Abstract
Background
Genetic tests for hereditary hearing loss inform clinical management of patients and can provide the first step in the development of therapeutics. However, comprehensive genetic tests for deafness genes by Sanger sequencing is extremely expensive and time-consuming. Next-generation sequencing (NGS) technology is advantageous for genetic diagnosis of heterogeneous diseases that involve numerous causative genes.
Methods
Genomic DNA samples from 58 subjects with hearing loss from 15 unrelated Japanese families were subjected to NGS to identify the genetic causes of hearing loss. Subjects did not have pathogenic GJB2 mutations (the gene most often associated with inherited hearing loss), mitochondrial m.1555A>G or 3243A>G mutations, enlarged vestibular aqueduct, or auditory neuropathy. Clinical features of subjects were obtained from medical records. Genomic DNA was subjected to a custom-designed SureSelect Target Enrichment System to capture coding exons and proximal flanking intronic sequences of 84 genes responsible for nonsyndromic or syndromic hearing loss, and DNA was sequenced by Illumina GAIIx (paired-end read). The sequences were mapped and quality-checked using the programs BWA, Novoalign, Picard, and GATK, and analyzed by Avadis NGS.
Results
Candidate genes were identified in 7 of the 15 families. These genes were ACTG1, DFNA5, POU4F3, SLC26A5, SIX1, MYO7A, CDH23, PCDH15, and USH2A, suggesting that a variety of genes underlie early-childhood hearing loss in Japanese patients. Mutations in Usher syndrome-related genes were detected in three families, including one double heterozygous mutation of CDH23 and PCDH15.
Conclusion
Targeted NGS analysis revealed a diverse spectrum of rare deafness genes in Japanese subjects and underscores implications for efficient genetic testing.
Similar content being viewed by others
Background
Hearing loss is a common sensory defect, affecting approximately one in 500 to 1000 newborns [1]. Approximately 50% of congenital hearing loss cases and 70% of childhood hearing loss cases are attributed to genetic mutations [1]. The remaining 50% of congenital cases are attributable to other factors such as prenatal exposure to measles, cytomegalovirus, premature birth, and newborn meningitis. Genetic tests for hereditary hearing loss assist in the clinical management of patients and can provide the first step in the development of therapeutics [2]. For example, early diagnosis of Usher syndrome, which comprises congenital hearing loss and late-onset retinitis pigmentosa, provides important information to choose communication modalities. However, causes of hereditary hearing loss are highly heterogeneous; more than 60 genes have been identified as responsible for nonsyndromic hearing loss [3], and several hundreds of syndromic diseases, such as Pendred syndrome and Usher syndrome, are accompanied by hearing loss. GJB2 mutations are the most common cause of childhood hearing loss worldwide [1], followed by SLC26A4 mutations [4]. OTOF mutations are common in patients with auditory neuropathy, which is characterized by normal outer hair cell function and abnormal neural conduction [5]. The prevalence of childhood hearing loss patients with mutations in other deafness-related genes is likely to be less than 1% [1]. Such high heterogeneity of hearing loss makes it impractical to perform genetic tests by Sanger sequencing. This is also the case for some types of syndromic hearing loss. For example, nine genes have been reported to cause Usher syndrome, and all are large and difficult to analyze using Sanger sequencing.
Next-generation sequencing (NGS) technology has been applied to genetic diagnosis of nonsyndromic hearing loss [6–8] and exploring the causes of hearing loss [9–11]. These studies have revealed that it is technically feasible to identify causative genes for nonsyndromic and syndromic hearing loss using targeted NGS [6, 8]. In this study, we used targeted NGS to identify the genetic basis of hearing loss in Japanese families.
Methods
Subjects
This was a multi-center study of 58 subjects (36 subjects with hearing loss and 22 subjects with normal hearing) from 15 unrelated Japanese families in which at least two family members had bilateral hearing loss. All subjects were patients at the National Hospital Organization Tokyo Medical Center or a collaborating hospital. Medical histories were obtained and physical, audiological, and radiological examinations were carried out for the subjects and family members. Subjects with hearing loss related to environmental factors were excluded. Subjects with GJB2 mutations or mitochondrial m.1555A>G or 3243A>G mutations were excluded. Subjects with enlarged vestibular aqueduct, which is often associated with SLC26A4 mutations, and subjects with clinical features that suggested syndromic hearing loss were excluded. Subjects with auditory neuropathy were tested for OTOF mutations, which are associated with auditory neuropathy [12], and subjects with OTOF mutations were excluded. The Ethics Review Committees of the National Hospital Organization Tokyo Medical Center and all collaborating hospitals approved the study procedures. All procedures were conducted after written informed consent had been obtained from each subject or their parents.
