Abstract
Habitat fragmentation can reduce population viability, especially for area-sensitive species. The Minimum Area Requirements (MAR) of a population is the area required for the population’s long-term persistence. In this study, the response of occupancy probability of giant pandas against habitat patch size was studied in five of the six mountain ranges inhabited by giant panda, which cover over 78% of the global distribution of giant panda habitat. The probability of giant panda occurrence was positively associated with habitat patch area, and the observed increase in occupancy probability with patch size was higher than that due to passive sampling alone. These results suggest that the giant panda is an area-sensitive species. The MAR for giant panda was estimated to be 114.7 km2 based on analysis of its occupancy probability. Giant panda habitats appear more fragmented in the three southern mountain ranges, while they are large and more continuous in the other two. Establishing corridors among habitat patches can mitigate habitat fragmentation, but expanding habitat patch sizes is necessary in mountain ranges where fragmentation is most intensive.
Similar content being viewed by others
Introduction
Habitat fragmentation poses serious threats to many species and to global biodiversity1,2,3, including reductions in population viability4,5,6. Animal species absent or rare in small habitat patches are called ‘area-sensitive’ species7,8,9. Researchers have documented the ‘area-sensitive’ species in almost all taxonomic groups of wildlife, including insects10,11, fish12, reptiles and amphibians13, mammals5,14 and birds15,16.
The most effective way to conserve area-sensitive species is to maintain habitat patches large enough for the persistence of local populations. Therefore, estimating habitat patch area requirements17,18 is an essential component of conservation plans. The Minimum Area Requirements (MAR) of a species is defined as the amount of space (suitable habitat) that is required for the long-term persistence of a population19. Recently, an increasing number of studies have focused on MAR for protecting animals and highlighted the importance of this measure for conservation decisions12,20,21,22. Despite the conservation significance of MAR, empirical studies estimating it remain limited19.
The giant panda (Ailuropoda melanoleuca) is regarded as one of the most imperiled mammals in the world23. Populations of giant panda originally extended throughout most of southern and eastern China, northern Myanmar, and northern Vietnam24. Giant panda habitat is dramatically degrading due to increasing human activities and natural catastrophes25,26,27,28. Current remnant populations are restricted to six separate regions scattered throughout rugged mountain ranges at the eastern edge of the Tibetan Plateau29,30. Within those regions, the habitats are highly fragmented31,32,33,34, which drives further decreases in total giant panda numbers35.
Many studies have been conducted on the habitat of giant panda, and most of them focused on habitat selection36,37,38, habitat quality assessment39,40,41 and the impacts of human activity27,42,43. However, little work has been done on the effects of habitat patch size on giant panda populations. Giant pandas are generally regarded as an area-sensitive species in previous studies44, but empirical evidence is still lacking. This study seeks to understand how giant panda population presence depends on patch size and to test whether the giant panda are area-sensitive. We use extensive empirical datasets on giant panda presence and environmental variables to evaluate the MAR for giant panda in five mountain ranges, covering more than 78.7% of giant panda habitat and supporting more than 74.4% of giant pandas remaining in the wild45. We also examine the characteristics of remnant habitat patches in these mountain ranges, which collectively comprise the most important giant panda habitat in the world.
Results
Using the giant panda presence records and environmental variables, we estimated habitat suitability index (HSI). The area under the curve (AUC) value of 0.843 indicated that the Maximum Entropy (MaxEnt) model of giant panda presence had a good discriminatory ability, while the true skill statistic (TSS) score of 0.772 indicated an excellent discriminatory ability of MaxEnt. By the cut-off value that maximized the TSS score, we identified 5569 habitat patches with a total area of 15475.53 km2. The habitat patches clustered in five mountain ranges: Minshan Mountains (MS Mountains), Qionglaishan Mountains (QLS Mountains), Daxiangling Mountains (DXL Mountains), Xiaoxiangling Mountains (XXL Mountains) and Liangshan Mountains (LS Mountains) (Table 1).
A logistic regression model for the response of occupancy probability to patch area showed a positive association between occupancy probability and habitat area (P < 0.001) (Table 2, Fig. 1). We tested whether the giant panda was area-sensitive by testing whether the occupancy probability of giant panda in a patch of a given size was greater than the probability driven only by passive sampling, in which large patches have a higher probability of being occupied at random than smaller patches46,47,48. We used a curve, for the response of occupancy rate to patch area only due to passive sampling, as the null response curve. Occupancy probability to patch area exceeded the null response curve after 40.4 km2 (Fig. 1), suggesting area-sensitivity of giant panda.
Relationship between the probability of giant panda presence and habitat size.
Open circles are patches without indicators of giant panda presence and filled circles are patches where giant pandas were present. The black line is the logistic regression fit, and the grey region shows 95% confidence intervals. The red line represents the null response curve derived from pure passive sampling.
