Abstract
Background
Health-related quality of life (HRQOL) is a key outcome in arthritis, but few population-based studies have examined the relationship of specific arthritic conditions, such as osteoarthritis (OA) and rheumatoid arthritis (RA) with HRQOL.
Methods
Older adults in Pennsylvania completed a mail version of the Centers for Disease Control and Prevention (CDC) HRQOL modules. Medicare data were used to identify subjects with OA, RA, and no arthritis diagnosis. We compared HRQOL responses among these groups, and we also examined relationships of demographic characteristics to HRQOL among subjects with arthritis.
Results
In analyses controlling for demographic characteristics and comorbidity, subjects with OA and RA had poorer scores than those without arthritis on all HRQOL items, including general health, physical health, mental health, activity limitation, pain, sleep, and feeling healthy and full of energy. HRQOL scores were also lower for those with RA compared to OA. Among individuals with arthritis, all subject characteristics (including age, race, sex, nursing home residence, marital status, income, and comorbid illnesses) were significantly related to at least one HRQOL item. Older age, nursing home residence, and greater comorbidity were the most consistently associated with poorer HRQOL.
Conclusions
Results of this study show that both OA and RA have a significant impact on multiple dimensions of HRQOL among older adults. Results also suggest the CDC HRQOL items are suitable for use among older adults and in mail surveys. Due to the rising number of older adults in many countries, the public health burden of arthritis is expected to increase dramatically. Efforts are needed to enhance access to medical care and disseminate self-management interventions for arthritis.
Similar content being viewed by others
Background
Arthritis is one of the most common chronic diseases and a leading cause of disability in the United States (U.S.), and other developed countries [1–8]. In the U.S., 55% of individuals age 65 and older report having arthritis or chronic joint symptoms [9]. Due to the increasing number of older adults in many countries, the prevalence of arthritis is expected to rise dramatically [1, 8, 10–13]. In the U.S., it is estimated that the prevalence of arthritis will rise from 43 million in 1997 to 60 million in 2020 [12, 13]. In Canada, it is projected that the prevalence of arthritis will increase from 2.9 million to 6.5 million between 1991 and 2031 [8]. These estimates portend a mounting public health burden.
Because of this growing epidemic, it is important to understand and monitor the impact of arthritis on health outcomes. One health outcome of primary interest within the context of arthritis is health-related quality of life (HRQOL). Arthritis can affect a variety of physical and psychological health domains, and HRQOL measures provide a broad picture of this impact. Therefore HRQOL is now a standard, widely accepted measure in both clinical trials and observational studies. A number of large population- and community-based studies have examined the effects of arthritis on disability and physical function [14–19], but few have assessed overall HRQOL [20–22]. One challenge to population-based assessment of HRQOL is that the length of most traditional measures makes widespread distribution (via telephone or mail) cumbersome. However, the Centers for Disease Control and Prevention (CDC) has developed a brief set of HRQOL items that are now used routinely in the U.S. Behavioral Risk Factor Surveillance System (BRFSS) and other national surveys [23]. Analyses of BRFSS data have shown that individuals with self-reported arthritis or chronic joint symptoms have poorer HRQOL than those without arthritis [20, 24]. These HRQOL items have also been shown to perform well in a clinical population of individuals with rheumatic diseases[25].
In this study, we used the CDC measure to examine the relationship of HRQOL with osteoarthritis (OA) and rheumatoid arthritis (RA) among a statewide sample of older adults. We also examined the relationship of demographic characteristics with HRQOL among older adults with OA and RA. This study adds to previous findings in several important ways. First, this study compared HRQOL scores among a large sample of individuals with physician diagnoses of specific diseases (OA and RA) to individuals without these diagnoses. Previous studies of HRQOL and arthritis have either relied on self reported diagnosis [20–22, 24] or have not included a group of individuals without rheumatic disease [25]. Second, this study specifically involved older adults. Since older adults are the demographic group most commonly affected by arthritis, it is important to understand how OA and RA influence HRQOL in this population. Third, this study tested the feasibility of large-scale mail administration of the CDC HRQOL items, which are most commonly administered via phone. Fourth, this study involved linkage of HRQOL survey data with diagnosis codes from administrative medical databases. This methodology can be used by health care systems and government organizations to examine HRQOL in large samples of individuals with other chronic diseases.
