Abstract
Aims
The aim of our study was to evaluate the relationship between four CFTR variations and the congenital bilateral absence of the vas deferens (CBAVD).
Methods
A systematic search was performed in the literature databases for the case–control studies of CFTR variations with the risk of CBAVD. A total of 29 studies among 1139 controls and 1562 CBAVD patients were gathered for the meta-analyses of three commonly tecsted variations (5T, ΔF508 and M470V) with CBAVD.
Results
Our meta-analyses observed significant associations between CBAVD and all the three variations, including 5T (P < 0.001, OR = 8.35, 95% CI = 6.68-10.43), M470V (P = 0.027, OR = 0.74, 95% CI = 0.60-0.91) and ΔF508 (P < 0.001, OR = 22.20, 95% CI = 7.49-65.79).
Conclusion
In the current study, we demonstrated a significant association between CFTR variations and CBAVD. Our results showed that the 5T variation was a risk factor of CBAVD in French, Spanish, Japanese, Chinese, Iranian, Indian, Mexican and Egyptian populations. CFTR ΔF508 was another important risk factor in Caucasians, including Slovenians, Canadians, Iranians, and Egyptians. In addition, M470V was a protective factor among French, Chinese, Italian and Iranian populations.
Similar content being viewed by others
Introduction
Congenital bilateral absence of the vas deferens (CBAVD, MIM: #277180) is one of the abnormal forms in the male reproductive system, causing obstruction to sperm outflow into the urethra. CBAVD is responsible for 1-2% of male infertility [1]. CBAVD was widely considered as an atypical symptom of cystic fibrosis (CF, MIM: #219700) [2, 3], which was a severe recessive disease characterized by obstructive chronic lung disease, pancreatic disease and abnormal concentrations of electrolytes in the sweat in clinical studies [4]. Approximately 97% of the male CF patients also suffered from CBAVD [5].
CF is a severe genetic disease with a incidence of 1/2500 births every year in Caucasians [6]. CF patients are often carriers of CFTR gene variations [7] that were originally found in CBAVD patients [8, 9]. The CFTR gene encodes a glycosylated transmembrane protein that is a chloride channel conducts the regulation of other transport pathways. CFTR was widely expressed in epithelial cells of exocrine tissues, such as the sweat glands, lungs, and vas deferens [10]. Published studies have shown evidence that abnormal expression of CFTR contributed to the dysfunction of body organs, including the sweat glands, respiratory system and reproductive systems. CBAVD is one of the generally acknowledged monosymptomatic CFTR-related disorders [11].
Located on chromosome 7q31.2, the CFTR gene contains 27 exons and 1945 polymorphisms according to the Cystic Fibrosis Variations Database [12]. There are four commonly tested variations (ΔF508, 5T, M470V and R1117H) in CBAVD.
The abnormal fold of the △F508-CFTR protein triggered the activity of proteasomes for degradation, which caused a lower open probability than normal [13]. Another common variations, IVS8-Tn, had three forms containing five, 7 seven, or 9 nine thymidine residues in this locus, which is considered an incomplete penetrance [14]. The 5T allele especially produces high levels of CFTR transcript without exon 9, resulting in variable phenotypes which were observed in CBAVD or mild CF patients [4]. Experimental evidence indicates that 5T carriers have a decreased splicing efficiency of intron 8 [15, 16]. M470V (1540A/G in exon 10) is in a strong linkage disequilibrium with the 5T locus in the CBAVD population, and has become a commonly tested locus [17–19]. The 470V-CFTR protein has lower function than the 470M-CFTR protein [20]. There were arguments on the role of M470V in the CBAVD, although M470V might increase the penetrance of 5T and lead to an obvious association between 5T and Val470 alleles in CBAVD but not in fertile males.
CBAVD incidence is rare (1-2%) in the patients with male infertility [1]. Most genetic studies only involved a few number of individuals. Meanwhile, there was large ethnic difference for the CFTR variations that were more frequent in Caucasians than in other populations, such as Asians. Meta-analysis has an advantage in the sample size by pooling all the relational studies. In the present study, we performed a comphrehensive search for the eligible case–control studies on the association between CBAVD and three CFTR variations, including F508del, IVS8-Tn, and M470V. The goal of our study was to evaluate the overall contribution of the three CFTR variations to the risk of CBAVD in different ethnic groups through systematic meta-analyses.
