Abstract
Background
French Guiana (FG) is a French overseas territory where malaria is endemic. The current incidence rate is 0.74‰ inhabitants, and Plasmodium vivax is widely predominating even though Plasmodium falciparum is still present due to imported cases mainly from Africa. In FG, rapid diagnostic test (SD Malaria Ag P.f/Pan®) is based on the detection of pan-pLDH, PfHRP2, and PfHRP3 antigens, while in South America, the share of deletion of PfHRP2 gene is significantly increasing. Accordingly, the study questions the reliability of RDTs in the Amazonian context.
Methods
The study is retrospective. It is conducted over 4 years and analysed 12,880 rapid diagnostic tests (RDTs) compared to concomitant Blood Film Tests (BFTs) sampled for malaria diagnosis.
Results
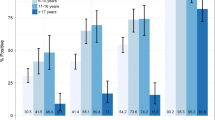
The global assessment of the accuracy of SD Malaria Ag P.f/Pan® in the diagnostic of malaria shows both Positive and Negative Predictive Values (PPV and NPV) higher than 95%, except for PPV in the diagnosis of malaria to P. falciparum (88%). Overall, the concordance rate between RDT and BFT (positive/positive; negative/negative) was 99.5%. The PPV of the RDT in the follow-up of patients diagnosed with P. falciparum was the lowest during the first 28 days. The PPV of the RDT in the follow-up of patients diagnosed with P. vivax was the lowest during the first 21 days. The global sensitivity of SD Malaria Ag P.f/Pan® test was, on average, 96% (88.2–100) for P. falciparum and 93% (90.6–94.2) for P. vivax. The global specificity was 99.8% (99.5–100) for all included species.
Conclusion
SD Malaria Ag P.f/Pan® is a reliable rapid test used for the first-line diagnosis in remote healthcare centres. The test results should be interpreted in the light of patient’s recent medical history and the date of arrival to FG.
Similar content being viewed by others
Background
WHO classified malaria as a public health concern. In 2017, malaria was the most prevalent parasitic disease, with 1.4 billion people at risk worldwide [1]. The year after, the number of malaria cases was estimated at 228 million, with 405,000 recorded deaths worldwide, mostly in the African continent. In the Americas, the malaria incidence has been increasing since 2016, mainly due to the epidemic situation in Venezuela [2, 3] with 75% of cases caused by Plasmodium vivax [2]. In French Guiana (FG) malaria is endemic [4]. Since the 1950s, efforts to fight malaria coupled with the control strategies implemented in the Guiana Shield [5] have significantly reduced the incidence of the disease [6]. Indeed, the malaria incidence recorded in 2019 in FG was the lowest during the ten previous years (0.74‰ inhabitants) [7].
Previously in FG, Plasmodium falciparum was accountable for the majority of cases. However, within the last 20 years, the distribution of Plasmodium species changed with a vast rate of diagnosed P. vivax [6, 8]. Despite a very encouraging figure with a very low malaria transmission rate in the coastal and urban areas, the risk of malaria remains endemic in FG. The country's inland sites continue to experience high levels, mainly in socially marginalized and isolated populations.
Among laboratory methods used for malaria parasite detection and exposure, only rapid diagnostic tests (RDT) and blood films tests (BFT) are currently available for daily practice. Molecular detection of low-density P. falciparum infections is a crucial point for surveillance studies to steer malaria control strategies in countries where malaria is near to almost elimination [9]. RDT is based on the detection of pan-pLDH, PfHRP2 and PfHRP3 antigens. However, there is an increasing rate of deletion of the PfHRP2 gene worldwide, threatening the ability to diagnose patients infected with P. falciparum, and causing false negative RDT results [10]. In South America, the distribution of PfHRP2 gene deletion varies with free countries and areas where the deletion rate is over 30% [11]. In FG, the RDT used is SD Malaria Ag P.f/Pan® and P. falciparum can present PfHRP3 deletion (4.5% of cases) but no PfHRP2 deletion [12].