Targeted capture and DNA sequencing
We selected coding exons and proximal flanking intronic sequences of 84 genes, including 17 genes responsible for autosomal dominant nonsyndromic hearing loss (DFNA), 32 genes responsible for autosomal recessive nonsyndromic hearing loss (DFNB), 8 genes responsible for both DFNA and DFNB, one gene responsible for auditory neuropathy, 3 genes responsible for X-linked hearing loss, and 23 genes responsible for syndromic hearing loss. A list of the targeted genes responsible for nonsyndromic or syndromic hearing loss is provided in the supporting material [Additional file 1]. More than 90% of the target genomic sequences were successfully designed to be captured by the SureSelect Target Enrichment System (Agilent Technologies, CA, USA) (data not shown). Genomic DNA was extracted from whole blood using the Genetra Puregene DNA isolation kit (QIAGEN, Hilden, Germany) and checked for quality using Qubit (Life technologies, CA, USA). Genomic DNA (3 μg) was fragmented into approximately 150 base pairs and used to capture the targeted genomic sequences. The captured DNA was subjected to the paired-end read sequencing system (GAIIx system; Illumina, CA, USA).
Sequence analysis
Sequence analysis initially focused on the 61 genes responsible for nonsyndromic hearing loss. If no candidate mutations were detected among these genes, the 23 genes responsible for syndromic hearing loss were subjected to sequence analysis.
The sequences were mapped and quality-checked with the programs BWA, Novoalign, Picard, and GATK using the human reference sequence hg19/GRCh37. Single and multiple nucleotide variants, including small insertion or deletions that would affect amino acid sequences or could affect splice sites, were annotated by Avadis NGS v.1.4.5 (Strand Life Sciences, Bangalore, India). Variants already known as pathogenic mutations or detected with <1% frequency in public databases (dbSNP135 [13], 1000GENOME [14], NHLBI Exome Variant Server [15]) were extracted and further subjected to segregation analysis within each family. If no candidate variants were found, the 23 genes responsible for syndromic hearing loss were subjected to the same procedures.
Selected variants were classified as known mutations, possible pathogenic mutations, or variants with unknown pathogenicity; the latter classification was made if there were reports of a controversial finding of pathogenicity or >1% allele frequency in the in-house database of 95 (up to 189) Japanese subjects with normal hearing. Conservation of the corresponding mutated amino acid was compared across nine primate, 20 mammal, and 13 vertebrate species by UCSC Conservation [16]. Functional pathogenic effects of the variants were predicted by PolyPhen-2 [17] and PROVEAN [18]. Effect on splice-site mutations was predicted by NNSPLICE [19].
All the variants and their segregation in each family were confirmed by Sanger sequencing. The specific primer sets were selected from the resequencing amplicon probe sets (NCBI) or designed originally by Primer-BLAST (NCBI). The genotype of each individual and segregation in the family was characterized using DNASIS Pro (Hitachisoft, Tokyo, Japan).
Structural modeling
To find sequences homologous to ACTG1 and MYO7A that could be used as the structural templates for the modeling exercise, we searched the Protein Data Bank (PDB) using Gapped BLAST [20] and PDBsum [21]. The crystal structure of Limulus polyphemus filamentous actin (PDB: 3B63) and the 4.1 protein-ezrin-radixin-moesin (FERM) domain of Mus musculus myosin VIIa in complex with Sans protein (PDB: 3PVL) were utilized as the templates to model ACTG1 with the p.G268S mutation and MYO7A with the p.W2160G mutation, respectively. The models were built using SWISS-MODEL [22–24] in the automatic modeling mode and with default parameters. The quality of the models was evaluated using the Verify_3D Structure Evaluation Server [25, 26]. The α-carbon frames and ribbon models were superimposed using Chimera [27].
Results
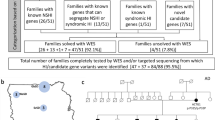
Pedigrees of the seven families are shown in Figure 1; clinical features are described in Table 1 and supplemental materials [Additional file 2 and Additional file 3]. In this targeted NGS study, the mean read depth of the target regions was more than 100× for all subjects (data not shown). Table 2 summarizes the number of variants detected from the 61 or 84 targeted genes for each subject. The number of variants was consistent across subjects (339–435 variants per subject for 61 genes, 539–607 variants per subject for 84 genes), which supported the reproducibility and reliability of our technical procedures and analytical pipeline. After excluding frequent variants (>1%) in public databases, 12 variants of 9 genes co-segregated with symptoms and were selected as possible pathogenic mutations (Table 3) or variants with uncertain pathogenicity in 7 families (Table 4).
Pedigrees of the seven families with hearing loss. Double horizontal bars above a symbol indicate individuals who underwent genetic analysis by targeted next-generation sequencing. Single horizontal bars above a symbol indicate individuals who underwent analysis by Sanger sequencing. A-G denote pedigrees of family 1-7, respectively.
