Abstract—
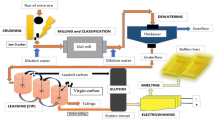
This paper presents results of analysis of the patent, scientific, and technical literature concerned with the physicochemical and technological foundations of the preparation of high-purity arsenic-containing compounds from various raw materials. Considerable attention is paid to processing of unconventional raw materials (lewisite detoxification products, nonferrous metals industry waste, and III–V semiconductor materials fabrication waste). Arsenic-containing substances (elemental arsenic, arsenic trichloride, arsenic trioxide, and arsine) containing less than 1 ppm by weight of regulated impurities have been prepared and characterized.



Similar content being viewed by others
REFERENCES
Fedorov, V.A. and Churbanov, M.F., Preparation of high-purity arsenic, in Vysokochistye veshchestva (High-Purity Substances), Churbanov, M.F. , Eds., Moscow: Nauchnyi Mir, 2018, pp. 566–592.
Fedorov, V.A., Menshchikova, T.K., Vargunin, A.I., Brekhovskikh, M.N., and Myslitskii, O.E., Processes and apparatuses for the extraction and purification of elemental arsenic and its compounds, Khim. Tekhnol., 2021, no. 1, pp. 44–48.
Zhukov, E.G., Nikolashin, S.V., Babitsyna, A.A., Polulyak, E.S., and Fedorov, V.A., Purification of crude arsenic by vacuum sublimation, Vysokochist. Veshchestva, 1995, no. 5, pp. 49–75.
Fedorov, V.A., Zhukov, E.G., Nikolashin, S.V., Potolokov, V.N., Serov, A.V., and Smetanin, A.V., Sublimation purification of crude arsenic recovered from nonferrous waste, Inorg. Mater., 2001, vol. 37, no. 10, pp. 1011–1016.
Veviorskii, A.P., Ivanov, V.N., Krol’, A.L., and Mosalev, O.V., USSR Patent 1632996, 1991.
Masato, K. and Satoshi, N., Jpn. Patent 5331569, 1993.
Lingqi Cao, CN Patent 1096058, 1994.
Zorin, A.D., Karataev, E.N., Stepanova, L.V., Sidorov, Yu.V., Sidnev, V.I., Kosyak, A.M., Kapashin, V.P., Kholstov, V.I., and Anastasov, A.D., RF Patent 2170279, 2001.
Fedorov, V.A., Efremov, A.A., Grinberg, E.E., Zhukov, E.G., Baranov, Yu.I., Kuznetsov, B.A., Potepalov, V.P., and Kholstov, V.I., Problems in obtaining ultrapure arsenic and arsenic compounds using lewisite, Ross. Khim. Zh., 1994, vol. 38, no. 2, pp. 25–33.
Pashkov, V.M., Ultrapurification of arsenic by directional solidification, Vysokochist. Veshchestva, 1988, no. 1, pp. 123–126.
Fedorov, V.A., Gasanov, A.A., Potolokov, N.A., Menshchikova, T.K., and Brekhovskikh, M.N., Ultrapurification of arsenic by crystallization, Inorg. Mater., 2018, vol. 54, no. 10, pp. 1027–1032.
Fedorov, V.A., Potolokov, N.A., Menshchikova, T.K., and Brekhovskikh, M.N., Hydride process for the preparation of high-purity arsenic, Inorg. Mater., 2018, vol. 54, no. 10, pp. 1033–1038.
Radu, I.O., Dumbac, A., and Dragan, E.V., RO Patent 96547, 1989.
Bessarabov, A.M., Nikolashin, S.V., Zhukov, E.G., Efremov, V.A., and Fedorov, V.A., Modeling of the degassing of industrial arsenic, a lewisite detoxification product, Theor. Found. Chem. Eng., 2004, vol. 38, no. 1, pp. 67–71.
Nisel’son, L.A., Gasanov, A.A., and Yaroshevskii, A.G., Liquid–vapor equilibrium in the arsenic–selenium system, Vysokochist. Veshchestva, 1993, no. 4, pp. 56–61.