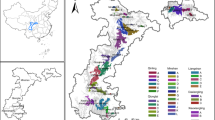
We calculated the value of EDp, i.e. the patch area at which it is likely that pandas were present with probability of p%. Both counts and total sizes of habitat patches with size over EDp decreased with the increasing of p, but they did not change from ED80 to ED99 (Fig. 2). At the 90% effective dose (ED90, indicating the patch area at which it is 90% likely that pandas were present), the value of MAR was estimated as 114.7 km2. Ten habitat patches exceeded the estimated value of MAR, which covered 86.1% of the habitat of giant panda. Six habitat patches greater than the MAR were in the MS and QLS Mountains, including the four largest patches. Two patches whose sizes exceed the MAR threshold were located in the LS Mountains, and another two patches in the DXL Mountains just reached the MAR threshold. In contrast, the habitat in the XXL Mountains was highly fragmented, consisting of 1,326 habitat patches, none of which exceeded the MAR threshold (Fig. 3).
Discussion
Using the latest and most complete empirical data of giant panda presence and land cover, we analyzed the response of occupancy probability of giant panda against habitat patch size. The occupancy probability of giant panda was positively associated with habitat patch areas, and the changes in occupancy as a function of patch area exceeded effects due only to sampling. These results suggest that giant panda is indeed area-sensitive, implying that the size of individual habitat patches is important to giant panda conservation, as is total habitat and population sizes.
Though area-sensitive species are believed to be absent from small habitat fragments, the area-sensitivity of species are mostly identified by the relationship between the patch-specific density of individuals and patch size49,50,51,52. For sparsely distributed species, where calculated densities can be strongly influenced by small numbers of individuals, occupancy rather than density is the preferred metric for assessing area-sensitivity. Generally, low probability of occurrence for wildlife in small patches is attributed to demographic stochasticity53,54, environmental stochasticity and catastrophic events55,56, inbreeding and loss of heterozygosity57, edge effects58, food shortages in small patches15, and landscape structure59. Our study went further, comparing the occurrence-patch size curve with a null response curve, demonstrating that area-sensitivity in giant pandas is real and not just an effect of patch size sampling.
The MAR was estimated to be 114.7 km2 using a logistic regression of habitat patch size on giant panda occupancy. MAR can also be estimated from Population Viability Analyses (PVAs)19. A MAR of 156–248 km2 for giant panda was estimated from its PVA (40 individuals)60 and home range size (3.9–6.2 km2)29. The MAR value estimated from PVA and home range size is higher than that estimated from occupancy patterns and patch area. This discrepancy may arise because giant panda home ranges often overlap61,62, which will inflate estimates of required space in PVAs. MAR estimated from occupancy patterns are believed to be sensitive to transient dynamics, while MAR estimated using the PVA-based approach enable us to consider time horizon and extinction probability19. As a mechanism-driven model, the plausibility of the PVA-based approach is dependent upon the reality of the modeling assumptions and the robustness of model behavior when population and environment parameters cannot be accurately determined, both of which are, often untested. Meanwhile, occupancy data is an empirical synthetic result of all ecological factors and processes in the research area. MAR estimated from occupancy data, together with that estimated by a PVA-based approach, can produce more applicable conservation planning and policy.
Giant panda habitats are large and continuous in the MS and QLS Mountains, preserving approximately 45 and 36% of giant panda habitat in the five mountain ranges, respectively. Residential areas and farmland along roads and valleys segment the habitat into several patches, of which Patch A, B, C and D (in Fig. 3) are large enough to hold relatively viable populations. The gaps between Patch A, B and D are narrow, therefore establishing corridors could further enhance the situation of populations in those patches. It is interesting that giant panda have not been found in patch C at least since the 1980s, even though this patch exceeds the MAR threshold and has a high habitat suitability index. This absence of giant panda may occur because patch C is isolated from other habitat patches in the MS and QLS Mountains by the Minjiang River (Fig. 3), a major tributary, and by human communities along those rivers. Patch E accounted for about 85% of the total area of habitat patches in the QLS Mountains. Several habitat patches, each smaller than the MAR but still containing giant panda presence records, were distributed around Patch E. Those records may stem from ‘spillover’ of giant panda from patch E rather than separate viable populations63. Therefore, we urge connection of these small habitat patches to Patch E to enhance the persistence of giant panda in the region.
Though habitats are fragmented in the DXL and LS Mountains44, four habitat patches (Patches G-J) exceed the giant panda’s MAR. Establishing corridors to link Patches G and H with other patches in the QLS Mountains could be particularly beneficial for giant panda by creating a single patch substantially larger than the MAR. Such corridors may be possible because there is no major river or other barrier separating the patches in question. In contrast, habitat patches in the LS Mountains have been isolated from adjacent giant panda habitat in the DXL and XXL Mountains due to the Dadu River and a wide stretch of human settlements. As a result, connecting patches in the LS Mountains to those in other mountain ranges appears infeasible. Nevertheless, within the LS Mountains, the gap between Patches I and J is very narrow, suggesting it might be possible to link the patches with corridors.