Candidate mutations in each family
In family 1 (Figure 1A), subjects III:3 and IV:2 with hearing loss had a unique heterozygous missense mutation of ACTG1 (c.802G >A; p.G268S), whereas subject III:4 with normal hearing did not. ACTG1 encodes actin gamma 1 and is responsible for DFNA20/26 (OMIM 604717) [28]. The glycine residue at 268 of actin gamma 1 is located on a hydrophobic loop that has been suggested to be critical for polymerization of the actin monomers into a filament (Figures 2A and 2B) [29]. Molecular modeling predicted that the p.G268S mutation would disrupt the hydrophobic interactions that are important for polymerization of actin gamma 1 (Figures 2C and Figure 2D). The p.G268S mutant would weaken polymerization of actin gamma 1, which could result in destabilized cytoskeletal structure of stereocilia and dysfunction of the sensory hair cells.
Molecular modeling of ACTG containing the p.G268S mutation. (A) Ribbon model of filamentous actin gamma 1. (B) Magnified ribbon model of filamentous actin gamma 1. Glycine residue 268 is shown in red and indicated by an arrow. Regions in yellow and green indicate the hydrophobic loop (262–274; a) and the corresponding interactive residues (281–289; b), respectively. (C and D) Vertical views of the regions a and b superimposed with predicted surface hydrophobicity in the wild type (C) and the p.G268S mutant (D).
Family 2 (Figure 1B) had two candidate genes with possible pathogenic mutations: A unique heterozygous POU4F3 frameshift mutation, c.1007delC (p.A336Vfs), was detected in subjects III:1 and IV:3 with hearing loss, and a unique heterozygous DFNA5 nonsense mutation, c.781C >T (p.R261X), was detected in subjects III:2 and IV:3 with hearing loss, whereas subject IV:1 with normal hearing had neither of these mutations. Sanger sequencing revealed that subject IV:2 with hearing loss had both the heterozygous mutations. POU4F3 is responsible for DFNA15 (OMIM 602459) [30, 31], and DFNA5 is responsible for DFNA5 (OMIM 600994) [32]. A frameshift mutation in DFNA5, which would lead to decreased expression, has been reported not to cause hearing loss [33]; therefore, the cause of hearing loss in subjects IV:2 and IV:3 is more likely to POU4F3 with the p.A336Vfs mutation derived from subject III:1, rather than DFNA5 with p.R261X mutation derived from subject III:2.
In family 3 (Figure 1C), subjects III:1 and III:2 with hearing loss had compound heterozygous SLC26A5 with c.209G >A (p.W70X) and c.390A >C (p.R130S) mutations, whereas subjects II:1 and II:2 with normal hearing had a heterozygous p.W70X mutation and a heterozygous p.R130S mutation, respectively. SLC26A5 encodes prestin, a member of the SLC26A/SulP transporter family, and is responsible for DFNB61 (OMIM 613865) [34].
In family 4 (Figure 1D), subjects I:2 and II:1 with hearing loss did not have candidate mutations in the first 61 genes. Analysis of the additional 23 genes indicated a heterozygous SIX1 mutation, c.328C >T (p.R110W), in the subjects with hearing loss but not in subject I:1 with normal hearing. SIX1 is responsible for DFNA23 (OMIM 605192) and Branchio-otic syndrome 3 (BOS3, OMIM 608389). The p.R110W mutation was previously reported in two BOS3 families [35]. To make the clinical diagnosis of branchiootorenal syndrome or branchiootic syndrome, major and minor criteria of these syndromes must be present [36]. In the affected subjects of the present study, clinical histories were thoroughly evaluated and physical examination of the ear, nose, throat, head and neck, and audiological tests were performed. In addition, CT of the temporal bone was evaluated in subject II:1. With these examinations, the affected subjects did not present clinical features of the major and minor criteria other than hearing loss. Therefore, family 4 was considered to have non-syndromic hearing loss, DFNA23, based on the clinical information available at the time of this study.
In family 5 (Figure 1E), subjects III:1 and III:2 with hearing loss had compound heterozygous MYO7A mutations, c.6439-2A >G (intron 51) and c.6478T >G (p.W2160G). Subjects II:2 and II:4 with normal hearing had a heterozygous c.6439-2A >G mutation and a heterozygous p.W2160G mutation, respectively. MYO7A is responsible for DFNA11 (OMIM 601317) [37], DFNB2 (OMIM 600060) [38], and Usher syndrome 1B (OMIM 276900) [39]. Tryptophan 2160 in myosin 7A was found to be located in a carboxyl-terminal FERM domain in the myosin-tail (Figures 3A and Figure 3B); this domain reportedly associates with filamentous actin [40] and contributes to hair bundle formation. Molecular modeling predicted that the p.W2160G mutation would reduce hydrophobic interactions among residues in the center of the F3 subdomain of the FERM domain (Figures 3C and 3D). The p.W2160G mutation would destabilize the structure of the F3 domain and could result in disrupted protein interaction and stereocilial degeneration of the sensory hair cells [41, 42].
