Abstract
The kinetic characteristics, mechanisms of activity, and relationship between the antioxidant activity and the molecular and supramolecular structure of fullerene C60 and some of its N-monosubstituted amino acid derivatives have been studied. The introduction of an amino acid substituent in the C60 fullerene molecule led to an increase in its antiradical activity in the free radical oxidation of a water-soluble target (fluorescein). The mechanism of the antioxidant activity of amino acid derivatives of fullerene is not associated with the hydrogen atom transfer, electron donation, or catalysis of peroxide decomposition. It was demonstrated that the structure of the amino acid substituent does not affect the antiradical properties, which are thus determined only by the effective total surface area of the nanoparticles of the fullerene C60 derivatives and increase when their size decreases. This surface can be characterized as nanowalls on which the radical death occurs. A change in the concentration of the compounds in solution does not lead to a change in the relative antiradical activity and hence in the nanoparticle size. The results of this study are important for understanding the biological activity of this group of compounds.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
INTRODUCTION
Free radical processes are an integral part of metabolism in all living systems. On the one hand, the excessive formation of free radicals leads to a damage to cell structures and can lead to the development of various diseases. On the other hand, free radicals generated by phagocytising cells are an important factor in nonspecific immune defense. In addition, free radical processes are involved in the regulation of many important biological processes, including cell proliferation and apoptosis [1].
Of particular interest are the antiradical properties of various nanomaterials, including carbon-based nanostructures such as fullerene C60. It was noted that the antiradical properties of fullerene C60 affect the kinetics of radical polymerization of styrene and methyl methacrylate until the polymerization is completely suppressed [2, 3]. The kinetics and stoichiometry of the reaction of fullerene C70 with ozone were described in [4]. Many publications demonstrated the unique properties of fullerenes C60 and their derivatives in interactions with living organisms. It was shown that fullerene derivatives with amino acids such as proline, ε-aminocaproic acid, and arginine exhibit membranotropic activity and percolate through cell membranes in ionized form, thereby causing cell depolarization [5]. An immunofluorescence microscopy study of carboxylated fullerene C61(CO2H)2 showed that the water-soluble derivatives of fullerene C60 can penetrate through the membrane layer of the cell and that the above compound is predominantly localized in the membrane and mitochondria-rich fractions of the cell [6]. As mitochondria are the main primary sources of free radicals, while fullerene C60 derivatives have antiradical properties due to which they are often called “radical sponges” [7], these compounds can be involved in the regulation of oxidative stress, as indicated by the data of [6].
Therefore, it is important to study in detail the inhibitory effect of fullerene C60 and its derivatives with respect to various free radicals and to reveal the relationship between the chemical structure of fullerene compounds and their antiradical activity. The activity with the superoxide radical was first studied in fullerenols [8] and hexasulfobutyl fullerenes [9]. In these studies, the superoxide radical anion was generated using the xanthine/xanthinoxidase system; the consumption of the superoxide radical was judged from the intensity of chemiluminescence of lucigenin. It was found that hexasulfobutyl fullerenes had high activity with the superoxide radical and inhibited 60 and 96% of free radicals at concentrations of 50 and 100 μM, respectively, while fullerenols inhibited only 59 and 70% at 50 and 100 μM, respectively. It was concluded that the derivatives with fewer substituents had higher activity as the system of conjugated π electrons was retained in this case. It was also demonstrated in [8] that the presence of fullerenol at concentrations of 0–47 µM in the xantine/xanthinoxidase system did not affect the enzyme activity, which eliminated the ambiguity of interpretation of studies using this system.
The introduction of a substituent from the group of organic acids significantly increased the antiradical activity of fullerene in a nonpolar medium (a model system of initiated oxidation of ethylbenzene) [10].
The antioxidant properties of fullerene adducts with malonic acid were investigated at approximately the same time. An electron paramagnetic resonance (EPR) study using the xanthine/xanthinoxidase system as a source of the superoxide radical and 5,5-dimethyl-1-pyrroline-N-oxide as a spin trap showed that the triadducts of fullerene with malonic acid completely inhibited the superoxide radical anions [11].
A computer simulation of the mechanism by which the superoxide radical is inhibited by a fullerene C60 derivative with three malonic acid residues was suggested in [12]. According to this model, the reaction occurs in several stages:
(1) \({\text{O}}_{2}^{{\bullet - }}\) transfers the unpaired electron to the fullerene nucleus with formation of O2 and the radical anion of the fullerene derivative.