Of all habitats in the five mountain ranges, those in the XXL Mountains are the most fragmented. No patch in the XXL Mountains exceeds the MAR, and the largest patch therein was a mere 81.7 km2. Unsurprisingly, the population of giant panda in the XXL Mountains is the smallest and most endangered of all. To help improve this population, giant panda have been translocated from captivity to the XXL Mountains64. However, Veitch65 suggested that revealing and repairing the original cause of population degrading is the most important prerequisite to successful translocation. Our MAR analyses imply that these translocation efforts are insufficient to guarantee the long-term survival of local populations, unless efforts are also made to reduce habitat fragmentation. Two parallel efforts would be beneficial. One effort should focus on restoring habitats to increase patch size and suitability for giant pandas. The other should focus on creating ‘corridor groups’66 to connect highly fragmented patches inside the XXL Mountains. Together these two restoration strategies would consolidate several small patches into a bigger one, thereby helping to relieve the problem of giant panda habitat fragmentation.
This study has examined only the effects of habitat patch area on giant panda occupancy. Habitat isolation is another key factor that can influence occupancy patterns in other systems5,67,68,69. However, we did not explore the effects of isolation on giant panda occupancy here because there was a strong negative correlation between patch size and distance to the nearest patch. Nevertheless, isolation should be considered in future studies of giant panda occupancy that explicitly adopt a more mechanistic perspective on giant panda dispersal.
We determined the value of MAR based on the response of giant panda occurrence probability against habitat patch size. The landscapes and forest habitats in which giant panda live have changed considerably in the five mountain ranges over the last few decades, and the impacts of these changes on populations may lag behind the physical changes themselves. For example, because wild giant pandas may live as long as 13.3 years70, a shrinking habitat patch can still be occupied by giant pandas for many years beyond when the patch size falls below the true MAR, due to extinction debt71 or just because of the stochastic nature of extinction72,73. This time lag effect introduces a downward bias on the estimated MAR because the occurrence patterns have not equilibrated to habitat changes. In contrast, though rare, landscapes where dispersal is sufficient to maintain long-term connections between nearby patches (e.g., landscapes where big river gorges, farming areas, communities and roads introduce transient but not permanent barriers to connectivity; for example, Patch C) would bias the estimated MAR upwards. Because time lags, rare dispersal events, and other factors will influence giant panda occupancy patterns over the long-term, our calculated MAR values are merely estimates of the relationship between giant panda occupancy and patch size. Nevertheless, these estimates provide helpful guides to assist in identifying priority areas for restoration efforts.
We identified habitat patches using HSI, but this does not negate the importance of areas outside the patches for the viability of giant panda. Almpanidou et al.74 suggest that conservation planning should consider the entire habitat rather than isolated patches of high quality, because less suitable areas can be indispensable connections among habitat patches. That connection is essential for the viability of populations, especially those in small patches75. Future work should analyze habitat connectivity and plan corridors to enhance the viability of giant panda populations76, particularly in southern mountain ranges where most habitat patches are smaller than the MAR. Because fragmentation of habitats may be aggravated in the future due to possible global climate change76,77,78, it is necessary to evaluate their status within that context and investigate potential solutions.
Methods
Data and data sources
All of the presence records (feces, footmark and forage traces) for giant panda in the five mountain ranges (i.e. Minshan Mountains, Qionglaishan Mountains, Daxiangling Mountains, Xiaoxiangling Mountains and Liangshan Mountains) were obtained from the Fourth National Giant Panda Survey (NGPS4) in Sichuan province. The survey was carried out between 28°10′ N and 33°48′ N, 101°50′ E and 105°28′ E, from 2011 to 2013, and covered about 330,000 km2.
Giant panda habitat patches were identified by 29 spatially explicit environmental variables and classified into two categories, namely geography and land use (Supplementary Information). Elevation data were obtained from the Digital Elevation Model (DEM) (30 m × 30 m), provided by the International Scientific & Technical Data Mirror Site, Computer Network Information Center, Chinese Academy of Sciences (http://www.gscloud.cn). Other geographic variables (slope, curvature, topography position index (TPI), aspect, solar radiation index, and latitude) were derived from the DEM. Information on land use was obtained from the Second National Forest Inventory (NFI2) and revised by the NGPS4 dataset.
Habitat patch identification
The habitat suitability index was evaluated using MaxEnt modeling79,80. This technique, which has its origins in statistical mechanics81, builds a map of a species’ likelihood distribution by estimating the probability distribution of maximum entropy82,83. The presence records of giant pandas were thinned randomly to consolidate presence records from sites separated by less than 1,125 m. The distance of 1,125 m was roughly determined by the value of the smallest home range of giant pandas (3.9 km2)29. Overall, 4,224 records of giant panda presence were consolidated to 1,421 valid presence sites.
We did not try to reduce the collinearity by eliminating strongly correlated environmental variables, because high collinearity is not a significant problem for machine learning methods if the goal is predicting presence rather than interpreting the response of presence to environmental variables84. Moreover, eliminating variables may discard critical information for prediction, even when those variables are strongly correlated with other variables. For example, the variable of slope is strongly correlated with altitude (r = 0.8469, P < 0.01) at the scale of the five mountain ranges, whereas slope is apparently indispensable for predicting giant panda presences at finer scales.
