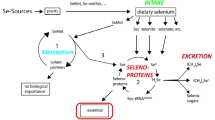
Abstract—The trace element selenium, which was discovered by Berzelius in 1817, remains poorly investigated and exhibits diverse and surprising functions. We consider it unique for several reasons. First, it is present not only in organic or inorganic compounds but also in the amino acids selenocysteine and selenomethionine, as a key component, and in selenoproteins, which are found in all Domains of life. Second, selenocysteine is the 21st proteinogenic amino acid. Its uniqueness lies not only in the fact that it is encoded by one of the three stop codons of translation but in that its biosynthesis possesses unique features and that this process involves unique cis- and trans-active factors that are necessary for the recognition of this triplet as selenocysteine-encoding to avoid premature translation termination and synthesize full-sized selenoproteins. The maintenance of these cis- and trans-active factors is energy consuming, which suggests the crucial importance of selenoproteins for an organism. In addition, the pathways of amino-acid and selenoprotein biosynthesis have some distinctive features in different Domains of life. Third, the processes and effects of selenium compounds of different origins, in which this micronutrient plays key roles, are strikingly diverse, especially in the regulation of vital functions in mammals. This review uses the most recent data to present a comprehensive view of the properties and functions of selenium and to provide insight into the uniqueness of this micronutrient.