This study aims to assess the relevance of SD Malaria Ag P.f/Pan® in the diagnosis of malaria in French Guiana.
Methods
Our study is a retrospective analysis conducted over 4 years (January 2016 to December 2019) in the microbiology laboratory of the Cayenne General Hospital. It includes all RDT and BFT sampled for malaria diagnosis. Cayenne General Hospital is a 742-beds health facility that provides first-line medical care for an urban population of 150,000 inhabitants. It manages 18 delocalized prevention and healthcare centers providing care for almost 50,000 inhabitants. Thereby, it is also a referral centre for a larger population from all over French Guiana and other border countries [13].
Data collection
Data were collected from the computerized database of the microbiology laboratory of the Cayenne General Hospital. They include the date of the tests, and the results of the RDT and the concomitant BFT. In a first step, files with a RDT and a concomitant BFT were included and files with a BFT without a concomitant RDT were excluded. Also files where concomitant RDT/BFT were sampled during the six months following the malaria diagnosis were separately analysed.
Microbiological technique
RDT was based on the SD Malaria Ag P.f/Pan® (Standard Diagnostics Inc.) which detects the presence of pan-pLDH and PfHRP2 antigens [12]. BFT is based on microscopic examination of blood and represents the gold standard for the malaria diagnosis [14]. Two sorts of blood film are traditionally used. Thick films allow the screening of a larger blood volume and are about eleven times more sensitive than thin films. It enables the diagnosis of infection with a low level of parasites. In contrast, thin films allow better identification of the responsible parasite. Both smears are recommended when attempting to make a definitive diagnosis of malaria [15].
Thick and thin blood films are prepared within one hour of blood collection. Thick blood films are stained with Giemsa diluted at 10%, while thin blood films are stained using a rapid method (RALH 555, RAL Diagnostics). Two hundred fields of the thin blood film are examined before classifying the thin smear-negative, and 1000 counted white blood cells (WBCs) from the thick smear are observed before classifying the sample as negative [12]. The parasite density estimation is based on an assumed 6000 WBC/ml of blood [12].
Statistical analysis
Results are reported as mean and standard deviation, or numbers and percentages. We calculated the sensitivity (Ss), specificity (Sp), positive and negative predictive values (PPV and NPV), Youden test, and the Q coefficient of Yule to assess the diagnostic accuracy of RDT in the diagnosis of confirmed malaria by BFT. All statistical analyses were carried out using Excel (2010 Microsoft corporation, Redmond, USA) and IBM SPSS Statistics for Windows, version 24 (IBM Corp., Armonk, NY, USA).
Ethical consideration
The study is retrospective that did not require individual consent according to the French law regarding research conforming to MR-003 (JORF no. 0160 du 13 juillet 2018. texte no. 109). The database has been registered at the Commission National de l'Informatique et des Libertés (registration no 2219819), in compliance with French law on electronic data sources.
Results
During the study period, 12,984 samples of blood films for the diagnostic of malaria were recorded. A concomitant BFT and RDT were sampled in 12,880 cases. Amongst them, 10,873 files (84.4%) fulfilled our inclusion criteria (Fig. 1).
The average number of RDTs and BFTs per year was 2718 ± 394 tests. The average number of RDTs and BFTs per month was 1073 ± 105 tests. RDT was performed in 10,873 cases and was positive in 773 cases (7.1%) with identification of P. falciparum in 125 cases (16.1% of positive tests) and P. vivax in 648 cases (83.9% of positive tests). BFT was performed in 10,873 cases and was positive in 791 cases (7.3%) with identification of P. falciparum in 105 cases (13.3% of positive tests), P. vivax in 673 cases (85.1% of positive tests), both of them in 10 cases (1.3% of positive tests), and Plasmodium ovale in 3 cases (0.4% of positive tests).
