Abstract
Background
Breast cancer (BC) is the most common malignant tumor among women worldwide. Tissue transglutaminase 2 (TG2) has been reported as a major player across several types of cancer. However, the effects of TG2 in breast cancer are less known.
Methods
The expression of TG2 in patients with BC was detected by immunochemistry staining and RT-qPCR. The correlation of TG2 expression and clinicopathological factors or overall survival (OS) was analyzed by Chi-square test, Kaplan-Meier, and Cox-regression analysis. The effects of TG2 on cell proliferation and glycolysis were investigated in vivo and in vitro by gain- and loss-of-function experiments.
Result
Both mRNA and protein levels of TG2 were overexpressed in BC tissues and cultured cells. Clinical stage (p = 0.011), molecular subtype (p<0.001) and survival status (p<0.001) were significantly correlated with TG2 expression. Specifically, TG2 expression was positively associated with the clinical stage (r = 0.193, p = 0.005) and OS (r = 0.230, p = 0.001), while negatively associated with molecular subtype (r = − 0.161, p = 0.020). Overexpressed TG2 was a prognostic factor of poor OS by Cox-regression analysis. Gain- and loss-of-function experiments indicated that cell proliferation and glycolysis were regulated by TG2 via the MEK/ERK/LDH pathway. TG2-induced activation of the MEK/ERK/LDH pathway and glycolysis were attenuated by MEK inhibitor U0126.
Conclusion
TG2 is overexpressed in BC, which can serve as an independent prognostic factor for OS. TG2 promotes tumor cell proliferation and increases glycolysis associated with the activation of the MEK/ERK/LHD pathway.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Breast cancer (BC) is the most common malignant tumor among women worldwide and the second leading cause of cancer-related deaths [1]. Despite major advances in the diagnosis and treatment of BC in recent decades, the mechanism underlying tumorigenesis and progression is still unknown. Cancer is a multistep malignancy associated with mutation or abnormal expression of specific oncogenes and/or tumor suppressor genes [2]. The main cancer-promoting signaling pathways in BC are PI3K/AKT/mTOR and RAS/RAF/MEK, which control cell proliferation, invasion, and other biological functions [3, 4]. In addition, the mitogen-activated protein kinase (MEK) and extracellular signal-related protein kinase (ERK) pathway regulate lactate dehydrogenase (LDH) to reprogram cancer cell metabolism, which promotes glycolysis during cancer progression [5, 6]. Studies have revealed that while lactic acid promotes tumor metastasis, elevated LDH as a key enzyme in cancer metabolism acts as a poor prognostic biomarker [7, 8].
Tissue transglutaminase (TG) has been reported as a major player, across several types of cancer. TG can bind and hydrolyze GTP, as well as catalyze the enzymatic transamidation reaction which crosslinks primary amines to glutamine residues. TG adopts two vastly different conformations, depending on whether functioning as a GTP-binding protein or a crosslinking enzyme [9]. To date, nine human TGs have been identified since the discovery of the first TG (TG2 or TGM2) by Heinrich Waelsch in 1957 [10]. Recent studies have shown that TG expression has relation to various biological functions which included extracellular matrix formation, apoptosis, cell migration, cell adhesion, and signal transduction [11]. Among the TGs, TG2 is the most widely distributed and extensively studied. Several reports have supported increased expression of TG2 in multiple cancer types, including BC, associated with poor disease outcome, increased drug resistance, and increased incidence of metastasis [12, 13]. These observations imply that TG2 plays a crucial role in promoting an aggressive phenotype in mammary epithelial cells [14]. However, the underlying mechanism of TG2 in tumors, especially in BC, is less studied.
In the present study, we found that both mRNA and protein levels of TG2 are overexpressed in BC tissues and cultured cells, which also served as a poor prognostic factor of overall survival (OS). Gain- and loss-of-function experiments indicated that cell proliferation and glycolysis were regulated by TG2 via the MEK/ERK/LDH pathway.
Material and methods
Cell lines and reagents
The human breast epithelium cell line MCF-10A was grown in DMED/HAM F12 1:1 mixed medium (Hyclone, Logan, UT, USA), which contains 5% horse serum (Gibco, Billings, MT, USA), hydrocortisone, cholera toxin, insulin (Sigma-Aldrich, St. Louis, MO, USA) and EFG (PeproTech, Cranbury, NJ, USA). The human BC cell lines SK-BR-3, BT-474 and MCF-7 were cultured in RPMI 1640, DMEM F12 or DMEM H21 (Hyclone), respectively, comprising 10% fetal bovine serum (Gibco). All cell lines were obtained from the American Type Culture Collection (Manassas, VA, USA) and cultured in a humidified atmosphere with 5-% CO2 at 37 °C. MEK inhibitor U0126 and DMSO was purchased from Sigma-Aldrich. A final concentration of 50 μM (1:1000, v/v) U0216 or the same volume of DMSO was applied to treat the cells.
