Abstract
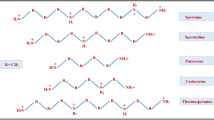
Modulation of differential gene expression and change of polyamine content by salt stress are analyzed for the first time in a well-known medicinal plant, Panax ginseng C.A. Meyer. Three ginseng genes (PgSPD, PgSAMDC, and PgADC) involved in polyamine biosynthesis showed differential up-regulation patterns after 1 and 7 days of salt treatments. The modulation of gene expression resulted in the elevation of total polyamine content with relatively high levels of spermidine and spermine, while putrescine level diminished depending on the salt concentration. Conversely, salt stress led to a significant increase in diamine oxidase and subsequent decline in polyamine oxidase. The proline content caused by salinity follows a similar pattern as the total polyamine content and exogenous spermidine also resulted in the alleviation of proline content under salinity. Further, polyamine biosynthesis inhibitors, such as cyclohexylamine and methylglyoxal bis-(guanylhydrazone) mediated down-regulation of PgSPD and PgSAMDC, and affected cellular polyamine levels. Thus, polyamines may enhance the ginseng plant tolerance in response to the salt stress by increasing the levels of endogenous polyamines.
Similar content being viewed by others
Abbreviations
- CHA:
-
cyclohexylamine
- DAO:
-
diamine oxidase
- EST:
-
expressed sequence tag
- MGBG:
-
methylglyoxal bis-(guanylhydrazone)
- PA:
-
polyamine
- PAO:
-
polyamine oxidase
- Put:
-
putrescine
- qRT-PCR:
-
quantitative real-time Reverse Transcription-Polymerase Chain Reaction
- Spd:
-
spermidine
- SPD:
-
spermidine synthase
- Spm:
-
spermine
References
Sairam, R.K. and Tyagi, A., Physiology and Molecular Biology of Salinity Stress Tolerance in Plants, Curr. Sci., 2004, vol. 86, pp. 407–421.
Kuznetsov, Vl.V., Radyukina, N.L., and Shevyakova, N.I., Polyamines and Stress: Biological Role, Metabolism, and Regulation, Russ. J. Plant Physiol., 2006, vol. 53, pp. 583–604.
Groppra, M.D. and Benavides, M.P., Polyamines and Abiotic Stress: Recent Advances, Amino Acids, 2008, vol. 34, pp. 35–45.
Kusano, T., Berberich, T., Tateda, C., and Takahashi, Y., Polyamines: Essential Factors for Growth and Survival, Planta, 2008, vol. 228, pp. 367–381.
Santa-Cruz, A., Acosta, M., Perez-Alfocea, F., and Bolarin, M.C., Changes of Free Polyamine Levels Induced by Salt Stress in Leaves of Cultured and Wild Tomato Species, Physiol. Plant., 1999, vol. 101, pp. 341–346.
Walter, D.R., Resistance to Plant Pathogens; Possible Role of Free Polyamines and Polyamine Catabolism, New Phytol., 2003, vol. 159, pp. 109–115.
Duan, J., Li, J., Guo, S., and Kang, Y., Exogenous Spermidine Affects Polyamine Metabolism in Salinity-Stressed Cucumis sativus Roots and Enhances Short-Term Salinity Tolerance, J. Plant Physiol., 2008, vol. 165, pp. 1620–1635.
Parvin, S., Kim, Y.J., Pulla, R.K., Sathiyamoorthy, S., Miah, M.G., Kim, Y.J., Wasnik, N.G., and Yang, D.C., Identification and Characterization of Spermidine Synthase Gene from Panax ginseng, Mol. Biol. Rep., 2010, vol. 37, pp. 923–932.
Murashige, T. and Skoog, F., A Revised Medium for Rapid Growth and BioAssays with Tobacco Tissue, Physiol. Plant., 1963, vol. 15, pp. 473–497.
Parvin, S., Pulla, R.K., Shim, J.S., Kim, Y.J., Jung, D.Y., and Yang, D.C., Isolation and Characterization of Cinnamoyl-CoA Reductase Gene from Panax ginseng C. A. Meyer, J. Ginseng Res., 2008, vol. 32, pp. 232–237.
Sathiyamoorthy, S., In, J.G., Gayathri, S., Kim, Y.J., and Yang, D.C., Generation and Gene Ontology Based Analysis of Expressed Sequence Tags (EST) from a Panax ginseng C. A. Meyer Roots, Mol. Biol. Rep., 2009, vol. 37, pp. 3465–3472.