Wu Guoyuan, CN Patent 1155587, 1998.
Masasi, N., Naobuki, K., and Hiroyuki, T., Jpn. Patent 57-160914, 1982.
Koujirou, K., Reizou, N., and Akifumi, I., Jpn. Patent 55075923, 1980.
Hisasi, D., Hideo, S., and Sazo, A., Jpn. Patent 58-24378, 1983.
Isabaev, S.M., Polukarov, A.N., and Kuzgibekova, Kh., USSR Patent 937333, 1982.
Fedorov, V.A., Efremov, V.A., Kazanskii, L.N., Efremov, A.A., and Menshchikova, T.K., Liquid–vapor equilibria in the arsenic trichloride–acetyl chloride and arsenic trichloride–chloroacetyl chloride binary systems, Vysokochist. Veshchestva, 1994, no. 2, pp. 47–57.
Yuiti Ova, Masasi Nakano, and Nabesama Takeharu Nabesama, Jpn. Patent 62-100413, 1988.
Fedorov, V.A., Menshchikova, T.K., Vargunin, A.I., Brekhovskikh, M.N., and Myslitskii, O.E., Technological principles of arsenic extraction from various raw materials, Khim. Tekhnol., 2021, no. 6, pp. 283–288.
Yasuo Kuroda and Konti Yamagawa, Jpn. Patent 61-146716. 1986.
Fedorov, V.A., Efremov, A.A., Efremov, E.A., and Filippov, E.P., Preparation of extrapure-grade arsenic trichloride, Elektronika, 1976, no. 4.
Yuiti Ova, Tadasi Nakano, and Takezi Nabesima, Jpn. Patent 62-235216, 1987.
Yuiti Ova, Tadasi Nakano, and Takezi Nabesima, Jpn. Patent 62-235217, 1987.
Fedorov, V.A., Potolokov, N.A., Menshikova, T.K., and Brekhovskikh, M.N., Physicochemical aspects of the preparation of high-purity arsenic-containing substances from various raw materials, Inorg. Mater., 2017, vol. 53, no. 10, pp. 1024–1032.
Fedorov, V.A., Filippov, E.P., Lebedeva, R.N., Kuznetsova, N.P., Pashinkin, A.S., Sokolov, E.B., and Trushina, T.N., Behavior of organic impurities during heat treatment of arsenic trichloride, Zh. Prikl. Khim., 1974, vol. 47, no. 8, pp. 1750–1760.
Fedorov, V.A., Potolokov, N.A., Nikolashin, S.V., Borisov, S.A., Menshchikova, T.K., Shai, E.P., Khokhlov, R.V., Shevchenko, A.V., and Kholstov, V.I., Physical chemistry and technology of the preparation of arsenic from lewisite detoxification products, Ross. Khim. Zh., 2010, vol. 54, no. 4, pp. 56–66.
Borisov, S.A., Menshchikova, T.K., Potolokov, N.A., Fedorov, V.A., and Brekhovskikh, M.N., Physicochemical principles underlying the preparation of high-purity substances for microelectronic and optical applications, Inorg. Mater., 2014, vol. 50, no. 11, pp. 1151–1156.
Fedorov, V.A., Potolokov, N.A., Menshchikova, T.K., and Brekhovskikh, M.N., Physicochemical and methodological approaches to the development of integrated processes for the preparation of high-purity substances, Inorg. Mater., 2019, vol. 55, no. 12, pp. 1264–1272.
Fedorov, V.A., Manufacture of high-purity arsenic from an unconventional source, Russ. J. Inorg. Chem., 2001, vol. 46, suppl. 2, pp. 87–105.
Petkov, L. and Stoytcheva, M., Anodic reactions with As(III) participation, Bull. Electrochem., 2005, vol. 21, no. 2, pp. 93–96.
Devyatykh, G.G. and Zorin, L.D., Letuchie neorganicheskie gidridy osoboi chistoty (Ultrapure Volatile Inorganic Hydrides), Moscow: Nauka, 1974.
Schon, H., Heinicke, J., and Tzschach, A., DDR Patent 225684, 1985.