(2) The electron is transferred from the radical anion of the fullerene derivative to the second \({\text{O}}_{2}^{{\bullet - }}\) followed by the transfer of the proton H+ to it, forming –OOH.
(3) The transfer of the next hydrogen proton H+ to –OOH leads to the formation of hydrogen peroxide H2O2.
However, similar catalytic properties of the malonic derivative of fullerene have not been confirmed experimentally.
The addition of benzyl radicals to the fullerene C60 molecule in a toluene medium was shown by EPR using the 13C spin trap [13]. The existence of at least two intermediate radical adducts with three (\({{{\text{R}}}_{3}}{\text{C}}_{{60}}^{\bullet }\)) and five (\({{{\text{R}}}_{5}}{\text{C}}_{{60}}^{\bullet }\)) benzyl radicals stable at 50°C was found. It was assumed that up to 15 benzyl radicals could be attached to one fullerene molecule. It was also found by mass spectrometry that one fullerene molecule had up to 34 methyl groups attached to it.
Pulsed radiolysis studies revealed effective binding of the solvated electron, hydroxyl radical, and singlet oxygen by various carboxylated fullerenes. In the interaction with the solvated electron, the carboxylated fullerene derivatives are believed to form a radical monoanion [14].
The mechanisms proposed for the binding of the hydroxyl radical \({}^{\bullet }{\text{OH}}\) by hydroxylated fullerene derivatives are as follows: (1) \({}^{\bullet }{\text{OH}}\) captures H from the hydroxyl group, forming an ether bond on the surface of the fullerene core; (2) \({}^{\bullet }{\text{OH}}\) is attached to the double bond of the fullerene nucleus [15].
The inhibitory effect of hydroxyfullerene on the nitroxide \({\text{NO}}{}^{\bullet }\) was demonstrated both for chemical and biological systems [16]. In the latter case, the administration of hydroxyfullerene to mice previously injected with a source of the \({\text{NO}}{}^{\bullet }\) radical (sodium nitroprusside) prevented a decrease in the activity of the enzymes: superoxide dismutase (SOD), catalase, glutathione-s-transferase, and glutathione peroxidase.
To summarize the available data on the mechanisms of the inhibitory action of fullerene and its derivatives with respect to free radicals, we can conclude that studies of the antioxidant properties were performed for a limited number of structures, and the relationship between the chemical structure and the antiradical activity was studied insufficiently. The proposed mechanisms are mostly hypothetical. The activity of fullerene and its derivatives with such a biologically active form of oxygen as hydrogen peroxide was not considered either. Therefore, investigation of the mechanism of activity of various fullerene derivatives is an important challenge.
It is of interest to study fullerene conjugates with molecules involved in metabolism of biological systems, in particular, conjugates with proteinogenic amino acids [17] and amino acids that are neuromediators and/or have any other biological activity from the viewpoint of their use as regulators of free radical processes. The toxicity of fullerene C60 and its derivatives has long been the subject of debate. On the one hand, there is evidence that C60 fullerene has a toxic effect on biological objects in the presence of light [18]. On the other hand, there is a lot of evidence that injection of even high doses of fullerene C60 does not negatively affect the life activities of living organisms. Thus, the administration of fullerene to mice in a dose of 2.5 g/kg did not lead to the death of animals or any changes in their behavior [19]. Parenteral administration of N-monohydrofullerenyl-L-serine to mice in a dose of 80 μg/kg did not affect the behavior and viability of mice [20].
As is known, in aqueous solutions, fullerene and its derivatives are in the form of a colloidal solution of nanoparticles [21, 22]. Their physicochemical properties are determined by both the molecular structure and the size of nanoparticles [23, 24].
Here, we studied the mechanisms of the antioxidant action and correlations between the structure and antiradical activity of N-monoamino acid derivatives of fullerene C60 based on the analysis of their behavior in various model systems.
EXPERIMENTAL
Preparation of an Aqueous Dispersion of Fullerene С60
An aqueous dispersion of fullerene C60 was prepared by the procedure of [25]. A saturated solution in toluene was obtained, to which water was added, and the solution was treated in the dark in an ultrasonic bath (42 kHz, 50 W) for a few hours with intervals for cooling.