We measured model performance using the area under the receiver-operator curve (AUC)85 and the True Skill Statistic (TSS)86. A value of AUC greater than 0.90 was considered to be excellent, 0.8 to 0.9 good, 0.7 to 0.8 fair, and 0.6 to 0.7 poor87. For TSS values, greater than 0.75 was considered to be excellent, 0.4 to 0.75 good, and less than 0.4 poor88.
Using the reduced set of valid presence recodes and environmental variables, we performed a 10-fold cross-validation procedure to create MaxEnt models. The averaged AUC and TSS across the 10-fold test sets were calculated to determine the predictive power of the models. We delineated the averaged HSI into suitable and unsuitable habitat based on the threshold value that would maximize the TSS score, i.e., the sensitivity-specificity sum maximization approach89,90. There are many approaches to determining the thresholds to transform results of species distribution modeling from suitability for species occurrence to presences/absences. Liu et al.91 compared 12 methods and suggested that sensitivity-specificity sum maximization, which we adopted, was a good approach.
Those adjacent suitable habitat cells were joined together as a habitat patch following an 8-cell rule. The 8-cell rule considers all 8 adjacent cells, including the 4 orthogonal and 4 diagonal neighbors. A habitat patch is considered occupied if any presence record of giant panda (valid or not) is located in it.
Area-sensitivity identification
Identifying whether a species is area-sensitive is tricky. An occupancy rate increasing with patch size does not necessarily indicate area-sensitivity because occupancy of a species may be higher in bigger habitat patches than in smaller ones due to passive sampling. Specifically, if a patch has an area of a, and the occupancy rate of a species in that patch is q, then in a bigger patch with an area of na, the occupancy rate will be 1 − (1 − q)n in the absence of any area-related ecological effects. In contrast, a species that is truly area-sensitive should have an occupancy curve that increases faster than that of passive sampling as patch size increases. A variety of mechanisms contribute to this effect. For example, bigger patches can increase the availability of ‘core’ habitat area unaffected by edge effects or human disturbance, and thus are favorable for species that require interior habitat92. Therefore, an area-sensitive species should show a higher occupancy rate than that driven by passive sampling alone.
To test whether the occupancy rate of giant panda in patches of given size is higher than that driven by passive sampling, we compared the observed logistic occupancy curve with a null response curve. The null response curve is defined by the function  , where ab (corresponding to the minimum home range size of a giant panda29, namely 3.9 km2) and a (a > ab) is the patch size, qb is the occupancy rate in a patch with size of ab. The null response curve corresponds to the probability of a habitat patch of size a being occupied by giant panda, provided that patch is ecologically and functionally equivalent to
, where ab (corresponding to the minimum home range size of a giant panda29, namely 3.9 km2) and a (a > ab) is the patch size, qb is the occupancy rate in a patch with size of ab. The null response curve corresponds to the probability of a habitat patch of size a being occupied by giant panda, provided that patch is ecologically and functionally equivalent to  independent patches with areas of ab and shows no area-related ecological advantages over the set of small patches.
independent patches with areas of ab and shows no area-related ecological advantages over the set of small patches.
Estimating MAR
We progressed from occurrence estimates to estimates of giant panda’s area needs by leveraging dose-response curves from pharmacology93,94 in which the key measure is the effective dose (ED) that yields a particular probability. Thus, in the present study, EDpis the patch area at which it is likely that pandas were present with probability of p%. We used the ED90 as the value of MAR. The ED90 was derived from a logistic regression of probability of occurrence against patch size. The interpretation of ED90 is that 90% of patches of that size will support pandas. The ED90 is sometimes used as an estimation of the maximal effective dose in drug development95. Though we used a threshold of 90% to determine MAR, the choice of threshold may vary among conservation programs depending on specific objectives. Therefore, we plotted the function EDp (1% ≤ p% ≤ 99%) against p%, to show the effects of criterion choice on the amount of habitat patches matching the criteria.
Additional Information
How to cite this article: Qing, J. et al. The minimum area requirements (MAR) for giant panda: an empirical study. Sci. Rep. 6, 37715; doi: 10.1038/srep37715 (2016).
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
Andrén, H. Effects of Habitat Fragmentation on Birds and Mammals in Landscapes with Different Proportions of Suitable Habitat: A Review. Oikos 71, 355–366, doi: 10.2307/3545823 (1994).
Foley, J. A. et al. Global consequences of land use. Science 309, 570–574, doi: 10.1126/science.1111772 (2005).
Krauss, J. et al. Habitat fragmentation causes immediate and time-delayed biodiversity loss at different trophic levels. Ecol Lett 13, 597–605, doi: 10.1111/j.1461-0248.2010.01457.x (2010).