Materials and methods
Literature search and study selection
The search terms including CBAVD, SNP or variations and CFTR gene were used in the literature search on the PubMed. We investigated all publications on the association between CFTR variants and CBAVD disease from 1993 to 2013. Reference lists in the harvested literatures were explored for additional relevant studies suitable for inclusion in our meta-analysis. The selection criteria of studies for our meta-analysis were as follows: (1) CBAVD patients with typical CF were excluded; (2) complete data with allele or genotype in the case–control studies was available; (3) only CBAVD diagnosis for studies with physical examination, semen analysis and ultrasonography were considered. The retrieved information consisted of author, year of publication, ethnicity, and the number of participants with different alleles.
Statistical analysis
All the eligible case–control studies were involved in the meta-analysis done by Stata software (version 11.0, Stata Corporation, College Station, TX) [21]. Total odd ratios (OR) and 95% confidence intervals (CIs) were estimated to assess the association between CFTR variations and CBAVD risk. Cochran’s Q statistic and I2 test were used to test heterogeneity in our study. The Fixed-effect model was used for meta-analysis with moderate heterogeneity (I2 < 50%). Otherwise, a random-effect model was adopted for the meta-analysis with significant heterogeneity (I2 > =50%) [22, 23]. Z-test with a two-sided P value < 0.05 was used to determine the significance of the total ORs. The publication bias in the meta-analysis was evaluated by the funnel plot and Egger’s test. Summary analysis of the overall frequency of all the studies was executed to show the frequency for all fou variations [24]. Subgroup analysis was performed by ethnicity to evaluate the effect of different ethnicity on total ORs.
Results
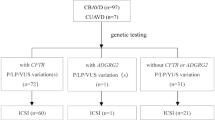
Our comprehensive literature search returned a total of 149 articles on the association between CFTR and CBAVD from PubMed and two studies from CNKI. A total of 104 studies was harvested after being filtered by their titles and abstracts. Among those articles, we removed four family studies and three reviews or meta-analysis. Consequently, 26 studies without obtainable allele or genotype data were removed. Among them, there were 29 studies on three CFTR variations. Our meta-analyses of three CFTR variations were performed among 1139 cases and 1563 controls (Figure 1, Table 1).
A strong association between the CFTR 5T variant and CBAVD was observed in the meta-analysis of 19 studies among 1069 cases and 1419 controls (OR = 8.35, 95% CI = 6.03-10.81, Figure 2). Our results also found that ΔF508 was a risk factor of CBAVD (OR = 22.2, 95% CI = 7.49-65.79, Figure 2). Our meta-analysis showed the M470V variation was associated with CBAVD (OR = 0.74, 95% CI = 0.60-0.91, Figure 2). Future study is required to explore whether there are other causal variants in high linkage disequilibrium with M470V.
As shown in Figure 2, moderate heterogeneity was observed for CFTR 5T (I2 = 44.0%), M470V (I2 = 61.0%) and ΔF508 (I2 = 0.00%). Furthermore, subgroup meta-analyses of the 5T variations by ethnicity showed that the heterogeneity mainly came from Asians (I2 = 71.5%) instead of Caucasians (I2 = 30.0%). Our results indicated that 5T was a risk factor of CBAVD in Caucasians (OR = 8.07, 95% CI = 6.03-10.81, P = 0.00, Figure 2), as well as in Asians (OR = 9.31, 95% CI = 6.42-13.49, P = 0.00, Figure 2). As shown in the funnel plot, no obvious publication bias was observed for the meta-analyses (Figure 3). The results of meta-analyses were robust after the removal of individual studies (Figure 4).
Discussion
In the present study, we carried out a systematic overview of the studies for a genetic link with CBAVD. Significant evidence was observed (that showed) the strong association with CBAVD onset with the risk of CBAVD for all the three variations, including 5T (OR = 8.35, 95% CI = 6.03-10.81, Figure 2), M470V (OR = 0.74, 95% CI = 0.60-0.91, Figure 2) and ΔF508 (OR = 22.2, 95% CI = 7.49-65.79, Figure 2).