Patients and samples
The tissue samples of 210 female patients with BC who had received breast-conserving surgery or modified radical mastectomy during the period from January 2012 to December 2015 at China-Japan Union Hospital of Jilin University were enrolled in our study. The patients who received chemotherapy, radiotherapy, or endocrine therapy before surgery were excluded. The cases with recurrent or metastatic cancer at the time of diagnosis were also eliminated. The last date of follow-up was January 2020. Furthermore, a total of 40 normal or para-tumor tissues were acquired as controls. The study was approved by the Research Ethics Committee of the China-Japan Union Hospital of Jilin University and conducted with written informed consent from all patients.
RT-qPCR
Total RNA extraction was isolated using TRIzol reagent (Ambion, Austin, TX, USA). The reverse transcription was performed with a 5 × PrimeScript RT Master Mix for Real-Time kit (TaKaRa, Tokyo, Japan). qRT-PCR was done using an SYBR® Premix DimerEraser™ Perfect Real-Time kit (TaKaRa). All operating procedures followed the supplier’s instructions. RT-qPCR analyses were done with a Mastercycler (Eppendorf, Hamburg, Germany). The primer sequences were as follow: TGM2, F: 5′- CAG CAG GGC TTT ATC TAC CA-3′, R: 5′- GAT CCC ATC TTC AAA CTG CC-3′; LDHA, F: 5′- TTC CAG TGT GCC TGT ATG G-3′ R: 5′- TTA TCA GTC CCT AAA TCT GGG TG-3′; LDHB, F: 5′- ACA ATA AGA TCA CTG TAG TGG G-3′, R: 5′- CAT CAG CCA GAG ACT TTC C-3′; GAPDH, F: 5′-GAA GGC TGG GGC TCA TTT GCA GGG-3′, R: 5′-GGT GCA GGA GGC ATT GCT GAT GAT-3′. The relative fold-change was normalized to GAPDH and calculated by the 2-ΔΔCt method.
Western blot
All the antibodies were obtained from Abcam (Cambridge, UK), except if stated otherwise. The cells were harvested in a lysis buffer and the proteins were extracted by RIPA buffer. The protein concentration was quantitated using the Bio-Rad (Hercules, CA, USA) method. Proteins (25 μg) were loaded into each well and separated on a 10% SDS-PAGE gel, and transferred to a nitrocellulose membrane. Blots were cut prior to hybridization with antibodies. After blocking with 5% fatty-free milk, the membranes were incubated with either TG2, p-MEK/MEK, p-ERK/ERK, LDHA/B, or GAPDH at a dilution of 1:1000 overnight. Anti-rabbit secondary antibodies were coupled to horseradish peroxidase (Proteintech, Wuhan, China). Immunoreactivity was detected using enhanced chemiluminescence (Beyotime, Shanghai, China) and visualized through autoradiography. Protein levels were normalized against GAPDH and analyzed.
Immunochemistry staining (IHC)
To identify the level of TG2 in BC tissues, IHC staining was performed. Briefly, 3 μm thick sections were deparaffinized, rehydrated and submerged into EDTA for antigen retrieval. All sections were then treated with hydrogen, heated, and incubated in bovine serum albumin. Subsequently, they were incubated with TG2 antibody (Abcam) overnight at 4 °C. Negative controls were employed with normal goat serum. The sections were washed and incubated with secondary antibody and incubated with streptavidin-horseradish peroxidase complex (Invitrogen, Waltham, MA, USA). Each section was immersed in 3-amino-9-ethyl carbazole followed by counterstaining with Mayer’s hematoxylin, dehydrated, and then mounted.
The plasma staining of TG2 was considered positive. The intensity of staining was categorized as follows: 0 (no staining), 1 (light yellow), 2 (yellow-brown), and 3 (brown). The percentage of positive cells were grouped as: 0% = 0, 1–10% = 1, 11–25% = 2, 25–50% = 3, and > 50% = 4. IHC intensity, with a range between 0 and 12, was calculated by the score of the staining indexes which equals the staining intensity × proportion of positive cells. The staining score of TG2 level was grouped as: 0, no level; 1–3, weak; 4–6, mild; and > 6, strong.
Establishment of TG2 overexpression and shRNA knockdown cell colonies
Empty vector (EV), wildtype TG2 for overexpression (OE), scramble control (SC), and shRNA targeting TG2 (knockdown, KD) were constructed into lenti-virus (Hanbio, Shanghai, China). BT-474 cells were infected with wildtype TG2 or EV. The cell line SK-BR-3 was infected with shRNA or SC. Concisely, 2 × 105 cells were incubated with lenti-virus and polybrene (Hanbio) for 8 h. Fresh growth medium was added, and stable cell lines were gained in selected medium containing puromycin. Prepared cells were then used for further analysis as described.
Cell proliferation assay
Cell proliferation was evaluated using a cell counting kit-8 (CCK-8, Meilunbio, Dalian, China). Cells were suspended into a 96-well plate in a 100 μL growth medium. After incubation for 24, 48, and 72 h, CCK-8 was added and further cultured for 4 h. The optical density (OD) value was obtained at 450 nm using a microplate reader (BioTek, Winooski, VT, USA).
