Abstract
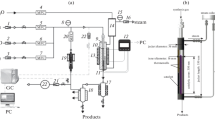
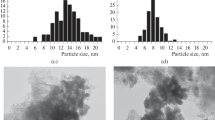
The results of a study on the deactivation of an industrial Co–Al2O3/SiO2 catalyst under the conditions of highly productive Fischer–Tropsch synthesis of long-chained hydrocarbons at high pressure (6 MPa) are presented. It was found that an increase in the synthesis temperature led to a decrease in the rate of catalyst deactivation. The effect of the thermal agglomeration of cobalt particles and the carbonization of catalyst surface on the activity of the catalyst under the test conditions was insignificant. A correlation between the rate of catalyst deactivation and the selectivity for С19+ hydrocarbons was found; this correlation indicated the blocking of chain growth centers by synthesized waxes as the main reason for the rapid loss of catalyst activity.






Similar content being viewed by others
REFERENCES
Martinelli, M., Gnanamani, M.K., LeViness, S., Jacobs, G., and Shafer, W.D., Appl. Catal., A, 2020, vol. 608, no. 117740.
Van de Loosdrecht, J., Botes, F., Ciobica, I., Ferreira, A., Gibson, P., Moodley, D., Saib, A., Visagie, J., Weststrate, C.J., and Niemantsverdriet, J., Comprehensive Inorganic Chemistry II: From Elements to Applications, Amsterdam: Elsevier, 2013, p. 525.
Savost’yanov, A.P., Yakovenko, R.E., Sulima, S.I., Bakun, V.G., Narochnyi, G.B., Chernyshev, V.M., and Mitchenko, S.A., Catal. Today, 2017, vol. 279, p. 107.
Yakovenko, R.E., Savost’yanov, A.P., Narochniy, G.B., Soromotin, V.N., Zubkov, I.N., Papeta, O.P., Svetogorov, R.D., and Mitchenko, S.A., Catal. Sci. Technol., 2020, vol. 10, no. 22, p. 7613.
Claeys, M., Dry, M.E., Steen, E.V., Berge, P., Booyens, S., Crous, R., Helden, P., Labuschagne, J., Moodley, D., and Saib, A., Am. Chem. Soc. Catal., 2015, vol. 5, no. 2, p. 841.
Keyvanloo, K., Fisher, M.J., Hecker, W.C., Lancee, R.J., Jacobs, G., and Bartholomew, C.H., J. Catal., 2015, vol. 327, p. 33.
Jiang, Z., Zhao, Y., Huang, C., Song, Y., Li, D., Liu, Z., and Liu, Z., Fuel, 2018, vol. 226, p. 213.
Wolf, M., Gibson, E.K., Olivier, E.J., Neethling, J.H., Catlow, C.R.A., Fischer, N., and Claeys, M., Am. Chem. Soc. Catal., 2019, vol. 9, no. 6, p. 4902.
Ma, W., Jacobs, G., Shafer, W.D., Pendyala, V.R.R., Xiao, Q., Hu, Y., and Davis, B.H., Catal. Lett., 2016, vol. 146, no. 7, p. 1204.
Pölmann, F., Kern, C., Röbler, S., and Jess, A., Catal. Sci. Technol., 2016, vol. 6, p. 6593.
Savost’yanov, A.P., Yakovenko, R.E., Narochniy, G.B., Sulima, S.I., Bakun, V.G., Soromotin, V.N., and Mitchenko, S.A., Catal. Commun., 2017, vol. 99, p. 25.
Jinglin, Y., Xuejin, F., Yuebing, X., and Xiaohao, L., Catal. Sci. Technol., 2020, vol. 10, no. 4, p. 1182.
Rahmati, M., Safdari, M., Fletcher, T., Argyle, M., and Bartholomew, C.H., Chem. Rev., 2020, vol. 120, no. 10, p. 4455.
Yakovenko, R.E., Zubkov, I.N., Narochnyi, G.B., Papeta, O.P., Denisov, O.D., and Savost’yanov, A.P., Kinet. Catal., 2020, vol. 61, no. 2, p. 310.
Mitchenko, S.A., Savost’yanov, A.P., Narochnyi, G.B., Yakovenko, R.E., Bakun, V.G., Sulima, S.I., and Yakuba, E.O., Kinet. Catal., 2017, vol. 58, no. 1, p. 81.
Moodley, D., loosdrecht, J., Saib, A., Overett, M., Datye, A., and Niemantsverdriet, J., Appl. Catal., A, 2009, vol. 354, nos. 1–2, p. 102.
Choudhury, H., Cheng, X., Afzal, S., Prakash, A.V., Tatarchuk, B.J., and Elbashir, N.O., Catal. Today, 2020, vol. 343, p. 112.
Savost’yanov, A.P., Eliseev, O.L., Yakovenko, R.E., Narochniy, G.B., Maslakov, K.I., Zubkov, I.N., Soromotin, V.N., Kozakov, A.T., Nicolskii, A.V., and Mitchenko, S.A., Catal. Lett., 2020, vol. 150, no. 7, p. 1932.
PDF-2. The powder diffraction file TM. International Center for Diffraction Data (ICDD), 2012. http://www.icdd.com.
Loosdrecht, J., Balzhinimaev, B., Dalmon, J., Niemantsverdriet, J., Tsybulya, S., Saib, A., Berge, P., and Visagie, J., Catal. Today, 2007, vol. 123, nos. 1–4, p. 293.
Todic, B., Ma, W., Jacobs, G., Davis, B.H., and Bukur, D.B., J. Catal., 2014, vol. 311, p. 325.
Rytter, E., Tsakoumis, N.E., and Holmen, A., Catal. Today, 2016, vol. 261, p. 3.
Moodley, D., Claeys, M., van Steen, E., van Helden, P., Kistamurthy, D., Weststrate, K.-J., Niemantsverdriet, H., Saib, A., Erasmus, W., and van de Loosdrecht, J., Catal. Today, 2020, vol. 342, p. 59.
Weststrate, C.J., Ciobica, I.M., Saib, A.M., Moodley, D., and Niemantsverdriet, J., Catal. Today, 2014, vol. 228, p. 106.
Bian, G.-Z., Fujishita, N., Mochizuki, T., Ning, W.-S., and Yamada, M., Appl. Catal., A, 2003, vol. 252, no. 2, p. 251.
Funding
This study was supported by the Russian Foundation for Basic Research (project no. 20-33-90155), funded by the Ministry of Education and Science of the Russian Federation (state contract no. 2019-0990), and carried out with the use of the equipment of the Nanotechnology Center for Collective Use at the Platov South-Russian State Polytechnic University (NPI).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflicts of interest.
Additional information
Translated by V. Makhlyarchuk
Abbreviations and notation: FTS, Fischer–Tropsch synthesis; TEM, transmission electron microscopy; XRD, X-ray diffraction; GHSV, gas hourly space velocity; DSC, differential scanning calorimetry; DTA, differential thermal analysis.
Rights and permissions
About this article
Cite this article
Soromotin, V.N., Yakovenko, R.E., Medvedev, A.V. et al. Reasons for the Rapid Deactivation of a Cobalt Catalyst in the High-Efficiency Fischer–Tropsch Synthesis of C19+ Hydrocarbons. Kinet Catal 62, 845–852 (2021). https://doi.org/10.1134/S002315842106015X
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S002315842106015X