Among the tested variations, M470V was the only one with the protective role of CBAVD. V470 carriers tended to have a higher fertility than other carriers, suggesting that there might be competing and evolutionary forces acting on the CFTR gene [25]. Furthermore, the interaction of the variations might affect the risk of CBAVD [26, 27]. In addition, the haplotypes of these variations can significantly contribute to the susceptibility of CBAVD alone or together with the SPINK1 locus [11]. Further detailed studies are required to provide evidence that independent CBAVD rather than CBAVD with CF is associated with CFTR variations.
Several limitations in our meta-analyses were taken into consideration as followed. Firstly, some studies did not reveal whether CBAVD patients were isolated CBAVD patients or patients with CF symptoms for the incomplete information. This might potentially interfere with the results of our meta-analyses. Secondly, the power of our meta-analyses is still low due to low allele frequency of these variations. Under a moderate risk of OR (OR = 1.2), power analysis showed a lack of power in the meta-analyses, including 5T (47.3%), ΔF508 (7.1%) of LPL, and M470V (52.1%). Thirdly, there was a lack of haplotype information among these loci in most of involved studies. Reports have shown there were interactions among these variations [28–30]. We discontinued the further analysis for examining variables, including interaction between the two or three loci.
In conclusion, the present study demonstrated an association between CFTR variants and CBAVD risk. Our meta-analysis among 1069 cases and 1418 controls showed that 5T was a risk factor of CBAVD in the combined populations, including French, Spanish, Japanese, Chinese, Iranian, Indian, Mexican and Egyptian. Meta-analysis among 60 cases and 133 controls confirmed that CFTR ΔF508 was another important risk factor in Caucasian populations including Slovenian, Canadian, Iranian and Egyptian. In addition, M470V was shown to be a protective factor by our meta-analysis among 10 cases and 11 controls from French, Chinese, Italian and Iranian populations.
Abbreviations
- SNP:
-
Single nucleotide polymorphism
- CBAVD:
-
Congenital bilateral absence of the vas deferens
- CF:
-
Cystic fibrosis
- OR:
-
Odds ratio
- 95% CI:
-
95% confidence interval
- I2:
-
Inconsistency index.
References
Grangeia A, Barro-Soria R, Carvalho F, Damas AM, Mauricio AC, Kunzelmann K, Barros A, Sousa M: Molecular and functional characterization of CBAVD-causing mutations located in CFTR nucleotide-binding domains. Cell Physiol Biochem. 2008, 22: 79-92. 10.1159/000149785.
Holsclaw DS, Perlmutter AD, Jockin H, Shwachman H: Genital abnormalities in male patients with cystic fibrosis. J Urol. 1971, 106: 568-574.
Kaplan E, Shwachman H, Perlmutter AD, Rule A, Khaw KT, Holsclaw DS: Reproductive failure in males with cystic fibrosis. N Engl J Med. 1968, 279: 65-69. 10.1056/NEJM196807112790203.
Ghorbel M, Baklouti-Gargouri S, Keskes R, Sellami-Ben Hamida A, Feki-Chakroun N, Bahloul A, Fakhfakh F, Ammar-Keskes L: Screening of DeltaF508 mutation and IVS8-poly T polymorphism in CFTR gene in Tunisian infertile men without CBAVD. Andrologia. 2012, 44 (Suppl 1): 376-382.
Wang Z, Milunsky J, Yamin M, Maher T, Oates R, Milunsky A: Analysis by mass spectrometry of 100 cystic fibrosis gene mutations in 92 patients with congenital bilateral absence of the vas deferens. Hum Reprod. 2002, 17: 2066-2072. 10.1093/humrep/17.8.2066.
Kerem B, Chiba-Falek O, Kerem E: Cystic fibrosis in Jews: frequency and mutation distribution. Genet Test. 1997, 1: 35-39.
Riordan JR, Rommens JM, Kerem B, Alon N, Rozmahel R, Grzelczak Z, Zielenski J, Lok S, Plavsic N, Chou JL, et al: Identification of the cystic fibrosis gene: cloning and characterization of complementary DNA. Science. 1989, 245: 1066-1073. 10.1126/science.2475911.
