Abstract
We investigated sex-related differences in the prognosis of patients with hypertrophic cardiomyopathy (HCM) using the Korea National Health Insurance Service database. From 2010 to 2016, 9524 patients diagnosed with HCM and had more than 1-year follow-up period were analyzed. The primary endpoint was the composite of cardiovascular death or new-onset heart failure (HF) admission. Propensity score-matching analysis was performed to adjust for different baseline characteristics. With a 4.4-years’ median follow-up interval (range 2.0–6.6 years) and male predominance (77.6%), women with HCM were older (52.6 ± 9.7 vs. 51.4 ± 9.1, p < 0.001), had lower incomes, more comorbidities based on Charlson comorbidity index. Women with HCM had a higher incidence of the primary endpoint than men (incidence rate: 34.15 vs. 22.83 per 1000 person-years, log-rank p < 0.001). Multivariable Cox analysis showed that female sex was a poor prognostic factor for the primary endpoint (HR 1.43, 95% CI 1.24–1.64, p < 0.001). This was mainly driven by a higher incidence of new-onset HF admission (HR 1.55, 95% CI 1.34–1.80). However, there was no difference in the incidence of cardiovascular death between the sexes. This result was concordant in the propensity score-matched cohort. In conclusion, women with HCM have worse prognosis, which was mainly driven by a higher new-onset HF admission.
Similar content being viewed by others
Introduction
Hypertrophic cardiomyopathy (HCM) is a genetic cardiac disease that is transmitted in an autosomal dominant fashion with extreme diversity in clinical presentation and natural history1,2. Despite an autosomal dominant transmission, earlier investigations of the disease frequently reported male predominance in prevalence3,4,5,6,7, suggesting sex differences in clinical expression. Although prognostic differences according to sex in a variety of acquired cardiac diseases including coronary artery disease, atrial fibrillation, valvular heart disease, and aortic diseases have been reported8,9,10,11,12, there have been conflicting results regarding sex differences in the prognosis of HCM6,7,13,14,15. Given the improved prognosis of HCM patients in the contemporary management era, a larger number of HCM patients may be required to draw more solid conclusion. Differences in the baseline characteristics, especially older age in women than men, also made it difficult to draw a definite conclusion3,5,6,7,16. Therefore, this study was designed to investigate sex differences in the prognosis of HCM by using a large database of the Korea National Health Insurance Service (NHIS). In addition, we utilized the propensity score (PS)-matching analysis to balance the differences in the baseline characteristics of both sexes.
Results
Baseline characteristics of the study subjects
A total of 9524 patients were finally included in this study (mean age 51.7 ± 9.3 years); the study population was predominantly male (77.6%, n = 7388). The baseline clinical characteristics of the patients are summarized in Table 1. Generally, Women were one year older than men (52.6 ± 9.7 vs. 51.4 ± 9.1, p < 0.001) and had a higher Charlson comorbidity index (CCI) (2.31 ± 1.98 vs. 1.95 ± 1.89, p < 0.001), but were less likely to have a history of chronic kidney disease, ischemic heart disease (IHD) requiring coronary intervention, and myocardial infarction.
Clinical outcomes in the original cohort
The median follow-up duration was 4.4 (interquartile range [IQR] 2.0–6.6) years or 39,267 patient-years (4.4 [IQR 1.9–6.1] years for women; 4.5 [IQR 2.1–6.7] years for men). During follow-up, the primary endpoint was reached in 956 (10.0%) patients; cardiovascular death in 227 (2.4%) patients, and new-onset heart failure (HF) admission in 804 (8.4%) patients. Seventy-five patients experienced both new-onset HF admission and cardiovascular death events, for whom the first event, i.e., new-onset HF admission, was included in the primary endpoint analysis. All-cause death was observed in 379 patients (4.0%).
According to the Kaplan–Meier survival curves (Fig. 1), women had a higher incidence of new-onset HF admission than men (p < 0.001), whereas the incidences of cardiovascular death in women and men were similar (p = 0.265). Therefore, the significantly higher occurrence of the primary endpoint in women was mainly driven by a greater number of new-onset HF admission (34.15 vs. 22.83 per 1000 person-years for women and men, respectively, p < 0.001) (Table 2). There was no difference between the sexes regarding all-cause death (p = 0.546) (Fig. 1D).
