Abstract
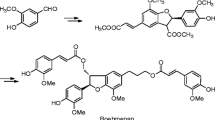
Three synthetic approaches to [2+1] conjugates of betulic acid with α,ω-diols have been tested. The best results have been obtained in the reaction of betulic acid with α,ω-dibromides in the presence of potassium carbonate in DMF.



Similar content being viewed by others
REFERENCES
Poumale, H.M.P., Amadou, D., Shiono, Y., Deccaux, G., Kapche, W.F., and Ngadjui, B.T., Asian J. Chem., 2011, vol. 23, p. 525.
Ryu, S.Y., Lee, C.K., Ahn, J.W., Lee, S.H., and Zee, O.P., Arch. Pharm. Res., 1993, vol. 16, p. 339. https://doi.org/10.1007/BF02977528
Flekhter, O.B., Nigmatullina, L.R., Baltina, L.A., Karachurina, L.T., Galin, F.Z., Zarudii, F.S., Tolstikov, G.A., Boreko, E.I., Pavlova, N.I., Nikolaeva, S.N., and Savinova, O.V., Pharm. Chem. J., 2002, vol. 36, p. 484. https://doi.org/10.1023/A:1021844705853
Mukherjee, R., Jaggi, M., Rajendran, P., Siddiqui, M.J.A., Srivastava, S.K., Vardhan, A., and Burman, A.C., Bioorg. Med. Chem. Lett., 2004, vol. 14, p. 2181. https://doi.org/10.1016/j.bmcl.2004.02.044
Kim, J.Y., Koo, H.-M., and Kim, D.S.H.L., Bioorg. Med. Chem. Lett., 2001, vol. 11, p. 2405. https://doi.org/10.1016/S0960-894X(01)00460-7
Tolstikov, G.A., Flekhter, O.B., Shultz, E.E., Baltina, L.A., and Tolstikov, A.G., Chem. Sustainable Develop., 2005, vol. 13, p. 1.
Xiong, J., Kashiwada, Y., Chen, C.-H., Qian, K., Morris-Natschke, S.L., Lee, K.-H., and Takaishi, Y., Bioorg. Med. Chem., 2010, vol. 18, p. 6451. https://doi.org/10.1016/j.bmc.2010.06.092
Pokrovsky, A.G., Plyasunova, O.A., Ilyicheva, T.N., Borisova, O.A., Fedyuk, N.V., Petrenko, N.I., Petukhova, V.Z., Shults, E.E., and Tolstikov, G.A., Chem. Sustainable Develop., 2001, vol. 9, p. 485.
Bębenek, E., Chrobak, E., Wietrzyk, J., Kadela, M., Chrobak, A., Kusz, J., Książek, M., Jastrzębska, M., and Boryczka, S., J. Mol. Struct., 2016, vol. 1106, p. 210. https://doi.org/10.1016/j.molstruc.2015.10.102
Urban, M., Sarek, J., Tislerova, I., Dzubak, P., and Hajduch, M., Bioorg. Med. Chem., 2005, vol. 13, p. 5527. https://doi.org/10.1016/j.bmc.2005.07.011
Jie, Y., CN Patent no. CN 101519423B, 2011.
Sczepek, R., Nitsche, C., Heller, L., Siewert, B., Schãfer, R., Flemming, F., Otgonbayar, C., and Csuk, R., Mediterranean J. Chem., 2015, vol. 4, p. 126. https://doi.org/10.13171/mjc.4.3.2015.05.26.14.18/csuk
Leunis, J.-C. and Couche, E., WO Patent Appl. no. WO2007101873 A3, 2006.
Popov, S.A., Semenova, M.D., Baev, D.S., Sorokina, I.V., Zhukova, N.A., Frolova, T.S., Tolstikova, T.G., Shults, E.E., and Turks, M., Steroids, 2019, vol. 150, p. 108443. https://doi.org/10.1016/j.steroids.2019.108443
Kogay, T.I., Svirskaya, N.M., Ivanchenko, N.M., and Kuznetsova, B.N., Zh. Sibir. Fed. Univer. Khim., 2014, vol. 7, p. 79.