Synthesis of Amino Acid Derivatives of Fullerene (ADFs)
The following compounds: N-(monohydrofullerenyl)-L-alanine potassium salt (Н–C60–L-Ala–ОK), N-(monohydrofullerenyl)-D-alanine potassium salt (Н–C60–D-Ala–ОK), N-(monohydrofullerenyl)-L-valine potassium salt (Н–C60–L-Val–ОK), N-(monohydrofullerenyl)-D-valine potassium salt (Н–C60–D-Val–ОK), N-(monohydrofullerenyl)-L-aspartic acid dipotassium salt (Н–C60–L-Asp–(ОK)2), N-(monohydrofullerenyl)-D-aspartic acid dipotassium salt (Н–C60–D-Asp–(ОK)2), N-(monohydrofullerenyl)-ε-aminocaproic acid potassium salt (Н–C60–ε-ACA–ОK) were obtained as a result of a one-stage synthesis by direct addition of the amino acid residue to the fullerene nucleus by the procedure of [26].
Fluorimetry
The kinetic curves of the time dependences of fluorescence intensity of fluorescein were measured on a FluoroLog 3.21 spectrofluorimeter (HORIBA Scientific, United States) using a cell compartment thermostatted at 37 ± 0.2°C. The observed fluorescence was detected at a wavelength of 515 nm with an excitation wavelength of 491 nm. The sample temperature was maintained using a 12108-15 liquid thermostat (Cole-Parmer, United States).
The relative antioxidant activity was determined by the method presented in [27].
The solutions of fluorescein and 2,2'-azobis(2-amidinopropane) dihydrochloride (AAPH) were prepared in a phosphate buffer (75 mM, pH 7.4) and then incubated for 10 min at 37°C. The solutions of fluorescein and the test sample were the first to be mixed in the fluorimeter cell; the AAPH solution was introduced last, and the recording of the kinetic curve of fluorescence intensity started immediately. The final concentrations of the mixture components in the cell were: 9.6 mM (AAPH), 10–8 M (fluorescein), and 2.5 × 10–6 M (test solution). The phosphate buffer (75 mM) was used for blank experiment; trolox (6‑hydroxy-2,5,7,8-tetramethylchromane-2-carboxylic acid) at a concentration of 1.2 × 10–6 M was used as a reference drug. The relative antiradical activity (RAA) was calculated as the ratio of the differences in the areas under the fluorescence quenching curves for the test sample (Ssamp) and trolox (Str) including the areas in the blank experiment (without addition of antioxidants) (Sbl), as well as the concentrations of trolox (Ctr) and test samples (Csamp):
Voltammetry
The TsvetYauza-01-AA amperometric unit (Khimavtomatika, Russia) is an electrochemical cell with a glassy carbon anode and a stainless steel cathode, to which a potential difference in the range 0.1–2.0 V can be applied [28]. The analyte was injected into the eluent flow (distilled water) pumped through the electrochemical cell at a rate of 1.2 mL/min using a six-way dosing valve with a loop volume of 20 μL. When the sample passed through the cell, the current of electrochemical oxidation was recorded (nA); its time curve (s) was displayed on the monitor, and the area S (nA s) was determined for the samples. The dependence of S on the applied voltage was plotted based on the data obtained at different anode potentials, and the ionization potential of the test substance was found.
Model Reaction with the DPPH Radical
To evaluate the antioxidant activity by the mechanism associated with elimination of a hydrogen atom, we observed the reaction of the compounds under study with the stable chromogen radical 2,2-diphenyl-1-picrylhydrazyl (DPPH) [29].
The test sample in various volumes was added to the DPPH solution mixed with water in a 1 : 1 ratio to the total volume of the reaction system of 3.6 mL. The variation of optical density was recorded at a wavelength of 517 nm (maximum absorption in the visible region of the spectrum of DPPG) on a PE 5400VI spectrophotometer (EKROSKHIM, Russia).
Potentiometric Study of the Decomposition Kinetics of Hydrogen Peroxide
A three-electrode printed circuit consisting of a reference electrode, auxiliary electrode, and working electrode modified with Prussian blue and nickel hexacyanoferrate by the procedure of [30] was placed, while constantly stirring, in a 5-mL cell filled with a 1.6 × 10–5 М solution of the sample in a phosphate buffer solution (0.05 М KH2PO4/K2HPO4, 0.1 M KCl, pH 6.0). The measurements were performed at a working potential of 0 V relative to the built-in reference electrode. The background current was recorded for 5–7 min until it reached a constant level; then hydrogen peroxide was injected to a final concentration of 5 × 10–5 M in the cell, and variation of its concentration was recorded according to the potential drop on the electrode.