Methods
Subjects
All subjects were enrolled in Pennsylvania's Pharmaceutical Assistance Contract for the Elderly (PACE). PACE is a state program that helps to pay for prescription medications for Pennsylvanians age 65 and older whose annual income does not exceed $17,200 if married or $14,000 if single. During 1997, HRQOL surveys were included with PACE applications mailed to cardholders who were scheduled to renew their coverage. Among the 161,851 individuals who reapplied for coverage, 84,065 returned completed surveys. Due to complications in survey processing during April, surveys were not sent out with all PACE renewal applications during this month. Therefore, individuals scheduled to reapply for PACE benefits during April were excluded when calculating a survey response rate. Out of the 144,819 individuals who reapplied for PACE benefits in months other than April, 83,471 returned surveys, resulting in a 57.6 % response rate. Respondents and non-respondents were similar with respect to demographic characteristics including age, gender, race, residential status, marital status, and annual income.
Arthritis diagnoses for the current study were based on Medicare Part B (Physician/Supplier) claims occurring within one year prior to subjects' survey completion. Only subjects with complete Medicare Part B data for the year prior to survey completion were eligible for this study. Approximately 49% of survey respondents were excluded because they were either not Medicare eligible for the full year prior to survey completion or were members of a health maintenance organization (HMO) that processed their Medicare claims. This resulted in a final study sample of 41,467. HMO enrollment was a considerable source of ineligibility. For example, approximately 24% of survey respondents were enrolled in an HMO for at least one month of 1997. Subjects with complete Medicare data were similar to subjects excluded due to incomplete Medicare data with regard to age, race, income, and residential subjects. However, subjects with complete Medicare data were more likely to be male (23.6% vs. 20.5%, p < 0.001) and were less likely to be married and living with their spouse (16.9% vs. 4.4%, p < 0.001).
Arthritis Diagnoses
OA and RA were identified on the basis of International Classification of Diseases, Version 9 (ICD-9) codes (715 and 714, respectively) from both principal and secondary diagnosis fields within Medicare Physician/Supplier data files. Individuals were placed into three categories: no arthritis diagnosis, OA diagnosis, and RA diagnosis. Approximately 1% of individuals in sample had ICD-9 codes for both OA and RA during the year prior to survey completion. HRQOL scores of individuals with both OA and RA were similar to those for individuals with an RA diagnosis code only. Because of the small sample size of this group, they were combined with individuals with an RA diagnosis code only.
Demographic Factors and Comorbidity
Demographic characteristics included in these analyses were age, gender, annual income (treated as a continuous variable), marital status (married and living with spouse vs. other), residential status (nursing home vs. community dwelling), and race. The sample was predominantly (94%) white, with small proportions of other racial/ethnic groups (5% African American, and <1% of each of the following groups: Asian, Hispanic, Indian, and other – not otherwise described). Therefore we examined race as a dichotomous variable – white and non-white. All demographic factors were obtained from the PACE Enrollment History File. Comorbidity was assessed via the Charlson Comorbidity Score, which uses ICD-9 codes to identify specific diseases [26, 27]. The Charlson Score gives greater weight to mores severe illnesses. For this study, subjects' outpatient visits (Medicare Part B) occurring within one year prior to participants' survey completion date were used to derive the Charlson Score. The Charlson Score was treated as a continuous variable in all analyses.
Survey Instrument
HRQOL was assessed via a mail version of the CDC's Core and Optional HRQOL Modules. In this study, we examined nine items from the CDC HRQOL measure (Appendix A; See Additional File 1). These HRQOL items have been shown to be internally consistent and to identify unmet health-related needs, health disparities, and health behaviors [23, 28–30]. Four of these items (numbers 1–4 in Appendix A) are part of the Core HRQOL module, which is administered to all BRFSS participants. The five remaining questions are part of a 10-item Optional Module that measures more detailed aspects of HRQOL. We selected these additional five items because of their relevance to arthritis. For the general health item (number 1 in Appendix A), we grouped responses into two categories: excellent, very good, or good health compared to fair or poor health. Two additional items, presence of a health-related activity limitation (number 8 in Appendix A) and needing help with personal care (number 9 in Appendix A), were treated as binary (yes/no) variables. The remainder of the HRQOL items were treated as continuous variables, with a minimum score of 0 days and a maximum score of 30 days.