The global assessment of the accuracy of RDT in the diagnosis of malaria shows an overall PPV and NPV higher than 95%, except for PPV in the diagnosis of malaria to P. falciparum (88%). Figure 2 shows the accuracy of RDT in the diagnosis of malaria independently of the Plasmodium species identification.
The yearly assessment of the accuracy of RDT in the diagnosis of malaria to P. falciparum and P. vivax is reported in Table 1. The yearly assessment of the accuracy of RDT in the diagnosis of malaria to P. falciparum shows a drop-down in the PPV in 2017, but an increase after that (Fig. 3).
Overall, the concordance rate between RDT and BFT (positive/positive; negative/negative) was 99.5%. It was the highest in 2016 (99.8%) and the lowest in 2017 (99.1%) (Fig. 4). The concordance rate between RDT and BFT in the diagnosis of P. falciparum was 99.8%. It was at 99.6% in 2017 (the lowest) and 100% in 2018–2019. The concordance rate between RDT and BFT in the diagnosis of P. vivax was 99.3%. It was at 99.1% in 2017 (the lowest) and 99.5% in 2016 and 2019.
During the study period, RDTs and BFTs were performed as part of the follow-up (in patients previously diagnosed with malaria) in 2007 cases (15.6%). The PPV value of RDT in this context was the lowest during the 42 days of follow-up (Fig. 5). The PPV of the RDT in the follow-up of patients diagnosed with P. falciparum was the lowest during the 28 first days. The PPV of the RDT in the follow-up of patients diagnosed with P. vivax was the lowest during the 21 first days (Table 2).
Discussion
The objective of the study was to assess the accuracy of SD Malaria Ag P.f/Pan® test, which is the RDT currently used in remote healthcare centers in FG, in the rapid diagnosis of malaria. The SD Malaria Ag P.f/Pan® test was sufficiently accurate in diagnosing malaria in suspected patients, in routine monitoring, and in detecting passive cases in malaria low transmission areas [16, 17]. Please move to references Rapid diagnosis of malaria is essential to introduce early curative treatment and prevent a severe outcome. High-quality RDTs may be used, as a first measure, thanks to their efficiency, large availability, and low cost [18]. In FG remote areas, the only way to quickly diagnose malaria disease remains RDTs. The choice of the SD Malaria Ag P.f/Pan® was based on the comparison of performance provided by the WHO reports [16, 17].
In FG, the implementation of malaria control strategies allowed to reduce the number of recorded cases by 82% during the last years [6, 19]. In 2011, among the 1209 reported cases, P. falciparum and P. vivax represented 31% and 68.5%, of cases, respectively [12]. In 2019, among the 212 recorded malaria cases, 6% were caused by P. falciparum, 42 cases were hospitalized, and only 2 cases developed a complicated clinical picture (one with P. falciparum and one with P. vivax) [20]. In the current study, the average share of confirmed P. falciparum malaria cases decreased significantly with 13.3% of positive tests (minus 17.7%), while confirmed P. vivax cases increased to 85.1% of positive tests (plus 16.6%).
The current study analysed all performed RDTs and compared them to BFTs regardless of whether they were first-line diagnostic tests or monitoring tests during treatment. The global sensitivity of SD Malaria Ag P.f/Pan® test was 96% (88.2–1) for P. falciparum and 93% (90.6–94.2) for P. vivax. The sensitivity can be affected by a low rate of parasitaemia [17]. The global specificity was high, with a rate of 99.8% (99.5–1) for all species included. In our database, among 12,880 concomitant RDT and BFT, 10,873 (84.4%) represented first-line diagnosis tests, and 2007 RDTs were performed during the follow-up period. The latter affected results of the VVP, which was on average 97% (94.5–98.9), but significantly different for P. falciparum and P. vivax with 88% (80.4–1) and 98% (94.7–99.1), respectively.