Glucose consumption and lactate production
The glucose/lactate assay kit (RSBio, Shanghai, China) was used to detect the levels of glucose and lactate in the cell culture medium following the manufacturer’s instructions. Briefly, 3 × 105 cells/well were seeded into 6-well plates and incubated for 2 days. The medium was then replaced with a fresh medium and incubated for another 2 days. The medium was collected, and the glucose or lactate level was measured at the end of the incubation. The difference between the concentration of glucose or lactate and the controls (0 h) was used to calculate the glucose or lactate content. Glucose consumption, as well as lactate production, was normalized by cell numbers [15].
Gene set enrichment analysis (GSEA)
The mRNA profile data were obtained from the TCGA database and included 1104 BC tissues and 114 normal tissues. The mRNA level of TG2 was divided into a low- or high-level group based on the median ratio. Briefly, gene sets (h.all.v6.2.symbols.gmt) were obtained from the Molecular Signatures Database and analyzed using GSEA v3.0. Normalized enrichment scores (NES) were obtained from 1000 site permutations with a p-value < 0.05 and false discovery rate (FDR) < 0.25 considered statistically significant.
Mice xenograft model
Six female nude mice (Huafukang Bioscience, Beijing, China) aged 4–5 weeks were randomly separated into two groups. SK-BR-3 cells (4 × 106, either SC or KD) were injected into the fourth mammary fat pad of each mouse. The tumor size was measured using calipers every 3 days for 1 month. At the end of the experimental period, the mice xenograft tumors were excised and weighed after euthanasia. Organs such as the brain, heart, lung, and liver were fixed in formalin and pathologically investigated for signs of toxicity and metastatic lesions. All procedures of animal handling were approved by the Laboratory Animal Care and Use Committee of Jilin University.
Statistical analysis
The statistical data were analyzed using SPSS v21.0 (IBM, Armonk, NY, USA). The Chi-square test as well as Spearman’ s correlation analysis was performed for analysis of the relationship between clinicopathological parameters and TG2 expression. Survival curves drawn by the Kaplan-Meier method were compared using the log-rank test. Univariate and multivariate Cox-regressive analyses were performed for further assessment. Student’s t-test or one-way ANOVA was adopted to investigate data among or between groups. A p-value < 0.05 signified statistical significance. All the in vitro experimentations are performed independently at least three times, and the data were expressed as mean ± standard deviation.
Results
TG2 overexpressed in patients with BC and an independent prognostic factor for OS
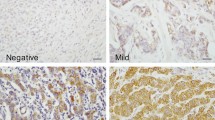
The present study enlisted 40 normal or adjacent samples and 210 BC samples. IHC staining showed negative or weak levels of TG2 in normal or adjacent samples, while positive staining was observed in 183 out of 210 BC samples. According to the intensity of staining, the cohort was divided into two groups: negative to weak staining (0–1+) or mild to strong staining (2–3+) (Fig. 1A–H). In addition, higher levels of TG2 mRNA were confirmed by RT-qPCR in 40 BC samples compared with the paired adjacent samples (Fig. 1I). Clinicopathological parameters were evaluated according to TG2 expression using univariate analysis (Table 1). Clinical stage (p = 0.011), molecular subtype (p<0.001), and survival status (p<0.001) were significantly correlated with TG2 expression. Next, Spearman correlation analysis was employed to estimate the correlation between TG2 expression and clinicopathological characteristics in patients with BC (Table 2). TG2 expression was positively associated with the clinical stage (r = 0.193, p = 0.005) and OS (r = 0.230, p = 0.001), while negatively associated with molecular subtype (r = − 0.161, p = 0.020). Kaplan-Meier analyses indicated that patients with high TG2 expression represented significantly poorer OS (Fig. 1J). Furthermore, univariate and multivariate Cox-regression analysis demonstrated that high expression of TG2 was a significant and independent prognostic factor for OS in patients with BC (Table 3).
TG2 is overexpressed in breast cancer tissues and correlated with poor survival. The representative images of immunochemistry staining of breast cancer tissues for negative (normal tissues, A, B), weak (1+, C, D), mild (2+, E, F) and strong (3+, G, H) TG2 expression. The lower panel represents the magnification (× 200) of squared area from the upper panel (× 80). I the mRNA expression of TG2 was performed in 40 paired adjacent and tumor samples by RT-qPCR. J High expression of TG2 is correlated with poor overall survival in Kaplan-Meier analysis. (* p<0.05)
TG2 promotes BC cell proliferation in vitro and in vivo
To understand the function of TG2 in BC, the mRNA level of TG2 was screened in several cell lines. Compared with normal breast epithelium MCF-10A cells, TG2 expression was downregulated in BT-474, while higher in T47D, ZR-75, MDA-MB-231, MCF-7, and SK-BR-3 cells (Fig. 2A). Next, TG2-KD or OE cell lines were successfully generated in SK-BR-3 (mRNA level decreased by 73% in TG2-KD, p < 0.01) and BT-474 (mRNA level increased 2.13 times in OE, p < 0.01) cell lines, respectively (Fig. 2B–C, Fig. 3B). The cell proliferation ability was increased in TG2-OE (OD450nm at 72 h, EV vs OE: 1.67 ± 0.12 vs 2.34 ± 0.13, p < 0.05) cell line (Fig. 2D), while inhibited in TG2-KD cell line (OD450nm at 72 h, SC vs TG2-KD: 2.87 ± 0.07 vs 2.13 ± 0.09, p < 0.05) (Fig. 2E). Moreover, the xenograft model confirmed the inhibited growth in the TG2-KD cell line (Tumor weight: 1.21 ± 0.08 g vs 0.42 ± 0.07 g, tumor volume: 125.40 ± 8.26 mm3 vs 53.47 ± 5.10 mm3, p < 0.01) (Fig. 2F–H). Besides, no signs of toxicity and metastatic lesions were observed in the brain, heart, lung, liver, and other organs.