Statistical Analyses
First, we examined relationships of arthritis diagnoses to each HRQOL item. We used Analysis of covariance (ANCOVA) models to compare continuous HRQOL items among subjects with no arthritis, OA diagnosis, and RA diagnosis, while controlling for covariates listed above. Similarly, we used logistic regression models (forward selection) to compare dichotomous HRQOL items across arthritis diagnosis groups, controlling for all covariates. Second, we examined relationships of demographic characteristics and comorbidity with HRQOL among subjects with OA and RA. For these analyses, we examined five representative HRQOL items, including the four Core CDC items and the pain item (because of its particular relevance to arthritis). We used logistic regression (forward selection) to examine relationships of subject characteristics with the dichotomous general health variable, and we used ANCOVA models to examine the four continuous HRQOL items. Statistical significance was set at p < 0.05 for all analyses, which were performed using Statistical Analysis System (SAS) version 8.0.
Results
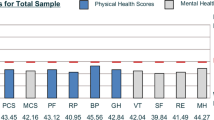
Among the 41,467 eligible survey respondents, 26.4% (N = 10,923) had an ICD-9 code for OA and/or RA during the year prior to survey completion (Table 1). Specifically, 24% of respondents had a diagnosis code for OA and 2.4% had a diagnosis code for RA (approximately half of whom also had a diagnosis code for OA). In analyses adjusted for demographic characteristics and comorbidity, subjects in both arthritis groups had significantly poorer HRQOL on all six "days" items (Table 2) compared to subjects with no arthritis diagnosis. For subjects in both arthritis groups, the HRQOL item that differed most from subjects with no arthritis was the number of days of pain. Compared to those with no arthritis, subjects with OA reported 3.7 more days of pain, and subjects in the RA group reported 6.6 more days of pain. The smallest group differences were observed in the mental health item, with subjects in the arthritis groups reporting approximately one additional day of "not good" mental health compared to subjects with no arthritis. In addition to differences between subjects with arthritis and no arthritis, there were significant differences in the "days" HRQOL items between subjects in both arthritis groups. Subjects in the RA group had poorer scores on each item, and these differences were statistically significant for all items except for mental health.
There were also statistically significant differences among diagnosis groups for each of the categorical HRQOL items. Forty-nine percent of individuals with no arthritis diagnosis reported fair or poor general health, compared with 55% of subjects with OA and 66% of subjects with RA (p < 0.001). In the logistic regression model controlling for demographic characteristics and comorbidity, there were statistically significant differences between all three groups (p < 0.01; Table 3). Fifty-seven percent of subjects without arthritis reported that their activities were limited because of some impairment or health problem, compared with 67% of subjects with OA and 79% of subjects with RA (p < 0.001). In a multivariable logistic regression model, there were statistically significant differences among all three groups (p < 0.01). Twenty-two percent of subjects with no arthritis diagnosis reported needing help with personal care, compared with 25% of subjects with OA and 32% of subjects with RA (p < 0.001). In the multivariable logistic regression model, subjects with RA had significantly greater odds of needing help with personal care than subjects without arthritis and those with OA (p < 0.01). However, there were no statistically significant differences between subjects with OA and those without arthritis.
Among subjects with arthritis (OA and/or RA), there were some differences in HRQOL according to demographic characteristics and comorbidity (Table 4). Subjects who were more likely to report fair or poor general health included those who were older, non-whites, nursing home residents, married subjects, those with lower income, and those with more comorbid illnesses. Subjects who were 85 years and older, male, residing in a nursing home, married, and had lower income and more comorbid illnesses reported poorer scores on one or more of the "days" HRQOL items. Subject race was not significantly related to any of the "days" items.
