Abdoli et al.‘s study, the Persian version of the questionnaire was employed, indicating acceptable validity (alpha = 0.88) and reliability (r = 0.64) specifically for measuring state anxiety [34].
The researchers employed the SF-36 questionnaire to evaluate the quality of life of individuals in this study. The SF-36 is a frequently employed survey that individuals fill out on their own, providing a comprehensive assessment of their quality of life related to health. It consists of 36 items grouped into eight subscales, which are further classified into two broad components: the physical component summary (PCS) and the mental component summary (MCS). The physical component summary comprises evaluations of 10 items related to physical functioning, 4 items related to role-physical, 2 items related to bodily pain, and 5 items related to general health perceptions. The mental component summary comprises the following subscales: social functioning (2 items), role-emotional (3 items), mental health (5 items), and vitality (4 items). Additionally, there is a single item that separately assesses changes in health. The scores for all items are assigned numerical codes, summed together, and then transformed into a scale ranging from 0 to 100. In this scale, a score of 0 represents the poorest condition, while a score of 100 indicates the best condition [35]. In 2006, Montazeri et al. conducted a study in Tehran, Iran, to validate the SF-36 questionnaire among a healthy population aged 15 years and above. The study confirmed the reliability of the questionnaire by calculating Cronbach’s alpha coefficients, which ranged from 0.77 to 0.90 for the subscales and the overall scale [36].
Statistical analyses
The quantitative data were expressed as the mean value accompanied by the standard deviation. On the other hand, qualitative data was presented in the form of percentages and numerical counts to describe the status of these measurements. The Kolmogorov-Smirnov test was employed to validate the normal distribution. The study employed the Independent T-test and chi-square test to evaluate and draw comparisons between groups’ results at the beginning and conclusion of the study. Changes from the initial baseline were analyzed within each group using a paired samples t-test. The analysis of covariance (ANCOVA) test was used to account for confounding factors. All ANCOVA models were adjusted for weight, serum magnesium levels at baseline, and calorie intake. The data underwent analysis with SPSS19 software, and if the P-value was less than 0.05, it was deemed statistically significant.
Results
Patients
Out of the 110 patients evaluated for qualification, 64 met the criteria and were randomly assigned to either the magnesium group or the placebo group. Some participants couldn’t be included because of the following reasons: pregnancy, taking antioxidant supplements, admission to the intensive care unit and participants’ hesitancy to take part in the study after knowing about the objectives of the study. Out of 64 patients participating in the study, two patients from the intervention group were removed from the study because they were not willing to continue their cooperation and nausea after taking magnesium supplements. Also, 2 people from the control group were withdrawn from this project due to their resistance to further involvement in the study, and the final analysis was performed on 60 people (30 people in each group). Table 1 presents a concise overview of the information about demographic characteristics of the participants involved in this study. The mean (SD) age of the patients was 47.8 (9.50) years and mean weight was 72.47 ± 8.75 kg. Also, 48.34% of participants were male and the rest were female. The participants in the study were from three ethnics: Arabs (21.67%), Fars (21.67%), and Lor/Bakhtiari (31.66%). The mean duration of COVID-19 in magnesium group was 9.90 ± 3.40 days and in control group was 9.10 ± 2.14, without any significant differences (P = 0.68). The length of time patients stayed in the hospital was 7.32 ± 5.07 days in the magnesium group and 7.28 ± 4.25 days in the placebo group, and no notable distinction was found between the two groups (P = 0.84). Also, in term of Moreover, no substantial disparities were detected between the two groups in term of other baseline characteristics.
Dietary intake of participants showed in Table 1. The mean of calorie intake in magnesium group was 1843.18 ± 436.25 kcal/day and in the control, group was 1812.52 ± 373.51 kcal/day, without any significant differences (P = 0.65). There weren’t any significant differences between two groups in term of other macro and micronutrients (P > 0.05).
Effects of magnesium supplementation on serum concentration of magnesium
The average level of magnesium in the blood at the start of the investigation was 2.25 ± 0.28 mg/dl in the intervention group and 2.28 ± 0.37 mg/dl in the placebo group, and there was no substantial variation noticed between the two groups (P = 0.55). At the end of the study, we found a significant difference between two groups in term of serum magnesium concentration in the crude (P = 0.004) and adjusted model (P = 0.001) (Table 2).
