Abstract
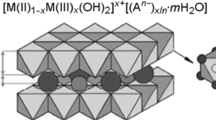
The possibility of removing dyes from aqueous solutions containing 137Cs, 90Sr, 90Y, and U(VI) using layered double oxides (LDOs) and hydroxides (LDHs) of Mg and Al was examined. The use of LDO-Mg-Al allows rapid and efficient pretreatment of radioactive solutions containing Cs and Sr radionuclides to remove dyes for the subsequent recovery of Cs and Sr with other selective sorbents. The use of LDO-Mg-Al, LDH-Mg-Al-OH, and LDH-Mg-Al-CD (CD is β-cyclodextrin) allows simultaneous removal from aqueous solutions of UO2(NO3)2 (initial concentration <10−2 M), 90Y (an analog of trivalent f elements), and dye (degree of removal >90%).
Similar content being viewed by others
References
Kritskii, V.G. and Rodionov, Yu.A., Dezaktivatsiya ob”ektov yadernogo toplivnogo tsikla (Decontamination of Nuclear Fuel Cycle Facilities), St. Petersburg; KSI, 2013.
Milyutin, V.V., Alekhina, M.B., and Ryabchikov, B.E., Sovremennye metody ochistki tekhnogennykh stochnykh vod ot toksichnykh primesei (Modern Methods for Treatment of Industrial Wastewater to Remove Toxic Impurities), Moscow: Ross. Khimiko-Tekhnol. Univ. im. D.I. Mendeleeva, 2016.
Belovskii, I., Radiation will be painted white, https://www.gazeta.ru/social/2011/04/04/3574025.shtml (Visited Oct. 30, 2018).
O’Shea, K.E. and Dionysiou, D.D., J. Phys. Chem. Lett., 2012, vol. 3, no. 15, pp. 2112–2113.
Ahmed, M.B., Zhou, J.L., Huu Hao Ngo, et al., J. Hazard. Mater., 2017, vol. 323, part A, pp. 274–298.
The, C.Y., Budiman, P.M., Shak, K.P.Y., and Wu, T.Y., Ind. Eng. Chem. Res., 2016, vol. 55, no. 16, pp. 4363–4389.
Miller, D.J., Dreyer, D.R., Bielawski, Ch.W., et al., Angew. Chem. Int. Ed., 2017, vol. 56, no. 17, pp. 4662–4711.
Perlov, A.G., Tekhnologii ochistki prirodnykh vod (Processes for Natural Water Treatment), Moscow: ASV, 2016.
Ali, I., Chem. Rev., 2012, vol. 112, no. 10, pp. 5073- 5091.
Dremicheva, E.S. and Laptedul’che, N.K., Voda: Khim. Ekol., 2015, no. 8, pp. 63–68.
Khajeh, M., Laurent, S., and Dastafkan, K., Chem. Rev., 2013, vol. 113, no. 10, pp. 7728–7768.
Bo, A., Sarina, S., Liu, H., et al., ACS Appl. Mater. Interfaces, 2016, vol. 8, no. 25, pp. 16503–16510.
Zhou, J.Z., Wu, Y.Y., Liu, C., et al., Environ. Sci. Tech-nol., 2010, vol. 44, no. 23, pp. 8884–8890.
Palmer, S.J. and Frost, R.L., Ind. Eng. Chem. Res., 2010, vol. 49, no. 19, pp. 8969–8976.
Yu, S., Wang, X., Chen, Z., et al., J. Hazard. Mater., 2017, vol. 321, pp. 111–120.
Klimovich, I.V., Synthetic analogs of hydrotalcite in localization of radioactive elements from solutions, Cand. Sci. (Chem.) Dissertation, Moscow, 2013.
Majoni, S. and Hossenlopp, J.M., J. Phys. Chem. A, 2010, vol. 114, no. 49, pp. 12858–12869.
Radha, A.V., Vishnu Kamath, P., and Shivakumara, C., J. Phys. Chem. B, 2007, vol. 111, no. 13, pp. 3411–3418.