Most of RDTs carried during the follow-up period were performed during the two first years of the study, which can explain the annual variation of the PPV. Furthermore, there is a significant variation of the global PPV week after week during the first 28 days of follow-up. It is also explained by the number of false positive cases related to the persistence of the protein-encoding for Plasmodium in the blood. In this study, we have not collected clinical information. Consequently, immunological factor or infectious agents explaining the calculated PPV were not investigated [21, 22]. Thus, a positive result of the RDT must be confirmed by a BFT. Otherwise, there is a risk of underdiagnosing malaria.
The NPV of SD Malaria Ag P.f/Pan® in the malaria diagnosis was, 99.7% (99.3–99.5) without a significant difference between P. falciparum and P. vivax. It has been reported that a low parasitaemia could affect the accuracy of the test with false-negative results [23], but this was not the case in this study. Consequently, the negativity of SD Malaria Ag P.f/Pan®, used as a first-line diagnosis test, allows to reasonably rule out malaria even though performing BFT remains compulsory.
In the current study, 2007 concomitant RDT/BFT were carried during the follow-up period of patients under treatment. The results have shown that the RDT remains positive up to 28 days, even though parasites were no longer detectable with BFT. Previous findings suggest that PfHRP2 RDTs remain positive after treatment for longer than the combined or pLDH RDTs [24]. This is explained by a slower degradation of PfHRP2 compared to pLDH after the parasite elimination [24, 25]. Indeed, PfHRP2 antigen, is progressively eliminated from the blood and can be responsible for false positives [26, 27]. Contrarily, pLDH is quickly eliminated from the blood within 1 week of treatment [28]. Thus, the initial parasite density influence false-positive results and PfHRP2 persistence [25].
Furthermore, RDTs have a higher probability of remaining positive in patients receiving artemisinin-based combination therapy (ACT) than in those receiving non-ACT drugs [25]. In practice, the persistence of positivity of RDT after treatment makes it irrelevant for monitoring. Thus, the SD Malaria Ag P.f/Pan® test should not be used to assess the efficacy of the treatment set up in these conditions.
During the last 2 years, the majority of malaria cases in FG were autochthonous (83% first-quarter 2019, 78% first-quarter 2020) while the remaining cases were from Brazil (range 9 to 14%), Suriname (range 2 to 5%), or Africa (range 2 to 7%) [7]. Illegal gold miners (garimpeiros) inside the rainforest and native populations on border areas between Suriname and Brazil are at risk of malaria resurgence [6, 29, 30]. The epidemic of malaria experienced a sharp increase in 2017 on the Amazonian border between FG and Brazil. Multifactorial causes were pointed out, such as migration from Brazil and Venezuela and related issues, local politics, logistics issues, amongst others [31]. Most cases occurred in forested areas except Saint Georges d' Oyapock on the Brazilian border [29, 32]. In FG, the malaria incidence is low, so all patients with positive RDT are considered as recently infected. However, in 2015, Orpal-1, a study carried out on a population of 421 gold miners, has shown that the wide majority of malaria cases were Brazilian citizens (93.8%). This study has shown that the malaria prevalence in asymptomatic carriers, researched by PCR, ranged from 22.3 to 84%. Species identified were P. falciparum and P. vivax, 47.9% and 37.2%, respectively with 10.6% co-infections. Thus, there is a risk of periodic reintroduction of the disease in FG [33]. It is estimated that illegal gold miners are around 10,000 people. Thus, Orpal-1 investigated only 4.2% of the garimpeiros and might not be representative.
It is worth noting that, in the Brazilian Amazon basin, the lack of PfHRP2 protein varies from region to region. It is the highest in Acre state (31.6%) and absent in Para state [34]. However, P. falciparum HRP3 deletion was recorded in 100% of cases [11]. In 2013, CDC reported PfHPR2 deletion in 14% of cases in Brazil, 14.1% in Suriname, 33.3% in Peru, 7.5% in Colombia and 4% in Bolivia [35]. Moreover, there is no accurate data about imported malaria cases in FG, the origin of immigration waves and if patients come from endemic countries with a significant rate of PfHPR2 deletion. In theory, this can represent a risk of false-negative tests that should be better estimated by further studies [36].