Molecular modeling of MYO7A containing the p.W2160G mutation. (A) Structural motif of myosin 7A. Tryptophan 2160 on the C-terminal 4.1 protein-ezrin-radixin-moesin (FERM) domain is indicated by an arrow. Motor, myosin motor domain; IQ, Isoleucine-glutamine calmodulin-binding motif; CC, coiled-coil domain; MyTH4, myosin tail homology 4 domain; SH3, Src homology 3 domain. (B) Ribbon model of the C-terminal FERM domain consisting of three subdomains (F1, F2, F3) and an MyTH4 domain. (C, D) Magnified ribbon model of the F3 subdomain superimposed with predicted surface hydrophobicity in the wild type (C) and the p.W2160G mutant (D).
In family 6 (Figure 1F), subjects II:1 and II:2 with hearing loss had a heterozygous CDH23 mutation, c.719C>T (p.P240L), and a heterozygous PCDH15 mutation, c.848G >A (p.R283H). Sanger sequencing revealed that the other subject with hearing loss (subject II:3) also had both heterozygous CDH23 and PCDH15 mutations. A p.P240L mutation inCDH23 has been reported to be pathogenic [43]. Subject I:1 with normal hearing had a heterozygous mutation in CDH23 (p.P240L), and subject I:2 with normal hearing had a heterozygous mutation in PCDH15 (p.R283H). CDH23 is responsible for both DFNB12 (OMIM 601386) and Usher syndrome 1D (OMIM 601067) [44], whereas PCDH15 is responsible for both DFNB23 (OMIM 609533) and Usher syndrome 1F (OMIM 602083) [45]. Double heterozygous mutations of CDH23 and PCDH15 have been reported to be a digenic cause of hearing loss [46].
In family 7 (Figure 1G), subjects II:1 and II:2 with hearing loss did not have candidate mutations in the first 61 genes. Analysis of the additional 23 genes indicated a compound heterozygous USH2A variant or mutation, c.1346G >A(p.R449H) and c.12431delC (p.A4144GfsX23), in subjects with hearing loss, whereas subjects I:1 and II:2 with normal hearing had a heterozygous p.R449H variant and a heterozygous p.A4144GfsX23 mutation, respectively. USH2A is responsible for Usher syndrome 2A (OMIM 276901) [47]. Although USH2A with the p.R449H variant was not found on dbSNP135, 1000GENOME, or the Exome Variant Server, the allele frequency in Japanese control subjects with normal hearing was 1.3% (5/378).
In the remaining eight families, none of the detected variants co-segregated with hearing loss in the pedigrees (data not shown).
Discussion
In the present study we selected Japanese subjects that had hereditary hearing loss without GJB2 mutations, mitochondrial mutations, enlarged vestibular aqueduct or auditory neuropathy-associated OTOF mutations, and we aimed to detect the spectrum of rare deafness genes in these patients. Targeted NGS for 84 deafness genes resulted in identification of candidate genes in 7 of 15 families and revealed the diverse spectrum of rare deafness genes in Japanese subjects with nonsyndromic hearing loss for the first time. This is the first report of mutations in ACTG1, POU4F3, and SLC26A5 in Japanese families with hearing loss. Families 5, 6, and 7 appeared to have candidate mutations or variants in MYO7A, CDH23, PCDH15, and USH2A, all of which are associated with Usher syndrome [39, 44, 45, 47]. Our results are in contrast to an NGS study of a different ethnic group [48], which showed TMC1 mutations to be the prevalent candidate cause of hearing loss.
For the eight families without candidate genes, hearing loss could be attributable to mutations in non-captured regions including regulatory domains of the 84 genes, other unidentified deafness genes, unknown multigenic causes, copy number variations, or chromosomal structural change.
Double heterozygous mutations
In family 5, double heterozygous mutations of CDH23 and PCDH15 were detected as a candidate cause. This combination of double heterozygous mutations has been reported [46]. Cadherin 23 and protocadherin 15 consist of the upper and lower part of tip link, respectively, which is critical for proper function of mechanotransduction channels on the stereocilia of the sensory hair cells [49]. In addition, P240 of CDH23 is on the extracellular cadherin 1 domain, and R283 of PCDH15 is on the extracellular cadherin 2 domain, which are considered to interact with each other for tip-link bound [49], raising the possibility that the double heterozygous mutations could lead to a destabilized tip-link.