TG2 is necessary for breast cancer proliferation in vivo and in vitro. A The mRNA expression of TG2 was detected in breast cancer cell lines. B and C The TG2 knockdown and overexpression cell line was established in SK-BR-3 and BT-474 cells, respectively. D and E The proliferation ability was measured by CCK-8 assay in TG2 overexpression (BT-474) and knockdown (SK-BR-3) cell line. F-H The xenograft model was established in nude female mice by injection SK-BR-3 cells (scramble control [SC] and knockdown [KD]), orthotopically. (* p<0.05, ** p<0.01)
TG2 regulated glycolysis via controlling MEK/ERK/LDH pathway. A GSEA analysis shown that KRAS pathway enriched. B p-MEK, p-ERK, LDHA and LDHB was down-regulated in TG2 knockdown cell line (SK-BR-3), while upregulated in TG2 overexpression cell line (BT-474) by western blot. C and E The mRNA level of LDHA and LDHB was detected by RT-qPCR in TG2 knockdown and overexpression cell lines. D and F The consumption of glucose and production of lactate was measured in TG2 knockdown and overexpression cell lines. (* p<0.05, ** p<0.01)
MEK/ERK/LDH pathway and glycolysis were activated by TG2
To explore the potential mechanism by which TG2 promotes BC cell proliferation, the bioinformatic analysis was used based on public database. GSEA showed that the KRAS pathway was enriched (Fig. 3A). Since MEK/ERK is the main downstream pathway of KRAS, and glycolysis is regulated by LDH via the ERK pathway [16], these key proteins were detected by western blot. Lower phosphorylation of MEK (58% decreased, p < 0.05) and ERK (37% decreased, p < 0.05) were observed in TG2-KD cells (Fig. 3B). Decreased protein and mRNA levels of LDHA (46% decreased, p < 0.05) and LHDB (31% decreased, p < 0.05) was also detected in these cells (Fig. 3B–C). The opposite phenomena were observed in the TG2-OE cell line, in which the higher phosphorylation of MEK (1.65-fold increased, p < 0.05) and ERK (1.78-fold increased, p < 0.05) resulted in increased levels of LDHA (2.77-fold increased, p < 0.01) and LDHB (1.62-fold increased, p < 0.01) (Fig. 3B and E). TG2-OE resulted in increased glycolysis (i.e., increased consumption of glucose and production of lactate by 2.63 ± 0.16 and 2.17 ± 0.87 μmol/106 cells, p < 0.05), whereas TG2-KD decreased glycolysis (i.e., consumption of glucose and production of lactate decreased by 3.9 ± 1.04 and 3.3 ± 0.21 μmol/106 cells, p < 0.05) (Fig. 3D and F, statistical significance for the changes in protein levels are shown in Supplementary Fig. 1). Taken together, these finding demonstrated the MEK/ERK/LDH axis and glycolysis activation by TG2.
TG2-induced activation of MEK/ERK/LDH pathway and glycolysis attenuated by MEK inhibitor U0126
To further confirm our hypothesis that TG2 promotes BC cell proliferation by activation of the MEK/ERK/LDH pathway and reprogramming of cell metabolism, the MEK inhibitor U0126 was used to perform rescue experiments. The proliferation (OD450nm of TG2-OE at 72 h, DMSO vs U0126: 2.74 ± 0.22 vs 1.93 ± 0.16, p < 0.01) and glycolysis (△Glu. and △Lac. of TG2-OE ± U0126: − 5.93 ± 0.17 and 4.97 ± 0.60 μmol/106 cells, respectively, p < 0.01) promoted by TG2 was attenuated by U0126 treatment (Fig. 4A and D). Furthermore, the activation of p-MEK, p-ERK, LDHA and LDHB by TG2-OE was rescued by U0126 (Fig. 4B and Supplementary Fig. 2). The same pattern of mRNA levels of LDHA (64% decreased, p < 0.01) and LDHB (68% decreased, p < 0.01) was detected by RT-qPCR (Fig. 4C).