Kaplan–Meier curve of the cumulative incidence of clinical events in the original cohort of patients with hypertrophic cardiomyopathy. (A) The primary endpoint of cardiovascular death or new-onset HF admission; (B) cardiovascular death; (C) new-onset HF admission; (D) all-cause death. CV cardiovascular, HF heart failure.
Univariate Cox analysis revealed significant differences in baseline characteristics between men and women, including sex, age, CCI, income, hypertension, dyslipidemia, chronic kidney disease, IHD requiring coronary intervention, atrial fibrillation, and use of beta-blocker (Supplementary Table 1). Multivariate Cox regression analysis conducted after adjusting for the clinical differences in the univariate Cox analysis showed that the female sex remained a significant poor prognosticator for predicting the primary endpoint in two different multivariable models of the original cohort. Of note, this finding was mainly driven by the significantly higher incidence of new-onset HF admission among women with HCM, and there was no statistical difference in the incidences of cardiovascular death or all-cause death in two different multivariable models (Table 3).
Clinical outcomes in the propensity score-matched cohort
In the original HCM cohort, baseline clinical characteristics were significantly different between women and men (Table 1). Thus, to balance the baseline clinical characteristics of both sexes, we created a PS-matched HCM cohort. After PS-matching, each baseline clinical characteristic was well balanced (Supplementary Table 2).
In the PS-matched cohort, the rate of occurrence of the primary endpoint was also higher among women than among men (32.06 vs. 22.24 per 1,000 person-years, log-rank p < 0.001) (Fig. 2 and Supplementary Fig. 1A). The clinical outcomes of cardiovascular death, new-onset HF admission, and all-cause death were all consistent with those in the original cohort (Supplementary Fig. 1B–D). In the PS-matched cohort, multivariable Cox regression analysis showed that female sex remained a significant poor prognosticator for the primary endpoint (hazard ratio [HR] 1.43, 95% confidence interval [CI] 1.22–1.68; p < 0.001), a finding that was again driven by the higher incidence of new-onset HF admission among women (HR 1.54, 95% CI 1.30–1.82; p < 0.001) (Table 3).
Kaplan–Meier curve of the cumulative incidence of the primary endpoint in the original and propensity score-matched cohort of patients with hypertrophic cardiomyopathy. The rate of occurrence of the primary endpoint, a composite of cardiovascular death or new-onset heart failure, was higher among women than among men, and this result was concordant with that in the propensity score-matched cohort. PS, propensity score.
Sensitivity analysis of patients without hypertension
To eliminate the potential confounding effects of hypertension on increased ventricular wall thickness, we performed a sensitivity analysis for 5568 patients without the International Classification of Disease, Tenth Revision (ICD-10) code of hypertension in the original cohort. Baseline characteristics of the patients without hypertension were basically the same as those of the patients in the original cohort (Supplementary Table 3).
Sensitivity analyses of the numbers of events for each endpoint in this cohort are presented in Supplementary Table 4. Specifically, women showed a significantly higher incidence of the primary endpoint after multivariable Cox regression analysis, which was adjusted for age, CCI, income, and clinical variables including dyslipidemia, IHD requiring coronary intervention, chronic kidney disease, atrial fibrillation, and use of beta-blocker (HR 1.52, 95% CI 1.27–1.82; p < 0.001). This finding was also mainly influenced by the significantly higher incidence of new-onset HF admission (HR 1.66, 95% CI 1.37–2.01; p < 0.001). There was no significant difference in the incidences of cardiovascular death for both sexes (HR 1.31, 95% CI 0.90–1.89; p = 0.154). The incidence of all-cause death for women and men were also similar (p = 0.579).
Discussion
We here investigated the sex-related differences in the prognosis of patients with HCM. To the best of our knowledge, we have included the largest ever number of patients with HCM with the aim of evaluating the sex-related differences in their prognosis. In this study, women with HCM were generally older, had more comorbidities, and lower socioeconomic status, but were less likely to have IHD requiring coronary intervention and a history of myocardial infarction or chronic kidney disease than men with HCM. In the original cohort, the PS-matched cohort, and the patients without hypertension cohort (i.e., the cohort for sensitivity analysis), women with HCM consistently had poorer prognosis for the primary endpoint than men. This outcome was mainly due to a higher incidence of new-onset HF admission among women. However, there was no difference in the incidence of cardiovascular or all-cause death between the sexes.