Additional findings of double heterozygous mutations associated with hereditary hearing loss have been reported for KCNJ10 and SLC26A4[50] and for FOXI1 and SLC26A4[51], and some mutated genes may have a modifying effect [52]. Although most NGS pipelines, including ours, focus on identifying monogenic causes of disease, development of a detection strategy for digenic and oligogenic causes of disease should be considered in the future.
Discrimination of mutations from variants
The key challenge for the diagnostic application of NGS is to distinguish causal alleles from the numerous nonpathogenic variants present in each individual. In the present study, for example, the high allele frequency of USH2A with the p.R449H variant in Japanese control subjects implied that pathogenicity of this variant was unlikely. Ethnic diversity of genetic variance has been reported in deafness genes such as OTOF[12] and CDH23[43, 53], and integration of a database of genetic variants with allele frequencies in a specific ethnic group would increase the certainty of the causative nature of genetic mutations by filtering out variants that occur with high frequency. This would facilitate targeted NGS analysis for genetic diagnosis of hearing loss.
References
Morton CC, Nance WE: Newborn hearing screening–a silent revolution. N Engl J Med. 2006, 354: 2151-2164. 10.1056/NEJMra050700.
Kral A, O’Donoghue GM: Profound deafness in childhood. N Engl J Med. 2010, 363: 1438-1450. 10.1056/NEJMra0911225.
Hereditary hearing loss homepage. http://hereditaryhearingloss.org,
Hutchin T, Coy NN, Conlon H, Telford E, Bromelow K, Blaydon D, Taylor G, Coghill E, Brown S, Trembath R, Liu XZ, Bitner-Glindzica M, Mueller R: Assessment of the genetic causes of recessive childhood non-syndromic deafness in the UK - implications for genetic testing. Clin Genet. 2005, 68: 506-512. 10.1111/j.1399-0004.2005.00539.x.
Matsunaga T, Kumanomido H, Shiroma M, Goto Y, Usami S: Audiological features and mitochondrial DNA sequence in a large family carrying mitochondrial A1555G mutation without use of aminoglycoside. Ann Otol Rhinol Laryngol. 2005, 114: 153-160.
Shearer AE, DeLuca AP, Hildebrand MS, Taylor KR, Gurrola J, Scherer S, Scheetz TE, Smith RJ: Comprehensive genetic testing for hereditary hearing loss using massively parallel sequencing. Proc Natl Acad Sci USA. 2010, 107: 21104-21109. 10.1073/pnas.1012989107.
Shearer AE, Smith RJ: Genetics: advances in genetic testing for deafness. Curr Opin Pediatr. 2012, 24: 679-686. 10.1097/MOP.0b013e3283588f5e.
Brownstein Z, Bhonker Y, Avraham KB: High-throughput sequencing to decipher the genetic heterogeneity of deafness. Genome Biol. 2012, 13: 245-10.1186/gb-2012-13-5-245.
Delmaghani S, Aghaie A, Michalski N, Bonnet C, Weil D, Petit C: Defect in the gene encoding the EAR/EPTP domain-containing protein TSPEAR causes DFNB98 profound deafness. Hum Mol Genet. 2012, 21: 3835-3844. 10.1093/hmg/dds212.
Schraders M, Haas SA, Weegerink NJ, Oostrik J, Hu H, Hoefsloot LH, Kannan S, Huygen PL, Pennings RJ, Admiraal RJ, Kalscheuer VM, Kunst HP, Kremer H: Next-generation sequencing identifies mutations of SMPX, which encodes the small muscle protein, X-linked, as a cause of progressive hearing impairment. Am J Hum Genet. 2011, 88: 628-634. 10.1016/j.ajhg.2011.04.012.
Zheng J, Miller KK, Yang T, Hildebrand MS, Shearer AE, DeLuca AP, Scheetz TE, Drummond J, Scherer SE, Legan PK, Goodyear RJ, Richardson GP, Cheatham MA, Smith RJ, Dallos P: Carcinoembryonic antigen-related cell adhesion molecule 16 interacts with alpha-tectorin and is mutated in autosomal dominant hearing loss (DFNA4). Proc Natl Acad Sci USA. 2011, 108: 4218-4223. 10.1073/pnas.1005842108.
Matsunaga T, Mutai H, Kunishima S, Namba K, Morimoto N, Shinjo Y, Arimoto Y, Kataoka Y, Shintani T, Morita N, Sugiuchi T, Masuda S, Nakano A, Taiji H, Kaga K: A prevalent founder mutation and genotype-phenotype correlations of OTOF in Japanese patients with auditory neuropathy. Clin Genet. 2012, 82: 425-432. 10.1111/j.1399-0004.2012.01897.x.