The proliferation and glycolysis promoted by TG2 was attenuated by MEK inhibitor U0126. A and D The proliferation and glycolysis promoted by TG2 was attenuated by U0126 treatment. B The activation of p-MEK, p-ERK, LDHA and LDHB by overexpressed TG2 was rescued by U0126. C The mRNA level of LDHA and LDHB was detected by RT-qPCR. (* p<0.05, ** p<0.01)
Discussion
Our study found that TG2 was upregulated in both human BC tissues and cultured cells. A high mRNA level of TG2 could serve as an independent prognostic factor of OS. TG2-OE promoted BC cell proliferation in vitro and in vivo. Moreover, glucose consumption and lactate production were shown to be regulated by TG2. All the findings indicated that cell proliferation and glycolysis were induced by TG2 via activation of the MEK/ERK/LDH pathway. This hypothesis was further confirmed by rescue experiments in TG2-OE cells when treated with the MEK inhibitor U0126.
It is reported TG2 is mainly a cytosolic protein but is present on the plasma membrane, in the nucleus, and the extracellular environment [11]. Our findings are consistent with previous reports showing elevated TG2 expression in ovarian cancer [17], lung cancer [18], and glioblastoma [19]. Numerous studies indicated that elevated expression of TG2 in cancer tissues is related to poor survival, drug resistance, and metastasis [1]. For instance, Jiang et al. reported that normal breast tissues expressed low levels of TG2, while node-positive tumors exhibited significantly higher levels of TG2 [20]. Mehta et al. found up-regulated expression of TG2 in MDA-MB-231 cells and the metastatic lymph nodes of patients with BC [21]. Hettasch et al. reported up-regulated expression of TGs in intraductal and invasive BC, which its localization in the neovasculature and extracellular matrix suggested that TGs may be involved in the regulation of tumor growth and metastasis [22]. Furthermore, Kyparidou et al. reported that TG is an independent favorable prognostic factor for OS in a cohort of 68 patients with BC [23]. Although TG2 is extensively studied in many types of cancer, its prognostic role is controversial in BC and the mechanism by which it promotes the progression of BC is less explored. Our data indicated that TG2 may be a novel biomarker for BC.
To further explore the underlying mechanism of TG2 promotion of BC cell proliferation, we found that TG2-OE or -KD respectively elevated or reduced phosphorylation of MEK/ERK. These findings revealed that TG2 may promote BC proliferation by activating the MEK/ERK pathway. Rapidly growing tumor cells acquire genetic and epigenetic alterations to meet their oxygen requirements via upregulation of glycolysis. Once the cells are exposed to various stress stimuli, mechanisms such as metabolic reprogramming and autophagy will be induced to provide nutrients and metabolites to the stressed cells [24, 25]. A continuous supply of energy is required to produce precursors for de novo synthesis of biomacromolecules involved in cell growth and proliferation, including RNA, DNA, amino acids, fatty acids. This energy can be provided by the reprogramming of glucose metabolism. For instance, hypoxia-inducible factor-1 (HIF-1) is a transcription factor that can create a shift in energy production from mitochondria towards glycolytic sources in tumor hypoxia regions, and its upregulation forms the main mechanism of metabolic reprogramming [26]. TG2 reportedly increases the Warburg effect by inducing HIF-1α via NFκB in BC cells, resulting in increased glucose uptake, which could be abolished by KD of HIF-1α, NFκB, or TG2 [27,28,29]. Furthermore, glycolysis in tumor cells is reportedly regulated by LDH through the MEK/ERK pathway [6]. Accordingly, respective higher or lower expression of LDH was also seen in TG2-OE or -KD cells in the present study. This result implicates the involvement of the TG2/MEK/ERK/LDH axis during BC progression.
LDH, with two major subunits LDHA and LDHB, can catalyze a mutual reversible conversion between pyruvic acid and lactic acid. Research has shown that lactate acid, an end product of glycolysis, can versatilely promote tumor growth and participates in the metabolic symbiosis of tumors [30]. Recent studies have shown that the mitochondrial oxidative phosphorylation pathway is downregulated, while glucose consumption and lactate release rate increased in cancer cells, independent of oxygen availability (Warburg effect) [31]. Furthermore, anaerobic glycolysis produces large amounts of lactate, which is a primary substrate for the TCA cycle in cancer cells [32]. It is widely accepted that this metabolic reprogramming promotes the growth and survival of tumors. LDH is a key enzyme involved in cancer metabolism could serve as a prognostic index, as the elevated level is linked to poor prognosis [7, 33]. Previous research on reduced LDH expression has also shown that LDH participated in tumorigenesis, however, the relevant mechanism has not been elucidated [34]. Many molecules, such as MYC, KRAS, and the tumor suppressor TP53, are involved in the regulation of metabolic glycolysis as well as oxidative stress, although their underlying molecular mechanisms also remain unclear [35]. LDH may be an emerging target for cancer therapy in pharmacological approaches among the enzymes involved in glycolysis [36].