Moore, J. W., Yeakel, J. D., Peard, D., Lough, J. & Beere, M. Life-history diversity and its importance to population stability and persistence of a migratory fish: steelhead in two large North American watersheds. J Anim Ecol 83, 1035–1046, doi: 10.1111/1365-2656.12212 (2014).
Prugh, L. R., Hodges, K. E., Sinclair, A. R. & Brashares, J. S. Effect of habitat area and isolation on fragmented animal populations. Proceedings of the National Academy of Sciences 105, 20770–20775 (2008).
Vance, R. R. The Effect of Dispersal on Population Stability in One-Species, Discrete-Space Population-Growth Models. Am Nat 123, 230–254, doi: 10.1086/284199 (1984).
Lynch, J. F. & Whigham, D. F. Effects of Forest Fragmentation on Breeding Bird Communities in Maryland, USA. Biol Conserv 28, 287–324, doi: 10.1016/0006-3207(84)90039-9 (1984).
Lee, M., Fahrig, L., Freemark, K. & Currie, D. J. Importance of patch scale vs landscape scale on selected forest birds. Oikos 96, 110–118, doi: 10.1034/j.1600-0706.2002.960112.x (2002).
Fraser, G. S. & Stutchbury, B. J. M. Area-sensitive forest birds move extensively among forest patches. Biol Conserv 118, 377–387, doi: 10.1016/j.biocon.2003.06.006 (2004).
Tscharntke, T., Steffan-Dewenter, I., Kruess, A. & Thies, C. Characteristics of insect populations on habitat fragments: A mini review. Ecol Res 17, 229–239, doi: 10.1046/j.1440-1703.2002.00482.x (2002).
Buse, J. et al. Relative importance of pasture size and grazing continuity for the long-term conservation of European dung beetles. Biol Conserv 187, 112–119, doi: 10.1016/j.biocon.2015.04.011 (2015).
Liu, X. Q. & Wang, H. Z. Estimation of minimum area requirement of river-connected lakes for fish diversity conservation in the Yangtze River floodplain. Divers Distrib 16, 932–940, doi: 10.1111/j.1472-4642.2010.00706.x (2010).
Hager, H. A. Area-sensitivity of reptiles and amphibians: Are there indicator species for habitat fragmentation? Ecoscience 5, 139–147 (1998).
McAlpine, C. A. et al. The importance of forest area and configuration relative to local habitat factors for conserving forest mammals: A case study of koalas in Queensland, Australia. Biol Conserv 132, 153–165, doi: 10.1016/j.biocon.2006.03.021 (2006).
Zanette, L., Doyle, P. & Tremont, S. M. Food shortage in small fragments: Evidence from an area-sensitive passerine. Ecology 81, 1654–1666, doi: 10.1890/0012-9658(2000)081[1654:Fsisfe]2.0.Co;2 (2000).
Schmiegelow, F. K. A. & Monkkonen, M. Habitat loss and fragmentation in dynamic landscapes: Avian perspectives from the boreal forest. Ecological Applications 12, 375–389 (2002).
Brito, D. & Grelle, C. Estimating Minimum Area of Suitable Habitat and Viable Population Size for the Northern Muriqui (Brachyteles hypoxanthus). Biodiversity & Conservation 15, 4197–4210, doi: 10.1007/s10531-005-3575-1 (2006).
Fahrig, L. & Merriam, G. Habitat Patch Connectivity and Population Survival. Ecology 66, 1762–1768, doi: 10.2307/2937372 (1985).
Pe’er, G. et al. Toward better application of minimum area requirements in conservation planning. Biol Conserv 170, 92–102, doi: 10.1016/j.biocon.2013.12.011 (2014).
Prugh, L. R., Hodges, K. E., Sinclair, A. R. E. & Brashares, J. S. Effect of habitat area and isolation on fragmented animal populations. P Natl Acad Sci USA 105, 20770–20775, doi: 10.1073/pnas.0806080105 (2008).
Dardanelli, S., Nores, M. L. & Nores, M. Minimum area requirements of breeding birds in fragmented woodland of Central Argentina. Divers Distrib 12, 687–693, doi: 10.1111/j.1472-4642.2006.00266.x (2006).
Gurd, D. B., Nudds, T. D. & Rivard, D. H. Conservation of mammals in eastern North American wildlife reserves: How small is too small? Conserv Biol 15, 1355–1363, doi: 10.1046/j.1523-1739.2001.00188.x (2001).
Wei, F. W. et al. Giant pandas are not an evolutionary cul-de-sac: evidence from multidisciplinary research. Mol Biol Evol 32, 4–12, doi: 10.1093/molbev/msu278 (2015).
Lu, Z. et al. Patterns of genetic diversity in remaining giant panda populations. Conserv Biol 15, 1596–1607, doi: 10.1046/j.1523-1739.2001.00086.x (2001).
Li, Y. M., Guo, Z. W., Yang, Q. S., Wang, Y. S. & Niemela, J. The implications of poaching for giant panda conservation. Biol Conserv 111, 125–136 (2003).
