Abstract
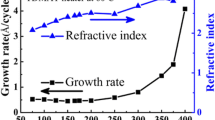
The annealing of amorphous TiOx films obtained by electron-beam evaporation under atmospheric conditions at temperatures from 300 to 400°C is found to lead to the formation of an anatase crystalline phase. According to Raman spectroscopy data, the increase in the fraction of the crystalline phase stops at an annealing temperature above 350°C. According to the results of X-ray diffraction analysis, the average crystallite diameter is about 23 nm. Electron-microscopy studies show that, upon annealing, the surface layer (15 nm thick) crystallizes in the films, and TiO2 nanocrystals with sizes from 4 to 10 nm are formed in the bulk. As the depth increases, the number of nanocrystals decreases.





Similar content being viewed by others
REFERENCES
J.-H. Lim, D. Bae, and A. Fong, J. Agric. Food Chem. 66, 13533 (2018). https://doi.org/10.1021/acs.jafc.8b06571
V. Moreno, M. Zougagh, and A. Rios, Anal. Chim. Acta 1050, 169 (2019). https://doi.org/10.1016/j.aca.2018.10.067
A. D. Modestov and O. Lev, J. Photochem. Photobiol., A 112, 261 (1998). https://doi.org/10.1016/s1010-6030(97)00269-4
B. A. Movchan, J. Mater. 48, 40 (1996). https://doi.org/10.1007/BF03223243
J. Singh, ASM J. 12, 27 (1996).
W. K. Halan and D. Lee, in High-Temperature Protective Coatings, Ed. by S. C. Singhal (Warrendale: Metall. Soc. AIME, 1983), p. 3.
R. Saravanan, F. Gracia, and A. Stephen, in Nanocomposites for Visible Light-induced Photocatalysis (Springer, Cham, 2017), p. 19. https://doi.org/10.1007/978-3-319-62446-4_2
W. Wang, B. Gu, L. Liang, W. A. Hamilton, D. J. Wesolowski, J. Phys. Chem. B 108, 14789 (2004). https://doi.org/10.1021/jp0470952
V. A. Lebedev, Candidate’s Dissertation in Chemistry (Moscow State Univ., Moscow, 2017).
G. Cacciato, M. Zimbone, F. Ruffino, and M. G. Grimaldi, in Green Nanotechnology—Overview and Further Prospects (InTech, London, 2016), p. 87. https://doi.org/10.5772/62620
E. Baranowska-Wojcik, D. Szwajgier, P. Oleszczuk, and A. Winiarska-Mieczan, Biol. Trace Elem. Res. 193, 118 (2019). https://doi.org/10.1007/s12011-019-01706-6
N. S. Kozhevnikova, E. S. Ul’yanova, E. V. Shalaeva, D. A. Zamyatin, A. O. Bokunyaeva, A. A. Yushkov, V. Yu. Kolosov, L. Yu. Buldakova, M. Yu. Yanchenko, T. I. Gorbunova, M. G. Pervova, A. N. Enyashin, and A. S. Vorokh, Kinet. Catal. 60, 325 (2019). https://doi.org/10.1134/S002315841903008X
X. Chen and S. S. Mao, Chem. Rev. 107, 2891 (2007). https://doi.org/10.1021/cr0500535
L. A. Dunyushkina, Introduction to Methods for Producing Film Electrolytes for Solid Oxide Fuel Cells (Inst. Vysokotemp. Elektrokhim. Ural. Otd. Ross. Akad. Nauk, Yekaterinburg, 2015) [in Russian].
S. G. Shymyrbek, MSc Thesis (Tomsk, 2018).
D. B. Zolotukhin, V. A. Burdovitsin, A.V. Tyun’kov, Yu. G. Yushkov, E. M. Oks, D. A. Golosov, and S. M. Zavadskii, Usp. Prikl. Fiz., No. 5, 442 (2017).
A. Niilisk, M. Moppel, M. Pars, I. Sildos, T. Jantson, T. Avarmaa, R. Jaaniso, and J. Aarik, Cent. Eur. J. Phys. 4, 105 (2006). https://doi.org/10.1007/s11534-005-0009-3
C.-P. Lin, H. Chen, A. Nakaruk, P. Koshy, and C. C. Sorrell, Energy Proc. 34, 627 (2013). https://doi.org/10.1016/j.egypro.2013.06.794
F. D. Hardcastle, J. Arkansas Acad. Sci. 65, 43 (2011). https://doi.org/10.54119/jaas.2011.6504
S. Nikodemski, A. A. Dameron, J. D. Perkins, R. P. O’Hayre, D. S. Ginley, and J. J. Berry, Sci. Rep. 6, 32830 (2016). https://doi.org/10.1038/srep32830
U. Balachandran and N. G. Eror, J. Solid State Chem. 42, 276 (1982). https://doi.org/10.1016/0022-4596(82)90006-8
S. Mamedov, Mater. Res. Soc. Symp. Proc. 1806, 1 (2015). https://doi.org/10.1557/opl.2015.377
M. N. Iliev, V. G. Hadjiev, and A. P. Litvinchuk, Vibr. Spectrosc. 64, 148 (2013). https://doi.org/10.1016/j.vibspec.2012.08.003
V. Swamy, M. C. Muddle, and Q. Dai, App. Phys. Lett. 89, 163118 (2006). https://doi.org/10.1063/1.2364123
K.-R. Zhu, M.-S. Zhang, Q. Chen, and Z. Yin, Phys. Lett. A 340, 220 (2005). https://doi.org/10.1016/j.physleta.2005.04.008
P. G. Li, M. Lei, and W. H. Tang, Mater. Lett. 64, 161 (2010). https://doi.org/10.1016/j.matlet.2009.10.032
X. Wei, G. Zhu, J. Fang, and J. Chen, Int. J. Photoenergy 18, 726872 (2013). https://doi.org/10.1155/2013/726872
G. M. Kuz’micheva, Tonk. Khim. Tekhnol. 10 (6), 5 (2015).
A. I. Gusev, Nanomaterials, Nanostructures, Nanotechnologies (Fizmatlit, Moscow, 2007) [in Russian].
H. A. Mahmoud, K. Narasimharao, T. T. Ali, and K. M. S. Khalil, Nanoscale Res. Lett. 13, 48 (2018). https://doi.org/10.1186/s11671-018-2465-x
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflicts of interest.
Rights and permissions
About this article
Cite this article
Nezhdanov, A.V., Zhukov, A.O., Shestakov, D.V. et al. Effect of the Annealing Conditions on the Formation of a Nanocrystalline Phase in TiOx Films. J. Surf. Investig. 17, 1078–1082 (2023). https://doi.org/10.1134/S1027451023050294
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1027451023050294