Tesaric, P., Skuta, D., and Nozka, K., CZ Patent 190113, 1981.
Piffard, J.-F. and Rade, J.-Y., Fr. Patent 2609978, 1988.
Efremov, A.A., Morozov, V.I., and Ryabenko, E.A., Behavior of impurities in monogermane synthesis, Vysokochist. Veshchestva, 1993, no. 5, pp. 71–81.
Marinasce, J.C., US Patent 4945857, 1989.
Denkhhaus, E., Beck, F., Buescchier, P., Gerhard, R., and Gololoch, A., Electrolytie hydride generation atomic absorption spectrometry for the determination of antimony, arsenic, selenium and tin – mechanistic aspects and figures of merit, J. Anal. Chem., 2001, vol. 370, pp. 735–743.
Bolea, E., Laborda, F., Castillo, J.R., and Sturgeon, R.E., Electrochemical hydride generation for the simultaneous determination of hydride forming elements by inductively coupled plasmas-atomic emission, Spectrochim. Acta, Part A, 2004, vol. 59, pp. 505–513.
Tomilov, A.P. and Chomutov, N.E., in Encyclopedia of Electrochemistry of the Elements: II. Arcenic, Bard, A, Ed., New York: Marcel Dekker, 1974, pp. 21–51.
Tomilov, A.P., Osadchenko, I.M., and Khomutov, N.E., Electrochemistry of arsenic and its compounds, Itogi Nauki Tekh. Elektrokhim., 1970, vol. 14, pp. 168–206.
Tomilov, A.P., Smetanin, A.V., Chernykh, I.N., and Smirnov, M.K., Electrode reactions involving arsenic and its inorganic compounds, Russ. J. Electrochem., 2001, vol. 37, no. 10, pp. 997–1011.
Efimov, E.A. and Erusalimchik, I.G., Electrochemical processes on an arsenic electrode, Elektrokhimiya, 1965, vol. 1, no. 9, pp. 1133–1137.
Salzberg, H.W. and Goldschmidt, B., Arsine evolution and water reduction at an arsenic cathode, J. Electrochem. Soc., 1960, vol. 107, pp. 348–353.
Valdes, J.L., Cadet, G., and Mitchell, J.W., On demand electrochemical generation of arsine, J. Electrochem. Soc., 1991, vol. 138, pp. 1654–1658.
Grube, G. and Kleber, H., Die Überspannung des Wasserstoffes an Arsenkathoden und das electromotorische Verhalten des Arsenwasserstoffes, Z. Elektrochem., 1924, vol. 30, nos. 21–22, pp. 517–523.
Vorotyntsev, V.M., Kozin, L.F., Zhylkamanova, K., Glushachenko, O.A., Abdrakhmanov, R.R., and Balabanov, V.V., Electrochemical synthesis of high-purity arsine: II. Cathode hydrogenation of elemental arsenic, Vysokochist. Veshchestva, 1995, no. 5, pp. 59–66.
Chernykh, I.N. and Tomilov, A.P., Electrochemical reduction of elemental arsenic, Zh. Prikl. Khim. (S.-Peterburg), 1995, vol. 68, no. 7, pp. 1208–1209.
Ayers, W.M., US Patent 5158656, 1992.
Cadet, G., Mitchell, J.W., and Valdes, J.L., US Patent 5474659, 1995.
Ayers, W.M., US Patent 6080297, 2000.
Buckley, D.N., Seabury, C.W., Valdes, J.L., Cadet, G., Mitchell, J.W., et al., Growth of InGaAs structures using in situ electrochemically generated arsine, Appl. Phys. Lett., 1990, vol. 57, pp. 1684–1686.
Machado, R.M., Hollen, J.E., Ryals, G.L., Hartz, Ch.L., and Mohr, R.J., EEC Patent 1845172, 2007.
Akbasova, A.D., Electrochemical reduction of arsenous acid, Tr. Minist. Vyssh. Uchebn. Zaved. Kaz. SSR, 1986, pp. 97–99.