Zhu, L. F., Hu, Y. B., Zhang, Z. J. & Wei, F. W. Effect of China’s rapid development on its iconic giant panda. Chinese Sci Bull 58, 2134–2139, doi: 10.1007/s11434-012-5647-4 (2013).
Liu, J. G. et al. A framework for evaluating the effects of human factors on wildlife habitat: the case of giant pandas. Conserv Biol 13, 1360–1370, doi: 10.1046/j.1523-1739.1999.98418.x (1999).
Bearer, S. et al. Effects of fuelwood collection and timber harvesting on giant panda habitat use. Biol Conserv 141, 385–393, doi: 10.1016/j.biocon.2007.10.009 (2008).
Hu, J. C., Schaller, G. B., Pan, W. S. & Zhu, J. The giant panda of Wolong. (Sichuan Publishing House of Science and Technology 1985).
Feng, T. T., van Manen, F. T., Zhao, N. X., Li, M. & Wei, F. W. Habitat Assessment for Giant Pandas in the Qinling Mountain Region of China. J Wildlife Manage 73, 852–858, doi: 10.2193/2008-186 (2009).
Qi, D. et al. Quantifying landscape linkages among giant panda subpopulations in regional scale conservation. Integr Zool 7, 165–174, doi: 10.1111/j.1749-4877.2012.00281.x (2012).
Wang, X., Xu, W. & Ouyang, Z. Integrating population size analysis into habitat suitability assessment: implications for giant panda conservation in the Minshan Mountains, China. Ecol Res 24, 1101–1109, doi: 10.1007/s11284-009-0589-2 (2009).
Xu, W., Ouyang, Z., Jiang, Z., Zheng, H. & Liu, J. Assessment of giant panda habitat in the Daxiangling Mountain Range, Sichuan, China. Chinese Biodiversity 14, 223–231 (2006).
Li, H. et al. Application of least-cost path model to identify a giant panda dispersal corridor network after the Wenchuan earthquake—Case study of Wolong Nature Reserve in China. Ecol Model 221, 944–952, doi: 10.1016/j.ecolmodel.2009.12.006 (2010).
Zhu, L. F. et al. Conservation Implications of Drastic Reductions in the Smallest and Most Isolated Populations of Giant Pandas. Conserv Biol 24, 1299–1306, doi: DOI 10.1111/j.1523-1739.2010.01499.x (2010).
Qi, D. et al. Different habitat preferences of male and female giant pandas. J Zool 285, 205–214, doi: 10.1111/j.1469-7998.2011.00831.x (2011).
Wei, F., Feng, Z., Wang, Z. & Li, M. Feeding strategy and resource partitioning between giant and red pandas. Mammalia 63, 417–430 (1999).
Zhang, Z. J. et al. What determines selection and abandonment of a foraging patch by wild giant pandas (Ailuropoda melanoleuca) in winter? Environ Sci Pollut R 16, 79–84, doi: 10.1007/s11356-008-0066-4 (2009).
Liu, J. G. Integrating ecology with human demography, behavior, and socioeconomics: Needs and approaches. Ecol Model 140, 1–8, doi: 10.1016/S0304-3800(01)00265-4 (2001).
Qi, D., Hu, Y., Gu, X., Li, M. & Wei, F. Ecological niche modeling of the sympatric giant and red pandas on a mountain-range scale. Biodivers Conserv 18, 2127–2141, doi: 10.1007/s10531-009-9577-7 (2009).
Xu, W. H. et al. Conservation of giant panda habitat in South Minshan, China, after the May 2008 earthquake. Front Ecol Environ 7, 353–358, doi: 10.1890/080192 (2009).
An, L. et al. Simulating demographic and socioeconomic processes on household level and implications for giant panda habitats. Ecol Model 140, 31–49, doi: 10.1016/S0304-3800(01)00267-8 (2001).
Linderman, M. A. et al. Modeling the spatio-temporal dynamics and interactions of households, landscapes, and giant panda habitat. Ecol Model 183, 47–65, doi: 10.1016/j.ecolmodel.2004.07.026 (2005).
Wang, T. J., Ye, X. P., Skidmore, A. K. & Toxopeus, A. G. Characterizing the spatial distribution of giant pandas (Ailuropoda melanoleuca) in fragmented forest landscapes. J Biogeogr 37, 865–878, doi: 10.1111/j.1365-2699.2009.02259.x (2010).
Foretry Department of Sichuan Province. The pandas in Sichuan: The 4th Survey Report on Giant Panda in Sichuan Province. (Sichuan Publishing House of Science and Technology, 2015).
Thornton, D., Branch, L. & Sunquist, M. Passive sampling effects and landscape location alter associations between species traits and response to fragmentation. Ecological Applications 21, 817–829, doi: 10.1890/10-0549.1 (2011).
Johnson, D. H. & Igl, L. D. Area requirements of grassland birds: a regional perspective. The Auk 118, 24–34 (2001).
Connor, E. F. & Mccoy, E. D. Statistics and Biology of the Species-Area Relationship. Am Nat 113, 791–833, doi: 10.1086/283438 (1979).