Conclusion
SD Malaria Ag P.f/Pan® is a reliable rapid test used for the first-line diagnostic of malaria in remote healthcare centres in FG. However, it must no longer be used during the follow-up period of patients diagnosed with malaria. The test reading should be interpreted with caution while considering the recent medical history of patients and their arrival date in FG. BFT must always confirm RDT results. In FG, uncontrolled migratory flows might increase the risk of importing new variants of Plasmodium and impact the efficiency of the test.
Availability of data and materials
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- ACT:
-
Artemisinin-based combination therapy
- BFT:
-
Blood Film Tests
- CDC:
-
Centers for Disease Control and Prevention
- FG:
-
French Guiana
- FN:
-
False negative
- FP:
-
False positive
- IT:
-
Information Technology
- Nb:
-
Number
- NPV:
-
Negative Predictive Value
- PPV:
-
Positive Predictive Value
- RDT:
-
Rapid diagnosis tests
- Sp:
-
Specificity
- Ss:
-
Sensitivity
- TN:
-
True negative
- TP:
-
True positive
- WBC:
-
White blood cells
- WHO:
-
World Health Organization
References
WHO. World malaria report 2018. Geneva, World Health Organization, 2018 [Internet]; [cited 2020 Sep 13]. Available from: http://www.who.int/malaria/publications/world-malaria-report-2018/report/en/
WHO. World malaria report 2019. Geneva, World Health Organization, 2019. [Internet]; [cited 2020 Sep 13]. Available from: https://www.who.int/publications-detail-redirect/9789241565721
Plan Maestro para el fortalecimiento de la respuesta al VIH, la tuberculosis y la malaria en la República Bolivariana de Venezuela desde una perspectiva de salud pública - Venezuela (Bolivarian Republic of) [Internet]. ReliefWeb. [cited 2020 Sep 13]. Available from: https://reliefweb.int/report/venezuela-bolivarian-republic/plan-maestro-para-el-fortalecimiento-de-la-respuesta-al-vih-la
Tarantola A, Eltges F, Ardillon V, Lernout T, Sissoko D, Kendjo E, et al. Malaria in France: mainland and territories. Med Mal Infect. 2011;41:301–6.
Breeveld FJV, Vreden SGS, Grobusch MP. History of malaria research and its contribution to the malaria control success in Suriname: a review. Malar J. 2012;11:95.
Musset L, Pelleau S, Girod R, Ardillon V, Carvalho L, Dusfour I, et al. Malaria on the Guiana Shield: a review of the situation in French Guiana. Mem Inst Oswaldo Cruz. 2014;109:525–33.
ARS Guyane. Surveillance du paludisme 1er trimestre 2020 : du 30 décembre 2019 au 29 mars 2020. Le Point Epidemio cellule Guyane. 2020;
Carme B, Ardillon V, Girod R, Grenier C, Joubert M, Djossou F, et al. [Update on the epidemiology of malaria in French Guiana](in French). Med Trop (Mars). 2009;69:19–25.
Grossenbacher B, Holzschuh A, Hofmann NE, Omar KA, Stuck L, Fakih BS, et al. Molecular methods for tracking residual Plasmodium falciparum transmission in a close-to-elimination setting in Zanzibar. Malar J. 2020;19:50.
Molina-de la Fuente I, Pastor A, Herrador Z, Benito A, Berzosa P. Impact of Plasmodium falciparum pfhrp2 and pfhrp3 gene deletions on malaria control worldwide: a systematic review and meta-analysis. Malar J. 2021;20:276.
Rachid Viana GM, Akinyi Okoth S, Silva-Flannery L, Lima Barbosa DR, Macedo de Oliveira A, Goldman IF, et al. Histidine-rich protein 2 (pfhrp2) and pfhrp3 gene deletions in Plasmodium falciparum isolates from select sites in Brazil and Bolivia. PLoS One. 2017;12:e0171150.