Initiated Oxidation of Liposomes
Liposomes based on phosphatidylcholine (PC) serve as models for the study of biochemical processes in biomembranes, in particular, lipid peroxidation (LPO) and regulation of oxidation with various substances. For the preparation of liposomes, a suspension of soybean PC (P 3644, Sigma) in a phosphate buffer (pH 7.4, ionic strength 1 mM) was used. PC liposomes were prepared according to [31] at PC concentration of 1 mg/mL. To initiate liposome oxidation, a water-soluble azo initiator (AAPH, Fluka, Germany) with a final liposome concentration of 0.33 mM in solution was used. Liposomes at a concentration of 0.1 mg/mL were oxidized in quartz cells thermostatted at a physiological temperature (37°C) using a spectrophotometer (Perkin Elmer, Germany). The kinetics of formation of the products of lipid peroxidation (diene conjugates, DCs) was recorded with time at a maximum absorption wavelength of DCs of 234 nm. To study the inhibitory effect of N-monohydrofullerenyl-D-alanine, a solution of this amino acid derivative at a concentration of 4.81 × 10–4 M in various volumes (30–80 μL) was introduced in the liposome solution before oxidation. For comparison with the effect of trolox, the oxidation of liposomes was also performed with addition of trolox (0.5–2 μL) at a concentration of 0.5 mM.
Statistical Data Processing
The statistical processing of the experimental data was performed using the Microsoft® Office Excel and OriginPro 8.5 computer programs.
RESULTS AND DISCUSSION
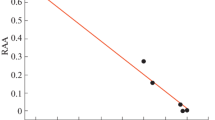
The results of screening of the relative antiradical activity (RAA) of fullerene C60 and six of its amino acid derivatives by fluorescence observation of inhibited free radical destruction of a fluorescent target molecule were presented in [32]. According to Fig. 1, the introduction of an amino acid substituent leads to a significant increase in the antiradical activity of compounds that form aggregates of similar size in the aqueous phase. At the same time, the amino acid derivatives of fullerene with different structures of substituent lie on the same regression line, which indicates that the structure of the amino acid substituent does not significantly affect the antiradical activity of the compounds. Further studies showed that the correlation dependence shown in Fig. 1 is also valid when the synthesis of the same compound gave nanoparticles with another average size. In this case, the corresponding point lies on the same regression line. Thus, N-monohydrofullerenyl-D-alanine obtained in the second synthesis and having a nanoparticle size similar to that of N-monohydrofullerenyl-L-valine also coincides with this compound in the antiradical activity.
Correlation between RAA and the nanoparticle size of the amino acid derivatives of fullerene C60: (1) C60; (2) H–C60–L-Ala–OK; (3) Н–C60–D-Ala–OK, synthesis 1; (3') H–C60–D-Ala–OK, synthesis 2; (4) H–C60–L-Val–OK; (5) H–C60–D-Val–OK; (6) Н–C60–L-Asp–(OK)2; and (7) Н–C60–D-Asp–(OK)2. The Pearson coefficient of pair correlation is rxy = – 0.981.
This correlation between the RAA and the nanoparticle size of amino acid derivatives of fullerene (ADFs) is apparently due to the steric factor: when the nanoparticle size increases, the number of molecules isolated from the radicals lying in the reaction medium increases in proportion to the cube of their radius, which just leads to a decrease in the effective RAA values.
To simulate the protective effect on biomembranes, we studied the inhibitory effect of fullerene derivatives on the initiated oxidation of PC in the liposomal model system. For this, N-(monohydrofullerenyl)-D-alanine obtained in synthesis 2 was used (Fig. 2).
Increase in the optical density (D) of diene conjugates (DCs) in the AAPH-induced (0.33 mM) oxidation of PC liposomes (0.1 mg/mL): (1) in a blank experiment; (2) after addition of trolox at a concentration of 0.17 μM; and (3) after addition of Н–C60–D-Ala–OK (synthesis 2) at a concentration of 4.81 μM.
