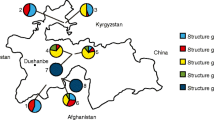
Abstract
Broomrapes (Orobanche spp.) are holoparasitic plants without chlorophyll, parasitising roots of a wide range of hosts. Some species are noxious weeds having a devastating effect on many important crops. Knowledge of the variability in the population genetics of weedy broomrapes is important in any attempt to develop resistance-breeding strategies for the relevant host crops against these parasites. The distribution of genetic variation in O. aegyptiaca, O. cumana, O. crenata, O. foetida, O. gracilis, O. hederae, O. minor and O. ramosa populations has been reviewed in relation to (1) the amount and structure of population genetic diversity as a consequence of the mating system, (2) the geographic differentiation as shaped by migration, (3) the spread of infestations into new areas followed by genetic drift, and (4) the host-differentiation owing to the host-induced selection. It has been shown that dominant markers such as RAPDs and AFLPs can be used efficiently in the analysis of predominant mating system and in the analysis of host-differentiation. As crop-seed exchange and transport play an important role in migration of seeds of Orobanche, geographical differentiation is difficult to discern from molecular data. Finally, in analysing genetic drift, co-dominant markers such as microsatellites are clearly needed
Similar content being viewed by others
References
Benharrat H, Veronesi C, Theodet C, Thalouarn P (2002) Orobanche species and population discrimination using intersimple sequence repeat (ISSR). Weed Research 42, 470–475. doi: 10.1046/j.1365-3180.2002.00305.x
Benharrat H, Boulet C, Theodet C, Thalouarn P (2005) Virulence diversity among branched broomrape (O. ramosa L.) populations in France. Agronomy for Sustainable Development 25, 123–128. doi: 10.1051/ agro:2004059
Brault M, Betsou F, Jeune B, Tuquet C, Sallé G (2007) Variability of Orobanche ramosa populations in France as revealed by cross infestations and molecular markers. Environmental and Experimental Botany 61, 272–280. doi: 10.1016/j.envexpbot.2007.06.009
Buschmann H, Gonsior G, Sauerborn J (2005) Pathogenicity of branched broomrape (Orobanche ramosa) populations on tobacco cultivars. Plant Pathology 54, 650–656. doi: 10.1111/j.1365-3059.2005.01211.x
Castejón-Muñoz M (1989) ‘Parasitismo del jopo (Orobanche cernua Loelf., O. cumana Wallr.) en girasol (Helianthus annuus L.), su respuesta al glifosato y variabilidad isoenzimatica intraespecifica.’ (University of Seville: Seville, Spain)
Castejón-Muñoz M, Suso MJ, Romero-Muñoz F, García-Torres L (1991) Isoenzymatic study of Orobanche (Orobanche cernua) populations infesting sunflower (Helianthus annuus). In ‘International symposium on parasitic weeds’. (Eds JK Ransom, LJ Musselman, AD Worsham, C Parker) pp. 313–319. (CIMMYT: Nairobi, Kenya)
Cornuet JM, Luikart G (1996) Description and power analysis of two tests for detecting recent population bottlenecks from allele frequency data. Genetics 144, 1119–1127.
Cubero JI, Moreno T (1979) Agronomic control and sources of resistance in Vicia faba to Orobanche crenata. In ‘Some current research on Vicia faba in Western Europe’. (Eds DA Bond, GT Scarascia-Mugnozza, MH Poulsen) pp. 41–80. (Commission of the European Communities: Luxemburg)
Dlugosch KM, Parker IM (2008) Founding events in species invasions: genetic variation, adaptive evolution, and the role of multiple introductions. Molecular Ecology 17, 431–449. doi: 10.1111/j.1365-294X.2007.03538.x
Dubois S, Cheptou PO, Petit C, Meerts P, Poncelet M, Vekemans X, Lefèbvre C, Escarré J (2003) Genetic structure and mating systems of metallicolous and nonmetallicolous populations of Thlaspi caerulescens. New Phytologist 3, 633–641. doi: 10.1046/j.1469-8137.2003.00684.x
Eizenberg H, Plakhine D, Landa T, Achdari G, Joel DM, Hershenhorn J (2004) First report of a new race of sunflower broomrape (Orobanche cumana) in Israel. Plant Disease 88, 1284. doi: 10.1094/PDIS.2004.88.11.1284C
Excoffier L, Smouse PE, Quattro JM (1992) Analysis of molecular variance inferred from metric distances among DNA haplotypes: application to human mitochondrial DNA restriction sites. Genetics 131, 479–491.