Lee, K.-H. and Morris-Natschke, S.L., Pure Appl. Chem., 1999, vol. 71, p. 1045. https://doi.org/10.1351/pac199971061045
Kaledin, V.I., Zhukova, N.A., Grek, O.R., Tolstikova, T.G., Popova, N.A., Nikolin, V.P., Sorokina, I.V., and Pozdnyakova, S.V., J. Clin. Experim. Med., 2005, p. 53.
Vedernikov, D.N. and Roshchin, V.I., Russ. J. Bioorg. Chem., 2012, vol. 38, p. 762. https://doi.org/10.1134/S1068162012070229
Yakovleva, M.P., Vydrina, V.A., Sayakhov, R.R., and Ishmuratov, G.Yu., Chem. Nat. Compd., 2018, vol. 54, p. 795. https://doi.org/10.1007/s10600-018-2479-8
Barycza, B., PL Patent no. PL 232662, 2019.
Li, J., Chang, L.-C., Hsieh, K.-Y., Hsu, P.-L., Capuzzi, S.J., Zhang, Y.-C., Li, K.-Po, Morris-Natschke, S.L., Goto, M., and Lee, K.-H., Bioorg. Med. Chem. Lett., 2019, vol. 27, p. 2871. https://doi.org/10.1016/j.bmc.2019.05.016
Kazakova, O.B., Giniyatullina, G.V., Tolstikov, G.A., Kataev, V.E., and Musin, R.Z., Russ. J. Bioorg. Chem., 2009, vol. 35, p. 645. https://doi.org/10.1134/S1068162009050173
Nader, N.P., Marziyeh, B., Ertan, Ş., and Tuncay, T., J. Iran. Chem. Soc., 2014. https://doi.org/10.1007/s13738-014-0415-9
Callies, O., Sánchez-Cañete, M.P., Gamarro, F., Jiménez, I.A., Castanys, S., and Bazzocchi, I., J. Med. Chem., 2016, vol. 59, p. 1880. https://doi.org/10.1021/acs.jmedchem.5b01429
Neises, B. and Steglich, W., Angew. Chem. Int. Ed., 1978, vol. 17, p. 522. https://doi.org/10.1002/anie.197805221
Boden, E.P. and Keck, G.E., J. Org. Chem., 1985, vol. 50, p. 2394. https://doi.org/10.1021/jo00213a044
Popov, S., Steroids, 2020, vol. 162, p. 108698. https://doi.org/10.1016/j.steroids.2020.108698
Liu, J.-H., Zhu, Z.-F., Tang, J., Jiang, A.-Q., Hua, L.-F., and Chen, L., Chin. Chem. Lett., 2015, vol. 26, p. 759. https://doi.org/10.1016/j.cclet.2015.04.002
Gordon, A. and Ford, R., Sputnik khimika, Moscow: Mir, 1976.
Petrenko, N.I., Elantseva, N.V., Petukhova, V.Z., Shakirov, M.M., Shul’ts, E.E., and Tolstikov, G.A., Chem. Nat. Compd., 2002, vol. 38, p. 331. https://doi.org/10.1023/A:1021621907515
Funding
This work was financially supported by the Russian Academy of Sciences, in the framework of the “Basic Principlesof Chemistry”, topic no. 8 “Chemo-, Regio-, and Stereoselective Transformations of Terpenoids, Steroids, and Lipids in the Directed Synthesis of Low-Molecular-Weight Bioregulators” (State Reg. no. AAAA-A17-117011910023-2, 2017), using the equipment of the “Chemistry” Center for Collective Use, Ufa Institute of Chemistry, Russian Academy of Sciences, and the “Agidel” Regional Center for Collective Use, Ufa Federal Research Center, Russian Academy of Sciences.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare no conflict of interest.
Additional information
Translated from Zhurnal Organicheskoi Khimii, 2021, Vol. 57, No. 11, pp. 1624–1632 https://doi.org/10.31857/S0514749221110082.
Rights and permissions
About this article
Cite this article
Yakovleva, M.P., Vydrina, V.A., Sayakhov, R.R. et al. Synthesis of [2+1] Conjugates of Betulic Acid with α,ω-Diols. Russ J Org Chem 57, 1861–1867 (2021). https://doi.org/10.1134/S1070428021110087
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070428021110087