Primary outcome
The main result of this study was the need to oxygen therapy (Table 3). The results of the present study showed that the number of people who needed oxygen therapy was significantly less in the magnesium group than in the control group (9 patients vs. 14 patients; P < 0.001).
Secondary outcomes
The average duration of hospital stay did not show any significant difference between the magnesium group and control group (7.32 ± 5.37 days vs. 7.28 ± 4.65 days; P = 0.84). In term of oxygen saturation of arterial blood, at the end of the study, we found a significant improvement in the magnesium group than control group (4.55 ± 2.35 vs. 1.8 ± 1.67; P < 0.001). However, we couldn’t find any significant differences between two groups in term of respiratory rate (P = 0.16), fever (P = 0.47), hs-CRP (P = 0.65) and TNF-α (P = 0.54). There were no notable distinctions observed in the significance of the results after adjusting for confounding variables (Table 4).
In term of mental health related factors and quality of life, we found a significant improvement in the SF-36 MCS score (15.09 ± 11.90 vs. 2.03 ± 10.52; P < 0.001) and SF-36 PCS score (16.78 ± 14.32 vs. 3.28 ± 12.35; P = 0.01). Also, in relation to the severity of depression, magnesium supplementation caused a significant reduction in the depression score compared to placebo (-5.24 ± 0.76 vs. -1.63 ± 0.68; P = 0.03). However, we couldn’t find any significant differences between groups in term of anxiety score (P = 104) (Table 5).
Discussion
The present study aimed to investigate the effect of magnesium supplementation on clinical and biochemical parameters in patients with COVID-19. The findings of this randomized clinical trial provide valuable insights into the potential benefits of magnesium supplementation in the management of COVID-19 patients. The results of the present study showed that magnesium supplementation among the patients with COVID-19 led to a significant reduction in the need for oxygen therapy, oxygen saturation of arterial blood, quality of life and depression scores. However, we couldn’t find any significant effects on other clinical and biochemical variables.
Magnesium, a vital micronutrient, plays multifaceted roles in cellular metabolism, immune modulation, and pulmonary health [37]. Our study aimed to investigate the impact of magnesium supplementation on both clinical outcomes and biochemical parameters in COVID-19 patients. While the observed improvements in quality of life and mental health may appear to precede changes in hematological factors, it is essential to consider the complex and dynamic nature of magnesium’s effects on physiological processes. The involvement of magnesium in cellular and tissue metabolism is intricate and influenced by multiple factors [26, 38]. Growing evidence indicates that magnesium primarily serves as an important signaling element and metabolite in cellular physiology. Previous findings have emphasized the significance of magnesium in maintaining appropriate immune, vascular, and pulmonary function. In this context, magnesium plays a vital role in the following aspects: ensuring the effective functioning of neutrophils and macrophages, enhancing the cytotoxic activity of T lymphocytes, activating immune cells, and restricting viral replication [27, 39, 40].
Our results demonstrated a significant reduction in the number of patients requiring oxygen therapy in the magnesium supplementation group compared to the control group. This outcome suggests that magnesium supplementation may have a protective effect on respiratory function in COVID-19 patients. These results align with prior research that have highlighted the role of magnesium in maintaining airway smooth muscle tone [41], bronchodilation [42], and reducing airway inflammation [1, 43]. Magnesium’s ability to modulate calcium homeostasis and impact ion channels may contribute to its beneficial effects on respiratory function in COVID-19 patients [18]. As far as we know, in individuals with COVID-19, similar to other viral infections, there may be a higher prevalence of airway hyperreactivity [44]. In relation to this, it is important to consider enhancing ventilation and decreasing airway resistance [45, 46]. Certain medications like β2 agonists and anti-muscarinic agents are employed to alleviate these symptoms. An alternative treatment available for patients who are hospitalized is the use of nebulized magnesium sulfate. Existing literature suggests that magnesium sulfate can induce bronchodilation in individuals with asthma through multiple mechanisms, such as inhibiting calcium influx into the cytosol, preventing acetylcholine secretion, inhibiting histamine release, and enhancing β2 receptor affinity. This, in turn, enhances the bronchodilator effect of β2 agonists [42, 45].