Vorotyntsev, V.M., Balabanov, V.V., Abdrakhmanov, R.R., and Malygina, L.S., Electrochemical synthesis of high-purity arsine: I. Current efficiency for arsine in the cathodic reduction of As2O3, Vysokochist. Veshchestva, 1993, no. 5, pp. 22–27.
Smirnov, M.K., Smetanin, A.V., Turygin, V.V., Khudenko, A.V., and Tomilov, A.P., Electroreduction of As(III) in acid environment, Russ. J. Electrochem., 2001, vol. 37, no. 10, pp. 1050–1053.
Nilov, A.P., Optimization of mass transport and Phase Formation Processes for the Electrochemical Reduction of As(III), Sb(III), and Ge(IV) Compounds, Extended Abstract of Doctoral (Chem.) Dissertation, Egor’evsk: ELATERA, 2001.
Gladyshev, V.P., Zebreva, A.I., Tulebaev, A.Zh., Kovaleva, S.V., Sarieva, L.S., et al., USSR Patent 962335, 1982.
Bouard, P., Labrune, Ph., and Cocolios, P., US Patent 5425857, 1995.
Smirnov, M.K., Turygin, V.V., Shalashova, N.N., Khudenko, A.V., and Tomilov, A.P., Electrochemical reduction of As(III) in acid media, Inorg. Mater., 2007, vol. 43, no. 1, pp. 25–29.
Porter, V.R., US Patent 4178224, 1979.
Smetanin, A.V., Smirnov, M.K., Chernykh, I.N., Turygin, V.V., Khudenko, A.V., Fedorov, V.A., and Tomilov, A.P., Electrochemical preparation of arsenic and its compounds, Inorg. Mater., 2003, vol. 39, no. 1, pp. 22–36.
Fedorov, V.A., Potolokov, N.A., Menshchikova, T.K., and Kholstov, V.I., Scientific principles of the preparation of high-purity substances and materials from secondary raw materials, Trudy XX Mendeleevskogo s"ezda po obshchei i prikladnoi khimii (Proc. XX Mendeleev Meet. on General and Applied Chemistry), Yekaterinburg, 2016, p. 214.
Tolstikov, V.P., Interrelationship between redox processes and reaction pH, Zh. Obshch. Khim., 1969, vol. 39, no. 2, pp. 240–244.
Meites, L., Polarographic characteristics of +3 and +5 arsenic in hydrochloric acid solutions, J. Am. Chem. Soc., 1954, vol. 76, pp. 5927–5931.
Dzhaparidze, Dzh.I., Dzhokhadze, G.M., and Siradze, R.V., Polarographic determination of \({{{\text{H}}}_{2}}{\text{AsO}}_{4}^{ - }\) anions, Soobshch. Akad. Nauk Gruz. SSR, 1975, vol. 79, pp. 105–107.
Zemtsova, A.G. and Kaplan, B.Ya., AC polarography in phosphate solutions, in Khimiya v Tadzhikistane (Chemistry in Tajikistan), Dushanbe: Donish, 1974, pp. 208–216.
Arnold, J.P. and Jonson, R.M., Polarography of arsenic, Talanta, 1969, vol. 16, pp. 1191–1207.
Zhdanov, S.I., Kryukova, T.A., and Vasil’eva, E.G., On the mechanism of arsenic reduction on a dropping mercury cathode, Elektrokhimiya, 1975, vol. 11, no. 5, pp. 767–770.
Akbasova, A.D. and Dzekunov, V.P., Fizikokhimicheskie metody razdeleniya, polucheniya i analiza metallov (Physicochemical Methods for the Separation, Preparation, and Analysis of Metals), Alma-Ata: Kaz. NIINTI, 1988, p. 28.
Akbasova, A.D., Dzekunov, V.P., and Pavlova, O.D., Electrochemical reduction of arsenic(V) on solid electrodes in the presence of a surfactants, Doklady 6-oi vsesoyuznoi konferentsii po elektrokhimii (Proc. 6th All-Union Conf. on electrochemistry), Moscow: VINITI, 1982, vol. 1, p. 172.