In complex heterogeneous systems that simulate biological membranes (PC liposomes), the compounds exhibit dose-dependent inhibitory activity, reducing the rate of DC formation during the oxidation of PC liposomes initiated by AAPH.
The protective effect of the D-alanine derivative (synthesis 2) in the liposome model, calculated from the areas above the PC oxidation curve (2) by the method similar to the one used for a homogeneous fluorescent system, was 0.03 for trolox, which is nine times lower than the value obtained for inhibited oxidation of fluorescein. This phenomenon is evidently associated with the increased affinity of the nanoparticles of fullerene substituted with amino acid for aqueous medium:
where АОА is the antioxidant activity in the model system of oxidation of PC liposomes; \(S_{{{\text{samp}}}}^{{\text{H}}}\), \(S_{{{\text{bl}}}}^{{\text{H}}}\), and \(S_{{{\text{tr}}}}^{{\text{H}}}\) are the areas over the sample oxidation curves in the absence of the sample and in the presence of trolox, respectively, calculated to the horizontal asymptote drawn at the level of the plateau of the oxidation curve; Ctr and Сsamp are the trolox and sample concentrations, respectively.
It is worthwhile to note that the kinetic curves of PC liposome oxidation in the presence of trolox and the test sample are different. In experiments with trolox, the oxidation curves are S-shaped, indicating that trolox is quickly and completely consumed along with almost complete inhibition of oxidation, and this is followed by uninhibited oxidation of lipid membranes. The shape of the oxidation curves in experiments with fullerene derivatives suggests partial decrease in the intensity of oxidations throughout the experiment with a slight change in the inhibitory effect of the test compound. Thus, fullerene derivatives act as inhibitors for much longer periods of time. This character of inhibition of free radical oxidation is preferable from the viewpoint of obtaining optimum biological effects (protection of living organisms from the oxidative stress) as free radicals in living systems play not only the negative role, but are also involved in normal biological processes.
The linear character of the dependence of the increase in the transformation time of 0.25 of the oxidized compound relative to the control experiment on the concentration of the fullerene derivative (Fig. 3) suggests that dilution does not affect the values of RAA and AOA and hence the rate constants of the interaction of monoamino acid derivatives of fullerene with radicals. The fact that the antiradical activity of the compounds is in negative linear correlation with respect to the size of aggregates formed by them leads, in turn, to the conclusion that dilution of solution does not cause a change in the size of nanoparticles formed by fullerene derivatives, in contrast to some published data [33].
Dependence of the increase in the conversion time of 0.25 of the initial oxidized compound relative to the control experiment (\({{t}_{{{{{0.25}}_{{{\text{samp}}}}}}}}\) – \({{t}_{{{{{0.25}}_{{{\text{bl}}}}}}}}\)) on the concentration of H–C60–Ala–OK in the reaction medium: (a) oxidation of fluorescein and (b) oxidation of PC liposomes.
The straightening of the damped exponential curves of fluorescein oxidation in the absence and in the presence of inhibitor in semilogarithmic coordinates indicates that the process is pseudo-first-order under the chosen experimental conditions (Fig. 4). This agrees with the results of our other experiments, which showed that the half-transformation time of fluorescein is independent of its initial concentration in the range from 10–8 to 5 × 10–8 M at a fixed AAPH concentration of 9.6 mM. This is also consistent with the theoretical assumptions because due to the low (1.3 × 10–6 s–1 [34]) rate constant of initiator decomposition, its amount does not markedly change, and thus the concentration of radicals in the reaction system is in the quasi-stationary equilibrium state.
(a) Kinetic curves of the decrease in the fluorescence intensity (I) of fluorescein as a result of its degradation in its interaction with the radicals formed during the decomposition of AAPH and (b) their semilogarithmic anamorphoses in a (1) blank experiment and in the presence of H–C60–D-Ala–OK in amounts of (2) 2.89, (3) 6.27, and (4) 9.41 μM. The initial concentrations of fluorescein and AAPH solutions are 0.01 and 9.6 mM, respectively.
The general kinetic equation of the pseudo-first-order reaction in the case of fluorometric observation of the consumption of the initial component has the form
where I0 and I are the initial and current intensities of fluorescence of fluorescein, \(k{\kern 1pt} '\) is the rate constant of the pseudo-first-order reaction, \({{k}_{{{\text{Fl}}}}}\) is the bimolecular rate constant of the interaction of fluorescein with radicals, and \({{C}_{{\text{R}}}}\) is the concentration of free radicals in the system.