As our understanding of COVID-19’s development has advanced, additional clues shed light on the possible involvement of imbalanced magnesium levels in both the prevention and progression of the disease. Magnesium plays a crucial role in stabilizing the membranes of mastocytes, which are found in the submucosa of the airways and alveolar septa. This function helps prevent their degranulation and the subsequent release of various mediators [28]. Also, this mineral controls the functioning of neutrophils and macrophages by suppressing their priming, inhibiting oxidative burst, and interfering with the Toll-like receptor 4/NFκB axis [47]. In lymphocytes, magnesium regulates the levels of Inositol Triphosphate and diacylglycerol, which are essential second messengers that become active upon the activation of B and T cell receptors. Moreover, magnesium plays a crucial role in defending the body against viral infections, as it is necessary to maintain sufficient levels of intracellular magnesium for the cytotoxic function of T lymphocytes and natural killer (NK) cells [48].
Regarding the secondary outcomes, we observed a significant improvement in oxygen saturation of arterial blood in the magnesium group compared to the control group. This improvement suggests enhanced oxygenation and respiratory efficiency in patients receiving magnesium supplementation. However, we did not observe significant differences between groups in terms of respiratory rate, fever, hs-CRP, and TNF-α levels. These results may indicate that magnesium supplementation primarily benefits respiratory function rather than directly affecting fever or inflammatory markers. In line with our findings, Simental-Mendia et al. in a systematic review and meta-analysis of 11 trials showed that magnesium supplementation hadn’t any significant effect on serum levels of CRP [49]. While our study did not find significant changes in hematological factors such as hs-CRP and TNF-α levels, it is important to acknowledge the multifactorial nature of COVID-19 pathogenesis [50]. The observed clinical improvements may be mediated through alternative pathways, such as magnesium’s neuroprotective properties and its role in neurotransmitter regulation [51]. The reduction in depression scores observed in the magnesium group underscores the potential neuropsychiatric benefits of magnesium supplementation, which may be independent of its effects on inflammatory markers.
In relation to factors impacting quality of life and mental health, our study demonstrated significant improvements in the SF-36 MCS and PCS score in the magnesium group compared to the control group. These findings indicate that magnesium supplementation may have positive effects on mental well-being and overall quality of life in COVID-19 patients. Magnesium’s involvement in neurochemical processes and its neuroprotective properties may contribute to these observed effects [52]. Furthermore, we noted a considerable decrease in depression scores in the magnesium group, indicating a potential role of magnesium in alleviating depressive symptoms in COVID-19 patients. Nevertheless, no notable disparities were detected in anxiety scores between the two groups. In line with our findings, it has been reported in some observational studies that there was an inverse correlation between dietary magnesium intake and risk of depression [53, 54]. Animal studies have definitively demonstrated that magnesium exhibits antidepressant properties. Previous findings indicate that supplementing with magnesium leads to an antidepressant-like effect, reducing the severity of depressive disorders in experimental animal tests and models [55, 56]. Tarleton et al. in a RCT study among the 126 adults showed that magnesium supplementation with daily dose of 248 mg after a duration of 6 weeks, there was a substantial positive improvement in the levels of depression and anxiety [57]. While the link between magnesium and depression is widely acknowledged, the specific mechanism remains uncertain. Nevertheless, magnesium actively participates in various pathways, enzymes, hormones, and neurotransmitters that are intricately involved in the regulation of mood [58]. This compound functions as a calcium antagonist and a voltage-dependent blocker of the N-methyl-D-aspartate (NMDA) channel, which is responsible for controlling the influx of calcium into the nervous system [59]. When magnesium levels are low, an imbalance of calcium and glutamate can disrupt synaptic function, leading to symptoms of depression [43, 60].
The current study has some limitations that should be considered. Firstly, the sample size was relatively small, which might that findings may have limited applicability to a broader context. Conducting larger studies with diverse populations could strengthen the evidence supporting the effects of magnesium supplementation in COVID-19 patients. Secondly, the study duration was relatively short, and the long-term effects of magnesium supplementation on COVID-19 outcomes remain unknown. Studies with longer follow-up periods are needed to assess the sustainability of the observed benefits. Moreover, the study relied on self-reported dietary intake data, which may introduce recall bias and inaccuracies in assessing nutritional status. Future studies could consider using more objective measures of dietary intake, such as food diaries or biomarkers of nutrient status.