Discussion
In agreement with prior research [20, 22, 24, 31], results of this study show that arthritis has a significant impact on multiple dimensions of HRQOL. Specifically, older adults with OA and RA reported poorer general health, physical health, mental health, and sleep, as well as more activity limitation and pain, compared to those without arthritis. These results also confirm that arthritis differentially impacts specific aspects of HRQOL. Not surprisingly, pain showed the largest difference between subjects with arthritis and without arthritis. Although the sleep and mental health items showed statistically significant differences across groups, the magnitude of these differences was small (1–2 days) and of questionable clinical relevance. Mili et al. reported a similar difference in "not good" mental health days between subjects without arthritis and those reporting doctor-diagnosed arthritis in the BRFSS sample [24]. Yet studies using other measures have shown that depression is a substantial problem among individuals with arthritis particularly RA [32].
Subjects with OA and RA in this sample had considerably poorer HRQOL than individuals in the BRFSS sample who were ≥ 65 years of age and reported having arthritis [24]. For example, 40% of the BRFSS sample reported fair or poor general health, compared with 55% of subjects with OA and 66% of subjects with RA in our sample. In the BRFSS group, the mean number of days of activity limitation was 3.5, compared with 10 days in our OA group and 13 days in our RA group. These differences may be partially due to the definitions of arthritis used in these two studies. In the BRFSS study, the arthritis sample was defined as those reporting either the presence of chronic joint symptoms or a doctor's diagnosis of arthritis. The present study used ICD-9 codes from medical records, so all patients were under physician care for OA. Since all participants in the BRFSS sample did not report having a doctor's diagnosis, this may have been a sample with a lower average level of OA severity than our sample. However, demographic and health characteristics of the samples may also be related to the observed differences. Even among those without arthritis, HRQOL was poorer in the current study compared to the BRFSS sample. Our sample consisted of older adults with fairly low income levels, and they may been in generally poorer health than those in the BRFSS. It is significant that even in our sample of older adults with relatively low income and multiple comorbid illnesses, a diagnosis of arthritis was associated with poorer HRQOL. This suggests an important independent impact of arthritis on HRQOL among older adults.
Also in agreement with prior studies [21, 22, 25], this study revealed poorer HRQOL among individuals with RA compared to OA. Individuals with RA had poorer scores than those with OA on all HRQOL items, though this difference was not statistically significant for days of "not good" mental health. In general, RA is associated with greater disability than OA, though individuals with more severe OA and widespread pain may have comparable impairment [21]. Although HRQOL may be lower for individuals with RA than OA, this study shows that OA also has a significant impact on HRQOL. Furthermore, the high prevalence of OA makes this a considerable public health concern.
Among subjects with OA and RA in this sample, we found that HRQOL differed according to demographic characteristics and comorbidity. These associations were largely in the expected direction and similar to other research [24, 33], with older age, non-white race, nursing home residence, lower income, and greater comorbidity being associated with poorer scores on at least one HRQOL item. In contrast to other research [24], men in this sample reported a greater number of days of "not good" physical and mental health compared with women. The reason for this association is not clear, but men in this sample may have had greater arthritis severity or overall poorer health (which was not captured adequately by the Charlson Comorbidity Score) compared to women.
This study also confirmed the utility of the CDC's HRQOL items for mail distribution and among older adults. This scale differentiated between individuals with arthritis and without arthritis, and it also detected differences between those with OA and RA. Because the CDC HRQOL Modules are brief and easy to administer by telephone or mail, they are particularly useful for large-scale epidemiological studies.
There are several limitations so this study. Compared to the general U.S. elderly population, PACE enrollees are older and have a lower annual income. PACE enrollees also include a greater proportion of females, a smaller percentage of minorities, and a smaller proportion of married individuals. This sample was also restricted to subjects with complete Medicare data during the observation period. These individuals were also less likely to be married than subjects who were excluded because of incomplete Medicare data. These sample characteristics may limit generalizability, and additional studies are needed among different samples of older adults. Because of the very small numbers (<20) in some racial/ethnic groups, it was not possible to examine differences across specific groups. We used a broad race categorization (white vs. non-white) that may have masked differences in some specific racial/ethnic groups, and further study is needed on more racially diverse samples.