Trouvay M, Palazon G, Berger F, Volney B, Blanchet D, Faway E, et al. High performance of histidine-rich protein 2 based rapid diagnostic tests in French Guiana are explained by the absence of pfhrp2 gene deletion in P. falciparum. PLoS One. 2013;8:e74269.
Kallel H, Resiere D, Houcke S, Hommel D, Pujo JM, Martino F, et al. Critical care medicine in the French Territories in the Americas: current situation and prospects. Rev Panam Salud Publica. 2021;45:e46.
Krafts KP, Hempelmann E, Oleksyn BJ. The color purple: from royalty to laboratory, with apologies to Malachowski. Biotech Histochem. 2011;86:7–35.
Warhurst DC, Williams JEACP. Broadsheet no 148. 1996. Laboratory diagnosis of malaria. J Clin Pathol. 1996;49:533–8.
Willie N, Mehlotra RK, Howes RE, Rakotomanga TA, Ramboarina S, Ratsimbasoa AC, et al. Insights into the performance of SD Bioline Malaria Ag P.f/Pan Rapid Diagnostic Test and Plasmodium falciparum Histidine-Rich Protein 2 gene variation in Madagascar. Am J Trop Med Hyg. 2018;98:1683–91.
WHO. Malaria rapid diagnostic test performance. Results of WHO product testing of malaria RDTs: round 7 (2015–2016). Geneva, World Health Organization. [Internet]; [cited 2020 Sep 13]. Available from: http://www.who.int/malaria/publications/atoz/978924151268/en/
WHO. Malaria microscopy quality assurance manual. Version 2. Geneva: World Health Organization; 2016.
Pommier de Santi V, Dia A, Adde A, Hyvert G, Galant J, Mazevet M, et al. Malaria in French Guiana linked to illegal gold mining. Emerg Infect Dis. 2016;22:344–6.
Rousseau C, Andrieu A, Carvalho L, Guidarelli M, Labonte C, Prudhomme J, et al. Surveillance du paludisme 1er trimestre 2020 : du 30 décembre 2019 au 29 mars 2020. Guyane: Santé Publique France; 2020 Apr p. 2. Report No.: 1.
Lee J-H, Jang JW, Cho CH, Kim JY, Han ET, Yun SG, et al. False-positive results for rapid diagnostic tests for malaria in patients with rheumatoid factor. J Clin Microbiol. 2014;52:3784–7.
Mouatcho JC, Goldring JPD. Malaria rapid diagnostic tests: challenges and prospects. J Med Microbiol. 2013;62:1491–505.
Natama HM, Ouedraogo DF, Sorgho H, Rovira-Vallbona E, Serra-Casas E, Somé MA, et al. Diagnosing congenital malaria in a high-transmission setting: clinical relevance and usefulness of P. falciparum HRP2-based testing. Sci Rep. 2017;7:2080.
Dalrymple U, Arambepola R, Gething PW, Cameron E. How long do rapid diagnostic tests remain positive after anti-malarial treatment? Malar J. 2018;17:228.
Houzé S, Boly MD, Le Bras J, Deloron P, Faucher J-F. Pf HRP2 and Pf LDH antigen detection for monitoring the efficacy of artemisinin-based combination therapy (ACT) in the treatment of uncomplicated falciparum malaria. Malar J. 2009;8:211.
Iqbal J, Siddique A, Jameel M, Hira PR. Persistent histidine-rich protein 2, parasite lactate dehydrogenase, and panmalarial antigen reactivity after clearance of Plasmodium falciparum monoinfection. J Clin Microbiol. 2004;42:4237–41.
Biswas S, Tomar D, Rao DN. Investigation of the kinetics of histidine-rich protein 2 and of the antibody responses to this antigen, in a group of malaria patients from India. Ann Trop Med Parasitol. 2005;99:553–62.
