Abstract
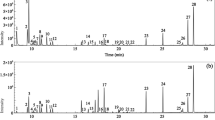
A comparative study was performed by solid phase microextraction and capillary gas chromatography to establish the ability of four polymer sorbents of different compositions to extract and concentrate volatile organic compounds from the gas phase above an aqueous solution. All polymer sorbents sorbed nonpolar monoterpene hydrocarbons via a cooperative mechanism with almost equal and high efficiency. Sorbents based on polymethyl disiloxane and its mixture with divinylbenzene were more effective in extracting acetates and sesquiterpenes. As the concentration of these compounds in the gas phase increased, their binding by sorbents decreased. It was found that the determination of polar compounds depended on the presence of a solvent in the system. Compounds that are highly soluble in water (alcohols, ketones, etc.) had low coefficients of distribution between gas and water phases. Consequently, their sorption to any of the polymer sorbents was negligible. In the absence of the solvent, the degree of their extraction from the gas phase above the sample was high. It was shown that the actual composition of compounds in the initial mixture of essential oils could significantly differ from their composition in the gas phase. This method is convenient and informative for the purpose of studying the composition of volatile compounds in the gas phase that determine the flavor of the product.
Similar content being viewed by others
References
Wardencky, W., Michulec, M., and Curylo, J., Int. J. Food Sci. Technol., 2004, vol. 39, pp. 703–717.
Roberts, D.D., Polien, P., and Milo, C., J. Agric. Food Chem., 2000, vol. 48, no. 6, pp. 2430–2437.
Misharina, T.A., Prikl. Anal. Khim., 2010, vol. 1, no. 2, pp. 12–19.
Ouyang, G. and Vuckovic, D., Pawliszyn, J. Chem. Rev., 2011, vol. 111, no. 4, pp. 2784–2815.
Arthur, C.L. and Pawliszyn, J., Anal. Chem., 1990, vol. 62, no. 7, pp. 1843–1852.
Ettre, L.S., in Headspace Analysis of Food and Flavours: Theory and Practice, Rouseff, R.L. and Cadwallader, K.R., Eds., New York: Academic Plenum Publ., 2001.
Nongonierma, A., Cayot, P., Le Quere, J.L., Springett, M., and Voilley, A.J., Food Rew. Int., 2006, vol. 22, no. 1, pp. 51–94.
Misharina, T.A., Terenina, M.B., and Krikunova, N.I., Appl. Biochem. Microbiol., 2017, vol. 53, pp. 5.
Zaitsev, V.N. and Zui, M.F., Zh. Anal. Khim., 2014, vol. 69, no. 8, pp. 787–801.
Savel'eva, E.I., Gavrilova, O.P., and Gagkaeva, T.Yu., Zh. Anal. Khim., 2014, vol. 69, no. 7, pp. 675–682.
Bajer, T., Ligor, M., Ligor, T., and Buszewski, D., J. Separation Sci., 2016, vol. 39, no. 4, pp. 769–774.
Souza, H.A.L. and Bragagnolo, N., J. Agric. Food Chem., 2014, vol. 62, no. 3, pp. 590–599.
Du, L., Wang Ch., Li, J., Xiao, D., Li, Ch., and Xu, Y., J. Agric. Food Chem., 2013, vol. 61, no. 2, pp. 561–568.
Golovnya, R.V. and Misharina, T.A., Usp. Khim., 1980, vol. 49, pp. 171–191.
Terenina, M.B. and Misharina, T.A., Appl. Biochem. Microbiol., 2005, vol. 41, no. 4, pp. 407–412.
Misharina, T.A., Terenina, M.B., Krikunova, N.I., and Kalinchenko, M.A., Appl. Biochem. Microbiol., 2006, vol. 42, no. 1, pp. 111–116.
Misharina, T.A., Samusenko, A.L., and Kalinchenko, M.A., in From Polysaccharides to Granules, Simple and Mixture Gels, Yuryev, V.P. and Tomasik, P., Eds., New York: Nova Sci. Publ., 2004, pp. 245–257.
Moradi, M., Kaykhali, M., Ghiasvand, A.R., Shadabi, S., and Salehinia, A., Phytochem. Anal., 2012, vol. 23, no. 2, pp. 379–386.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © T.A. Misharina, M.B. Terenina, N.I. Krikunova, I.A. Mihailov, 2018, published in Prikladnaya Biokhimiya i Mikrobiologiya, 2018, Vol. 54, No. 1, pp. 89–98.
Rights and permissions
About this article
Cite this article
Misharina, T.A., Terenina, M.B., Krikunova, N.I. et al. Effect of the Compositions of Sample and Polymer Sorbents on the Extraction of Volatile Compounds by Solid-Phase Microextraction. Appl Biochem Microbiol 54, 89–97 (2018). https://doi.org/10.1134/S0003683818010052
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0003683818010052