Another limitation concerns the specificity of ICD-9 codes for OA. Studies have reported the sensitivity of ICD-9 codes for OA to be between 0.32 and 1.0, and the positive predictive value between 0.76 and 0.90 [34, 35]. Sensitivity and positive predictive value are both greater when codes are used to identify OA in general (as was done in this study), rather than site-specific OA. Because of these known issues regarding ICD-9 code accuracy, there may have been some misclassification in this study. However, there are also inaccuracies associated with self-reported diagnosis of arthritis, and radiographic evidence is not feasible for samples of this size.
A limitation of all survey studies is the potential for non-response bias. The response rate for this survey was similar to the average response rate in other published studies involving mail surveys [36]. Non-respondents were similar to respondents on a variety of demographic characteristics. Though this survey was brief (16 questions), older adults with poorer health or greater functional limitation may have been less likely to complete the survey. Therefore this sample may under-represent older adults with poorer health and possibly greater arthritis severity.
In summary, this study revealed a substantial impact of OA and RA on HRQOL, as measured by the CDC items. Both pharmacological and non-pharmacological treatments can yield improvements in arthritis symptoms and HRQOL [37]. However, both medical treatment and non-pharmacological interventions are underutilized [38–40], and some evidence suggests that medications are often prescribed at doses that may be too low to optimally affect symptoms [41]. Efforts are needed to enhance access to medical care for OA, ensure optimal pharmacological management, and disseminate self-management interventions.
References
Helmick CG, Lawrence RC, Pollard RA, Lloyd E, Heyse SP: Arthritis and other rheumatic conditions: Who is affected now, Who will be affected later? Arthritis Care and Research 1995, 8: 203–211.
Centers for Disease Control and Prevention: Prevalence of disabilities and associated health conditions among adults - United States, 1999. Morbidity and Mortality Weekly Report 2001, 50: 120–125.
Guccione AA, Felson DT, Anderson JJ, Anthony JM, Zhang Y, Wilson PW, Kelly-Yayes M, Wolf PA, Kreger BE, Kannel WB: The effects of specific medical conditions on the functional limitations of elders in the Framingham Study. American Journal of Public Health 1997, 84: 351–358.
Dunlop D, Manheim LM, Yelin EH, Song J, Chang RW: The costs of arthritis. Arthritis & Rheumatism 2003, 49: 101–113. 10.1002/art.10913
Lawrence RC, Helmick CG, Arnette FC, Deyo RA, Felson DT, Giannini EH, Heyse SP, Hirsch R, Hochberg MC, Hunder GG, Liang MH, Pillemer S, Steen VD, Wolfe F: Estimates of the prevalence of arthritis and selected musculoskeletal disorders in the United States. Arthritis and Rheumatism 1998, 41: 778–799. 10.1002/1529-0131(199805)41:5<778::AID-ART4>3.0.CO;2-V
March LM, Bachmeier CJM: Economics of OA: a global perspective. Ballieres Clinical Rheumatology 1997, 11: 817–834.
Carmona L, Ballina J, Gabriel R, Lafffon A: The burden of musculoskeletal disease in the general population of Spain: results from a national survey. Annals of the Rheumatic Diseases 2001, 60: 1040–1045. 10.1136/ard.60.11.1040
Badley EM, Wang PP: Arthritis and the aging population: projections of arthritis prevalence in Canada 1991 to 2031. Journal of Rheumatology 1998, 25: 138–144.
Mili F, Helmick CG, Zack MM: Prevalence of arthritis: analysis of data from the U.S. Behavioral Risk Factor Surveillance System, 1996–1999. Journal of Rheumatology 2002, 29: 1981–1988.
Centers for Disease Control and Prevention: Public health and aging: projected prevalence of self-reported arthritis or chronic joint symptoms among persons aged >65 years -- United States, 2005–2030. Morbidity and Mortality Weekly Report 2003, 52: 489–491.
Elders MJ: The increasing impact of arthritis on public health. Journal of Rheumatology Supplement 2000, 60: 6–8.
