Abstract
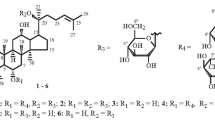
The aim of the study was to expand the substrate scope of Gliocladium deliquescens NRRL 1086 and generate coumarin glycosides using 4-methylumbelliferone (4-MU) as a substrate. The obtained results indicated that 4-MU can be metabolized to its glucoside (M1) and sulfate conjugate (M2), and the structures of the metabolites were elucidated by spectroscopic or enzymatic methods. Time course experiments detected that nearly 90% of 4-MU could be metabolized within 24 h, and the maximum yield of M1 could reach as high as 32%. Further tests revealed that the glucose concentration in the medium had little effect on the yield of M1 but time of 4-MU-adding could markedly affect the glycosylation procedure, and the favorable time to accumulate M1 was in the 12 or 24 h-old stage II culture, while in the 36 h-old or even older stage II culture, the substrate was almost metabolized to M2. The attempts to alter the ratio between M1 and M2 were performed by addition of quercetin and S-tetrahydroberberrubine or reduction of the sulfate concentration in the culture medium. Herein we describe, to the best of our knowledge, the first example of simultaneously microbial glycosylation and sulfation of coumarins.
Similar content being viewed by others
References
Hoult, J.R.S. and Paya, M., Gen. Pharmacol, 1996, vol. 27, no. 4, pp. 713–722.
Borges, F., Roleira, F., Milhazes, N., Santana, L., and Uriarte, E., Curr. Med. Chem., 2005, vol. 12, no. 8, pp. 887–916.
Karbownik, M.S. and Nowak, J.Z., Pharmacol. Rep., 2013, vol. 65, no. 5, pp. 1056–1074.
Benitez, A., Yates, T.J., Shamaldevi, N., Bowen, T., and Lokeshwar, V.B., J. Urology, 2013, vol. 190, no. 1, pp. 285–290.
Kakizaki, I., Kojima, K., Takagaki, K., Endo, M., Kannagi, R., Ito, M., et al., J. Biol. Chem., 2004, vol. 279, no. 32, pp. 33281–33289.
Urakawa, H., Nishida, Y., Wasa, J., Arai, E., Zhuo, L., Kimata, K., et al., Int. J. Cancer, 2012, vol. 130, no. 2, pp. 454–466.
Thuan, N.H. and Sohng, J.K., J. Ind. Microbiol. Biot., 2013, vol. 40, no. 12, pp. 1329–1356.
Kren, V. and Martínková, L., Curr. Med. Chem., 2001, vol. 8, no. 11, pp. 1303–1328.
Lacy, A. and O’Kennedy, R., Curr. Pharm. Des., 2004, vol. 10, no. 30, pp. 3797–3811.
Fylaktakidou, K.C., Hadjipavlou–Litina, D.J., Litinas, K.E., and Nicolaides, D.N., Curr. Pharm. Des., 2004, vol. 10, no. 30, pp. 3813–3833.
Zahri, S., Razavi, S.M., and Moatamed, Z., Nat. Prod. Res., 2012, vol. 26, no. 6, pp. 540–547.
Touisni, N., Maresca, A., McDonald, P.C., Lou, Y., Scozzafava, A., Dedhar, S., et al., J. Med. Chem., 2011, vol. 54, no. 24, pp. 8271–8277.
Woodley, J.M., Trends Biotechnol., 2008, vol. 26, no. 6, pp. 321–327.
Pollard, D.J. and Woodley, J.M., Trends Biotechnol., 2007, vol. 25, no. 2, pp. 66–73.
Li, W., Koike, K., Asada, Y., Yoshikawa, T., and Nikaido, T., Tetrahedron Lett., 2002, vol. 43, no. 32, pp. 5633–5635.
Xue, B.L., Zhou, L.B., Liu, J.W., and Yu, R.M., Pharmazie, 2012, vol. 67, no. 5, pp. 467–471.
Zhou, L.B., Tian, T., Xue, B.L., Song, L.Y., Liu, L., and Yu, R.M., Biosci. Biotechnol., Biochem., 2012, vol. 76, no. 5, pp. 1008–1010.
Chen, N.-D., Zhang, J., Liu, J.-H., and Yu, B.-Y., Appl. Microbiol. Biotechnol., 2010, vol. 86, no. 2, pp. 491–497.
Ge, H.-X., Zhang, J., Kai, C., Liu, J.-H., and Yu, B.-Y., Appl. Microbiol. Biotechnol., 2012, vol. 93, no. 6, pp. 2357–2364.
Du, C.H., Zhang, J., Xu, S.H., Wang, X.D., Kou, J.P., and Yu, B.Y., J. Mol. Catal. B: Enzym., 2014, vol. 99, pp. 85–88.
Ge, H.-X., Zhang, J., Dong, Y., Cui, K., and Yu, B.-Y., Chem. Commun., 2012, vol. 48, no. 49, pp. 6127–6129.
Betts, R.E., Walters, D.E., and Rosazza, J.P., J. Med. Chem., 1974, vol. 17, no. 6, pp. 599–602.
Golbeck, J.H. and Cox, J.C., Biotechnol. Bioeng., 1984, vol. 26, no. 5, pp. 434–441.
Lukey, M.J., Parkin, A., Roessler, M.M., Murphy, B.J., Harmer, J., Palmer, T., et al., J. Biol. Chem., 2010, vol. 285, no. 6, pp. 3928–3938.
Williams, S.J., Senaratne, R.H., Mougous, J.D., Riley, L.W., and Bertozzi, C.R., J. Biol. Chem., 2002, vol. 277, no. 36, pp. 32606–32615.
Wang, L.–Q. and James, M.O., Curr. Drug. Metab., 2006, vol. 7, no. 1, pp. 83–104.
Tranchimand, S., Tron, T., Gaudin, C., and Iacazio, G., FEMS Microbiol. Lett., 2005, vol. 253, no. 2, pp. 289–294.
Medina, M.L., Kiernan, U.A., and Francisco, W.A., Fungal. Genet. Biol., 2004, vol. 41, no. 3, pp. 327–335.
Fia, G., Giovani, G., and Rosi, I., J. Appl. Microbiol., 2005, vol. 99, no. 3, pp. 509–517.
Kwapiszewski, R., Skolimowski, M., Ziolkowska, K., Jedrych, E., Chudy, M., Dybko, A., and Brzozka, Z., Biomed. Microdevices, 2011, vol. 13, no. 3, pp. 431–440.
Franco, P.G., Pérez, M.J., Aranda, C., Adamo, A., and Silvestroff, L., Clin. Chim. Acta., 2015, vol. 446, pp. 86–92.
Smith, R.V. and Rosazza, J.P., J. Pharm. Sci., 1975, vol. 64, no. 11, pp. 1737–1759.
Griffiths, D.A., Best, D.J., and Jezequel, S.G., Appl. Microbiol. Biotechnol., 1991, vol. 35, no. 3, pp. 373–381.
Rosazza, J.P., Kammer, M., Youel, L., Smith, R.V., Erhardt, P.W., Truong, D.H., and Leslie, S.W., Xenobiotica, 1977, vol. 7, no. 3, pp. 133–143.
Author information
Authors and Affiliations
Corresponding author
Additional information
The article is published in the original.
Rights and permissions
About this article
Cite this article
Fan, N., Du, C.H., Xu, J.Q. et al. Glycosylation and sulfation of 4-methylumbelliferone by Gliocladium deliquescens NRRL 1086. Appl Biochem Microbiol 53, 85–93 (2017). https://doi.org/10.1134/S0003683817010033
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0003683817010033