Fernández-Martínez JM, Melero-Vara J, Muñoz-Ruz J, Ruso J, Domínguez J (2000) Selection of wild and cultivated sunflower for resistance to a new broomrape race that overcomes resistance to Or 5 gene. Crop Science 40, 550–555.
Frost CC, Musselman LJ (1980) Clover broomrape (Orobanche minor) in the United States. Weed Science 28, 119–122.
Gagne G, Roeckel-Drevet P, Grezes-Besset B, Shindrova P, Ivanov P, Grand-Ravel C, Vear F, Tourvieille de Labrouhe D, Charmet G, Nicolas P (1998) Study of the variability and evolution of Orobanche cumana populations infesting sunflower in different European countries. Theoretical and Applied Genetics 96, 1216–1222. doi: 10.1007/s001220050859
Hamrick JL, Godt MJW (1989) Allozyme diversity in plant species. In ‘Plant population genetics, breeding and genetic resources’. (Eds AHD Brown, MT Clegg, AL Kahler, BS Weir) pp. 43–63. (Sinauer Associates: Sunderland, MA)
Hamrick JL, Godt MJW (1997) Allozyme diversity in cultivated crops. Crop Science 37, 26–30.
Hamrick JL, Nason JD (1996) Consequences of dispersal in plants. In ‘Population dynamics in ecological space and time’. (Eds EO Rhodes, RK Chesser, MH Smith) pp. 203–236. (The University of Chicago Press: Chicago, IL)
Hassan EA (1998) Broomrape species in Egypt, a recent survey in relation to geographical distribution. In ‘Comptes rendu 6ème symposium Méditerranéen EWRS’. (Ed. J Maillet) pp. 13–15. (ENSA: Montpellier, France)
James RW (1976) Apreliminary note on the control of broomrape (Orobanche minor) in flue-cured tobacco. New Zealand Tobacco Growers Journal May, 7–8.
Joel DM (1987) Detection and identification of Orobanche seeds using fluorescence microscopy. Seed Science and Technology 15, 119–124.
Joel DM, Steffens JC, Mattews DE (1994) Germination of weedy root parasites. In ‘Seed development and germination’. (Eds J Kigel, G Galili) pp. 567–598. (Marcel Dekker: New York)
Joel DM, Portnoy V, Tzuri G, Katzir N (1998a) Use of DNA fingerprinting for soil-borne seed identification. Aspects of Applied Biology 51, 23–27.
Joel DM, Benharrat H, Portnoy VH, Thalouarn P (1998b) Molecular markers for Orobanche species — New approach and their potential uses. In ‘Current problems of Orobanche researches. Proceedings of the fourth international workshop on Orobanche’. (Eds K Wegmann, LJ Musselman, DM Joel) pp. 115–124. (Institute for Wheat and Sunflower: Albena, Bulgaria)
Joel DM, Hershenhorn Y, Eizenberg H, Aly R, Ejeta G, Rich PJ, Ransom JK, Sauerborn J, Rubiales D (2007) Biology and management of weedy root parasites. Horticultural Reviews 33, 267–349. doi: 10.1002/9780470 168011.ch4
Kharrat M, Halila MH, Linke KH, Haddar T (1992) First report of Orobanche foetida Poiret on faba bean in Tunisia. FABIS Newsletter 30, 46–47.
Lu YH, Melero-Vara JM, Garcia-Tejada JA, Blanchard P (2000) Development of SCAR markers linked to the gene Or 5 conferring resistance to broomrape (Orobanche cumana Wallr.) in sunflower. Theoretical and Applied Genetics 100, 625–632.
Manel S, Schwartz MK, Luikart G, Taberlet P (2003) Landscape genetics: combining landscape ecology and population genetics. Trends in Ecology & Evolution 18, 189–197. doi: 10.1016/S0169-5347(03)00008-9
Manen J-F, Habashi C, Jeanmonod D, Park J-M, Schneeweis GM (2004) Phylogeny and intraspecific variability of holoparasitic Orobanche (Orobanchaceae) inferred from plastid rbcL sequences. Molecular Phylogenetics and Evolution 33, 482–500. doi: 10.1016/j.ympev. 2004.06.010
Musselman LJ (1986) Taxonomy of Orobanche. In ‘Proceedings of a workshop on the biology and control of Orobanche’. (Ed. SJ ter Borg) pp. 2–20. (LH/VPO: Wageningen, The Netherlands)
Musselman LJ (1994) Taxonomy and spread of Orobanche. In ‘Biology and management of Orobanche. Proceedings of the third international workshop of Orobanche and related Striga research’. (Eds AH Pieterse, JAC Verkleij, SJ ter Borg) pp. 27–35. (Royal Tropical Institute: Amsterdam, The Netherlands)
Musselman LJ, Parker C (1982) Preliminary host ranges of some strains of economically important broomrapes (Orobanche). Economic Botany 36, 270–273.