Using Eq. (4) we obtain
Integrating the areas under the kinetic curves, we derive the relationship between the increase in the conversion time of 0.25 of fluorescein relative to the control experiment and RAA:
where \({{t}_{{{\text{ind}}}}}\) is the induction period for trolox, and \(k_{0}^{'}\) is the rate constant of the pseudo-first-order reaction in the control experiment (without addition of antioxidants).
As
where Wi is the rate of generation of radicals in the system, substitution of (7) and (8) into (1) gives
where \({{C}_{{{\text{AO}}}}}\) is the antioxidant concentration in the system.
Thus, the linear relationship between (\({{t}_{{{{{0.25}}_{{{\text{samp}}}}}}}}\) – \({{t}_{{{{{0.25}}_{{{\text{bl}}}}}}}}\)) and CAO (Fig. 3a) indicates that the RAA does not change when the concentration of the fullerene derivative in solution changes in the given range of concentrations.
The electron-donating ability of ADFs was studied by amperometry, recording the area under the current curve S with an increase in the potential difference between electrodes U [30]. According to Fig. 5, significant electrochemical oxidation of monoamino acid derivatives starts at anodic potentials of more than 1.2 V.
The highly active antioxidants such as flavonoids start to donate electrons already at anode potentials of 0.4–0.6 V [35]. As is known, flavonoids can exhibit antioxidant activity both by the radical mechanism (hydrogen atom transfer (HAT)) and ionic mechanisms based on electron transfer (sequential proton loss–electron transfer (SPLET) and electron transfer–proton transfer (ET–PT)) [36]. Based on the voltammograms obtained for fullerene C60 derivatives (Fig. 5), it can be concluded that these compounds do not exhibit antiradical activity by the mechanism associated with electron transfer because of the high oxidation potential. The different values of the slope are due to the change in the electrode response on different dates of experiment.
The observation of the optical density of the solution of the stable 2,2-diphenyl-1-picrylhydrazyl chromogen radical after addition of Н–C60–D-Ala–ОK and Н–C60–ε-ACA–ОK to the reaction system showed that they were inactive with respect to this radical. The stable shielded DPPH radical interacts only with antioxidants that can easily donate a hydrogen atom. Thus, amino acid derivatives of fullerene do not exhibit activity according to the above-mentioned HAT mechanism.
The antiperoxide activity (the ability to destroy peroxide compounds by various mechanisms) of the D-alanine derivative was studied by adding it to a buffer system containing hydrogen peroxide, whose concentration was controlled potentiometrically. According to Fig. 6, after the mixing of the components of the reaction system (vertical section of the curve), the measured value of the potential, which was proportional to the concentration of hydrogen peroxide, did not change over the whole time of experiment. Thus, the fullerene derivative under study is not a catalyst for peroxide decomposition and thus cannot promote or prevent the branching of chain peroxidation reactions.
CONCLUSIONS
The data obtained by voltammetry, potentiometry, and DPPH analysis indicate that the antioxidant mechanism of action of amino acid derivatives of fullerene is not associated with electron donation or hydrogen atom transfer and that these compounds are not catalysts for decomposition of peroxides. At the same time, the linear relationship between the antiradical properties of the N-monosubstituted amino acid derivatives of fullerene in the fluorescent model and the sizes of nanoparticles formed by them in aqueous solutions suggests that the total surface area of nanoparticles is the main factor that determines the antioxidant activity of these compounds. The introduction of an N-amino acid substituent significantly increases the inhibitory efficiency of the compounds with respect to the oxidation of a water-soluble target (fluorescein).
The structure of the amino acid substituent has no significant effect on the antiradical activity of nanoparticles, as confirmed by the comparison of the RAA of compounds with different substituents with the RAA of the same compound from different syntheses, which formed aggregates with different average sizes. The linear dependence of the inhibitory effect of nanoparticles on the concentration in both the fluorescence and liposomal models indicates that a change in the concentration does not lead to a change in the size characteristics of nanoparticles. Fullerene and its derivatives are electron-deficient molecules possessing a system of conjugated double bonds. The accessibility of the π-bonds of atoms is evidently just the factor that determines the possibility of radical addition reactions for nanoparticles of the amino acid derivatives of fullerene. The active surface of these nanoparticles can be characterized as nanowalls on which free radicals die.