Prasanna, S.V. and Vishnu Kamath, P., Ind. Eng. Chem. Res., 2009, vol. 48, no. 13, pp. 6315–6320.
Koilraj, P. and Srinivasan, K., Ind. Eng. Chem. Res., 2011, vol. 50, no. 11, pp. 6943–6951.
Chitrakar, R., Sonoda, A., Makita, Y., and Hirotsu, T., Ind. Eng. Chem. Res., 2011, vol. 50, no. 15, pp. 9280–9285.
Xue, X., Gu, Q., Pan, G., et al., Inorg. Chem., 2014, vol. 53, no. 3, pp. 1521–1529.
Shan, R.-r., Yan, L.-g., Yang, Y.-m., et al., J. Ind. Eng. Chem., 2015, vol. 21, pp. 561–568.
Ahmed, I.M. and Gasser, M.S., Appl. Surf. Sci., 2012, vol. 259, pp. 650–656.
Lei, C., Zhu, X., Zhu, B., et al., J. Hazard. Mater., 2017, vol. 321, pp. 801–811.
Benselka-Hadj Abdelkader, N., Bentouami, A., Der-riche, Z., et al., Chem. Eng. J., 2011, vol. 169, nos. 1–3, pp. 231–238.
Kulyukhin, S.A., Krasavina, E.P., Gredina, I.V., et al., Radiochemistry, 2008, vol. 50, no. 5, pp. 493–501.
Kulyukhin, S.A., Krasavina, E.P., Rumer, I.A., and Gredina, I.V., INCS News, 2009, vol. 6, no. 3, pp. 26–31.
Kulyukhin, S.A., Krasavina, E.P., Rumer, I.A., and Klimovich, I.V., Radiochemistry, 2014, vol. 56, no. 6, pp. 593–606.
Kulyukhin, S.A., Krasavina, E.P., Gredina, I.V., and Mizina, L.V., Radiochemistry, 2010, vol. 52, no. 6, pp. 653–661.
Kulyukhin, S.A. and Krasavina, E.P., Radiochemistry, 2016, vol. 58, no. 4, pp. 405–408.
Tretyakov, Yu.D., Lukashin, A.V., and Eliseev, A.A., Russ. Chem. Rev., 2004, vol. 73, no. 9, pp. 899–921.
Measurement of concentrations of harmful substances in air of a working zone, MUK 4.1.803-4.1.878-99: Sb. metodicheskikh ukazanii Gos. sistemy sanitarno-epide-miologicheskogo normirovaniya RF (Coll. of Methodical Instructions of the State System of Sanitary-Epi-demiological Regulation of the Russian Federation), Moscow: Minzdrav Rossii, 1999, issue 35.
Bogolepov, A.A., Pshinko, G.N., and Kornilo-vich, B.Yu., J. Water Chem. Tekhnol., 2007, vol. 29, no. 1, pp. 9–14.
Raschet ravnovesii v analiticheskoi khimii (Calculation of Equilibria in Analytical Chemistry), www.chemequ.ru, Visited Oct. 30, 2018.
Funding
The study was financially supported in part by the Ministry of Science and Higher Education of the Russian Federation (project no. AAAA-A16-116 110 910 010-3).
Author information
Authors and Affiliations
Corresponding author
Additional information
Conflict of Interest
The authors declare that they have no conflict of interest.
Russian Text © The Author(s), 2019, published in Radiokhimiya, 2019, Vol. 61, No. 5, pp. 409–414.
Rights and permissions
About this article
Cite this article
Kulyukhin, S.A., Krasavina, E.P., Rumer, I.A. et al. Use of Layered Double Oxides and Hydroxides of Mg and Al for Removing Dyes from Aqueous Solutions Containing 137Cs, 90Sr, 90Y, and U(VI). Radiochemistry 61, 585–591 (2019). https://doi.org/10.1134/S1066362219050102
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1066362219050102