Nybom H (2004) Comparison of different nuclear DNA markers for estimating intraspecific genetic diversity in plants. Molecular Ecology 13, 1143–1155. doi: 10.1111/j.1365-294X.2004.02141.x
Paran I, Gidoni D, Jacobsohn R (1997) Variation between and within orobanche (Orobanche) species revealed by RAPD markers. Heredity 78, 68–74. doi: 10.1038/hdy.1997.8
Parker C, Riches CR (1993) ‘Parasitic weeds of the world: biology and control.’ (CAB International: Wallingford, UK)
Pazy B (1998) Diploidization failure and apomixis in Orobanchaceae. Botanical Journal of the Linnean Society 128, 99–103.
Pérez-Vich B, Akhtouch B, Knapp SJ, Leon AJ, Velasco L, Fernández-Martínez JM, Berry ST (2004) Quantitative trait loci for broomrape (Orobanche cumana Wallr.) resistance in sunflower. Theoretical and Applied Genetics 109, 92–102. doi: 10.1007/s00122-004-1599-7
Pieterse AH (1979) The broomrapes (Orobanchaceae): a review. Abstracts on Tropical Agriculture 5, 9–35.
Plitmann U (2002) Agamospermy is much more common than conceived: a hypothesis. Israel Journal of Plant Sciences 50, 111–117. doi: 10.1560/ 4T32-TWGY-T5NP-JDAE
Pritchard JK, Stephens M, Donnelly P (2000) Inference of population structure using multilocus genotype data. Genetics 155, 945–959.
Radwan MS, Abdalla MMF, Fischbeck G, Metwally AA, Darwish DS (1988) Variation in reaction of faba bean lines to different accessions of Orobanche crenata Forsk. Plant Breeding 101, 208–216. doi: 10.1111/ j.1439-0523.1988.tb00289.x
Román B, Rubiales D, Torres AM, Cubero JI, Satovic Z (2001) Genetic diversity in Orobanche crenata populations from southern Spain. Theoretical and Applied Genetics 103, 1108–1114. doi: 10.1007/ s001220100644
Román B, Satovic Z, Rubiales D, Torres AM, Cubero JI, Katzir N, Joel DM (2002) Variation among and within populations of the parasitic weed Orobanche crenata from Spain and Israel revealed by inter simple sequence repeat markers. Phytopathology 92, 1262–1266. doi: 10.1094/ PHYTO.2002.92.12.1262
Román B, Alfaro C, Torres AM, Moreno MT, Satovic Z, Pujadas A, Rubiales D (2003) Genetic relationships among Orobanche species as revealed by RAPD analysis. Annals of Botany 91, 637–642. doi: 10.1093/aob/mcg060
Román B, Hernandez R, Pujadas A, Cubero JI, Rubiales D, Satovic Z (2007a) Genetic diversity in two variants of Orobanche gracilis Sm. [var. gracilis and var. deludens (Beck) A.Pujadas] from different regions of Spain. Electronic Journal of Biotechnology 10, 221–229. doi: 10.2225/vol10-issue2-fulltext-6
Román B, Satovic Z, Alfaro C, Moreno MT, Kharrat M, Perez de Luque A, Rubiales D (2007b) Host differentiation in Orobanche foetida Poir. Flora 202, 201–208.
Rubiales D (2003) Parasitic plants, wild relatives and the nature of resistance. New Phytologist 160, 459–461. doi: 10.1046/j.1469-8137.2003.00929.x
Rubiales D, Sadiki M, Román B (2005) First report of Orobanche foetida on common vetch (Vicia sativa) in Morocco. Plant Disease 89, 528. doi: 10.1094/PD-89-0528A
Ruso J, Sukno S, Dominguez-Jiménez J, Melero-Vara JM, Fernández-Martínez JM (1996) Screening of wild Helianthus species and derived lines for resistance to several populations of Orobanche cernua. Plant Disease 80, 1165–1169.
Sackston WE (1992) On a treadmill: breeding sunflower for resistance to disease. Annual Review of Phytopathology 30, 529–551. doi: 10.1146/ annurev.py.30.090192.002525
Schneeweiss GM, Colwell A, Park J-M, Jang CG, Stuessy TF (2004) Phylogeny of holoparasitc Orobanche (Orobanchaceae) inferred form nuclear ITS sequences. Molecular Phylogenetics and Evolution 30, 465–478. doi: 10.1016/S1055-7903(03)00210-0
Slatkin M (1980) The distribution of mutant alleles in a subdivided population. Genetica 95, 503–524.