Hamback, P. A. et al. Habitat specialization, body size, and family identity explain lepidopteran density-area relationships in a cross-continental comparison. Proc Natl Acad Sci USA 104, 8368–8373, doi: 10.1073/pnas.0611462104 (2007).
Villard, M. A. On forest-interior species, edge avoidance, area sensitivity, and dogmas in avian conservation. Auk 115, 801–805 (1998).
Winter, M. & Faaborg, J. Patterns of area sensitivity in grassland-nesting birds. Conserv Biol 13, 1424–1436, doi: 10.1046/j.1523-1739.1999.98430.x (1999).
Brotons, L., Monkkonen, M. & Martin, J. L. Are fragments islands? Landscape context and density-area relationships in boreal forest birds. Am Nat 162, 343–357, doi: 10.1086/376887 (2003).
Gilpin, M. E. Minimum viable populations: processes of species extinction. Conservation biology: the science of scarcity and diversity, 19–34 (1986).
Traill, L. W., Bradshaw, C. J. A. & Brook, B. W. Minimum viable population size: A meta-analysis of 30 years of published estimates. Biol Conserv 139, 159–166, doi: 10.1016/j.biocon.2007.06.011 (2007).
Shaffer, M. L. Minimum Population Sizes for Species Conservation. Bioscience 31, 131–134, doi: 10.2307/1308256 (1981).
Shaffer, M. Minimum viable populations: coping with uncertainty. Viable populations for conservation 69, 86 (1987).
Frankham, R., Briscoe, D. A. & Ballou, J. D. Introduction to conservation genetics. (Cambridge University Press, 2002).
Fletcher, R. J. Multiple edge effects and their implications in fragmented landscapes. J Anim Ecol 74, 342–352, doi: 10.1111/j.1365-2656.2005.00930.x (2005).
Dover, J. & Settele, J. The influences of landscape structure on butterfly distribution and movement: a review. J Insect Conserv 13, 3–27, doi: 10.1007/s10841-008-9135-8 (2009).
Yang, Z., Hu, J. & Liu, N. The influence of dispersal on the metapopulation viability of Giant Panda (Aliuropoda melanoleuca) in the Minshan Mountains. Acta Zoologica Academiae Scientiarum Hungaricae 53, 169–184 (2007).
Pan, W. et al. A chance for lasting survival. Beijing University (2001).
Jetz, W., Carbone, C., Fulford, J. & Brown, J. H. The scaling of animal space use. Science 306, 266–268 (2004).
Xu, W. H. et al. Designing a conservation plan for protecting the habitat for giant pandas in the Qionglai mountain range, China. Divers Distrib 12, 610–619, doi: 10.1111/j.1366-9516.2006.00236.x (2006).
Chen, Z... J. . et al. The Genetic Structure of Small Giant Panda Population in the Liziping Nature Reserve after Reintroduction. Journal of Hechi University 36, 1–7 (2016).
Veitch, C. R. Habitat repair: A necessary prerequisite to translocation of threatened birds. Reintroduction Biology of Australian and New Zealand Fauna, 97–104 (1995).
Qing, J. et al. The corridor design of giant panda in Xiaoxiangling Mountains. Acta Ecologica Sinica 36, 1125–1133 (2016).
Maag, N., Karpati, T. & Bollmann, K. Semi-natural river system maintains functional connectivity and gene flow of the critically endangered gravel grasshopper (Chorthippus pullus). Biol Conserv 158, 88–97, doi: 10.1016/j.biocon.2012.08.015 (2013).
Elmhagen, B. & Angerbjörn, A. The applicability of metapopulation theory to large mammals. Oikos 94, 89–100 (2001).
Hanski, I. Metapopulation ecology. (Oxford University Press, 1999).
Wei, F. et al. A study on the life table of wild giant pandas. Acta Ecologica Sinica 9, 81–86 (1989).
Tilman, D., May, R. M., Lehman, C. L. & Nowak, M. A. Habitat Destruction and the Extinction Debt. Nature 371, 65–66, doi: 10.1038/371065a0 (1994).
Schoener, T. W. & Spiller, D. A. High Population Persistence in a System with High Turnover. Nature 330, 474–477, doi: 10.1038/330474a0 (1987).
Terborgh, J. & Winter, B. Some causes of extinction. Conservation biology: an evolutionary-ecological perspective. Sinauer Associates, Sunderland, Massachusetts, 119–133 (1980).
Almpanidou, V. et al. Providing insights on habitat connectivity for male brown bears: A combination of habitat suitability and landscape graph-based models. Ecol Model 286, 37–44, doi: 10.1016/j.ecolmodel.2014.04.024 (2014).
Beier, P. & Noss, R. F. Do habitat corridors provide connectivity? Conserv Biol 12, 1241–1252, doi: 10.1046/j.1523-1739.1998.98036.x (1998).
Wei, F. W. et al. Progress in the ecology and conservation of giant pandas. Conserv Biol 29, 1497–1507, doi: 10.1111/cobi.12582 (2015).
