Abstract
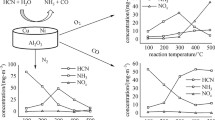
Catalytic decomposition of N2O in an air flow at at temperatures of ~420 to ~755 K in the presence of γ-Al2O3-based catalysts was studied. It was found that the degree of N2O decomposition in the presence of catalysts containing Mg, Ni, Co, and Fe does not exceed ~25% at a composite temperature of ~735 K and contact duration of the gas flow with the composite of 5–6 s. It was shown that the Al2O3–2RuO2–400 composite enables a nearly full decomposition of N2O in an air flow at temperatures of 738–753 K and contact durations of 7–15 s.




Similar content being viewed by others
REFERENCES
Ustinov, O.A, Kulyukhin, S.A., Voskresenskaya, Yu.A., et al., At. Energ., 2014, vol. 117, no. 6, p. 329.
Kulyukhin, S.A., Shadrin, A.Yu., Voskresenskaya, Yu.A., et al., J. Radioanal. Nucl. Chem., 2015, vol. 304, no. 1, p. 425.
Kosonen, H., Heinonen, M., Mikola, A., et al., Environ. Sci. Technol., 2016, vol. 50, no. 11, p. 5547.
Zil’berman, B.Ya., Ryabkov, D.V., and Mishina, N.E., RF Patent no. 2 596 816, Appl. 30.06.2015, Publ. 10.09.2016, Byull. no. 25
Ustinov, O.A., Shadrin, A.Yu., Batashov, M.V., et al., At. Energ., 2018, vol. 124, no. 2, p. 86.
Ustinov, O.A. and Yakunin, S.A., At. Energ., 2016, vol. 120, no. 2, p. 112.
Konsolakis, M., ACS Catalysis, 2015, vol. 5, no. 11, p. 6397.
Cornelissen, G., Rutherford, D.W., Arp H.P.H., et al., Environ. Sci. Technol., 2013, vol. 47, no. 14, p. 7704.
Kaczmarczyk, J., Zasada, F., Janas, J., et al., ACS Catal., 2016, vol. 6, no. 2, p. 1235.
Ivanova, Yu.A., Sutormina, E.F., Isupova, I.A., and Vovk, E.I., Kinet. Catal., 2017, vol. 58, no. 6, p. 793.
Seliverstov, A.F., Lagunova, Yu.O., Kulyukhin, S.A., and Ershov, B.G., Russ. J. Appl. Chem., 2017, vol. 90, no. 3, p. 393. https:/doi.org/10.1134/S107042721702011.
Baza dannykh IK spektrov (Database of IR Spectra), IR-Spektrensammlung der ANSYCO GmbH http://www.ansyco.de. Access on 10.02.2019
NIST Standard Reference Database Number 69, http://webbook.nist.gov/chemistry/. Access on 10.02.2019
Leont’ev, A.V., Fomicheva, O.A., Proskurnina, M.V., and Zefirov, N.S., Russ. Chem. Rev., 2001, vol. 70, no. 2, p. 91. https://doi.org/10.1070/RC2001v070n02ABEH000631
Entsiklopediya znanii Pandia.ru. Azota okisly (Knowledge Encyclopedia Pandia.ru). http://www.pandia.ru/135128, access on 10.02.2019
Mateski, A. and Mateski, B., Thermochim. Acta, 2006, vol. 446, p. 113.
Tada, S., Yokoyama, M., Kikuchi, R., et al., J. Phys. Chem. C, 2013, vol. 117, no. 28, p. 14652.
Yan, L., Ren, T., Wang, X., et al., Appl. Catal. B, 2003, vol. 45, p. 85.
Yan, L., Zhang, X., Ren, T., et al., Chem. Commun., 2002, p. 860.
Dacquin, J.P., Lancelot, C., Dujardin, C., et al., Appl. Catal., B, 2009, vol. 91, nos. 3–4, p. 596.
Wu, Y., Dujardin, C., Granger, P., et al., J. Phys. Chem. C, 2013, vol. 117, no. 27, p. 13989.
Piskorz, W., Zasada, F., Stelmachowski, P., et al., J. Phys. Chem. C, 2013, vol. 117, no. 36, p. 18488.
Ohnishi, C., Asano, K., Iwamoto, S., et al., Catal. Today, 2007, vol. 120, p. 145.
Shimizu, A., Tanaka, K., and Fujimori, M., Chemosphere, 2000, vol. 2, p. 425.
Giecko, G., Borowiecki, T., Gac, W., and Kruk, J., Catal. Today, 2008, vol. 137, p. 403.
Gonc¸alves, A.A.S., Costa, M.J.F., Zhang, L. ,et al., Chem. Mater., 2018, vol. 30, no. 2, p. 436.
Zasada, F., Stelmachowski, P., Maniak, G., et al., Catal. Lett., 2009, vol. 127, nos. 1–2, p. 126.
Chellam U., Xu, Z.P., and Zeng, H.C., Chem. Mater., 2000, vol. 12, p. 650.
Yan, L., Ren, T., Wang, X., et al., Catal. Commun., 2003, vol. 4, p. 505.
Spetsow, E.A., Russ. J. Appl. Chem., 2015, vol. 88, no. 9, p. 1381. https://doi.org/10.1134/S1070427215090013
Komvokisa, V.G., Martic, M., Delimitis, A., et al., Appl. Catal., B, 2011, vol. 103, nos. 1–2, p. 62.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
FUNDING
The study was financially supported in part by the Ministry of Science and Higher Education of the Russian Federation (research topic no. AAAA-A18-118021990023-6).
CONFLICT OF INTERESTS
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Kulyukhin, S.A., Rumer, I.A., Gorbacheva, M.P. et al. Catalytic Decomposition of N2O in Air Flows of Gas-Purification Systems in Radiochemical Industries. Radiochemistry 62, 177–188 (2020). https://doi.org/10.1134/S1066362220020058
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1066362220020058