1000GENOME. http://www.1000genomes.org/,
NHLBI exome variant server. http://evs.gs.washington.edu/EVS/,
UCSC conservation. http://genome.ucsc.edu/index.html,
PolyPhen-2. http://genetics.bwh.harvard.edu/pph2/,
PROVEAN. http://provean.jcvi.org/index.php,
Altschul SF, Madden TL, Schaffer AA, Zhang J, Zhang Z, Miller W, Lipman DJ: Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucleic Acids Res. 1997, 25: 3389-3402. 10.1093/nar/25.17.3389.
PDBsum. http://www.ebi.ac.uk/pdbsum/,
Arnold K, Bordoli L, Kopp J, Schwede T: The SWISS-MODEL workspace: a web-based environment for protein structure homology modelling. Bioinformatics. 2006, 22: 195-201. 10.1093/bioinformatics/bti770.
Kiefer B, Riemann M, Buche C, Kassemeyer HH, Nick P: The host guides morphogenesis and stomatal targeting in the grapevine pathogen plasmopara viticola. Planta. 2002, 215: 387-393. 10.1007/s00425-002-0760-2.
Peitsch MC, Tschopp J: Comparative molecular modelling of the Fas-ligand and other members of the TNF family. Mol Immunol. 1995, 32: 761-772. 10.1016/0161-5890(95)00016-8.
Bowie JU, Luthy R, Eisenberg D: A method to identify protein sequences that fold into a known three-dimensional structure. Science. 1991, 253: 164-170. 10.1126/science.1853201.
Luthy R, Bowie JU, Eisenberg D: Assessment of protein models with three-dimensional profiles. Nature. 1992, 356: 83-85. 10.1038/356083a0.
Pettersen EF, Goddard TD, Huang CC, Couch GS, Greenblatt DM, Meng EC, Ferrin TE: UCSF chimera–a visualization system for exploratory research and analysis. J Comput Chem. 2004, 25: 1605-1612. 10.1002/jcc.20084.
Morin M, Bryan KE, Mayo-Merino F, Goodyear R, Mencia A, Modamio-Hoybjor S, del Castillo I, Cabalka JM, Richardson G, Moreno F, Rubenstein PA, Moreno-Pelayo MA: In vivo and in vitro effects of two novel gamma-actin (ACTG1) mutations that cause DFNA20/26 hearing impairment. Hum Mol Genet. 2009, 18: 3075-3089. 10.1093/hmg/ddp249.
Shvetsov A, Musib R, Phillips M, Rubenstein PA, Reisler E: Locking the hydrophobic loop 262–274 to G-actin surface by a disulfide bridge prevents filament formation. Biochemistry. 2002, 41: 10787-10793. 10.1021/bi020205f.
Collin RW, Chellappa R, Pauw RJ, Vriend G, Oostrik J, van Drunen W, Huygen PL, Admiraal R, Hoefsloot LH, Cremers FP, Xiang M, Cremers CW, Kremer H: Missense mutations in POU4F3 cause autosomal dominant hearing impairment DFNA15 and affect subcellular localization and DNA binding. Hum Mutat. 2008, 29: 545-554. 10.1002/humu.20693.
Vahava O, Morell R, Lynch ED, Weiss S, Kagan ME, Ahituv N, Morrow JE, Lee MK, Skvorak AB, Morton CC, Blumenfeld A, Frydman M, Friedman TB, King MC, Avraham KB: Mutation in transcription factor POU4F3 associated with inherited progressive hearing loss in humans. Science. 1998, 279: 1950-1954. 10.1126/science.279.5358.1950.
Bischoff AM, Luijendijk MW, Huygen PL, van Duijnhoven G, De Leenheer EM, Oudesluijs GG, Van Laer L, Cremers FP, Cremers CW, Kremer H: A novel mutation identified in the DFNA5 gene in a Dutch family: a clinical and genetic evaluation. Audiol Neurootol. 2004, 9: 34-46. 10.1159/000074185.
Van Laer L, Meyer NC, Malekpour M, Riazalhosseini Y, Moghannibashi M, Kahrizi K, Vandevelde A, Alasti F, Najmabadi H, Van Camp G, Smith RJ: A novel DFNA5 mutation does not cause hearing loss in an Iranian family. J Hum Genet. 2007, 52: 549-552. 10.1007/s10038-007-0137-2.
Liu XZ: Prestin, a cochlear motor protein, is defective in non-syndromic hearing loss. Hum Mol Genet. 2003, 12: 1155-1162. 10.1093/hmg/ddg127.
Ruf RG, Xu PX, Silvius D, Otto EA, Beekmann F, Muerb UT, Kumar S, Neuhaus TJ, Kemper MJ, Raymond RM, Brophy PD, Berkman J, Gattas M, Hyland V, Ruf EM, Schwartz C, Chang EH, Smith RJ, Stratakis CA, Weil D, Petit C, Hildebrandt F: SIX1 mutations cause branchio-oto-renal syndrome by disruption of EYA1-SIX1-DNA complexes. Proc Natl Acad Sci USA. 2004, 101: 8090-8095. 10.1073/pnas.0308475101.