REFERENCES
Kondakova, I.V., Doctoral Sci. (Med.) Dissertation, Tomsk, 2005.
Yumagulova, R.Kh., Medvedeva, N.A., Kuznetsov, S.I., Kraikin, V.A., and Kolesov, S.V., Kinet. Catal., 2014, vol. 55, no. 1, p. 67.
Yumagulova, R.Kh., Kuznetsov, S.I., Diniakhmetova, D.R., Frizen, A.K., Kraikin, V.A., and Kolesov, S.V., Kinet. Catal, 2016, vol. 57, no. 3, p. 383.
Razumovskii, S.D., Bulgakov, R.G., Ponomareva, Yu.G., and Budtov, V.P., Kinet. Catal. 2006, vol. 47, no. 3, p. 347.
Andreev, I.M., Romanova, V.S., Petrukhina, A.O., and Andreev, S.M., Fiz. Tverd. Tela, 2002, vol. 44. no. 4, p. 683.
Fol Foley, S., Growley, C., Smaihi, M., Bonfils, C., Erlanger, B.F., Seta, P., and Larroque, C., Biochem. Biophys. Res. Commun., 2002, vol. 294, p. 116.
Beuerle, F., Lebovitz, R., and Hirsch, A., Medicinal Chemistry and Pharmacological Potential of Fullerenes and Carbon Nanotubes. Carbon Materials: Chemistry and Physics, Cataldo, F. and Da Ros, T., Eds., Dordrecht: Springer, 2008, vol. 1, p. 51. https://doi.org/10.1007/978-1-4020-6845-4_3
Chiang, L.Y., Lu, F-J., and Lin, J-T., Chem. Commun., 1995, no. 12, p. 1283.
Chi, Y., Bhonsle, J.B., Canteenwala, T., Huang, J-P., Shiea, J., Chen, B-J., and Chiang, L.Y., Chem. Lett., 1998, vol. 27, no. 5, p. 465.
Yakupova, L.R., Sakhautdinov, I.M., Malikova, R.N., and Safiullin, R.L., Kinet. Catal., 2019, vol. 60, no. 1, p. 21.
Dugan, L.L., Turetsky, D.M., Du, C., Lobner, D., Wheeler, M., Almli, C.R., Shen, C.K-F., Luh, R-Y., Choi, D.W., and Lin, T-S, Proc. Natl. Acad. Sci. USA, 1997, vol. 94, no. 17, p. 9434.
Osuna, S., Swart, M., and Solà, M., Chem. Eur. J., 2010, vol. 16, no. 10, p. 3207. https://doi.org/10.1002/chem.200902728
Kru Krustic, P.J., Wasserman, E., Keizer, P.N., Morton, J.R., and Preston, K.F., Science, 1991, vol. 254, no. 5035, p. 1183.
Bensasson, R.V., Brettreich, M., Frederiksen, J., Göttinger, H., Hirsch, A., Land, E.J., Leach, S., McGarvey, D.J., and Schönberger, H., Free Radic. Biol. Med., 2000, vol. 29, no. 1, p. 26.
Wang, Z., Wang, S., Lu, Z., and Gao, X., J. Cluster Sci., 2015, vol. 26, p. 375.
Mirkov, S.M., Djordjevic, A.N., Andric, N.L., Andric, S.A., Kostic, T.S., Bogdanovic, M.B., Vojinovic-Miloradov, M.B., and Kovacevic, R.Z., Nitric Oxide, 2004, vol. 11, no. 2, p. 201. https://doi.org/10.1016/j.niox.2004.08.003
Sun, T. and Xu, Z., Bioorg. Med. Chem. Lett., 2006, vol. 16, no. 14, p. 3731.
Piotrovskii, L.B., Eropkin, M.Yu., Eropkina, E.M., Dumpis, M.A., and Kisilev, O.I., Psikhofarmokologiya i Biologicheskaya Narkologiya, 2007, vol. 7, no. 2, p. 1548.
Moussa, F., Trivin, F., Ceolin, R., Hadchouel, M., Sizaret, P.Y., Greugny, V., Fabre, C., Rassat, A., and Szwarc, H., Fullerene Sci. Technol., 1996, vol. 4, no. 1, p. 21.