Conclusion
In summary, the findings of this randomized clinical trial suggest that oral magnesium supplementation in COVID-19 patients may have several beneficial effects. Magnesium supplementation was associated with a reduction in the need for oxygen therapy, improved arterial oxygen saturation levels, enhanced mental health-related factors and quality of life, and a decrease in depression scores. However, further research is warranted to confirm these findings, explore the underlying mechanisms, and examine the lasting impacts of magnesium supplementation in larger and more diverse patient populations. The potential benefits of magnesium supplementation in COVID-19 patients make it a promising area for future investigations and could have important implications for clinical management strategies.
Data availability
The data that support the findings of this study are available if anyone wants.
References
Tang C-F, et al. Possibility of magnesium supplementation for supportive treatment in patients with COVID-19. Eur J Pharmacol. 2020;886:173546.
Guerrero-Romero F, et al. Importance of Magnesium Status in COVID-19. Biology. 2023;12(5):735.
Sun J, et al. COVID-19: epidemiology, evolution, and cross-disciplinary perspectives. Trends Mol Med. 2020;26(5):483–95.
Gooruee R, et al. Evaluation of the effect of kefir supplementation on inflammatory markers and clinical and hematological indices in COVID-19 patients; a randomized double-blined clinical trial. Advances in Integrative Medicine; 2023.
Ayivi R, et al. COVID-19: human immune response and the influence of food ingredients and active compounds. Bioactive Compd Health Disease-Online ISSN: 2574– 0334; Print ISSN: 2769– 2426. 2021;4(6):100–48.
Vahdat M, et al. Effects of resistant starch interventions on circulating inflammatory biomarkers: a systematic review and meta-analysis of randomized controlled trials. Nutr J. 2020;19:1–10.
Rahimlou M, et al. Effects of ginger supplementation on anthropometric, glycemic and metabolic parameters in subjects with metabolic syndrome: a randomized, double-blind, placebo-controlled study. J Diabetes Metab Disord. 2019;18(1):119–25.
Ponce J, et al. Impact of malnutrition on clinical outcomes in patients diagnosed with COVID-19. J Parenter Enter Nutr. 2022;46(8):1797–807.
Liu H, et al. Malnutrition is associated with hyperinflammation and immunosuppression in COVID-19 patients: a prospective observational study. Nutr Clin Pract. 2021;36(4):863–71.
Parsi A, et al. The effects of crocus sativus extract on serum lipid profile and liver enzymes in patients with non-alcoholic fatty liver disease: a randomized placebo-controlled study. Obes Med. 2020;17:100165.
Hajibeygi R, et al. Effect of a diet based on Iranian traditional medicine on inflammatory markers and clinical outcomes in COVID-19 patients: a double-blind, randomized, controlled trial. Eur J Integr Med. 2022;55:102179.
Hashemi R, et al. Investigating the effect of DASH diet on blood pressure of patients with type 2 diabetes and prehypertension: randomized clinical trial. Volume 13. Diabetes & Metabolic Syndrome: Clinical Research & Reviews; 2019. pp. 1–4. 1.
Firouzi S, et al. The effect of vitamin C and zn supplementation on the immune system and clinical outcomes in COVID-19 patients. Clin Nutr Open Sci. 2022;44:144–54.
KARAOĞUL E, et al. Foods that improving the Immune System against Covid-19 and its infectiousness: Foods against Covid-19 and its infectiousness. Int J Curr Med Biol Sci. 2021;1(2):58–68.
Pahlavani N, et al. Effects of melatonin and propolis supplementation on inflammation, oxidative stress, and clinical outcomes in patients with primary pneumosepsis: a randomized controlled clinical trial. Complement Med Res. 2022;29(4):275–85.
Butler MJ, Barrientos RM. The impact of nutrition on COVID-19 susceptibility and long-term consequences. Brain Behav Immun. 2020;87:53–4.