Sukno S, Melero-Vara JM, Fernández-Martínez JM (1999) Inheritance of resistance to Orobanche cernua Loefl. in six sunflower lines. Crop Science 39, 674–678.
Sweigart AL, Willis JH (2003) Patterns of nucleotide diversity in two species of Mimulus are affected by mating system and asymmetric introgression. Evolution 11, 2490–2506.
Tang S, Heesacker A, Kishore VK, Fernandez A, Sadik ES, Cole G, Knapp SJ (2003) Genetic mapping of the Or 5 gene for resistance to Orobanche race E in sunflower. Crop Science 43, 1021–1028.
Teryokhin ES (1997) ‘Weed broomrapes - systematics, ontogenesis, biology, evolution’. (Aufstieg-Verlag: Germany)
Turner MG, Gardner RH, O’Neill RV (2001) ‘Landscape ecology in theory and practice: pattern and process.’ (Springer-Verlag: New York)
Uhlich H, Pusch J, Barthel KJ (1995) ‘Die Sommerwurzarten Europas.’ (Westarp Wissenschaften: Magdeburg, Germany)
Vaz Patto MC, Díaz-Ruiz R, Satovic Z, Román B, Pujadas-Salvà AJ, Rubiales D (2008) Genetic diversity of Moroccan populations of Orobanche foetida: from wild parasitic plants to parasitic weeds. Weed Research 48, 179–186. doi: 10.1111/j.1365-3180.2008.00621.x
Verkleij JAC, Pieterse AH (1994) Genetic variability of Orobanche (broomrape) and Striga (witchweed) in relation to host specificity. In ‘Biology and management of Orobanche. Proceedings of the third international workshop of Orobanche and related Striga research’. (Eds AH Pieterse, JAC Verkleij, SJ ter Borg) pp. 67–79. (Royal Tropical Institute: Amsterdam, The Netherlands)
Verkleij JAC, Egbers WS, Pieterse AH (1991a) Allozyme variations in populations of Orobanche crenata from Syria. In ‘Progress in Orobanche Research. Proceedings of the international workshop on Orobanche research’. (Eds K Wegmann, LJ Musselman) pp. 304–317. (Eberhard-Karls University: Tübingen, Germany)
Verkleij JAC, Koevoets P, López-Granados F, Egbers WS, García-Torres L, Pieterse AH (1991b) Genetic variability in populations of Orobanche crenata from Spain. In ‘Proceedings of the fifth international symposium on parasitic weeds’. (Eds JK Ransom, LJ Musselman, AD Worsham, C Parker) pp. 462–469. (CIMMYT: Nairobi, Kenya)
Vrânceanu AV, Pirvu N, Stoanescu FM, Pacureanu M (1986) Some aspects of the interaction Helianthus annuus L.2 /Orobanche cumana, and its implications in sunflower breeding. In ‘Proceedings of a workshop on the biology and control of Orobanche’. (Ed. SJ ter Borg) pp. 181–189. (LH/VPO: Wageningen, The Netherlands)
Wegmann K (1998) The Orobanche problem in tobacco. In ‘Current problems of Orobanche researches. Proceedings of the fourth international workshop on Orobanche’. (Eds K Wegmann, LJ Musselman, DM Joel) pp. 21–24. (Institute for Wheat and Sunflower: Albena, Bulgaria)
Westwood JH, Fagg CM (2004) ISSR characterization of Orobanche minor populations in the US. In ‘Proceedings of the 8th international parasitic weeds symposium’. (Eds DM Joel, H Bouwmeester, P Delavault, G Ejeta, F Kanampiu, M Press, B Román, MP Timko, JAC Verkleij, JH Westwood, K Yoneyama, WJ Zhou) p. 15. (IPPS: Durban, South Africa)
Wolfe AD, Randle CP, Liang L, Steiner KE (2005) Phylogeny and biogeography of Orobanchaceae. Folia Geobotanica 40, 115–134. doi: 10.1007/BF02803229
Zeid M, Madkour M, Koraiem Y, Nawar A, Soliman M, Zaitoun F (1997) Molecular studies on Orobanche. Journal of Phytopathology 145, 351–355. doi: 10.1111/j.1439-0434.1997.tb00413.x
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Satovic, Z., Joel, D.M., Rubiales, D. et al. Population genetics in weedy species of Orobanche . Australasian Plant Pathology 38, 228–234 (2009). https://doi.org/10.1071/AP08100
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1071/AP08100