Centers for Disease Control and Prevention: Arthritis prevalence and activity limitations -- United States, 1990. Morbidity and Mortality Weekly Report 1994, 43: 433–438.
Centers for Disease Control and Prevention: Prevalence of arthritis - United States, 1997. Morbidity and Mortality Weekly Report 2001, 50: 334–336.
Hochberg MC, Kasper J, Williamson JD, Skinner A, Fried LP: The contribution of osteoarthritis to disability: Preliminary data from the Women's Health and Aging Study. Journal of Rheumatology 1995, 22: 16–18.
Verbrugge LM, Lepkowski JM, Konkol LL: Levels of disability among U.S. adults with arthritis. Journals of Gerontology. Series B, Psychological Sciences and Social Sciences 1991, 46: 571–583.
Hopman-Rock M, Odding E, Hofman A, Kraaimaat FW, Bijlsma JWJ: Physical and psychosocial disability in elderly subjects in relation to pain in the hip and/or knee. Journal of Rheumatology 1996, 23: 1037–1044.
Ettinger WH, Davis MA, Neuhaus JM, Mallon KP: Long-term physical functioning in persons with knee osteoarthritis from NHANES I: effects of comorbid medical conditions. Journal of Clinical Epidemiology 1994, 47: 809–815.
Jordan JM, Luta G, Renner JB, Dragomir A, Hochberg MC, Fryer JG: Ethnic differences in self-reported functional status in the rural south: The Johnston County Osteoarthritis Project. Arthritis Care and Research 1996, 9: 483–491.
Jordan JM, Luta G, Renner JB, Linder GF, Dragomir A, Hochberg MC, Fryer JG: Self-reported functional status in osteoarthritis of the knee in a rural southern community: The role of sociodemographic factors, obesity, and knee pain. Arthritis Care and Research 1996, 9: 273–278.
Centers for Disease Control and Prevention: Health-related quality of life among adults with arthritis - Behavioral Risk Factor Surveillance System, 11 states, 1996–1998. Morbidity and Mortality Weekly Report 2000, 49: 366–369.
Hagen KB, Kvien TK, Bjorndal A: Musculoskeletal pain and quality of life in patients with noninflammatory joint pain compared to rheumatoid arthritis: a population survey. Journal of Rheumatology 1997, 24: 1703–1709.
Hill CL, Parsons J, Taylor A, Leach G: Health related quality of life in a population sample with arthritis. Journal of Rheumatology 1999, 26: 2029–2035.
Moriarty DG, Zack MM, Kobau R: The Centers for Disease Control and Prevention's Healthy Days Measures - Population tracking of perceived physical and mental health over time. Health and Quality of Life Outcomes 2003., 1:
Mili F, Helmick CG, Zack MM, Moriarty DG: Health related quality of life among adults reporting arthritis: analysis of data from the Behavioral Risk Factor Surveillance System, U.S. 1996–1999. Journal of Rheumatology 2003, 30: 160–166.
Currey SS, Rao JK, Winfield JB, Callahan LF: Performance of a generic health-related quality of life measure in a clinic population with rheumatic disease. Arthritis & Rheumatism 2003, 49: 658–664. 10.1002/art.11381
Charlson ME, Pompei P, Ales KL, MacKenzie CR: A new method of classifying prognostic comorbidity in longitudinal studies: Development and validation. Journal of Chronic Diseases 1987, 40: 373–383.
Deyo RA, Cherkin DC, Ciol MA: Adapting a clinical comorbidity index for use with ICD-9-CM administrative databases. Journal of Clinical Epidemiology 1992., 45:
Ounpuu S, Kreuger P, Vermeulen M, Chambers L: Behavioral risk factor surveillance system's health-related quality of life survey tool in a Canadian city. Canadian Journal of Public Health 1998, 91: 67–72.
Andresen E, Fouts B, Romeis J, Brownson C: Performance of health-related quality of life instruments in a spinal cord injured population. Archives of Physical Medicine and Rehabilitation 1999, 80: 877–884.