Moreover, the presence of TG2 in the extracellular environment is crucial for tumor progression. It is reported that high levels of TG2 and fibrillar fibronectin detected in BC-derived extracellular vesicle promotes BC cell growth in a TG2-dependent model [12]. In addition, fibronectin could promote BC cell metastasis as well [37, 38], while autocrine fibronectin inhibits BC metastasis [39]. It is reported that TG2 forms a complex with fibronectin to play a role in fibronectin-mediated BC cell attachment, growth, and survival functions [40]. However, the interaction between TG2 and fibronectin needs further investigation. Considering the complication of crosstalk between the main signaling pathways and their feedback loops, future research also needs to investigate the interaction between TG2 with RAS/RAF or other pathways.
Another limitation of our study is that the cell lines used for gain- or loss-of-function were based on the level of TG2. BT-474 was isolated from primary tumors which is an HR− and Her-2+ subtype, while SK-BR-3 was isolated from metastatic tissue, which is an HR+ and Her-2+ subtype. As these receptors may be important in the activation of the MEK/ERK/LDH pathway, further study is needed. It is reported that EGFR is mediated by TG2 phosphorylation and suggested the targeting TG2 as a novel strategy to downregulate EGFR signaling [41]. According to these findings, our further plan is exploring the effects of TG2 on HER2 or EGFR, and the sensitivity of tyrosine kinase inhibitors or HER2 target reagents such as trastuzumab etc.
Noticeable, our results showed that MEK inhibitor U0126 decreased p-MEK while MEK is phosphorylated by RAF kinase (Fig. 4B). Xuening et al. reported that phosphorylation of RAF-1 can be directly blocked by compounds specific inhibitors of MEK activation [42]. Kinase suppressor of Ras (KSR-1), a kinase upstream of Raf-1, is a substrate of ERK. U0126 as well as PD 098059 inhibits the phosphorylation activity of KSR-1 at concentrations similar to those that inhibit MEK-1. In addition to its target RAF-1, KSR-1 has been reported to associate with ERK and MEK proteins. Moreover, Eppstein et al. explored MAPK signaling and growth response in three neuroblastoma cell types after U0126 inhibition [43]. p-ERK levels decreased in dose response to U0126 at 1 and 24 hours in all lines. Conversely, p-MEK levels increased with increasing U0126 concentrations at 24 hours in all lines. The shRNA targeting MEK1/2 may be applied for further investigation. In addition, the effect of U0126 on KSR-1 and the association of KSR-1 with MEK/ERK or TG2 in breast cell lines also need more research.
In conclusion, our present study showed that TG2 is overexpressed in BC and acts as an independent prognostic factor for OS. Gain- and loss-of-function experiments indicated that TG2 promotes tumor cell proliferation and increases glycolysis by activating the MEK/ERK/LHD pathway.
Availability of data and materials
All data generated or analyzed during this study are included in this published article.
Abbreviations
- BC:
-
Breast cancer
- CCK-8:
-
Cell counting kit-8
- ERK:
-
Extracellular signal-related protein kinase
- EV:
-
Empty vector
- FDR:
-
False discovery rate
- GSEA:
-
Gene set enrichment analysis
- HIF-1:
-
Hypoxia-inducible factor-1
- IHC:
-
Immunochemistry staining
- KD:
-
Knockdown
- LDH:
-
Lactate dehydrogenase
- MEK:
-
Mitogen-activated protein kinase kinase
- NES:
-
Normalized enrichment scores
- OD:
-
Optical density
- OE:
-
Overexpression
- OS:
-
Overall survival
- SC:
-
Scramble control
- TG:
-
Tissue transglutaminase
References
Agnihotri N, Kumar S, Mehta K. Tissue transglutaminase as a central mediator in inflammation-induced progression of breast cancer. Breast Cancer Res. 2013;15(1):202.
Eckert RL, Fisher ML, Grun D, Adhikary G, Xu W, Kerr C. Transglutaminase is a tumor cell and cancer stem cell survival factor. Mol Carcinog. 2015;54(10):947–58.
Vasan N, Toska E, Scaltriti M. Overview of the relevance of PI3K pathway in HR-positive breast cancer. Ann Oncol. 2019;30(Suppl 10):x3–x11.
Neuzillet C, Tijeras-Raballand A, de Mestier L, Cros J, Faivre S, Raymond E. MEK in cancer and cancer therapy. Pharmacol Ther. 2014;141(2):160–71.
Amann VC, Ramelyte E, Thurneysen S, Pitocco R, Bentele-Jaberg N, Goldinger SM, et al. Developments in targeted therapy in melanoma. Eur J Surg Oncol. 2017;43(3):581–93.
Yuan J, Dong X, Yap J, Hu J. The MAPK and AMPK signalings: interplay and implication in targeted cancer therapy. J Hematol Oncol. 2020;13(1):113.
Xiang J, Zhou L, Zhuang Y, Zhang J, Sun Y, Li S, et al. Lactate dehydrogenase is correlated with clinical stage and grade and is downregulated by si-SAΤB1 in ovarian cancer. Oncol Rep. 2018;40(5):2788–97.
El Hassouni B, Granchi C, Valles-Marti A, Supadmanaba IGP, Bononi G, Tuccinardi T, et al. The dichotomous role of the glycolytic metabolism pathway in cancer metastasis: Interplay with the complex tumor microenvironment and novel therapeutic strategies. Semin Cancer Biol. 2020;60:238–48.
