Abstract
Animal populations are influenced strongly by fluctuations in weather conditions, but long-term fitness costs are rarely explored, especially in short-lived avian species. We evaluated the relative contributions of individual characteristics and environmental conditions to lifetime reproductive success (LRS) of female tree swallows (Tachycineta bicolor) from two populations breeding in contrasting environments and geographies, Saskatchewan and British Columbia, Canada. Female swallows achieved higher LRS by breeding early in the season and producing more fledglings. Other measures of female quality had virtually no influence on LRS. Genetic factors did not predict LRS, as there was no correlation between life-history components for sister pairs nor between mothers and their daughters. Instead, climate variability—indexed by spring pond density (i.e., abundance of wetland basins holding water) during years when females bred—had strong positive effects on female LRS in more arid Saskatchewan but only weak positive effects of moisture conditions were detected in wetter British Columbia. Overall, several life history trait correlates of LRS were similar between populations, but local environmental factors experienced by individuals while breeding produced large differences in LRS. Consequently, variable and extreme environmental conditions associated with changing climate are predicted to influence individual fitness of distinct populations within a species’ range.
Similar content being viewed by others
Introduction
Fluctuations in weather conditions play a major role in dynamics of animal populations by affecting survival and reproduction1, and these effects may be particularly pronounced in years with extreme weather events2. For instance, severe heat, cold or drought can reduce survival3,4 and result in reproductive failure5. The frequency and severity of extremes in weather conditions are predicted to increase in the future6, with heavy precipitation, warmer temperatures, extreme floods and drought projected for the northern hemisphere7. Studies assessing the effects of weather variability generally focus on short-term effects on survival and reproduction, whereas the effects on the long-term fitness of animals remain underexplored8. One approach to evaluating how weather variability and climate extremes influence avian fitness is to determine their individual and combined effects on the total numbers of offspring produced by individuals over their lifetime that contribute to future generations, i.e., lifetime reproductive success (LRS)9,10. A better understanding of the major drivers affecting LRS is urgently needed to predict population responses to a rapidly changing environment.
Decades of research across a variety of taxa have demonstrated large variation among individuals in reproductive performance, with relatively few individuals producing most of the surviving offspring9,10. Local weather conditions vary geographically, influencing the production of offspring by some individuals11, and emphasizing the importance of evaluating populations across contrasting environments. Alternatively, the success of individuals at producing offspring that contribute to future generations compared to those producing few to no offspring in their lifetime may also reflect underlying differences in individual quality12,13,14. For example, some individuals in a population may be consistently successful at producing young, even in the face of contrasting environmental conditions15. Indeed, some studies have demonstrated that individual traits reflecting quality, such as body size16, timing of breeding17,18, age of first reproduction19, and longevity20,21 are positively related to LRS. The relative contribution of these individual characteristics to variation in LRS is nonetheless often weak, suggesting variation in LRS may instead be driven by stochastic processes22,23.
Ultimately, there are still unresolved debates on whether the traits of individuals or environmental stochasticity are the most important drivers of variation in LRS14,23,24. Part of the uncertainty may arise from the context of events. For example, in southern fulmars (Fulmarus glacialoides), when conditions were favorable, environmental stochasticity influenced LRS, but during poor conditions, heterogeneity in individual traits explained greater variation in LRS25. Additionally, the majority of studies focus on long-lived species, whereas short-lived species that breed only once or twice in their lifetime may be less able to contend with fluctuating or extreme environmental conditions. Lastly, measurements of LRS, at least in avian studies, commonly assume that the “number of fledglings” (survive to fledging age) is a reliable approximation of fitness instead of more accurately determining the “number of recruits” (survive to breeding age), which may obscure the interpretation of LRS drivers16,20.