White, M.C. and Bard, A.J., Polarography of metal pyrogallol complexes, Anal. Chem., 1966, vol. 38, pp. 61–63.
Popova, T.G. and Khamudkhanova, Sh.Z., Electrodeposition of arsenic, Doklady 6-oi vsesoyuznoi konferentsii po elektrokhimii (Proc. 6th All-Union Conf. on Electrochemistry), Moscow: VINITI, 1982, vol. 1, p. 288.
Menshchikova, T.K., Vargunin, A.I., Brekhovskikh, M.N., Fedorov, V.A., and Myslitskii, O.E., Physicochemical properties of arsenic-containing lewisite detoxification products, Inorg. Mater., 2020, vol. 56, no. 11, pp. 1174–1178.
Ramberg, L., Arseniksyras och arseniksyrlighets elektrolytiska reduktion till arsenikvate vid katoder av olika metaller, Lunds Univ. Arsskr., Avd. 2, 1978, vol. 14, no. 21.
Gopalakrishnan, V., Gnanasekaran, K.S., Narasimham, K.C., and Udupa, N.V.K., Investigation of the reduction of arsenates, Trans. SAEST, 1976, vol. 11, pp. 251–258.
Chernykh, I.N., Tomilov, A.P., Smetanin, A.V., and Khudenko, A.V., Electrochemical reduction of arsenic acid, Russ. J. Electrochem., 2001, vol. 37, no. 9, pp. 9542–946.
Baranov, Yu.I., Smetanin, A.V., Turygin, V.V., Tomilov, A.P., Khudenko, .V., and Chernykh, I.N., RF Patent 2203983, 2003.
Turygin, V.V., Smirnov, M.K., Smetanin, A.V., Zhukov, E.G., Fedorov, V.A., and Tomilov, A.P., Electrochemical arsenic extraction from nonferrous metals industry waste, Inorg. Mater., 2008, vol. 44, no. 9, pp. 946–953.
Turygin, V.V., Smirnov, M.K., Berezkin, M.Yu., Khudenko, A.V., Tomilov, A.P., Kotkov, A.P., Grishnova, N.D., Polezhaev, D.M., Petukhov, G.G., and Gromov, A.V., Electrochemical preparation and impurity composition of arsine, Materialy XVI koordinatsionnogo nauchno-tekhnicheskogo seminara po SVCh tekhnike (Proc. XVI Science and Technology Coordination Seminar on Microwave Engineering), Nizhny Novgorod, 2009, pp. 187–189.
Turygin, V.V., Physicochemical principles underlying the technology of electrochemical synthesis of arsenic, phosphorus, and germanium hydrides, Doctoral (Chem.) Dissertation, Moscow, 2012.
Uryadov, A.A., Kotkov, A.P., Grishnova, N.D., Polezhaev, D.M., Petukhov, G.G., Turygin, V.V., Berezkin, M.Yu., and Khudenko, A.V., Impurity composition of electrochemically synthesized arsine, Doklady XIV konferentsii “Vysokochistye veshchestva i materialy. Poluchenie, analiz, primenenie” (Proc. XIV Conf. High-Purity Substances and Materials: Preparation, Analysis, and Application), Nizhny Novgorod, 2011, p. 103.
Veviorskii, A.P., Ivanov, V.N., and Frolov, A.V., USSR Patent 1004259, 1983.
Efremov, A.A., Morozov, V.I., and Fedorov, V.A., An absorption–adsorption process for ultrapurification of volatile inorganic hydrides, Vysokochist. Veshchestva, 1991, no. 1, pp. 115–123.
Kvaratskheli, Yu.K., Shatalov, V.V., Demin, Yu.V., Kondrat’ev, A.G., and Khorozova, O.D., RF Patent 2369666, 2010.
Cao Huazhen, Ahan Haipeng, Ruan Huimin, and Zheng Guoqu, A study on the evolution of arsenic electrodeposition. The influence of ammonium citrate, Electrochem. Commun., 2012, vol. 23, pp. 44–47.
Uany Dunfen, CN Patent 103896212, 2010.
Uany Dunfen, CN Patent 103896211, 2010.
