Abstract
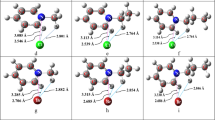
The density functional theory method (DFT) has been applied to investigate the competitive coordination upon formation of the bivariant (with respect to the coordination node composition; MN2O2 or MN2S2) isomers of Be(II) and Hg(II) bischelates based on thioxoindanone derivatives of azomethines, azines, and azoles. The simulation-revealed effects of the properties of the central metal atom and the composition of the ligating donor atoms on the relative stability of the complexes isomers (the MN2O2 or MN2S2 node) has been in agreement with the prediction based on the empirical Pearson’s hard and soft acids and bases (HSAB) principle.
Similar content being viewed by others
References
Garnovskii, A.D., Garnovskii, D.A., Vasil’chenko, I.S., Burlov, A.S., Sadimenko, A.S., and Sadekov, I.D., Russ. Chem. Rev., 1997, vol. 66, no. 5, p. 389. DOI: 10.1070/RC1997v066n05ABEH000258.
Garnovskii, A.D., Nivorozhkin, A.L., and Minkin, V.I., Coord. Chem. Rev., 1993, vol. 126, p. 1. DOI: 10.1016/00108545(93)85032-Y.
Bourget-Merle, L., Lappert, M.F., and Severn, J.R., Chem. Rev., 2002, vol. 102, p. 3031. DOI: 10.1021/cr010424r.
Kogan, V.A., Kharabaev, N.N., Osipov, O.A., and Kochin, S.G., Zh. Strukt. Khim., 1981, vol. 22, no. 1, p. 126.
Kharabaev, N.N., Koord. Khim., 1991, vol. 17, no. 5, p. 579.
Kharabayev, N.N., Starikov, A.G., and Minkin, V.I., Doklady Chem., 2014, vol. 458, no. 2, p. 181. DOI: 10.1134/S0012500814100024.
Pearson, R.G., J. Am. Chem. Soc., 1963, vol. 85, no. 22, p. 3533. DOI: 10.1021/ja00905a001.
Pirson, R.G., Russ. Chem. Rev., 1997, vol. 40, no. 7. DOI: 10.1070/RC1971v040n07ABEH003854.
Pearson, R.G., Coord. Chem. Rev., 1990, vol. 100, p. 403. DOI: 10.1016/0010-8545(90)85016-L.
Parr, R. and Yang, W., Density-Functional Theory of Atoms and Molecules, New York: Oxford University Press, 1989.
Sousa, S.F., Fernandes, P.A., and Ramos, M.J., J. Phys. Chem. (A), 2007, vol. 111, no. 42, p. 10439. DOI: 10.1021/jp0734474.
Burke, K. and Wagner, L.O., Int. J. Quantum Chem., 2013, vol. 113, no. 2, p. 96. DOI: 10.1002/qua.24259.
Tsipis, A.C., Coord. Chem. Rev., 2014, vol. 272, p. 1. DOI: 10.1016/jccr.2014.02.023.
Perdew, J.P., Burke, K., and Ernzerhof, M., Phys. Rev. Lett., 1996, vol. 77, p. 3865. DOI: 10.1103/PhysRevLett.77.3865.
Tao, J., Perdew, J.P., Staroverov, V.N., and Scuseria, G.E., Phys. Rev. Lett., 2003, vol. 91, p. 146401. DOI: 10.1103/PhysRevLett.91.146401.
Becke, A.D., Phys. Rev., 1988, no. 38, p. 3098. DOI: 10.1103/PhysRevA.38.3098.
Lee, C., Yang, W., and Parr, R.G., Phys. Rev., 1988, no. 37, p. 785. DOI: 10.1103/PhysRevB.37.785.
Sennikova, E.V., Korshunov, O.Yu., Borodkin, G.S., Korobov, M.S., Vlasenko, V.G., Kharabaev, N.N., and Garnovskii, A.D., Russ. J. Gen. Chem., 2007, vol. 77, no. 10, p. 1802. DOI: 10.1134/S1070363207100222.
Frisch, M.J., Trucks, G.W., Schlegel, H.B., Scuseria, G.E., Robb, M.A., Cheeseman, J.R., Scalmani, G., Barone, V., Mennucci, B., Petersson, G.A., Nakatsuji, H., Caricato, M., Li, X., Hratchian, H.P., Izmaylov, A.F., Bloino, J., Zheng, G., Sonnenberg, J.L., Hada, M., Ehara, M., Toyota, K., Fukuda, R., Hasegawa, J., Ishida, M., Nakajima, T., Honda, Y., Kitao, O., Nakai, H., Vreven, T., Montgomery, J.A., Jr., Peralta, J.E., Ogliaro, F., Bearpark, M., Heyd, J.J., Brothers, E., Kudin, K.N., Staroverov, V.N., Keith, T., Kobayashi, R., Normand, J., Raghavachari, K., Rendell, A., Burant, J.C., Iyengar, S.S., Tomasi, J., Cossi, M., Rega, N., Millam, J.M., Klene, M., Knox, J.E., Cross, J.B., Bakken, V., Adamo, C., Jaramillo, J., Gomperts, R., Stratmann, R.E., Yazyev, O., Austin, A.J., Cammi, R., Pomelli, C., Ochterski, J.W., Martin, R.L., Morokuma, K., Zakrzewski, V.G., Voth, G.A., Salvador, P., Dannenberg, J.J., Dapprich, S., Daniels, A.D., Farkas, O., Foresman, J.B., Ortiz, J., Cioslowski, J., and Fox, D.J., GAUSSIAN’09, Revision D.01. Gaussian, Inc., Wallingford CT, 2013.
Zhurko, G.A. and Zhurko, D.A., Chemcraft, Version 1.6. http://wwwchemcraftprogcom.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © N.N. Kharabayev, A.G. Starikov, V.I. Minkin, 2015, published in Zhurnal Obshchei Khimii, 2015, Vol. 85, No. 11, pp. 1890–1895.
Rights and permissions
About this article
Cite this article
Kharabayev, N.N., Starikov, A.G. & Minkin, V.I. The Pearson’s HSAB principle in the quantum-chemical model of formation of the MN2O2 or MN2S2 coordination node in the bischelates of Be(II) and Hg(II) with ambidentate ligands based on azomethines and their cyclic analogs. Russ J Gen Chem 85, 2629–2633 (2015). https://doi.org/10.1134/S1070363215110201
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070363215110201