For the last two decades, several studies were conducted on patients from different groups and of different ethnicities to determine whether women with HCM have poorer prognosis than men with HCM3,6,7,13,15,17,18,19,20. In a recent study by Geske et al., which had a large sample size and the longest follow-up duration so far, women with HCM had a significantly higher incidence of all-cause mortality than both men with HCM and the general population6. However, two other large studies by Olivotto et al. and Rowin et al. did not show any difference in survival rate between the sexes; the mean ages of the patients in the latter two studies were lower than that in the study by Geske et al. (mean age 42 and 46 years vs. 55)3,6,7. Our study’s mean age of the patients was 51, which may explain the insignificant difference in survival rate between the sexes. There are some plausible reasons why there was no sex-related difference in mortality rates. First, we focused on the association of sex with clinical course and survival in HCM. The age of patients in the original cohort was slightly younger, and thus the incidence of cardiovascular or all-cause death was so lower than that of the elderly general population that there may be no statistical difference between the two groups. Second, although the incidence of HF was higher among women, the incidence of HF-related death, one of the main causes of HCM-related death, might have decreased recently7. In a previous study of the HCM population in Korea, the proportion of implantable cardioverter defibrillator (ICD) implanted for primary prevention purposes was similar to that of large HCM cohorts of other countries (2.9% each)7,21. Since the number of patients with HCM in our study was greater than those of other previous studies, we could more actively adjust for the older age of women with HCM or referral bias using several statistical methods. Nevertheless, a well-designed prospective cohort study of patients of different ethnicities will be needed to determine the association of sex with the prognosis of HCM patients.
As reported in previous studies, our data showed a higher incidence of new-onset HF admission among women with HCM than among men7,22. One of the possible reasons for this might be the differences in cardiac chamber sizes between sexes. Investigations conducted using transthoracic echocardiography23 and cardiac magnetic resonance imaging before and after correction for body surface area showed that both women in the healthy population24,25 and HCM mutation carriers20 have smaller left ventricular (LV) end-diastolic dimensions and volumes than men. Nevertheless, there is no sex-related difference in the diagnostic criterion for HCM, i.e., a maximal LV wall thickness of at least 15 mm (or 13 mm in first-degree relatives)26. If the LV wall thickness of women reached a cutoff value enough to elicit a diagnosis of HCM, the LV dimensions and volumes in women with HCM would be relatively smaller than that of men. This may be the cause of the higher peak LV outflow gradient at initial diagnosis recorded in previous studies6,7,22 and higher incidence of new-onset HF admission in the present study. Second, women have a higher prevalence of HF with preserved ejection fraction (HFpEF) than men in the general population due to lower diastolic reserve, more frequent age-associated ventricular-arterial uncoupling, and smaller vessel size27,28. This trend was also observed in the HCM cohort; women with HCM had higher early mitral inflow velocity/early mitral annular velocity ratio and elevated right ventricular systolic pressure, both of which are parameters used for estimating LV filling pressure6,18. The beneficial effect of beta-blocker in patients with HCM was noted in a pre-specified subgroup analysis (Supplementary Fig. 2), which may be mediated by decreased inotropy with a decreased LV outflow pressure gradient and reduced heart rate with increased time for diastolic filling6,29. Third, in the present study, a higher proportion of women had income below the lowest 20% than men. In a previous study, the socioeconomic status affected the incidence and long-term outcomes of patients with HF30. Another international multicenter study also showed that women had higher Gini coefficients, which means more income inequality and can result in worse HF outcomes, even though they had fewer comorbidities than men31. Besides, decreased estrogen levels after menopause can partially explain the possible mechanisms underlying the poor HF outcomes in women with HCM. Specifically, LV mass in female mice with LV hypertrophy has been reported to increase after ovariectomy32. In another animal study, estrogen compounds had a proapoptotic effect and were not protective in the setting of HCM33. Although the lack of echocardiographic and biological data in the NHIS database makes it difficult to demonstrate the mechanisms underlying the poor HF outcome among the women with HCM in the present study, our findings are concordant with those of the previous studies7,22. The HR of the largest sample size in the present study also had a greater magnitude than those of previous studies after adjustment with several statistical methods.