Morvaridzadeh M, et al. Effect of ginger (Zingiber officinale) supplementation on oxidative stress parameters: a systematic review and meta-analysis. J Food Biochem. 2021;45(2):e13612.
Iotti S et al. The COVID-19 pandemic: is there a role for magnesium? Hypotheses and perspectives. Magnes Res, 2020. 33(2).
Faryadi Q. The magnificent effect of magnesium to human health: a critical review. Int J Appl. 2012;2(3):118–26.
Caspi R, et al. The MetaCyc database of metabolic pathways and enzymes and the BioCyc collection of Pathway/Genome databases. Nucleic Acids Res. 2014;42(D1):D459–71.
Morvaridzadeh M, et al. Effect of soy products and isoflavones on oxidative stress parameters: a systematic review and meta-analysis of randomized controlled trials. Food Res Int. 2020;137:109578.
Barbagallo M, Dominguez L. Magnesium and aging. Curr Pharm Design. 2010;16(7):832–9.
Rahimlou M, et al. Effects of ginger supplementation on anthropometric, glycemic and metabolic parameters in subjects with metabolic syndrome: a randomized, double-blind, placebo-controlled study. J Diabetes Metabolic Disorders. 2019;18:119–25.
Schuchardt J, Hahn A. Intestinal absorption and factors influencing bioavailability of magnesium—an update. Curr Nutr Food Sci. 2017;13:260–78.
Konrad M, Schlingmann KP, Gudermann T. Insights into the molecular nature of magnesium homeostasis. Am J Physiology-Renal Physiol. 2004;286(4):F599–605.
Feeney KA, et al. Daily magnesium fluxes regulate cellular timekeeping and energy balance. Nature. 2016;532(7599):375–9.
de Jesus JR, et al. Trace element homeostasis in the neurological system after SARS-CoV-2 infection: insight into potential biochemical mechanisms. J Trace Elem Med Biol. 2022;71:126964.
Maier JA, et al. Magnesium and inflammation: advances and perspectives. Semin Cell Dev Biol. 2021;115:37–44.
Rahimlou M, et al. Association between dietary glycemic index and glycemic load with depression: a systematic review. Eur J Nutr. 2018;57:2333–40.
Chaigne-Delalande B, et al. Mg2 + regulates cytotoxic functions of NK and CD8 T cells in chronic EBV infection through NKG2D. Science. 2013;341(6142):186–91.
Tan CW, et al. Cohort study to evaluate the effect of vitamin D, magnesium, and vitamin B(12) in combination on progression to severe outcomes in older patients with coronavirus (COVID-19). Nutrition. 2020;79–80:111017.
Hamidi R, et al. Validity and reliability Beck Depression Inventory-II among the Iranian elderly population. J Sabzevar Univ Med Sci. 2015;22(1):189–98.
Segal DL, et al. Psychometric properties of the Beck Depression Inventory—II (BDI-II) among community-dwelling older adults. Behav Modif. 2008;32(1):3–20.
Abdoli N, et al. Reliability and validity of Persian version of state-trait anxiety inventory among high school students. East Asian Archives Psychiatry. 2020;30(2):44–7.
Mohamadzadeh Tabrizi Z, et al. COVID-19 anxiety and quality of life among Iranian nurses. BMC Nurs. 2022;21(1):27.
Ghabaee NZ. Quality of life and its relationship to the job stress in among nursing staff in hospitals of Sari, in 2015. J Nurs Educ. 2016;5(2):40–8.
Trapani V et al. The relevance of magnesium homeostasis in COVID-19 European journal of nutrition, 2022: pp. 1–12.
Iotti S, et al. Quantitative mathematical expressions for accurate in vivo assessment of cytosolic [ADP] and ∆G of ATP hydrolysis in the human brain and skeletal muscle. Volume 1708. Biochimica et Biophysica Acta (BBA)-Bioenergetics; 2005. pp. 164–77. 2.
Brandao K, et al. The role of mg 2 + in immune cells. Immunol Res. 2013;55:261–9.
Rahimlu M, Shab-Bidar S, Djafarian K. Body Mass Index and all-cause mortality in chronic kidney disease: a dose-response Meta-analysis of Observational studies. J Ren Nutr. 2017;27(4):225–32.
