Abstract
In the present study, we assess the impact of incentives provided by the government on the dynamics of infectious diseases that spread due to the direct contacts of susceptibles with the infectives and also through the environmental contamination of those diseases. To this, we develop a mathematical model which comprises susceptible individuals, infected individuals, environmental contamination and the incentive provided by the government healthcare officials as dynamic variables. The proposed epidemic model is analyzed mathematically as well as numerically. System’s dynamics have been mainly studied about the disease-free and interior equilibrium points. We perform some sensitivity tests to find out model parameters that can have major roles in regulating the epidemic pattern. Our findings show that the disease could be effectively controlled by reducing the contact rate of susceptibles with the infected individuals and also their exposure to the environmental contamination. This could be possible by raising awareness among the public and using disinfectants of high quality for removing contaminants from the environment. These results suggest to increase the incentive amount in the areas facing rapid incline in the number of infected cases and the level of environmental contamination.










Similar content being viewed by others
Data availibility
All data generated or analyzed during this study are included in this article.
References
M. Mandal, S. Jana, S.K. Nandi et al., A model based study on the dynamics of COVID-19: prediction and control. Chaos Solit. Fract. 136, 109889 (2022)
I. Ghosh, P.K. Tiwari, S. Mandal, M. Martcheva, J. Chattopadhyay, A mathematical study to control Guinea Worm Disease: a case study on Chad. J. Biol. Dyn. 12(1), 846–871 (2018)
A.K. Srivastav, P.K. Tiwari, M. Ghosh, Modeling the impact of early case detection on dengue transmission deterministic vs stochastic. Stoch. Anal. Appl. 39(3), 434–455 (2021)
X. Chang, M. Liu, Z. Jin, J. Wang, Studying on the impact of media coverage on the spread of COVID-19 in Hubei Province, China. Math. Biosci. Eng. 17(4), 3147–3159 (2020)
P. Dubey, U.S. Dubey, B. Dubey, Role of media and treatment on an SIR model. Nonlinear Anal. Model. Control 21, 185–200 (2016)
A.K. Misra, R.K. Rai, Y. Takeuchi, Modeling the control of infectious diseases: effects of TV and social media advertisements. Math. Biosci. Eng. 15(6), 1315–1343 (2018)
R.K. Rai, A.K. Misra, Y. Takeuchi, Modeling the impact of sanitation and awareness on the spread of infectious diseases. Math. Biosci. Eng. 16(2), 667–700 (2019)
A.K. Misra, R.K. Rai, A mathematical model for the control of infectious diseases: effects of TV and radio advertisements. Int. J. Bifurcat. Chaos 28(03), 1850037 (2018)
R.K. Rai, P.K. Tiwari, Y. Kang, A.K. Misra, Modeling the effect of literacy and social media advertisements on the dynamics of infectious diseases. Math. Biosci. Eng. 17(5), 5812–5848 (2020)
P.K. Tiwari, R.K. Rai, A.K. Misra, J. Chattopadhyay, Dynamics of infectious diseases: local versus global awareness. Int. J. Bifurcat. Chaos 31(7), 2150102 (2021)
A.K. Misra, A. Sharma, J.B. Shukla, Modeling and analysis of effects of awareness programs by media on the spread of infectious diseases. Math. Comp. Model. 53(5–6), 1221–1228 (2011)
A.K. Misra, A. Sharma, J. Li, A mathematical model for control of vector borne diseases through media campaigns. Discrete Cont. Dyn. Syst. B. 18(7), 1909–1927 (2013)
A. Sharma, A.K. Misra, Modeling the impact of awareness created by media campaigns on vaccination coverage in a variable population. J. Biol. Syst. 22(02), 249–270 (2014)
A.K. Misra, A. Sharma, J.B. Shukla, Stability analysis and optimal control of an epidemic model with awareness programs by media. BioSystems 138, 53–62 (2015)
H.F. Huo, S.R. Huang, X.Y. Wang, H. Xiang, Optimal control of a social epidemic model with media coverage. J. Biol. Dyn. 11(1), 226–243 (2017)
I. Ghosh, P.K. Tiwari, S. Samanta et al., A simple SI\(-\)type model for HIV/AIDS with media and self-imposed psychological fear. Math. Biosci. 306, 160–169 (2018)
P.K. Roy, S. Saha, F.A. Basir, Effect of awareness programs in controlling the disease HIV/AIDS: an optimal control theoretic approach. Adv. Differ. Equ. 2015(1), 1–8 (2015)