Our study has some limitations. First, since this was a nationwide cohort study conducted using the NHIS database, we could not evaluate the echocardiographic parameters, such as LV peak pressure gradient, or cardiac magnetic resonance imaging results of the patients enrolled. This is the inherent limitation of national claims data. Additionally, we could not analyze the morphological phenotype of HCM or the prevalence of obstructive HCM, either. However, the diagnosis of HCM in Korea is strictly controlled periodically by the NHIS and a medical expert from the Health Insurance Review and Assessment Service (HIRA)34. Further, the ICD-10 and Rare Intractable Disease (RID) code-based diagnosis of HCM was well-matched with a previous echocardiography-based diagnosis of HCM35. Second, a previous study suggested that women with HCM had a smaller LV cavity size than men and that elderly women with HCM had a higher peak LV outflow pressure gradient due to gender-dependent contractile force and different structural remodeling36. Annual trends in number of diagnoses and age at diagnosis are both increasing, which seems to be irrespective of geographical or ethnicity5,37. This phenomenon might be due to increased physician awareness, diagnostic sensitivity or unknown mechanism related with sex-difference according to age. The present study only enrolled patients aged < 65 years, and thus we cannot investigate gender-specific changes in LV systolic function and remodeling pattern with age. However, exclusion of older HCM patients may be reasonable because LV wall thickness tends to increase in older age due to hidden hypertension or unknown causes38. We could focus on the association of sex with prognosis in patients with HCM by enrolling relatively young patients. Third, the prevalence of hypertension, which complicates a strict diagnosis of HCM, looks higher. However, the proportion of patients with hypertension in the present study was similar to that in other previous studies of HCM cohorts6,7. Furthermore, the results in the original cohort were concordant with those in the HCM patients without hypertension of the sensitivity analysis.
In conclusion, women with HCM have a higher incidence of the primary endpoint than men with HCM. This result was mainly driven by a higher new-onset HF admission in women with no difference in all-cause or cardiovascular mortality between women and men.
Methods
Data source and study population
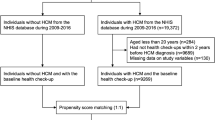
We conducted this nationwide population-based cohort study using the Korean NHIS claims database. The Korean NHIS is a mandatory universal health insurance program managed by the Korean government since 1989 and offers comprehensive medical care to 97% of the Korean population39. The remaining 3% of Koreans with evidence of low income are covered by the Medical Aid Program; information from this program has been incorporated into a single database since 2006. The NHIS database includes detailed information of each registered individual, including data on demographic characteristics, health behavior, diagnoses, prescriptions, surgeries or procedures, and health care utilization (i.e., hospitalization)39. This database has been previously used in many published studies, and its validity as a reliable data source has been established40,41,42. Additionally, the HIRA system provides regular evaluation data, quality control data, and feedback about the whole medical care system in Korea. Through the cooperation of the NHIS and HIRA, a large database of the medical information of all Koreans was established under the strict supervision of the Ministry of Health and Welfare34. This database encompasses information on demographics, medical facility utilization history, diagnoses, prescriptions, and the national health exam results of a given year43. From this database, we selected subjects who were aged ≥ 20 years between January 1, 2010, and December 31, 2016. To minimize the possibility of hypertrophic changes of the myocardium in older people38, 8676 patients aged > 65 years were excluded. We also excluded 364 patients who had less than a year of follow-up and 524 patients who had the ICD-10 code of HF in their record at the time of enrollment. A total of 9524 patients with HCM were finally included in the study (Fig. 3). This study complied with the provisions of the 2013 Declaration of Helsinki. The study was approved by the Institutional Review Board of Seoul National University Hospital (E-1092-058-1009), and the need for informed consent was waived by the Institutional Review Board of Seoul National University Hospital due to the retrospective nature of the study.
Diagnosis of HCM and its validity
HCM was defined as (1) record of at least one admission or outpatient clinic visit with ICD-10 code of I42.1 or I42.2; and (2) registration in the RID program (code of V127). In Korea, HCM falls under the RID category, in which patients are designated as special medical aid beneficiaries with expanding benefit of the NHIS. Since 2006, the Korean government has introduced an initiative covering 90% of all medical expenses claimed by these patients. Therefore, the diagnosis of HCM is strictly determined and monitored via thorough verification using clinical and imaging evidences, and periodical reviews by medical experts and health insurance professionals, according to an act established by the Ministry of Health and Welfare. Furthermore, the definition of HCM according to a diagnostic code was validated by our institution in a previous study, by reviewing medical records, including echocardiography or cardiac magnetic resonance imaging data, and comparing the diagnostic accuracy of the code35.
