Abstract
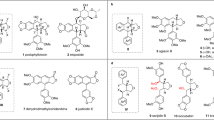
The first total synthesis of dilignan Boehmenan has been achieved. A biomimetic oxidative coupling of the ferulic acid methyl ester in the presence of silver oxide is the crucial step in the synthesis sequence, generating the dihydrobenzofuran skeleton. Hydroxyl group was protected with DHP and reducted with LiAlH 4 to afford the intermediate diol. The diol was condensated with the derivative of ferulic acid, then removed the protecting groups, to get Boehmenan. Meanwhile, a study on the ring-opening reaction of the intermediate dihydrobenzofuran neolignan under base conditions was described.

The first total synthesis of dilignan Boehmenan was achieved through eight steps from vanillin. Meanwhile, a study on ring-opening reaction of the intermediate dihydrobenzofuran neolignan under base conditions was described.



Similar content being viewed by others
References
. Seca A M L, Silva A M S, Silvestre A J D, Cavaleiro J A S, Domingues F M J and Pascoal-Neto C 2001 Phytochemistry 56 759
Wu P L, Wu T S, He C X, Su C H and Lee K H 2005 Chem. Pharm. Bull. 53 56
Rudiyansyah T and Garson M J 2006 J. Nat. Prod. 69 1218
Chin Y M, Jones W P, Rachman I, Riswan S, Kardono L B S, Chai H B, Farnsworth N R, Cordell G A, Swanson S M, Cassady J M and Kinghorn A D 2006 Phytother. Res. 20 62
Sasaki T, Li W, Morimura H, Li S, Li Q, Asada Y and Koike K 2011 Chem. Pharm. Bull. 59 1396
Ward R S 1995 Nat. Prod. Rep. 12 183
Koubaa I and Damak M 2003 Fitoterapia 74 18
Yang X W, Zhao P J, Ma Y L, Xiao H T, Zuo Y Q, He H P, Li L and Hao X J 2007 J. Nat. Prod. 70 521
Chen J J, Yang C S, Peng C F, Chen I S and Miaw C L 2008 J. Nat. Prod. 71 1016
Cutillo F, D’Abrosca B, DellaGreca M, Fiorentino A and Zarrelli A 2003 J. Agric. Food Chem. 51 6165
Xia Y M, Wang W, Yang F K and Chang L 2010 Chem. Res. Chin. Univ. 31 947
Pieters L, Dyck S V, Gao M, Bai R, Hamel E, Vlietinck A and Lemiere G 1999 J. Med. Chem. 42 5475
Ihara M, Suzuki M, Fukumoto K, Kametani T and Kabuto C 1988 J. Am. Chem. Soc. 110 1963
Kumar P, Raju S V N, Reddy R S and Pandey B 1994 Tetrahedron Lett. 35 1289
Orlandi M, Rindone B, Molteni G, Rummakko P and Brunow G 2001 Tetrahedron 57 371
Apers S, Paper D, Burgermeister J, Baronikova S, Dyck S V, Lemiere G, Vlietinck A and Pieters L 2002 J. Nat. Prod. 65 728
Zhou L X and Lin M 2000 Chin. Chem. Lett. 11 515
Patney H K 1991 Synth. Commun. 21 2329
Hon Y S, Lee C F, Chen R J and Szu P H 2001 Tetrahedron 57 5991
Eklund P C, Riska A and Sjoholm R E 2002 J. Org. Chem. 67 7544
Bringmaann G, Breuning M and Henschel P 2004 Org. Synth. 10 448
Ralph J, Quideau S, Grabber J H and HatfieId R D 1994 Chem. Soc. Perkin Trans. 1 3485
M, Orlandi M, Rindone B, Rummakko P and Zoia L 2006 J. Phys. Org. Chem. 19 592
Sako M, Hosokawa H, Ito T and Iinuma M 2004 J. Org. Chem. 69 2598
Acknowledgements
This work was supported by the National Natural Science Foundation of Shandong (No. ZR2010HM023), the Chinese Medicine Administration Bureau of Gansu Province (No.GZK-2011-62).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
XIA, Y., DAI, X., LIU, H. et al. First total synthesis of Boehmenan. J Chem Sci 126, 813–820 (2014). https://doi.org/10.1007/s12039-014-0599-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12039-014-0599-7