Brown DW, Balluz LS, Heath GW, Moriarty DG, Ford ES, Giles WH, Mokdad AH: Associations between recommended levels of physical activity and health-related quality of life: Findings from the 2001 Behavioral Risk Factor Surveillance System (BRFSS) survey. Preventive Medicine 2003, 37: 520–528. 10.1016/S0091-7435(03)00179-8
Kaplan RM, Alcaraz JE, Anderson JP, Weisman M: Quality-adjusted life years lost to arthritis: effects of gender, race, and social class. Arthritis Care and Research 1996, 9: 473–482.
Dickens C, Creed F: The burden of depression in patients with rheumatoid arthritis. Rheumatology 2001, 40: 1327–1330. 10.1093/rheumatology/40.12.1327
Ibrahim SA, Burant CJ, Siminoff LA, Stoller EP, Kwoh CK: Self-assessed global quality of life: A comparison of African-American and white older patients with arthritis. Journal of Clinical Epidemiology 2002, 55: 512–517. 10.1016/S0895-4356(01)00501-7
Fowles JB, Lawthers AG, Weinger JP, Garnick DW, Petrie DS, Palmer RH: Agreement between physicians' office records and Medicare Part B claims data. Health Care Financing Review 1995, 16: 189–199.
Katz JN, Barrett J, Liang MH, Bacon AM, Kaplan H, Kieval RI, Lindsey SM, Roberts WN, Sheff DM, Spencer RT, Weaver AL, Baron JA: Sensitivity and positive predictive value of Medicare Part B physician claims for rheumatologic diagnoses and procedures. Arthritis and Rheumatism 1997, 40: 1594–1600.
Asch DA, Jedrziewski MK, Christakis NA: Response rates to mail surveys published in medical journals. Journal of Clinical Epidemiology 1997, 50: 1129–1136. 10.1016/S0895-4356(97)00126-1
Altman RD, Hochberg MC, Moskowitz RW, Schnitzer TJ: Recommendations for the medical management of osteoarthritis of the hip and knee. Arthritis and Rheumatism 2000, 43: 1905–1915. 10.1002/1529-0131(200009)43:9<1905::AID-ANR1>3.0.CO;2-P
Rao JK, Callahan LF, Helmick CG: Characteristics of persons with self-reported arthritis and other rheumatic conditions who do not see a doctor. Journal of Rheumatology 1997, 24: 169–173.
Mazzuca SA, Brandt KD, Katz BP, Dittus RS, Freund D, Lubitz R, Hawker G, Eckert G: Comparison of general internists, family physicians, and rheumatologists managing patients with symptoms of osteoarthritis of the knee. Arthritis Care and Research 1997., 10:
Arthritis Foundation Association of State and Territorial Health Officials, Centers for Disease Control and Prevention: National Arthritis Action Plan: a public health strategy. Atlanta, Arthritis Foundation 1999.
Pahor M, Guralnik JM, Wan JY, Ferrucci L, Pennix BWJH, Lyles A, Ling S, Fried Linda P.: Lower body osteoarticular pain and dose of analgesic medications in older disabled women: The Women's Health and Aging study. American Journal of Public Health 1999, 89: 930–934.
Acknowledgements
The authors would like to thank PACE and PHC4 for providing the data used in this study. This research was funded by a cooperative agreement (TS213) from the Centers for Disease Control and the Association of Teachers of Preventive Medicine. This research was also supported in part by the Department of Veterans Affairs, Veterans Health Administration, HSR&D Service, Program 824 Funds to the first author. The views expressed in this paper are those of the authors and do not necessarily represent the views of the Department of Veterans Affairs.
Author information
Authors and Affiliations
Corresponding author
Additional information
Authors' Contributions
KD performed statistical analysis, drafted the manuscript, and assisted with survey processing and data management. FA, CG, and DH participated in study conception and design and assisted with drafting of the manuscript.
Electronic supplementary material
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Dominick, K.L., Ahern, F.M., Gold, C.H. et al. Health-related quality of life among older adults with arthritis. Health Qual Life Outcomes 2, 5 (2004). https://doi.org/10.1186/1477-7525-2-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/1477-7525-2-5