Covariates
Previously published detailed methods were adopted for this study35,44,45. Age and sex data were retrieved using resident identification numbers. Income level was dichotomized at the lowest 20% and was presented as a categorical variable. The detailed definition of comorbidities, including hypertension, diabetes mellitus, dyslipidemia, IHD (requiring stent insertion), myocardial infarction, peripheral artery disease, atrial fibrillation, ischemic/bleeding stroke, chronic kidney disease / end-stage renal disease is summarized in Supplementary Table 5. CCI was also calculated as previously described46,47. Prescription lists, including renin–angiotensin–aldosterone system blocker, beta-blocker, calcium channel blocker, statin, and antiplatelet agent, were ascertained.
Study outcome and follow-up
The primary endpoint was defined as a composite of cardiovascular death and new-onset HF. The secondary endpoint included the individual components of the primary endpoint and all-cause death. New-onset HF was defined as a newly acquired I50 diagnostic code on admission after the diagnostic code for HCM was established. The study subjects were followed up until the occurrence of each endpoint or until December 31, 2017, whichever came first.
Statistical analysis
Descriptive statistics were presented as means ± standard deviations or medians (IQRs) for continuous variables and numbers (percentages) for categorical variables. For the comparison between groups, the unpaired Student’s t-test was applied for continuous variables, and the χ2 test or Fisher’s exact test was used for categorical variables, as appropriate. The incidence rates of death or new-onset HF events were calculated by dividing the number of detected cases by the follow-up duration, and were presented as a value per 1000 person-years. Kaplan–Meier curves were used to present the cumulative incidence of the primary and secondary endpoints by using a log-rank test for statistical analysis. Multivariable Cox proportional-hazards analysis with two nested models were used to identify the independent risk factors for the primary and secondary endpoints, which were expressed as HRs and corresponding 95% CIs. The variables with a p-value of < 0.10 for the primary endpoint in the univariable Cox analysis were selected and included in the multivariable Cox analysis.
To adjust for the imbalance between the baseline characteristics of men and women, we used two different approaches. First, the multivariable Cox regression analysis was performed while adjusting for the baseline characteristics that were significantly different in the univariable analysis. Second, the PS-matching analysis was adopted to balance the differences in covariates. It was performed with a 3:1 greedy matching technique. We set a caliper for nearest-neighbor matching within the first four to eight digits. For sensitivity analysis, we excluded 3,956 patients with ICD-10 codes indicating hypertension (I10-13, I15) in their records and repeated the statistical analyses.
All statistical analyses were executed using the SAS software, Version 9.4 of SAS System for Windows, Copyright 2021 (SAS Institute Inc., Cary, NC), and R version 3.6.0 software (R Development Core Team, Vienna, Austria). A two-sided p-value of < 0.05 was considered a significant difference.
Data availability
The data is unavailable outside NHIS system.
References
Maron, B. J. et al. American College of Cardiology/European Society of Cardiology clinical expert consensus document on hypertrophic cardiomyopathy. A report of the American College of Cardiology Foundation Task Force on Clinical Expert Consensus Documents and the European Society of Cardiology Committee for Practice Guidelines. J. Am. Coll. Cardiol. 42, 1687–1713. https://doi.org/10.1016/s0735-1097(03)00941-0 (2003).
Maron, B. J. Hypertrophic cardiomyopathy: A systematic review. JAMA 287, 1308–1320. https://doi.org/10.1001/jama.287.10.1308 (2002).
Olivotto, I. et al. Gender-related differences in the clinical presentation and outcome of hypertrophic cardiomyopathy. J. Am. Coll. Cardiol. 46, 480–487. https://doi.org/10.1016/j.jacc.2005.04.043 (2005).
Lorenzini, M. et al. Mortality among referral patients with hypertrophic cardiomyopathy vs the general european population. JAMA Cardiol. https://doi.org/10.1001/jamacardio.2019.4534 (2019).