Similar content being viewed by others
REFERENCES
S. Menon, K. S. Shrudhi Devi, R. Santhiya, et al., Colloids Surf. B. Biointerfaces 170, 280 (2018).
E. G. Varlamova, M. V. Goltyaev, and E. E. Fesenko, Dokl. Biochem. Biophys. 468, 203 (2016).
E. G. Varlamova and I. V. Cheremushkina, J. Trace Elem-. Med. Biol. 39, 76 (2017).
E. G. Varlamova, M. V. Goltyaev, V. I. Novoselov, and E. E. Fesenko, Dokl. Biochem. Biophys. 476, 320 (2017).
E. G. Varlamova, J. Trace Elem. Med. Biol. 48, 172 (2018).
E. G. Varlamova, M. V. Goltyaev, J. P. Kuznetsova, Mol. Biol. (Moscow) 52, 446 (2018).
Y. P. Kuznetsova, M. V. Goltyaev, O. S. Gorbacheva, et al., Dokl. Biochem. Biophys. 480, 131 (2018).
U. Peters and Y. Takata, Mol. Nutr. Food Res. 52, 1261(2008)
J. P. Richie, Jr., A. Das, A. M. Calcagnotto, et al., Exp. Gerontol. 47, 223 (2012).
C. van Dronkelaar, A. van Velzen, M. Abdelrazek, et al., J. Am. Med. Dir. Assoc. 19, 6 (2018).
L. A. Wessjohann, A. Schneider, M. Abbas, and W. Brandt, J. Biol. Chem. 388, 997 (2007).
E. S. Arner, J. Exp. Cell Res. 316, 1296 (2010).
C. Jacob, G. I. Giles, N. M. Giles, et al., Angew. Chem. Int. Ed. Engl. 42, 4742 (2003).
L. Johansson, G. Gafvelin, and E. S. Arner, Biochim. Biophys. Acta 1726, 1 (2005).
M. Wada, S.-I. Nobuki, and Y. Tenkyuu, J. Organomet. Chem. 580, 282 (1999).
J. C. Pleasants, W. Guo, and D. L. Rabenstein, J. Am. Chem. Soc. 111, 6553 (1989).
X. M. Xu, B. A. Carlson, H. Mix, et al., PLoS Biol. 5, 122 (2007).
M. J. Berry, R. M. Tujebajeva, P. R. Copeland, et al., J. Biofactors 14, 17 (2001).
D. J. Klein, T. M. Schmeing, P. B. Moore, and T. A. Steitz, EMBO J. 20, 4214 (2001).
P. R. Copeland, J. E. Fletcher, B. A. Carlson, et al., EMBO J. 19, 306 (2000).
A. M. Diamond, B. Dudock, and D. L. Hatfield, Cell 25, 497 (1981).
D. A. G. Noble and H. Song, Cell Mol. Life Sci. 65, 1335 (2008).
G. R. Andersen, L. Pedersen, L. Valente, et al., Mol. Cell 6, 1261 (2000).
G. W. Martin, III, J. W. Harney, and M. J. Berry, RNA 2, 171 (1996).
G. W. Martin, III, J. W. Harne, and M. J. Berry, RNA 4, 65 (1998).
F. Werner, Chem. Rev. 113, 8331 (2013).
C. Sturchler, A. Lescure, G. Keith, et al., Nucleic Acids Res. 22, 1354 (1994).
C. Baron, E. Westhof, A. Bock, and R. Giege, J. Mol. Biol. 231, 274 (1993).
K. M. Holman, A. K. Puppala, J. W. Lee, et al., RNA 23, 1685 (2017).
Y. Itoh, M. J. Broecker, S.-I. Sekine, et al., Science 340, 75 (2013).
L. R. Manzine, V. H. B. Serrao, L. Lima, et al., FEBS Lett. 587, 906 (2013).
N. Noinaj, R. Wattanasak, D.-Y. Lee, et al., J. Bacte-riol. 194, 499 (2012).
T. Stock, M. Selzer, S. Connery, et al., Mol. Microbiol. 82, 734 (2011).
P. R. Copeland, V. A. Stepanik, and D. M. Driscoll, J. Mol. Cell Biol. 21, 1491 (2001).
J. Donovan and P. R. Copeland, Antioxid. Redox Signal. 12, 881 (2010).
L. Chavatte, B. A. Brown, and D. M. Driscoll, Nat. Struct. Mol. Biol. 12, 408 (2005).
P. R. Copeland, J. Genome Biol. 6, 221 (2005).
G. V. Kryukov, S. Castellano, S. V. Novoselov, et al., Science 300, 1439 (2003).
G. V. Kryukov and V. N. Gladyshev, EMBO Rep. 5, 538 (2004).
Y. Zhang, D. E. Fomenko, and V. N. Gladyshev, Genome Biol. 6, 37 (2005).
Q. A. Sun, Y. Wu, F. Zappacosta, et al., J. Biol. Chem. 274, 24522 (1999).
R. F. Burk and K. E. Hill, Bioessays 21, 231 (1999).
L. Flohe, W. A. Gunzler, and H. H. Schock, FEBS Lett. 32, 132 (1973).
J. T. Rotruck, A. L. Pope, H. E. Ganther, et al., Science 179, 588 (1973).
S. Gromer, J. Wissing, D. Behne, et al., J. Biochem. 332, 591 (1998).
M. P. Rayman, H. G. Infante, and M. Sargent, Br. J. Nutr. 100, 238 (2008).
P. D. Whanger, J. Am. Coll. Nutr. 21, 223 (2002).
Y. Dong, D. Lisk, E. Block, and C. Ip, Cancer Res. 61, 2923 (2001).