Andreev, S.M., Babakhin, A.A., Petrukhina, A.O., Romanova, V.S., and Petrov, R.V., Dokl. AN, 2000, vol. 370, no. 2, p. 261.
Bobylev, A.G., Pen’kov, N.V., Troshin, P.A., and Gudkov, S.V., Biophysics, 2015, vol. 60, no. 1, p. 30.
Andreev, S.M., Purgina, D.D., Bashkatova, E.N., Garshev, A.V., Maerle, A.V., and Khaitov, M.R., Nanotechnol. Russ., 2014, vol. 9, nos. 7–8, p. 369.
Burgess, S., Vishnyakov, A., Tsovko, C., and Neimark, A.V., J. Phys. Chem. Lett., 2018, vol. 9, no. 17, p. 4872. https://doi.org/10.1021/acs.jpclett.8b01696
Park, S., Xie, Y., and Weaver, M.J., Langmuir, 2002, vol. 18, no. 15, p. 5792. https://doi.org/10.1021/la0200459
Andrievsky, G.V., Kosevich, M.V., Vovk, O.M., Shelkovsky, V.S., and Vashchenko, L.A., J. Chem. Soc. Chem. Commun., 1995, no. 12, p. 1281.
Romanova, V.S., Tsyryapkin, V.A., Lyakhovetsky, Y.I., Parnes, Z.N., and Vol`pin, M.E., Russ. Chem. Bull., 1994, vol. 43, no. 6, p. 1090.
Ou, B., Hampsch-Woodill, M., and Prior, R.L., J. Agric. Food Chem., 2001, vol. 49, no. 10, p. 4619. https://doi.org/10.1021/jf010586o
Yashin, A.Ya., Ross. Khim. Zh., 2008, vol. 52, no. 2, p. 130.
Bondet, W., Brand-Williams, W., and Berset, C., Lebensm.-Wiss u.-Technol., 1997, vol. 30, no. 6, p. 609.
Karpova, E.V., Karyakina, E.E., and Karyakin, A.A., J. Electrochem. Soc., 2017, vol. 164, no. 5, p. 3056.
Sazhina, N.N., Antipova, A.S., Semenova, M.G., and Pal’mina, N.P., Russ. J. Bioorg. Chem., 2019, vol. 45, no. 2, p. 34.
Volkov, V.A., Yamskova, O.V., Shepel’, N.E., Romanova, V.S., Kurilov, D.V., Tregubov, A.V., Vyshivannaya, O.V., Voronkov, M.V., Yamskov, I.A., Misin, V.M., Zubareva, N.D., and Kustov, L.M., Russ. J. Phys. Chem. A, 2019, vol. 93, no. 11, p. 2152.
Timofeeva, G.I., Romanova, V.S., and Lopanova, L.A., Russ. Chem. Bull., 1996, vol. 45, no. 4, p.834.
Niki, E., Methods in Enzymology, 1990, vol.186, p. 100.
Yashin, A.Ya., Sorbtsionnye i Khromatograficheskie Protsessy, 2014, vol. 14, no. 3, p. 419.
Rimarčík, J., Lukeš, V., Klein, E., and Ilčin, M., J. Mol. Struct., 2010, vol. 952, nos. 1–3, p. 25.
Funding
This study was financially supported by the Ministry of Science and Education of the Russian Federation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflicts of interest.
Additional information
Translated by L. Smolina
Abbreviations and notation: AAPH, 2,2'-azobis(2-amidinopropane) dihydrochloride; ET–PT, sequential electron transfer–proton transfer; SPLET, sequential proton loss–electron transfer; AOA, antioxidant activity; ADFs, amino acid derivatives of fullerene; DCs, diene conjugates; DPPH, 2,2-diphenyl-1-picrylhydrazyl; RAA, relative antiradical activity; LPO, lipid peroxidation; SOD, superoxide dismutase; PC, phosphatidylcholine; EPR, electronic paramagnetic resonance.
Rights and permissions
Open Access. This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Volkov, V.A., Voronkov, M.V., Sazhina, N.N. et al. Mechanism of the Antioxidant Activity and Structure–Activity Relationship of N-Monosubstituted Amino Acid Derivatives of Fullerene С60 . Kinet Catal 62, 395–403 (2021). https://doi.org/10.1134/S0023158421030095
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0023158421030095