Dumur V, Gervais R, Rigot JM, Lafitte JJ, Manouvrier S, Biserte J, Mazeman E, Roussel P: Abnormal distribution of CF delta F508 allele in azoospermic men with congenital aplasia of epididymis and vas deferens. Lancet. 1990, 336: 512-
Oates RD, Amos JA: The genetic basis of congenital bilateral absence of the vas deferens and cystic fibrosis. J Androl. 1994, 15: 1-8.
O’Sullivan BP, Freedman SD: Cystic fibrosis. Lancet. 2009, 373: 1891-1904. 10.1016/S0140-6736(09)60327-5.
Steiner B, Rosendahl J, Witt H, Teich N, Keim V, Schulz HU, Pfutzer R, Lohr M, Gress TM, Nickel R, Landt O, Koudova M, Macek M, Farre A, Casals T, Desax MC, Gallati S, Gomez-Lira M, Audrezet MP, Ferec C, Des Georges M, Claustres M, Truninger K: Common CFTR haplotypes and susceptibility to chronic pancreatitis and congenital bilateral absence of the vas deferens. Hum Mutat. 2011, 32: 912-920. 10.1002/humu.21511.
Cystic Fibrosis Mutation Database. [http://www.genet.sickkids.on.ca/cftr/app]
Lukacs GL, Verkman AS: CFTR: folding, misfolding and correcting the DeltaF508 conformational defect. Trends Mol Med. 2012, 18: 81-91. 10.1016/j.molmed.2011.10.003.
Chillon M, Casals T, Mercier B, Bassas L, Lissens W, Silber S, Romey MC, Ruiz-Romero J, Verlingue C, Claustres M, et al: Mutations in the cystic fibrosis gene in patients with congenital absence of the vas deferens. N Engl J Med. 1995, 332: 1475-1480. 10.1056/NEJM199506013322204.
Chu CS, Trapnell BC, Curristin S, Cutting GR, Crystal RG: Genetic basis of variable exon 9 skipping in cystic fibrosis transmembrane conductance regulator mRNA. Nat Genet. 1993, 3: 151-156. 10.1038/ng0293-151.
Teng H, Jorissen M, Van Poppel H, Legius E, Cassiman JJ, Cuppens H: Increased proportion of exon 9 alternatively spliced CFTR transcripts in vas deferens compared with nasal epithelial cells. Hum Mol Genet. 1997, 6: 85-90. 10.1093/hmg/6.1.85.
de Meeus A, Guittard C, Desgeorges M, Carles S, Demaille J, Claustres M: Linkage disequilibrium between the M470V variant and the IVS8 polyT alleles of the CFTR gene in CBAVD. J Med Genet. 1998, 35: 594-596. 10.1136/jmg.35.7.594.
Wu CC, Alper OM, Lu JF, Wang SP, Guo L, Chiang HS, Wong LJ: Variations spectrum of the CFTR gene in Taiwanese patients with congenital bilateral absence of the vas deferens. Hum Reprod. 2005, 20: 2470-2475. 10.1093/humrep/dei077.
Groman JD, Hefferon TW, Casals T, Bassas L, Estivill X, Des Georges M, Guittard C, Koudova M, Fallin MD, Nemeth K, Fekete G, Kadasi L, Friedman K, Schwarz M, Bombieri C, Pignatti PF, Kanavakis E, Tzetis M, Schwartz M, Novelli G, D’Apice MR, Sobczynska-Tomaszewska A, Bal J, Stuhrmann M, Macek M, Claustres M, Cutting GR: Variation in a repeat sequence determines whether a common variant of the cystic fibrosis transmembrane conductance regulator gene is pathogenic or benign. Am J Hum Genet. 2004, 74: 176-179. 10.1086/381001.
Ni WH, Jiang L, Fei QJ, Jin JY, Yang X, Huang XF: The CFTR polymorphisms poly-T, TG-repeats and M470V in Chinese males with congenital bilateral absence of the vas deferens. Asian J Androl. 2012, 14: 687-690. 10.1038/aja.2012.43.