Katt WP, Antonyak MA, Cerione RA. The diamond anniversary of tissue transglutaminase: a protein of many talents. Drug Discov Today. 2018;23(3):575–91.
Sarkar NK, Clarke DD, Waelsch H. An enzymically catalyzed incorporation of amines into proteins. Biochim Biophys Acta. 1957;25(2):451–2.
Eckert RL, Kaartinen MT, Nurminskaya M, Belkin AM, Colak G, Johnson GV, et al. Transglutaminase regulation of cell function. Physiol Rev. 2014;94(2):383–417.
Shinde A, Paez JS, Libring S, Hopkins K, Solorio L, Wendt MK. Transglutaminase-2 facilitates extracellular vesicle-mediated establishment of the metastatic niche. Oncogenesis. 2020;9(2):16.
Chen F, Zhang Y, Varambally S, Creighton CJ. Molecular Correlates of Metastasis by Systematic Pan-Cancer Analysis Across The Cancer Genome Atlas. Mol Cancer Res. 2019;17(2):476–87.
Mangala LS, Fok JY, Zorrilla-Calancha IR, Verma A, Mehta K. Tissue transglutaminase expression promotes cell attachment, invasion and survival in breast cancer cells. Oncogene. 2007;26(17):2459–70.
Zeng Z, Jing D, Zhang X, Duan Y, Xue F. Cyclic mechanical stretch promotes energy metabolism in osteoblast-like cells through an mTOR signaling-associated mechanism. Int J Mol Med. 2015;36(4):947–56.
Wang CW, Purkayastha A, Jones KT, Thaker SK, Banerjee U. In vivo genetic dissection of tumor growth and the Warburg effect. Elife. 2016;5:e18126.
Singer CF, Hudelist G, Walter I, Rueckliniger E, Czerwenka K, Kubista E, et al. Tissue array-based expression of transglutaminase-2 in human breast and ovarian cancer. Clin Exp Metastasis. 2006;23(1):33–9.
Park KS, Kim HK, Lee JH, Choi YB, Park SY, Yang SH, et al. Transglutaminase 2 as a cisplatin resistance marker in non-small cell lung cancer. J Cancer Res Clin Oncol. 2010;136(4):493–502.
Gundemir S, Monteagudo A, Akbar A, Keillor JW, Johnson GVW. The complex role of transglutaminase 2 in glioblastoma proliferation. Neuro-Oncology. 2017;19(2):208–18.
Jiang WG, Ablin R, Douglas-Jones A, Mansel RE. Expression of transglutaminases in human breast cancer and their possible clinical significance. Oncol Rep. 2003;10(6):2039–44.
Mehta K, Fok J, Miller FR, Koul D, Sahin AA. Prognostic significance of tissue transglutaminase in drug resistant and metastatic breast cancer. Clin Cancer Res. 2004;10(23):8068–76.
Hettasch JM, Bandarenko N, Burchette JL, Lai TS, Marks JR, Haroon ZA, et al. Tissue transglutaminase expression in human breast cancer. Lab Invest. 1996;75(5):637–45.
Kyparidou E, Athanasiadis A, Xydakis E, Papadakou M, Panagos G. Correlation of tissue transglutaminase expression on breast cancer tissue with time to relapse, overall survival, and clinical and molecular prognostic factors: a preliminary report. J BUON. 2005;10(1):81–7.
Hardy SD, Shinde A, Wang WH, Wendt MK, Geahlen RL. Regulation of epithelial-mesenchymal transition and metastasis by TGF-beta, P-bodies, and autophagy. Oncotarget. 2017;8(61):103302–14.
Shinde A, Hardy SD, Kim D, Akhand SS, Jolly MK, Wang WH, et al. Spleen Tyrosine Kinase-Mediated Autophagy Is Required for Epithelial-Mesenchymal Plasticity and Metastasis in Breast Cancer. Cancer Res. 2019;79(8):1831–43.
Denko NC. Hypoxia, HIF1 and glucose metabolism in the solid tumour. Nat Rev Cancer. 2008;8(9):705–13.
Kumar A, Xu J, Sung B, Kumar S, Yu D, Aggarwal BB, et al. Evidence that GTP-binding domain but not catalytic domain of transglutaminase 2 is essential for epithelial-to-mesenchymal transition in mammary epithelial cells. Breast Cancer Res. 2012;14(1):R4.
Kumar S, Donti TR, Agnihotri N, Mehta K. Transglutaminase 2 reprogramming of glucose metabolism in mammary epithelial cells via activation of inflammatory signaling pathways. Int J Cancer. 2014;134(12):2798–807.
Rossin F, D'Eletto M, Falasca L, Sepe S, Cocco S, Fimia GM, et al. Transglutaminase 2 ablation leads to mitophagy impairment associated with a metabolic shift towards aerobic glycolysis. Cell Death Differ. 2015;22(3):408–18.
de la Cruz-Lopez KG, Castro-Munoz LJ, Reyes-Hernandez DO, Garcia-Carranca A, Manzo-Merino J. Lactate in the Regulation of Tumor Microenvironment and Therapeutic Approaches. Front Oncol. 2019;9:1143.