Moon, I. et al. Trends of the prevalence and incidence of hypertrophic cardiomyopathy in Korea: A nationwide population-based cohort study. PLoS ONE 15, e0227012. https://doi.org/10.1371/journal.pone.0227012 (2020).
Geske, J. B. et al. Women with hypertrophic cardiomyopathy have worse survival. Eur. Heart J. 38, 3434–3440. https://doi.org/10.1093/eurheartj/ehx527 (2017).
Rowin, E. J. et al. Impact of sex on clinical course and survival in the contemporary treatment era for hypertrophic cardiomyopathy. J. Am. Heart Assoc. 8, e012041. https://doi.org/10.1161/JAHA.119.012041 (2019).
Aggarwal, N. R. et al. Sex differences in ischemic heart disease: Advances, obstacles, and next steps. Circ. Cardiovasc. Qual. Outcomes 11, e004437. https://doi.org/10.1161/CIRCOUTCOMES.117.004437 (2018).
Emdin, C. A. et al. Atrial fibrillation as risk factor for cardiovascular disease and death in women compared with men: Systematic review and meta-analysis of cohort studies. BMJ 532, h7013. https://doi.org/10.1136/bmj.h7013 (2016).
Tomai, F. et al. Twenty year follow-up after successful percutaneous balloon mitral valvuloplasty in a large contemporary series of patients with mitral stenosis. Int. J. Cardiol. 177, 881–885. https://doi.org/10.1016/j.ijcard.2014.10.040 (2014).
Vassileva, C. M. et al. Gender differences in long-term survival of Medicare beneficiaries undergoing mitral valve operations. Ann. Thorac. Surg. 96, 1367–1373. https://doi.org/10.1016/j.athoracsur.2013.04.055 (2013).
Group, E. U. C. C. S. et al. Gender in cardiovascular diseases: impact on clinical manifestations, management, and outcomes. Eur Heart J 37, 24–34. https://doi.org/10.1093/eurheartj/ehv598 (2016).
Ho, H. H., Lee, K. L., Lau, C. P. & Tse, H. F. Clinical characteristics of and long-term outcome in Chinese patients with hypertrophic cardiomyopathy. Am. J. Med 116, 19–23. https://doi.org/10.1016/j.amjmed.2003.09.020 (2004).
Maron, M. S. et al. Contemporary natural history and management of nonobstructive hypertrophic cardiomyopathy. J. Am. Coll. Cardiol. 67, 1399–1409. https://doi.org/10.1016/j.jacc.2016.01.023 (2016).
Dimitrow, P. P., Czarnecka, D., Kawecka-Jaszcz, K. & Dubiel, J. S. Sex-based comparison of survival in referred patients with hypertrophic cardiomyopathy. Am. J. Med. 117, 65–66. https://doi.org/10.1016/j.amjmed.2004.02.038 (2004).
Husser, D. et al. Prevalence of clinically apparent hypertrophic cardiomyopathy in Germany—An analysis of over 5 million patients. PLoS ONE 13, e0196612. https://doi.org/10.1371/journal.pone.0196612 (2018).
Wang, Y. et al. Female sex is associated with worse prognosis in patients with hypertrophic cardiomyopathy in China. PLoS ONE 9, e102969. https://doi.org/10.1371/journal.pone.0102969 (2014).
Nijenkamp, L. et al. Sex differences at the time of myectomy in hypertrophic cardiomyopathy. Circ. Heart Fail. 11, e004133. https://doi.org/10.1161/CIRCHEARTFAILURE.117.004133 (2018).
Meghji, Z. et al. Survival differences in women and men after septal myectomy for obstructive hypertrophic cardiomyopathy. JAMA Cardiol. 4, 237–245. https://doi.org/10.1001/jamacardio.2019.0084 (2019).
van Driel, B., Nijenkamp, L., Huurman, R., Michels, M. & van der Velden, J. Sex differences in hypertrophic cardiomyopathy: New insights. Curr. Opin. Cardiol. 34, 254–259. https://doi.org/10.1097/HCO.0000000000000612 (2019).
Choi, Y. J. et al. Validation of the hypertrophic cardiomyopathy risk-sudden cardiac death calculator in Asians. Heart 105, 1892–1897. https://doi.org/10.1136/heartjnl-2019-315160 (2019).