Lian J, Huang Y, Huang RS, Xu L, Le Y, Yang X, Xu W, Huang X, Ye M, Zhou J, Duan S: Meta-analyses of four eosinophil related gene variants in coronary heart disease. J Thromb Thrombolysis. 2013, 36: 394-401. 10.1007/s11239-012-0862-z.
Higgins JP, Thompson SG, Deeks JJ, Altman DG: Measuring inconsistency in meta-analyses. BMJ. 2003, 327: 557-560. 10.1136/bmj.327.7414.557.
DerSimonian R, Laird N: Meta-analysis in clinical trials. Control Clin Trials. 1986, 7: 177-188. 10.1016/0197-2456(86)90046-2.
Yu J, Chen Z, Ni Y, Li Z: CFTR mutations in men with congenital bilateral absence of the vas deferens (CBAVD): a systemic review and meta-analysis. Hum Reprod. 2012, 27: 25-35. 10.1093/humrep/der377.
Kosova G, Pickrell JK, Kelley JL, McArdle PF, Shuldiner AR, Abney M, Ober C: The CFTR Met 470 allele is associated with lower birth rates in fertile men from a population isolate. PLoS Genet. 2010, 6: e1000974-10.1371/journal.pgen.1000974.
Tamburino L, Guglielmino A, Venti E, Chamayou S: Molecular analysis of mutations and polymorphisms in the CFTR gene in male infertility. Reprod Biomed Online. 2008, 17: 27-35. 10.1016/S1472-6483(10)60289-1.
Stuppia L, Antonucci I, Binni F, Brandi A, Grifone N, Colosimo A, De Santo M, Gatta V, Gelli G, Guida V, Majore S, Calabrese G, Palka C, Ravani A, Rinaldi R, Tiboni GM, Ballone E, Venturoli A, Ferlini A, Torrente I, Grammatico P, Calzolari E, Dallapiccola B: Screening of mutations in the CFTR gene in 1195 couples entering assisted reproduction technique programs. Eur J Hum Genet. 2005, 13: 959-964. 10.1038/sj.ejhg.5201437.
Boudaya M, Fredj SH, Haj RB, Khrouf M, Bouker A, Halouani L, Messaoud T: Cystic fibrosis transmembrane conductance regulator mutations and polymorphisms associated with congenital bilateral absence of vas deferens in a restricted group of patients from North Africa. Ann Hum Biol. 2012, 39: 76-79. 10.3109/03014460.2011.642892.
Sun W, Anderson B, Redman J, Milunsky A, Buller A, McGinniss MJ, Quan F, Anguiano A, Huang S, Hantash F, Strom C: CFTR 5T variant has a low penetrance in females that is partially attributable to its haplotype. Genet Med. 2006, 8: 339-345. 10.1097/01.gim.0000223549.57443.16.
Borden W, Berlin S: Gender, coping, and psychological well-being in spouses of older adults with chronic dementia. Am J Orthopsychiatry. 1990, 60: 603-610.
Acknowledgments
The research was supported by the grants from the Shanghai Committee of Science and Technology, China (Grant No. 09411964200), the Major State Basic Research Development Program of China (973 Program, No. 09411964200), National Natural Science Foundation of China (31100919, 81371469 and 81270744), Key Program of Shanghai Municipal Science and technology (10JC1409900, 10JC1410800 and 11411950100), Natural Science Foundation of Zhejiang Province (LR13H020003), Ningbo social development research projects (2012C50032) and K.C. Wong Magna Fund in Ningbo University.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Competing interests
All authors declare no competing financial interests.
Authors’ contributions
XTX drafted the manuscript; JZ and QL were responsible for analysis; HZ and HX participated in literature search. SJ and SD carried out language editing. All authors read and approved the final manuscript.
Xuting Xu, Jufen Zheng contributed equally to this work.
Authors’ original submitted files for images
Below are the links to the authors’ original submitted files for images.
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Xu, X., Zheng, J., Liao, Q. et al. Meta-analyses of 4 CFTR variants associated with the risk of the congenital bilateral absence of the vas deferens. J Clin Bioinform 4, 11 (2014). https://doi.org/10.1186/2043-9113-4-11
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/2043-9113-4-11