Liu, G., Guan, T. P., Dai, Q., Li, H. X. & Gong, M. H. Impacts of temperature on giant panda habitat in the north Minshan Mountains. Ecol Evol 6, 987–996, doi: 10.1002/ece3.1901 (2016).
Levinsky, I., Skov, F., Svenning, J. C. & Rahbek, C. Potential impacts of climate change on the distributions and diversity patterns of European mammals. Biodivers Conserv 16, 3803–3816, doi: 10.1007/s10531-007-9181-7 (2007).
Tittensor, D. P. et al. Predicting global habitat suitability for stony corals on seamounts. J Biogeogr 36, 1111–1128, doi: 10.1111/j.1365-2699.2008.02062.x (2009).
Davies, A. J. & Guinotte, J. M. Global habitat suitability for framework-forming cold-water corals. PloS One 6, e18483, doi: 10.1371/journal.pone.0018483 (2011).
Jaynes, E. T. Information Theory and Statistical Mechanics. Phys Rev 106, 620–630, doi: 10.1103/PhysRev.106.620 (1957).
Phillips, S. J., Anderson, R. P. & Schapire, R. E. Maximum entropy modeling of species geographic distributions. Ecol Model 190, 231–259, doi: 10.1016/j.ecolmodel.2005.03.026 (2006).
Hernandez, P. A. et al. Predicting species distributions in poorly-studied landscapes. Biodivers Conserv 17, 1353–1366, doi: 10.1007/s10531-007-9314-z (2008).
Elith, J. et al. A statistical explanation of MaxEnt for ecologists. Divers Distrib 17, 43–57 (2011).
Fielding, A. H. & Bell, J. F. A review of methods for the assessment of prediction errors in conservation presence/absence models. Environmental conservation 24, 38–49 (1997).
Allouche, O., Tsoar, A. & Kadmon, R. Assessing the accuracy of species distribution models: prevalence, kappa and the true skill statistic (TSS). J Appl Ecol 43, 1223–1232 (2006).
Swets, J. A. Measuring the accuracy of diagnostic systems. Science 240, 1285–1293 (1988).
Landis, J. R. & Koch, G. G. The measurement of observer agreement for categorical data. Biometrics 33, 159–174 (1977).
Manel, S., Williams, H. C. & Ormerod, S. J. Evaluating presence–absence models in ecology: the need to account for prevalence. J Appl Ecol 38, 921–931 (2001).
Cantor, S. B., Sun, C. C., Tortolero-Luna, G., Richards-Kortum, R. & Follen, M. A comparison of C/B ratios from studies using receiver operating characteristic curve analysis. Journal of clinical epidemiology 52, 885–892 (1999).
Liu, C., Berry, P. M., Dawson, T. P. & Pearson, R. G. Selecting thresholds of occurrence in the prediction of species distributions. Ecography 28, 385–393 (2005).
Brown, M. L., Donovan, T. M., Schwenk, W. S. & Theobald, D. M. Predicting impacts of future human population growth and development on occupancy rates of forest-dependent birds. Biol Conserv 170, 311–320 (2014).
Wei, W. Q. et al. Characterization of Statin Dose Response in Electronic Medical Records. Clin Pharmacol Ther 95, 331–338, doi: 10.1038/clpt.2013.202 (2014).
Thomas, N. Hypothesis testing and Bayesian estimation using a sigmoid E-max model applied to sparse dose-response designs. J Biopharm Stat 16, 657–677, doi: 10.1080/10543400600860469 (2006).
Ting, N. Dose finding in drug development. (Springer Science & Business Media, 2006).
Acknowledgements
We are grateful for comments from B. Fagan. Thanks to the team behind the Fourth National Giant Panda Survey (Sichuan). This work was supported by funds from the National Key Programme of Research and Development, Ministry of Science and Technology (2016YFC0503200) to the Z.Y., Z.Z., G.X., Y.X., Q.D., D.Q., the Fourth National Giant Panda Survey (Sichuan) to Z.Y., Z.Z., Q.D., W.Z. and D.Q., and the Reintroduction Program of Captive Giant Pandas to Z.Y. and Q.D.
Author information
Authors and Affiliations
Contributions
J.Q., Z.Y. and Q.D. conceived and designed the experiments; J.Q., Z.Y., Q.D. and K.H. analyzed the data; K.H., Z.Z., X.G., X.Y., B.Y. and D.Q. collected data; J.Q., Z.Y. and Z.Z. wrote the manuscript; W.Z. provided and revised the data of vegetation; All authors reviewed the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Qing, J., Yang, Z., He, K. et al. The minimum area requirements (MAR) for giant panda: an empirical study. Sci Rep 6, 37715 (2016). https://doi.org/10.1038/srep37715
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep37715
- Springer Nature Limited
This article is cited by
-
Spatial models of giant pandas under current and future conditions reveal extinction risks
Nature Ecology & Evolution (2021)
-
Quantifying the evidence for co-benefits between species conservation and climate change mitigation in giant panda habitats
Scientific Reports (2017)