Abba K, Kirkham AJ, Olliaro PL, Deeks JJ, Donegan S, Garner P, et al. Rapid diagnostic tests for diagnosing uncomplicated non-falciparum or Plasmodium vivax malaria in endemic countries. Cochrane Database Syst Rev. 2014;CD011431.
Douine M, Musset L, Corlin F, Pelleau S, Pasquier J, Mutricy L, et al. Prevalence of Plasmodium spp. in illegal gold miners in French Guiana in a hidden but critical malaria reservoir. Malar J. 2015;2016(15):315.
Ardillon V, Carvalho L, Prince C, Abboud P, Djossou F. Bilans 2013 et 2014 de la situation du paludisme en Guyane. p 16–20. [Internet]. Bulletin de veille sanitaire Antilles–Guyane; 2015. Available from: http://www.invs.sante.fr/fr/Publications-et-outils/Bulletin-de-veille-sanitaire/Tous-les-numeros/Antilles-Guyane/Bulletin-de-veille-sanitaire-Antilles-Guyane.-n-1-Janvier-2015
Mosnier E, Roux E, Cropet C, Lazrek Y, Moriceau O, Gaillet M, et al. Prevalence of Plasmodium spp. in the Amazonian border context (French Guiana-Brazil): associated factors and spatial distribution. Am J Trop Med Hyg. 2020;102:130–41.
Mosnier E, Dusfour I, Lacour G, Saldanha R, Guidez A, Gomes MS, et al. Resurgence risk for malaria, and the characterization of a recent outbreak in an Amazonian border area between French Guiana and Brazil. BMC Infect Dis. 2020;20:373.
Cohen JM, Smith DL, Cotter C, Ward A, Yamey G, Sabot OJ, et al. Malaria resurgence: a systematic review and assessment of its causes. Malar J. 2012;11:122.
Viana GMR, Silva-Flannery L, Lima Barbosa DR, Lucchi N, do Valle SCN, Farias S, et al. Field evaluation of a real time loop-mediated isothermal amplification assay (RealAmp) for malaria diagnosis in Cruzeiro do Sul, Acre, Brazil. PLoS One. 2018;13:e0200492.
Akinyi Okoth S, Abdallah JF, Ceron N, Adhin MR, Chandrabose J, Krishnalall K, et al. Variation in Plasmodium falciparum Histidine-Rich Protein 2 (Pfhrp2) and Plasmodium falciparum Histidine-Rich Protein 3 (Pfhrp3) gene deletions in Guyana and Suriname. PLoS ONE. 2015;10:e0126805.
Gendrot M, Fawaz R, Dormoi J, Madamet M, Pradines B. Genetic diversity and deletion of Plasmodium falciparum histidine-rich protein 2 and 3: a threat to diagnosis of P. falciparum malaria. Clin Microbiol Infect. 2019;25:580–5.
Acknowledgements
Not applicable.
Funding
Not applicable.
Author information
Authors and Affiliations
Contributions
JMP: Conceptualization, methodology, project administration, data curation, formal analysis. SL, AF: Data curation. SH, PP: Project administration. DB: Data curation, formal analysis. FD: Conceptualization. HK: Conceptualization, methodology, project administration, data curation, formal analysis. MD: Conceptualization, methodology, project administration. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Since our study is retrospective, it did not require individual consent according to the French law regarding research conforming to MR-003 (JORF no. 0160 du 13 juillet 2018. texte no. 109). Our database has been registered at the Commission National de l'Informatique et des Libertés (registration no 2219819), in compliance with French law on electronic data sources.
Consent for publication
At admission to our hospital, an information booklet was distributed to all patients or their relatives stating that their data can be used for research and publication purposes and that they can object to that. No person was unconsented.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Pujo, J.M., Houcke, S., Lemmonier, S. et al. Accuracy of SD Malaria Ag P.f/Pan® as a rapid diagnostic test in French Amazonia. Malar J 20, 369 (2021). https://doi.org/10.1186/s12936-021-03902-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12936-021-03902-z