Smith RJH, et al: Branchiootorenal spectrum disorders. Gene reviews. Edited by: Pagon RA, Adam MP, Bird TD. http://www.ncbi.nlm.nih.gov/books/NBK1380/,
Liu XZ, Walsh J, Tamagawa Y, Kitamura K, Nishizawa M, Steel KP, Brown SD: Autosomal dominant non-syndromic deafness caused by a mutation in the myosin VIIA gene. Nat Genet. 1997, 17: 268-269. 10.1038/ng1197-268.
Liu XZ, Walsh J, Mburu P, Kendrick-Jones J, Cope MJ, Steel KP, Brown SD: Mutations in the myosin VIIA gene cause non-syndromic recessive deafness. Nat Genet. 1997, 16: 188-190. 10.1038/ng0697-188.
Weil D, Kussel P, Blanchard S, Levy G, Levi-Acobas F, Drira M, Ayadi H, Petit C: The autosomal recessive isolated deafness, DFNB2, and the Usher 1B syndrome are allelic defects of the myosin-VIIA gene. Nat Genet. 1997, 16: 191-193. 10.1038/ng0697-191.
Yang Y, Baboolal TG, Siththanandan V, Chen M, Walker ML, Knight PJ, Peckham M, Sellers JR: A FERM domain autoregulates drosophila myosin 7a activity. Proc Natl Acad Sci USA. 2009, 106: 4189-4194. 10.1073/pnas.0808682106.
Adato A, Michel V, Kikkawa Y, Reiners J, Alagramam KN, Weil D, Yonekawa H, Wolfrum U, El-Amraoui A, Petit C: Interactions in the network of usher syndrome type 1 proteins. Hum Mol Genet. 2005, 14: 347-356.
Wu L, Pan L, Wei Z, Zhang M: Structure of MyTH4-FERM domains in myosin VIIa tail bound to cargo. Science. 2011, 331: 757-760. 10.1126/science.1198848.
Wagatsuma M, Kitoh R, Suzuki H, Fukuoka H, Takumi Y, Usami S: Distribution and frequencies of CDH23 mutations in Japanese patients with non-syndromic hearing loss. Clin Genet. 2007, 72: 339-344. 10.1111/j.1399-0004.2007.00833.x.
Bork JM, Peters LM, Riazuddin S, Bernstein SL, Ahmed ZM, Ness SL, Polomeno R, Ramesh A, Schloss M, Srisailpathy CR, Wayne S, Bellman S, Desmukh D, Ahmed Z, Khan SN, Kaloustian VM, Li XC, Lalwani A, Riazuddin S, Bitner-Glindzicz M, Nance WE, Liu XZ, Wistow G, Smith RJ, Griffith AJ, Wilcox ER, Friedman TB, Morell RJ: Usher syndrome 1D and nonsyndromic autosomal recessive deafness DFNB12 are caused by allelic mutations of the novel cadherin-like gene CDH23. Am J Hum Genet. 2001, 68: 26-37. 10.1086/316954.
Ahmed ZM, Riazuddin S, Ahmad J, Bernstein SL, Guo Y, Sabar MF, Sieving P, Riazuddin S, Griffith AJ, Friedman TB, Belyantseva IA, Wilcox ER: PCDH15 is expressed in the neurosensory epithelium of the eye and ear and mutant alleles are responsible for both USH1F and DFNB23. Hum Mol Genet. 2003, 12: 3215-3223. 10.1093/hmg/ddg358.
Zheng QY, Yan D, Ouyang XM, Du LL, Yu H, Chang B, Johnson KR, Liu XZ: Digenic inheritance of deafness caused by mutations in genes encoding cadherin 23 and protocadherin 15 in mice and humans. Hum Mol Genet. 2005, 14: 103-111.
Eudy JD, Weston MD, Yao S, Hoover DM, Rehm HL, Ma-Edmonds M, Yan D, Ahmad I, Cheng JJ, Ayuso C, Cremers C, Davenport S, Moller C, Talmadge CB, Beisel KW, Tamayo M, Morton CC, Swaroop A, Kimberling WJ, Sumegi J: Mutation of a gene encoding a protein with extracellular matrix motifs in usher syndrome type IIa. Science. 1998, 280: 1753-1757. 10.1126/science.280.5370.1753.
Brownstein Z, Friedman LM, Shahin H, Oron-Karni V, Kol N, Abu Rayyan A, Parzefall T, Lev D, Shalev S, Frydman M, Davidov B, Shohat M, Rahile M, Lieberman S, Levy-Lahad E, Lee MK, Shomron N, King MC, Walsh T, Kanaan M, Avraham KB: Targeted genomic capture and massively parallel sequencing to identify genes for hereditary hearing loss in middle eastern families. Genome Biol. 2011, 12: R89-10.1186/gb-2011-12-9-r89.