Spencer NY, Stanton RC. The Warburg Effect, Lactate, and Nearly a Century of Trying to Cure Cancer. Semin Nephrol. 2019;39(4):380–93.
Hui S, Ghergurovich JM, Morscher RJ, Jang C, Teng X, Lu W, et al. Glucose feeds the TCA cycle via circulating lactate. Nature. 2017;551(7678):115–8.
Burdett N, Desai J. New biomarkers for checkpoint inhibitor therapy. ESMO Open. 2020;5(Suppl 1):e000597.
Van Wilpe S, Koornstra R, Den Brok M, De Groot JW, Blank C, De Vries J, et al. Lactate dehydrogenase: a marker of diminished antitumor immunity. Oncoimmunology. 2020;9(1):1731942.
Pascale RM, Calvisi DF, Simile MM, Feo CF, Feo F. The Warburg Effect 97 Years after Its Discovery. Cancers. 2020;12(10):2819.
Varghese E, Samuel SM, Liskova A, Samec M, Kubatka P, Busselberg D. Targeting Glucose Metabolism to Overcome Resistance to Anticancer Chemotherapy in Breast Cancer. Cancers. 2020;12(8):2252.
Jun BH, Guo T, Libring S, Chanda MK, Paez JS, Shinde A, et al. Fibronectin-Expressing Mesenchymal Tumor Cells Promote Breast Cancer Metastasis. Cancers. 2020;12(9):2553.
Libring S, Shinde A, Chanda MK, Nuru M, George H, Saleh AM, et al. The Dynamic Relationship of Breast Cancer Cells and Fibroblasts in Fibronectin Accumulation at Primary and Metastatic Tumor Sites. Cancers. 2020;12(5):1270.
Shinde A, Libring S, Alpsoy A, Abdullah A, Schaber JA, Solorio L, et al. Autocrine Fibronectin Inhibits Breast Cancer Metastasis. Mol Cancer Res. 2018;16(10):1579–89.
Herman JF, Mangala LS, Mehta K. Implications of increased tissue transglutaminase (TG2) expression in drug-resistant breast cancer (MCF-7) cells. Oncogene. 2006;25(21):3049–58.
Wang Y, Ande SR, Mishra S. Overexpression of phospho mutant forms of transglutaminase 2 downregulates epidermal growth factor receptor. Biochem Biophys Res Commun. 2012;417(1):251–5.
Wang X, Studzinski GP. Phosphorylation of raf-1 by kinase suppressor of ras is inhibited by “MEK-specific” inhibitors PD 098059 and U0126 in differentiating HL60 cells. Exp Cell Res. 2001;268(2):294–300.
Eppstein AC, Sandoval JA, Klein PJ, Woodruff HA, Grosfeld JL, Hickey RJ, et al. Differential sensitivity of chemoresistant neuroblastoma subtypes to MAPK-targeted treatment correlates with ERK, p53 expression, and signaling response to U0126. J Pediatr Surg. 2006;41(1):252–9.
Acknowledgements
The authors would like to thank Professor Yaacov Ben-David (State Key Laboratory for Functions and Applications of Medicinal Plants, Guizhou Medical University, Guizhou, China) and Professor Jorge Filmus (Department of Biological Sciences, Sunnybrook Research Institute, University of Toronto, Ontario, Canada) for reviewing the manuscript, and providing critical insight during the preparation phase.
Funding
This study was supported by an NSFC (China) grant #81372456 (YJL), Program of Jilin Provincial Health Care Committee #2021LC022 (MH), Norman Bethune Program of Jilin University #2020B45 (MH).
Author information
Authors and Affiliations
Contributions
DHX performed the experiment and wrote the manuscript. NX analyzed the data. LS performed the in vivo experiments and provided support with experimental techniques. ZYY contributed to manuscript writing and revision. YJL conceived and designed the study and supervised all the experiments. All authors have read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
This study was approved by the Ethics Committee of China-Japan Union Hospital of Jilin University. Each patient signed informed consent to the use of their data in scientific research. All methods were performed following relevant guidelines and regulations. The study was carried out in compliance with the ARRIVE guidelines and all procedures of animal handling have received approval from the Laboratory Animal Care and Use Committee of Jilin University.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Additional file 1: Supplementary fig. 1.
Densitometric analysis for western bolt: A. TG2 knockdown in SK-BR-3; B. TG2 overexpress in BT-474. (*: p < 0.05). Supplementary fig. 2. Densitometric analysis for western bolt of U0126 treatment in TG2 overexpressed SK-BR-3 cells.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Xu, D., Xu, N., Sun, L. et al. TG2 as a novel breast cancer prognostic marker promotes cell proliferation and glycolysis by activating the MEK/ERK/LDH pathway. BMC Cancer 22, 1267 (2022). https://doi.org/10.1186/s12885-022-10364-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12885-022-10364-2