D.K. Das, S. Khajanchi, T.K. Kar, The impact of the media awareness and optimal strategy on the prevalence of tuberculosis. Appl. Math. Comput. 366, 124732 (2020)
A. Sharma, A.K. Misra, Backward bifurcation in a smoking cessation model with media campaigns. Appl. Math. Model. 39(3–4), 1087–1098 (2015)
X. Chang, J. Wang, M. Liu, Y. Yang, Stability analysis and optimal control of an epidemic model with multidimensional information of media coverage on networks. Math. Meth. Appl. Sci. 46(6), 6787–6802 (2023)
R.K. Rai, S. Khajanchi, P.K. Tiwari, E. Venturino, A.K. Misra, Impact of social media advertisements on the transmission dynamics of COVID-19 pandemic in India. J. Appl. Math. Comput. 68, 19–44 (2022)
P.K. Tiwari, R.K. Rai, S. Khajanchi, R.K. Gupta, A.K. Misra, Dynamics of coronavirus pandemic: Effects of community awareness and global information compaigns. Eur. Phys. J. Plus 136(10), 994 (2021)
F.T. Kobe, P.R. Koya, Modeling and analysis of effect of awareness programs by media on the spread of COVID-19 pandemic disease. Am. J. Appl. Math. 8(4), 223–229 (2020)
A.K. Misra, A. Sharma, V. Singh, Effect of awareness programs in controlling the prevalence of an epidemic with time delay. J. Biol. Syst. 19(02), 389–402 (2011)
S. Samanta, Effects of awareness program and delay in the epidemic outbreak. Math. Meth. Appl. Sci. 40(5), 1679–1695 (2017)
D. Greenhalgh, S. Rana, S. Samanta et al., Awareness programs control infectious disease - multiple delay induced mathematical model. Appl. Math. Comput. 251, 539–563 (2015)
A.K. Misra, R.K. Rai, P.K. Tiwari, M. Martcheva, Delay in budget allocation for vaccination and awareness induces chaos in an infectious disease model. J. Biol. Dyn. 15(1), 395–429 (2021)
R.K. Rai, P.K. Tiwari, S. Khajanchi, Modeling the influence of vaccination coverage on the dynamics of COVID-19 pandemic with the effect of environmental contamination. Math. Meth. Appl. Sci. (2023). https://doi.org/10.1002/mma.9185
H. Gaff, E. Schaefer, Optimal control applied to vaccination and treatment strategies for various epidemiological models. Math. Biosci. Eng. 6(3), 469–492 (2009)
S.M. Garba, J.M. Lubuma, B. Tsanou, Modeling the transmission dynamics of the COVID-19 pandemic in South Africa. Math. Biosci. 328, 108441 (2020)
P. van den Driessche, J. Watmough, Reproduction numbers and sub-threshold endemic equilibrium for compartmental models of disease transmission. Math. Biosci. 180(1–2), 29–48 (2002)
C. Castillo-Chavez, B. Song, Dynamical models of tuberculosis and their applications. Math. Biosci. Eng. 1(2), 361–404 (2004)
S.M. Blower, H. Dowlatabadi, Sensitivity and uncertainty analysis of complex models of disease transmission: an HIV model, as an example. Int. Stat. Rev. 62(2), 229–243 (1994)
S. Marino, I.B. Hogue, C.J. Ray, D.E. Kirschner, A methodology for performing global uncertainty and sensitivity analysis in systems biology. J. Theor. Biol. 254(1), 178–196 (2008)
S. Samanta, S. Rana, A. Sharma, A.K. Misra, J. Chattopadhyay, Effect of awareness programs by media on the epidemic outbreaks: a mathematical model. Appl. Math. Comput. 219(12), 6965–6977 (2013)
I. Ghosh, P.K. Tiwari, J. Chattopadhyay, Effect of active case finding on dengue control: Implications from a mathematical model. J. Theor. Biol. 464, 50–62 (2019)
M. Martcheva, An introduction to mathematical epidemiology (Springer, New York, 2015)
A. Abate, A. Tiwari, S. Sastry, Box invariance in biologically-inspired dynamical systems. Automatica 45(7), 1601–1610 (2009)
V. Lakshmikantham, S. Leela, A.A. Martynyuk, Stability analysis of nonlinear systems (Springer, Cham, 1989)
S. Wiggins, Introduction to applied nonlinear dynamical systems and chaos (Springer, New York, 1990)
Y. Li, J.S. Muldowney, On Bendixson’s criterion. J. Differ. Equ. 106, 27–39 (1993)
L. Perko, Differential equations and dynamical systems, 3rd edn. (Springer, Berlin, 2000)
P.K. Tiwari, S. Roy, G. Douglas, A.K. Misra, An optimal control model for the impact of Phoslock on the mitigation of algal biomass in lakes. J. Biol. Syst. 30(4), 945–984 (2022)
Acknowledgements
The authors express their gratitude to the associate editor and the reviewers whose comments and suggestions have helped the improvements of this paper.