Iny Enhua, U Fen, and Gun Czynminy, CN Patent 101857270, 2005.
Turygin, V.V. and Tomilov, A.P., Using electrochemistry for low-waste production development, Materialy vserossiiskoi konferentsii “Elektrokhimiya i ekologiya” (Proc. All-Russia Conf. on Electrochemistry and Ecology), Novocherkassk: YuRGU, 2008, p. 81.
Piffard, J.-F. and Rade, J.-Y., Fr. Patent 2609978, 1988.
Bouard, P., Labrune, Ph., and Cocolios, P., US Patent 5425857, 1995.
Ferguson, M.A., Hoffman, M.R., and Hering, J.G., TiO2-photocatalyzed As(III) oxidation in aqueous suspensions: reaction kinetics and effects of desorption, Environ. Sci. Technol., 2005, vol. 39, no. 6, pp. 1880–1886.
Devyatykh, G.G., Balabanov, V.V., and Zvereva, V.I., RF Patent 614615, 2015.
Veviorskii, A.P., Ivanov, V.N., and Frolov, A.V., USSR Patent 1004259, 1983.
CN Patent 103816787 A, 2015.
Alvarez, D., US Patent 6241955B1, 2010.
Dingjan Wu, Golden, T.C., Chun Christine Dong, and Batavio, P.J., US Patent Application 20050120877A1, 2005.
Watanabe, T. and Fraenkel, D., US Patent Application 20030097929 A1, 2003.
Alvarez, D., US Patent Application 20060188420 A1, 2006.
Slavutskii, V.P., Gur’ev, V.S., Kuklich, V.M., Ganka, G.F., and Ol’khovskaya, L.N., USSR Patent 1161159, 1988.
Kataoka Masakhiro, Niki Itikhvo, and Khatayuo Mamoru, Jpn. Patent 60-215601, 2010.
Devyatykh, G.G, Agliulov, N.Kh., Churbanov, M.F., Kalmykov, V.I., Balabanov, V.I., and Liverko, V.N., USSR Patent 1032708, 1992.
Katakhara Koiti, Simada Gakasi, Akita Noboru, and Nikhon Panonikusu, Jpn. Patent 63-238920, 2010.
Krylov, V.A., Chernova, O.Yu., Sozin, A.Yu., and Kotkov, A.P., Gas chromatography/mass spectrometry analysis of high-purity arsine, Zavod. Lab. Diagnostika Mater., 2010, vol. 76, no. 3, pp. 13–16.
Turygin, V.V., Smirnov, M.K., Berezkin, M.Yu., Sokhadze, L.A., Stepnova, N.P., Tomilov, A.P., Fedorov, V.A., and Potolokov, N.A., Physicochemical principles behind the preparation of high-purity arsenic compounds from lewisite detoxification products, Inorg. Mater., 2017, vol. 53, no. 4, pp. 397–399.
Baranov, Yu.I., Smetanin, A.V., Turygin, V.V., Tomilov, A.P., Khudenko, A.V, and Chernykh, I.N., RF Patent 2203983, 2001.
Smirnov, Yu.D., Vakher, V.F., Knyazev, B.A., and Tomilov, A.P., Electrochemical extraction of arsenic from arsenous acid esters, Inorg. Mater., 2005, vol. 41, no. 8, pp. 819–821.
Omiadze, A.P., Grinberg, E.E., and Efremov, A.A., Liquid–vapor phase equilibria in systems based on arsenic, silicon, boron, and antimony alkoxides and alcohols, Zh. Fiz. Khim., 1986, vol. 60, no. 6, pp. 1575–1582.
Funding
This work was supported by the Russian Federation Ministry of Science and Higher Education as part of the state research target for the Kurnakov Institute of General and Inorganic Chemistry, Russian Academy of Sciences.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fedorov, V.A., Menshchikova, T.K., Vargunin, A.I. et al. Processes for the Preparation of High-Purity Arsenic and Its Compounds. Inorg Mater 57, 1097–1108 (2021). https://doi.org/10.1134/S0020168521110042
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0020168521110042