Sotomayor M, Weihofen WA, Gaudet R, Corey DP: Structure of a force-conveying cadherin bond essential for inner-ear mechanotransduction. Nature. 2012, 492: 128-132. 10.1038/nature11590.
Yang T, Gurrola JG, Wu H, Chiu SM, Wangemann P, Snyder PM, Smith RJ: Mutations of KCNJ10 together with mutations of SLC26A4 cause digenic nonsyndromic hearing loss associated with enlarged vestibular aqueduct syndrome. Am J Hum Genet. 2009, 84: 651-657. 10.1016/j.ajhg.2009.04.014.
Yang T, Vidarsson H, Rodrigo-Blomqvist S, Rosengren SS, Enerback S, Smith RJ: Transcriptional control of SLC26A4 is involved in pendred syndrome and nonsyndromic enlargement of vestibular aqueduct (DFNB4). Am J Hum Genet. 2007, 80: 1055-1063. 10.1086/518314.
Riazuddin S, Castelein CM, Ahmed ZM, Lalwani AK, Mastroianni MA, Naz S, Smith TN, Liburd NA, Friedman TB, Griffith AJ, Riazuddin S, Wilcox ER: Dominant modifier DFNM1 suppresses recessive deafness DFNB26. Nat Genet. 2000, 26: 431-434. 10.1038/82558.
Miyagawa M, Nishio SY, Usami S: Prevalence and clinical features of hearing loss patients with CDH23 mutations: a large cohort study. PLoS One. 2012, 7: e40366-10.1371/journal.pone.0040366.
Acknowledgements
We are grateful to the families who participated in this study and to Dr. Shin Masuda at Hiroshima Prefectural Hospital, Hiroshima, Dr. Tomoko Sugiuchi at Kanto Rosai Hospital, Kanagawa, Dr. Hidenobu Taiji at the National Center for Child Health and Development, Tokyo, and Dr. Hirokazu Sakamoto at Kobe Children’s Hospital, Hyogo, Japan, who collected DNA samples and clinical data from the subjects. This work was supported by a Research on Applying Health Technology grant (H23-013) from the Ministry of Health and Labour and Welfare, Japan and a Grant-in-Aid for Clinical Research from the National Hospital Organization.
Author information
Authors and Affiliations
Corresponding author
Additional information
Competing interests
The authors declare that they have no competing interests.
Authors’ contributions
HM and NS carried out capturing and sequencing the DNA samples, interpreted the data, and drafted the manuscript. CT carried out capturing and sequencing the DNA samples. AS and JK worked on DNA sequencing and interpreting the data. KN carried out molecular modeling of gene products. KKosaki and TM designed the study and interpreted the data. NM, KKaga, and TM contributed to accumulation and interpretation of clinical data. TM finalized the manuscript. All authors read and approved the final manuscript.
Hideki Mutai, Naohiro Suzuki contributed equally to this work.
Electronic supplementary material
13023_2013_804_MOESM3_ESM.pdf
Additional file 3: Audiograms of subjects with hearing loss in the seven families in which candidate genes were detected. Figure legend: Hearing level as a function of frequency in subject IV:2 from family 1 (A), subject III:3 from family 1 (B), subject IV:3 from family 2 (C), subject III:1 from family 2 (D), subject III:2 from family 2 (E), subject III:1 from family 3 (F), subject II:1 from family 4 (G), subject III:1 from family 5 (H), subject II:2 from family 6 (I), subject II:3 from family 6 (J), and subject II:2 from family 7 (K). Open circles with solid lines represent air conduction thresholds of the right ear; crosses with dotted lines represent air conduction thresholds of the left ear; [ symbols represent bone conduction thresholds of the right ear; ] symbols represent bone conduction thresholds of the left ear; arrows pointing to the bottom left represent scale-out hearing level of the right ear; arrows pointing to the bottom right represent scale-out hearing level of the left ear. (PDF 132 KB)
Authors’ original submitted files for images
Below are the links to the authors’ original submitted files for images.
Rights and permissions
Open Access This article is published under license to BioMed Central Ltd. This is an Open Access article is distributed under the terms of the Creative Commons Attribution License ( https://creativecommons.org/licenses/by/2.0 ), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Mutai, H., Suzuki, N., Shimizu, A. et al. Diverse spectrum of rare deafness genes underlies early-childhood hearing loss in Japanese patients: a cross-sectional, multi-center next-generation sequencing study. Orphanet J Rare Dis 8, 172 (2013). https://doi.org/10.1186/1750-1172-8-172
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/1750-1172-8-172