Ommen, S. R. et al. Long-term effects of surgical septal myectomy on survival in patients with obstructive hypertrophic cardiomyopathy. J. Am. Coll. Cardiol. 46, 470–476. https://doi.org/10.1016/j.jacc.2005.02.090 (2005).
Lang, R. M. et al. Recommendations for cardiac chamber quantification by echocardiography in adults: An update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. J. Am. Soc. Echocardiogr. 28, 1–39. https://doi.org/10.1016/j.echo.2014.10.003 (2015).
Alfakih, K. et al. Normal human left and right ventricular dimensions for MRI as assessed by turbo gradient echo and steady-state free precession imaging sequences. J. Magn. Reson. Imaging 17, 323–329. https://doi.org/10.1002/jmri.10262 (2003).
Salton, C. J. et al. Gender differences and normal left ventricular anatomy in an adult population free of hypertension. A cardiovascular magnetic resonance study of the Framingham Heart Study Offspring cohort. J. Am. Coll. Cardiol. 39, 1055–1060. https://doi.org/10.1016/s0735-1097(02)01712-6 (2002).
Authors/Task Force, m et al. ESC Guidelines on diagnosis and management of hypertrophic cardiomyopathy: The Task Force for the Diagnosis and Management of Hypertrophic Cardiomyopathy of the European Society of Cardiology (ESC). Eur. Heart J. 35(2733–2779), 2014. https://doi.org/10.1093/eurheartj/ehu284 (2014).
Beale, A. L. et al. Sex differences in heart failure with preserved ejection fraction pathophysiology: A detailed invasive hemodynamic and echocardiographic analysis. JACC Heart Fail. 7, 239–249. https://doi.org/10.1016/j.jchf.2019.01.004 (2019).
Sheifer, S. E. et al. Sex differences in coronary artery size assessed by intravascular ultrasound. Am. Heart J. 139, 649–653. https://doi.org/10.1016/s0002-8703(00)90043-7 (2000).
Houston, B. A. & Stevens, G. R. Hypertrophic cardiomyopathy: A review. Clin. Med. Insights Cardiol. 8, 53–65. https://doi.org/10.4137/CMC.S15717 (2014).
Hawkins, N. M., Jhund, P. S., McMurray, J. J. & Capewell, S. Heart failure and socioeconomic status: Accumulating evidence of inequality. Eur. J. Heart Fail. 14, 138–146. https://doi.org/10.1093/eurjhf/hfr168 (2012).
Dewan, P. et al. Income inequality and outcomes in heart failure: A global between-country analysis. JACC Heart Fail. 7, 336–346. https://doi.org/10.1016/j.jchf.2018.11.005 (2019).
van Eickels, M. et al. 17beta-estradiol attenuates the development of pressure-overload hypertrophy. Circulation 104, 1419–1423. https://doi.org/10.1161/hc3601.095577 (2001).
Haines, C. D. et al. Estrogenic compounds are not always cardioprotective and can be lethal in males with genetic heart disease. Endocrinology 153, 4470–4479. https://doi.org/10.1210/en.2012-1391 (2012).
Lee, H. et al. Risk of end-stage renal disease in patients with hypertrophic cardiomyopathy: A nationwide population-based cohort study. Sci. Rep. 9, 14565. https://doi.org/10.1038/s41598-019-50993-5 (2019).
Choi, Y. J. et al. Temporal trends of the prevalence and incidence of atrial fibrillation and stroke among Asian patients with hypertrophic cardiomyopathy: A nationwide population-based study. Int. J. Cardiol. 273, 130–135. https://doi.org/10.1016/j.ijcard.2018.08.038 (2018).
Dimitrow, P. P., Czarnecka, D., Kawecka-Jaszcz, K. & Dubiel, J. S. The influence of age on gender-specific differences in the left ventricular cavity size and contractility in patients with hypertrophic cardiomyopathy. Int. J. Cardiol. 88, 11–16. https://doi.org/10.1016/s0167-5273(02)00323-6 (2003) (discussion 16–17).
Canepa, M. et al. Temporal trend of age at diagnosis in hypertrophic cardiomyopathy: An analysis of the international sarcomeric human cardiomyopathy registry. Circ. Heart Fail. 13, e007230. https://doi.org/10.1161/CIRCHEARTFAILURE.120.007230 (2020).