Funding
The research work of Kalyan Kumar Pal is supported by University Grants Commission, Government of India, New Delhi, in the form of National Fellowship for Other Backward Classes [No. F. 40-2/June 2021 (CSIR NET Fellowships)]. The work of Yun Kang is partially supported by NSF-DMS (Award Numbers 1716802 and 2052820) and The James S. McDonnell Foundation (10.37717/220020472).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interests regarding the publication of this article.
Ethical approval
The authors state that this research complies with ethical standards. This research does not involve either human participants or animals.
Appendices
Appendix A
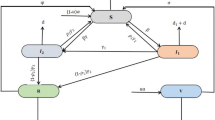
Let \(X=[S,I,E_c,M]\), then system (1) can also be written as follows:
where \(F=[\Lambda ,0,0,0]^T\) is a nonnegative vector and
with
Apparently, all the off-diagonal entries of matrix E(X) are nonnegative ensuring that the matrix is Metzler for all \(X\in {\mathbb {R}}_+^4\). Thus, system (1) is positively invariant in \({\mathbb {R}}_+^4\) [38], i.e., all the solution trajectories of the system starting from an initial state in \({\mathbb {R}}_+^4\) confine there for all \(t\ge 0\).
We obtain the following inequality from the second equation of system (3):
On integrating the above inequality from 0 to t, we get \(\displaystyle N(t)\le N_0e^{-dt}+\frac{\Lambda }{d}.\) This yields that \(\displaystyle \limsup _{t\rightarrow \infty } N(t)\le \dfrac{\Lambda }{d}.\) Thus, \(\displaystyle N(t)\le \dfrac{\Lambda }{d}\) for large \(t>0\). As \(I(t)\le N(t)\) at any time, so \(I(t) \le \dfrac{\Lambda }{d}\) for any large value of t.
Now, from the third equation of system (3), we have
Using the theory of differential inequality [39], we get \(\displaystyle \limsup _{t\rightarrow \infty} E_c(t)\le \dfrac{s_1 \Lambda }{ds_0}.\) This implies that \(\displaystyle E_c(t)\le \dfrac{s_1 \Lambda }{ds_0}\) for large \(t>0\).
Again, the last equation of system (3) yields the following inequality:
By using the comparison theorem from [39], we get
Thus, for any large value of t, we have
Thus, we find that the region \(\Omega\) is positively invariant, i.e., all the solutions of system (3) with initial conditions in \(\Omega\) remain therein for any large value of \(t>0\). Also, \(\Omega\) attracts all the solutions of system (3) with initial condition in \({\mathbb {R}}^4_+\). Hence, system (3) is mathematically well posed in the region \(\Omega\).
Appendix B
We find the Jacobian matrix of system (3) as \(J=[J_{ij}]_{4\times 4}\) with the following entries:
On evaluating the Jacobian matrix J at the equilibrium \(E_1\), we get two eigenvalues as \(-d\) and r whereas the remaining two will be obtained by solving the following equation:
Irrespective of signs of the reals parts of the roots of the above equation, the equilibrium point \(E_1\) is unstable as one eigenvalue, r, is always positive. Thus, we can say that having an equilibrium situation with susceptible only (no disease and no incentive) is practically impossible. Similarly, the equilibrium \(E_2\) with disease but no incentive is unconditionally unstable as one eigenvalue value of the Jacobian matrix J at this equilibrium point is \(r+\theta _1I_2+\theta _2E_{c2}\) (always positive) while the remaining three are roots of the following cubic equation:
where
The unstable nature of the incentive-free equilibrium \(E_2\) indicates that having such equilibrium situation is a realistic scenario in impossible, i.e., in a region of epidemic outbreak, government always provides some incentives for the prevention of the disease. On computing the matrix J at the equilibrium \(E_3\), we obtain the two eigenvalues as \(-r\) and \(-d\) , whereas the other two are roots of the equation below:
Clearly, if \({\mathcal {R}}_0<1\) and the condition (16) is satisfied, then the roots of above equation are either negative or have negative real parts leading to the stability of the equilibrium \(E_3\). Next, we evaluate the matrix J at the equilibrium \(E^*\) and obtain the associated characteristic equation as,
where
with
The roots of equation (26) are either negative or have negative real parts if and only if all the inequalities in (17) hold; in view of the Routh-Hurwitz criterion [42], these conditions imply the local asymptotic stability of the equilibrium \(E^*\). Note that the equilibrium \(E^*\) will be no longer stable if any of the conditions in (17) does not hold.