Barbagallo M, Veronese N, Dominguez LJ. Magnesium in aging, health and diseases. Nutrients. 2021;13(2):463.
Pooransari P, Pourdowlat G. Magnesium sulfate: a potential adjuvant treatment on COVID-19. Front Emerg Med. 2021;5(1):e1–1.
Mohammadifard N, et al. The effect of ketogenic diet on shared risk factors of cardiovascular disease and cancer. Nutrients. 2022;14(17):3499.
Abrams EM, W‘t Jong G, Yang CL. Asthma and COVID-19. CMAJ. 2020;192(20):E551–551.
Song W-J, Chang Y-S. Magnesium sulfate for acute asthma in adults: a systematic literature review. Asia Pac Allergy. 2012;2(1):76–85.
Morshedzadeh N, et al. Association between adipokines levels with inflammatory bowel disease (IBD): systematic reviews. Dig Dis Sci. 2017;62:3280–6.
Brandao K, et al. The role of Mg2 + in immune cells. Immunol Res. 2013;55(1–3):261–9.
Minton K. Immunodeficiency: magnesium regulates antiviral immunity. Nat Rev Immunol. 2013;13(8):548–9.
Simental-Mendia LE, et al. Effect of Magnesium supplementation on plasma C-reactive protein concentrations: a systematic review and Meta-analysis of Randomized controlled trials. Curr Pharm Des. 2017;23(31):4678–86.
Borczuk AC, Yantiss RK. The pathogenesis of coronavirus-19 disease. J Biomed Sci. 2022;29(1):87.
Coman AE, et al. The significance of low magnesium levels in COVID-19 patients. Medicina. 2023;59(2):279.
Szewczyk B, et al. The role of magnesium and zinc in depression: similarities and differences. Magnes Res. 2018;31(3):78–89.
Sun C, et al. Dietary magnesium intake and risk of depression. J Affect Disord. 2019;246:627–32.
Rahimlou M, et al. Association between dietary glycemic index and glycemic load with depression: a systematic review. Eur J Nutr. 2018;57(7):2333–40.
Haj-Mirzaian A, et al. Blockade of NMDA receptors reverses the depressant, but not anxiogenic effect of adolescence social isolation in mice. Eur J Pharmacol. 2015;750:160–6.
Poleszak E, et al. Effects of acute and chronic treatment with magnesium in the forced swim test in rats. Pharmacol Rep. 2005;57(5):654–8.
Tarleton EK, et al. Role of magnesium supplementation in the treatment of depression: a randomized clinical trial. PLoS ONE. 2017;12(6):e0180067.
Serefko A, et al. Magnesium in depression. Pharmacol Rep. 2013;65(3):547–54.
Iseri LT, French JH. Magnesium: nature’s physiologic calcium blocker. Am Heart J. 1984;108(1):188–93.
Derom ML, et al. Magnesium intake is not related to depression risk in Spanish university graduates. J Nutr. 2012;142(6):1053–9.
Acknowledgements
The author thanks the participants and their families who took part to the study.
Funding
There wasn’t any special funding for this project.
Author information
Authors and Affiliations
Contributions
SR provided formal analysis and writing (original draft). SAH, SR and SMA contributed to the investigation. RD and SAH contributed to the laboratory testing. SAH contributed to the project administration and funding acquisition. EM contributed to the statistical analysis and writing (review and editing). All authors approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
The study was conducted in compliance with the Declaration of Helsinki and the protocol of the research was approved by the Ahvaz Jundishapur University of Medical Sciences (Ethical code: IR.AJUMS.REC.1400.0255) and the protocol of this research work was registered in the Iranian Registry of Clinical Trials (IRCT) (IRCT code: IRCT20210413050957N1, URL: https://www.irct.ir/trial/55660, registration date: 01/05/2021). All participants signed voluntary written consent letter. Before the data collection, participants were explained the aims and methodology of the study and then were asked to sign a written informed consent letter.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Rostami, S., Alavi, S.M., Daghagheleh, R. et al. A randomized clinical trial investigating the impact of magnesium supplementation on clinical and biochemical measures in COVID-19 patients. Virol J 21, 91 (2024). https://doi.org/10.1186/s12985-024-02362-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12985-024-02362-6