Y. Kobayashi, Y. Ogra, and K. Ishiwata, Proc. Natl. Acad. Sci. U. S. A. 99, 15932 (2002).
Y. Anan, M. Kimura, M. Hayashi, et al., Chem. Res. Toxicol. 28, 1803 (2015).
E. Vetchinkina, E. Loshchinina, V. Kursky, and V. Nikitina, J. Microbiol. 51, 829 (2013).
J. F. Stolz, P. Basu, J. M. Santini, and R. S. Oremland, Annu. Rev. Microbiol. 160, 107 (2006).
J. F. Stolz and R. S. Oremland, FEMS Microbiol. Rev. 23, 615 (1999).
J. F. Stolz, P. Basi, and R. S. Oremland, Int. Microbiol. 5, 201 (2002).
B. Soboh, C. Pinske, and M. Kuhns, BMC Microbiol. 11, 173 (2011).
M. E. Losi and W. T. Frankenberger, Appl. Environ. Microbiol. 63, 3079 (1997).
H. Ridley, C. A. Watts, D. J. Richardson and C. S. Butler, Appl. Environ. Microbiol. 72, 5173 (2006).
J. Kessi, M. Ramuz, E. Wehrli, et al., Appl. Environ. Microbiol. 65, 4734 (1999).
F. A. Tomei, L. L. Barton, C. L. Lemanski, et al., J. Ind. Microbiol. 14, 329 (1995).
Y. V. Nancharaiah and P. N. L. Lens, Microbiol. Mol. Biol. Rev. 79, 61 (2015).
W. E. Hart, S. P. Marczak, A. R. Kneller, et al., J. Inorg. Biochem. 125, 1 (2013).
S. Ansar, M. Abudawood, S. S. Hamed, and M. M. Aleem, Biol. Trace Elem. Res. 175, 161 (2017).
Z. Shigemi, K. Manabe, and N. Hara, Chem. Biol. Interact. 266, 28 (2017).
M. Kieliszek, B. Lipinski, and S. Błazejak, Cells 24, 39 (2017).
E. D. Paskett, J. A. Dean, J. M. Oliveri, and J. P. Harrop, J. Clin. Oncol. 30, 3726 (2012).
N. Xiang, R. Zhao, and W. Zhong, Cancer Chemother. Pharmacol. 63, 351 (2009).
N. M. Corcoran, M. Najdovska, and A. J. Costello, J. Urol. 171, 907 (2004).
S. R. Shultz, D. K. Wright, P. Zheng, et al., Brain 138, 1297 (2015).
T. R. Schwartz, E. B. Kmiec, BMC Mol. Biol. 8, 7 (2007).
S. Cao, F. A. Durrani, K. Toth, and Y. M. Rustum, Br. J. Cancer 110, 1733 (2014).
S. Chintala, K. Toth, and S. Cao, Cancer Chemother. Pharmacol. 66, 899 (2010).
M. Puppo, F. Battaglia, and C. Ottaviano, Mol. Cancer Ther. 7, 1974 (2008).
M. E. Crosby, R. Kulshreshtha, M. Ivan, and P. M. Glazer, Cancer Res. 69, 1221 (2009).
A. Bhattacharya, K. Toth, and A. Sen, Clin. Colorectal Cancer 8, 155 (2009).
A. Kim, J. Lee, and M. S. Park, Arch. Pharmacol. Res. 38, 659 (2015).
V. G. Deepagan, S. Kwon, and D. G. You, Biomaterials 103, 56 (2016).
D. Bartolini, J. Commodi, M. Piroddi, Free Radic. Biol. Med. 88, 466 (2015).
A. P. Fernandes and V. Gandin, Biochim. Biophys. Acta 1850, 1642 (2015).
A. Liu, H. Liu, and Y. Li, Mol. Carcinog. 51, 303 (2012).
J. E. Spallholz, B. J. Shriver, and T. W. Reid, Nutr. Cancer 40, 34 (2001).
M. Tarrado-Castellarnau, R. Cortes, and M. Zanuy, Pharmacol. Res. 102, 218 (2015).
H. Zeng and M. Wu, Nutr. Cancer 67, 831 (2015).
B. Husbeck, R.S. Bhattacharyya, D. Feldman, and S. J. Knox, Mol. Cancer Ther. 5, 2078 (2006).
G. C. Mills, J. Biol. Chem. 229, 189 (1957).
G. Ravn-Haren, A. Olsen, and A. Tjonneland, Carcinogenesis 27, 820 (2006).
M. Cao, X. Mu, C. Jiang, et al., Tumour Biol. 35, 759 (2014).
J. A. Moscow, L. Schmidt, D. T. Ingram, et al., Carcinogenesis 15, 2769 (1994).
Z. Kote-Jarai, F. Durocher, S. M. Edwards, et al., Prostate Cancer Prostatic Dis. 5, 189 (2002).
I. Dokic, C. Hartmann, C. Herold-Mende, and A. Regnier-Vigouroux, Glia 60, 1785 (2012).
F. F. Chu, J. H. Doroshow, and R. S. Esworthy, J. Biol. Chem. 268, 2571 (1993).
F. F. Chu, R. S. Esworthy, and P. G. Chu, Cancer Res. 64, 962 (2004).
R. S. Esworthy, R. Aranda, M. G. Martin, et al., Am. J. Physiol. Gastrointest. Liver Physiol. 281, 848 (2001).
J. Walshe, M. M. Serewko-Auret, and N. Teakle, Cancer Res. 67, 4751 (2007).
S. B. Bull, H. Ozcelik, and D. Pinnaduwage, J. Clin. Oncol. 22, 86 (2004).
C. Schmutzler, B. Mentrup, L. Schomburg, et al., Biol. Chem. 388, 1053 (2007).