Arbab-Zadeh, A. et al. Effect of aging and physical activity on left ventricular compliance. Circulation 110, 1799–1805. https://doi.org/10.1161/01.CIR.0000142863.71285.74 (2004).
Lee, J., Lee, J. S., Park, S. H., Shin, S. A. & Kim, K. Cohort profile: The National Health Insurance Service-National Sample Cohort (NHIS-NSC), South Korea. Int. J. Epidemiol. 46, e15. https://doi.org/10.1093/ije/dyv319 (2017).
Lee, H. J. et al. Novel oral anticoagulants for primary stroke prevention in hypertrophic cardiomyopathy patients with atrial fibrillation. Stroke 50, 2582–2586. https://doi.org/10.1161/STROKEAHA.119.026048 (2019).
Lee, C. K. et al. Nationwide validation study of diagnostic algorithms for inflammatory bowel disease in Korean National Health Insurance Service database. J. Gastroenterol. Hepatol. 35, 760–768. https://doi.org/10.1111/jgh.14855 (2020).
Hwang, Y. J. et al. Validation of administrative big database for colorectal cancer searched by international classification of disease 10th codes in Korean: A retrospective big-cohort study. J. Cancer Prev. 23, 183–190. https://doi.org/10.15430/JCP.2018.23.4.183 (2018).
Cheol Seong, S. et al. Data resource profile: The National Health Information Database of the National Health Insurance Service in South Korea. Int. J. Epidemiol. 46, 799–800. https://doi.org/10.1093/ije/dyw253 (2017).
Choi, Y. J. et al. Reconsidering the cut-off diastolic blood pressure for predicting cardiovascular events: A nationwide population-based study from Korea. Eur. Heart J. 40, 724–731. https://doi.org/10.1093/eurheartj/ehy801 (2019).
Wu, C. S., Tsai, Y. T. & Tsai, H. J. Antipsychotic drugs and the risk of ventricular arrhythmia and/or sudden cardiac death: A nation-wide case-crossover study. J. Am. Heart Assoc. https://doi.org/10.1161/JAHA.114.001568 (2015).
Sundararajan, V. et al. New ICD-10 version of the Charlson comorbidity index predicted in-hospital mortality. J. Clin. Epidemiol. 57, 1288–1294. https://doi.org/10.1016/j.jclinepi.2004.03.012 (2004).
Charlson, M. E., Pompei, P., Ales, K. L. & MacKenzie, C. R. A new method of classifying prognostic comorbidity in longitudinal studies: Development and validation. J. Chronic Dis. 40, 373–383. https://doi.org/10.1016/0021-9681(87)90171-8 (1987).
Acknowledgements
The authors take responsibility for all aspects of the reliability and freedom from bias of the data presented and their discussed interpretation
Funding
None.
Author information
Authors and Affiliations
Contributions
M.K. conceptualized the work, data curation, and wrote the manuscript. B.K., Y.J.C., H.J.L., and K.D.H. performed data analysis and/or interpretation. H.L., J.B.P., S.P.L., and Y.J.K. reviewed/edited the manuscript. H.K.K. conceptualized, supervised, and reviewed/edited the manuscript. All authors approved the final approval of the manuscript to be published and agreed to be accountable for all aspects of the work by ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Corresponding author
Ethics declarations
Competing interests
HKK reports research grants from Actelion, Handok, Dae-Woong, Ildong, Samjin Pharm, and JW pharmaceutical. The remaining authors have nothing to disclose.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kim, M., Kim, B., Choi, YJ. et al. Sex differences in the prognosis of patients with hypertrophic cardiomyopathy. Sci Rep 11, 4854 (2021). https://doi.org/10.1038/s41598-021-84335-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-021-84335-1
- Springer Nature Limited
This article is cited by
-
Impact of coronary artery revascularization on long-term outcome in hypertrophic cardiomyopathy patients: a nationwide population-based cohort study
Scientific Reports (2023)
-
Comparison of mortality and cause of death between adults with and without hypertrophic cardiomyopathy
Scientific Reports (2022)
-
Sex Differences in Cardiomyopathy
Current Cardiovascular Risk Reports (2022)
-
Sex Differences in Hypertrophic Cardiomyopathy: Interaction With Genetics and Environment
Current Heart Failure Reports (2021)