Marce, M., Brown, D.S., Capelli, T., Figueras, X., and Tiburcio, A.F., Rapid High Performance Liquid Chromatographic Method for the Quantitation of Polyamines as Their Dansyl Derivatives: Application to Plant and Animal Tissues, J. Chromatogr., B, 1995, vol. 666, pp. 329–335.
Bates, L.S., Waldren, R.P., and Teare, I.D., Rapid Determination of Free Proline for Water Stress Studies, Plant Soil, 1973, vol. 39, pp. 205–207.
Su, G.X., An, Z.F., Zhang, W.H., and Liu, Y.L., Light Promotes the Synthesis of Lignin through the Production of H2O2 Mediated by Diamine Oxidases in Soybean Hypocotyls, J. Plant Physiol., 2005, vol. 162, pp. 1297–1303.
Korolev, S., Ikeguchi, Y., Sharina, T., Beasley, S., Arrowsmith, C., Edwards, A., Joachimiak, A., Pegg, A.E., and Savchenko, A., The Crystal Structure of Spermidine Synthase with a Multisubstrate Adduct Inhibitor, Nat. Struct. Biol., 2002, vol. 9, pp. 27–31.
Munns, R., Comparative Physiology of Salt and Water Stress, Plant Cell Environ., 2002, vol. 25, pp. 239–250.
Liu, J.H., Nada, K., Honda, C., Kitashiba, H., Wen, X.P., Pang, X.M., and Moriguchi, T., Polyamine Biosynthesis of Apple Callus under Salt Stress: Importance of the Arginine Decarboxylase Pathway in Stress Response, J. Exp. Bot., 2006, vol. 57, pp. 2589–2599.
Reggiani, R., Bozo, S., and Bertani, A., Changes in Polyamine Metabolism in Seedlings of Three Wheat (Triticum aestivum L.) Cultivars Differing in Salt Sensitivity, Plant Sci., 1994, vol. 102, pp. 121–126.
Jimenez-Bremont, J.F., Becerra, F.A., Hernandez-Lucero, E., Rodríguez-Kessler, M., Acosta-Gallegos, J.A., and Ramírez-Pimentel, J.G., Proline Accumulation in Two Bean Cultivars under Salt Stress and the Effect of Polyamines and Ornithine, Biol. Plant., 2006, vol. 50, pp. 763–766.
Goyal, M. and Asthir, B., Polyamine Catabolism Influences Antioxidative Defense Mechanism in Shoots and Roots of Five Wheat Genotypes under High Temperature Stress, Plant Growth Regul., 2010, vol. 60, pp. 13–25.
Roychoudhurya, A., Basub, S., and Senguptab, D.N., Amelioration of Salinity Stress by Exogenously Applied Spermidine or Spermine in Three Varieties of Indica Rice Differing in Their Level of Salt Tolerance, J. Plant Physiol., 2010, doi 10.1016/j.jplph.2010.07.009
Torrigiani, P., Serafini-Fracassini, D., and Bagni, N., Polyamine Biosynthesis and Effect of Dicyclohexylamine during the Cell Cycle of Helianthus tuberosus, Plant Physiol., 1986, vol. 84, pp. 148–152.
Mitchell, J.L.A., Mahan, D.W., McCann, P.P., and Qasba, P., Dicyclohexylamine Effect on HTC Cell Polyamine Content and Ornithine Decarboxylase Activity, Biochim. Biophys. Acta, 1985, vol. 840, pp. 309–315.
Bitonti, A.J., McCann, P.P., and Sjoerdsma, A., Restriction of Bacterial Growth by Inhibition of Polyamine Biosynthesis by Using Monofluoromethylornithine, Difluoromethylornithine and Dicyclohexylamine Sulphate, Biochem. J., 1982, vol. 208, pp. 435–441.
Feirer, R.P., Wann, S.R., and Einspahr, D.W., The Effects of Spermidine Synthase Inhibitors on In Vitro Plant Development, Plant Growth Regul., 1985, vol. 3, pp. 319–327.
Author information
Authors and Affiliations
Corresponding author
Additional information
This text was submitted by the authors in English.
Rights and permissions
About this article
Cite this article
Parvin, S., Lee, O.R., Sathiyaraj, G. et al. Modulation of polyamine levels in ginseng hairy root cultures subjected to salt stress. Russ J Plant Physiol 59, 757–765 (2012). https://doi.org/10.1134/S102144371206012X
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S102144371206012X