F. G. Ottaviano, S. S. Tang, D. E. Handy, and J. Loscalzo, Mol. Cell. Biochem. 327, 111 (2009).
X. Zhang, J. J. Yang, Y. S. Kim, et al., Int. J. Oncol. 36, 405 (2010).
Z. L. Yang, L. Yang, and Q. Zou, Dis. Markers 35, 163 (2013).
Y. Saga, M. Ohwada, and M. Suzuki, Oncol. Rep. 20, 1299 (2008).
F. Ursini, S. Heim, M. Kiess, et al., Science 277, 1393 (1999).
T. Pushpa Rekha, L. M. Burdsal, G. M. Chilsom, and D. M. Driscoll, J. Biol. Chem. 270, 26993 (1985).
M. V. Goltyaev, E. G. Varlamova, V. I. Novoselov, and E. E. Fesenko, Dokl. Biochem. Biophys. 457, 132 (2014).
E. G. Varlamova, Fundament. Issled. 9, 326 (2011).
A. Holmgren and M. Bjornstedt, Methods Enzymol. 252, 16644 (1995).
L. Zhong and A. Holmgren, J. Biol. Chem. 275, 1812 (2000).
C. Meplan, S. Rohrmann, A. Steinbrecher, et al., PloS One 7, e48709 (2012).
D. Su, S. V. Novoselov, Q. A. Sun, et al., J. Biol. Chem. 280, 26491 (2005).
A. S. H. Wu, J. E. Oldfield, L. R. Shull, and P. R. Cheeke, Biol. Reprod. 20, 793 (1979).
D. Behne, H. Weiler, A. Kyriakopoulos, J. Reprod. Fertil. 106, 291 (1996).
I. F. M. Marai, A. A. El-Darawany, E. A. Ismail, and M. A. M. Abdel-Hafez, Arch. Tierzucht 52, 402 (2009).
L. G. Shi, R. J. Yang, and W. B. Yue, Anim. Reprod. Sci. 118, 248 (2010).
M. Koga, H. Tanaka, K. Yomogida, et al., Biol. Reprod. 58, 261 (1998).
G. E. Olson, V. P. Winfrey, S. K. Nagdas, et al., Biol. Reprod. 73, 201 (2005).
E. G. Varlamova and V. I. Novoselov, Mol. Biol. (Moscow) 46, 819 (2012).
E. G. Varlamova and V. I. Novoselov, Mol. Biol. (Moscow) 46, 250 (2012).
E. G. Varlamova, S. V. Novoselov, and V. I. Novoselov, Mol. Biol. (Moscow) 49, 700 (2015).
S. Y. Cheng, J. L. Leonard, and P. J. Davis, Endocr. Rev. 31, 139 (2010).
P. H. Davies, M. C. Sheppard, and J. A. Franklyn, Mol. Cell Endocrinol. 129, 191 (1997).
E. L. Meyer, I. M. Goemann, J. M. Dora, et al., Mol. Cell Endocrinol. 289, 16 (2008).
R. Kukreja and A. Khan, Ind. J. Exp. Biol. 36, 203 (1998).
G. R. Hogan, J. Toxicol. Environ. Health 53, 113 (1998).
M. Roy, L. Kiremidjian-Schumacher, and H. I. Wis-he, Biol. Trace Elem. Res. 41, 103 (1994).
F. W. Hoffmann, A. C. Hashimoto, L. A. Shafer, et al., J. Nutr. 140, 1155 (2010).
G. Bermano, J. R. Arthur, and J. E. Hesketh, Biochem. J. 320, 891 (1996).
G. Bermano, J. R. Arthur, and J. E. Hesketh, FEBS Lett. 387, 157 (1996).
G. Bermano, F. Nicol, J. A. Dyer, et al., Biochem. J. 311, 425 (1995).
X. G. Lei, J. K. Evenson, K. M. Thompson, and R. A. Sunde, J. Nutr. 125, 1438 (1995).
Z. Touat-Hamici, Y. Legrain, A. L. Bulteau, and L. Chavatte, J. Biol. Chem. 289, 14750 (2014).
L. Latreche, S. Duhieu, Z. Touat-Hamici, et al., RNA Biol. 9, 681 (2012).
Y. Legrain, Z. Touat-Hamici, and L. Chavatte, J. Biol. Chem. 289, 6299 (2014).
M. A. Beck, P. C. Kolbeck, L. H. Rohr, et al., J. Med. Virol. 43, 166 (1994).
D. D. Hensrud, D. C. Heimburger, J. Chen, and B. Parpia, Eur. J. Clin. Nutr. 48, 455 (1994).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflict of interest. This article does not contain any studies involving animals or human participants performed by any of the authors.
Additional information
Translated by V. Gulevich
Abbreviations: Sec, selenocysteine; Cys, cysteine; SeMet, selenomethionine; SECIS element (SecInsertionSequence), cis-acting translation factor for selenoproteins; SBP2 (SECIS BindingProtein), trans-acting translation factor for selenoproteins; ROS, reactive oxygen species; TXNRD, thioredoxin reductase.
Rights and permissions
About this article
Cite this article
Varlamova, E.G., Maltseva, V.N. Micronutrient Selenium: Uniqueness and Vital Functions. BIOPHYSICS 64, 510–521 (2019). https://doi.org/10.1134/S0006350919040213
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0006350919040213