Appendix C
Considering only the variables I and \(E_c\), we get the following subsystem of system (3):
Defining \(\displaystyle h(I,E_c)=\dfrac{1}{IE_c},\) we have \(\displaystyle \Delta _{E_3}(I,E_c)=\dfrac{\partial }{\partial I}(hf_1)+\dfrac{\partial }{\partial E_c}(hf_2).\) Obviously, \(h(I,E_c)>0\) for all \(I,E_c>0\). Thus,
Obviously, \(\Delta _{E_3}(I,E_c)\) does not change its sign and is also not identically zero in the positive quadrant of the \(I-E_c\) plane. Thus, according to the Bendixson–Dulac criterion [40], system (3) could not exhibit any limit cycle in the positive quadrant of the \(I-E_c\) plane. As the disease-free equilibrium \(E_3\) is locally asymptotically stable whenever \({\mathcal {R}}_0<1\), so it is globally asymptotically stable in the positive quadrant of the \(I-E_c\) plane for \({\mathcal {R}}_0<1\). In the same line, one can show that the disease-free equilibrium \(E_3\) of system (3) is globally asymptotically in the other planes also [40].
Appendix D
Corresponding to system (3), we consider the following positive definite function:
where \(m_1,m_2\) and \(m_3\) are some positive constants to be chosen later. On calculating the time derivative of the function G along the solutions of system (3), choosing \(\displaystyle m_1=\dfrac{1}{\alpha }\left( \beta -\beta _1\dfrac{k_1M^*}{p+k_1M^*}\right)\) and rearranging the terms, we get
Notably, the derivative function \(\displaystyle \dfrac{{\text {d}}G}{{\text {d}}t}\) will be negative definite inside \(\Omega\) if the following inequalities are satisfied:
From inequalities (28) and (29), one can choose a positive value of \(m_2\) if inequality (19) holds. Also, from inequalities (30)–(33), one can get a positive value of \(m_3\) if the inequality (20) holds. Thus, one can conclude that \(\displaystyle \dfrac{{\text {d}}G}{{\text {d}}t}\) will be negative definite inside \(\Omega\) if inequalities (18)−(20) hold.
Appendix E
The second additive compound matrix associated with the matrix \(J_{E^*}\) is obtained as,
Define \(|Y|_\infty =\sup |Y_i|.\) The logarithmic norm \(\mu _\infty (J_{E^*}^{[2]})\) of the matrix \(J_{E^*}^{[2]}\) endowed with the vector norm \(|Y|_\infty\) is determined by the supremum of the following quantities:
Notably, (\(a_{11}+a_{22}+|a_{13}|+|a_{14}|)_{E^*}<0\) if
Also, \((a_{11}+a_{33}+|a_{34}|+|a_{12}|+|a_{14}|)_{E^*}<0\) if
Again, \((|a_{43}|+a_{11}+a_{44}+|a_{12}|+|a_{13}|)_{E^*}<0\) if
Further, \((|a_{31}|+|a_{21}|+a_{22}+a_{33}+|a_{34}|)_{E^*}<0\) if
Furthermore, \((|a_{41}|+|a_{21}|+|a_{43}|+a_{22}+a_{44})_{E^*}<0\) if
Finally, (\(|a_{41}+|+|a_{31}|+a_{33}+a_{44})_{E^*}<0\) if
Combining the inequalities (34)−(39), one can get the required condition (23).
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Pal, K.K., Rai, R.K., Tiwari, P.K. et al. Role of incentives on the dynamics of infectious diseases: implications from a mathematical model. Eur. Phys. J. Plus 138, 564 (2023). https://doi.org/10.1140/epjp/s13360-023-04163-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1140/epjp/s13360-023-04163-2