Here, we use confirmatory path analysis26 to simultaneously evaluate the relative importance of individual characteristics (i.e., age of first breeding, body condition, timing of breeding, and number of breeding attempts [longevity]) and environmental factors (i.e., number of wetland basins containing water [pond abundance], soil moisture conditions, and local cold weather severity) for LRS of adult female tree swallows (Tachycineta bicolor). While path analysis has been performed in relatively long-lived species20,27, this approach has not been used to disentangle whether individual traits or environmental factors are the dominant drivers of LRS in short-lived species. Apparent LRS, the number of recruited offspring to the study site, was calculated for large samples of known-age adult female tree swallows based on > 15 year longitudinal data sets from two geographically distinct populations in western Canada (Saskatchewan [SK] and British Columbia [BC]; > 1200 km apart) with distinct local environmental conditions (Table S1). The SK site in the prairies is more arid and experiences continental climate extremes driving fluctuations in pond abundances during drought-deluge cycles28, whereas the BC site has higher moisture and experiences unpredictable, severe cold and wet weather events, particularly early in the breeding season29. Under projected climate change scenarios, the frequency of these extreme events is predicted to increase6, warranting greater understanding of their effects. Tree swallows feed primarily on aerial insects that emerge from aquatic environments during warmer weather30 but are negatively affected by cold snaps31. We hypothesized that in years with more abundant ponds or warmer and wetter soil moisture conditions present when females bred would reflect increased food supply32 or higher quality food, resulting in the production of larger broods33. We also examined potential heritability of life-history components by assessing correlations in lifetime number of breeding attempts, eggs laid, fledglings produced, and timing of breeding for sister pairs and between mothers and their daughters.
Results
General patterns of reproduction and apparent LRS of known-age adult females
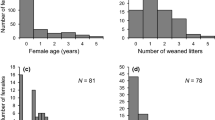
In SK, most female tree swallows were first detected breeding at either 1 (48%) or 2 (37%) years old; few were ≥ 3 (15%). In BC, most females were detected breeding for the first time as yearlings (90%) while the remaining females (10%) bred at ≥ 2 years old. In SK, 60% of female tree swallows made a single breeding attempt in their lifetime, while 21% bred twice, 10% bred three times, and fewer than 8% bred ≥ 4 times. The number of breeding attempts by females in BC was similar: 63%, 20%, 11% and 5% corresponding to 1, 2, 3, and ≥ 4 attempts, respectively. In both study areas, > 50% of adult females laid 5–7 eggs (Fig. 1a), and 50% and 35%, SK and BC respectively, fledged between 4–7 nestlings in their lifetime (Fig. 1b), reflecting the average for a single breeding attempt made by most females. The number of adult females failing to produce any fledged offspring differed by site: in SK, < 5% of females failed to fledge offspring, whereas 21% of females in BC produced no fledglings (Fig. 1b).
Apparent LRS varied widely among adult females at both study sites (Fig. 1c); the mean (± SE) number of recruits produced by females was 0.42 ± 0.04 at SK (range: 0 to 5) and 0.26 ± 0.03 at BC (range: 0 to 4). Low LRS of females suggests that the majority, 70% in SK and 81% in BC, produced no locally recruited offspring, whereas the remaining 30% of females in SK produced 1.41 ± 0.06 recruits and 19% in BC produced 1.40 ± 0.07 recruits.
Saskatchewan LRS path model
The original hypothesized path model for SK tree swallows had a poor fit to the data (Fisher’s C28 = 54.49, p = 0.002, n = 496). Two significant pathways were added (Relative Ponds → Total Eggs and Relative Ponds → LRS), and five non-significant pathways were removed (Total Breeding Attempts → LRS; Relative Initiation Date → Total Breeding Attempts; Relative Condition → Total Breeding Attempts; Age of First Breeding → Total Breeding Attempts; and Relative Ponds → Relative Initiation Date). Overall, the final path model had a good fit to the data (Fisher’s C24 = 23.39, p = 0.50, Fig. 2a).
Path diagram summarizing direct and indirect effects of explanatory variables on lifetime reproductive success of known-age adult female tree swallows breeding in (a) Saskatchewan and (b) British Columbia. Line thickness indicates the relative strength of the path. See “Materials and methods” for description of calculation of explanatory variables. Green represents individual characteristics of females, whereas blue represents environmental factors. Age of first breeding for females in Saskatchewan was 1 year old for 48%, 2 years old for 37%, and ≥ 3 years old for 15%, whereas in British Columbia, 90% of females bred at 1 year old and the other 10% bred at ≥ 2 years old.
The final path model revealed that the lifetime number of fledglings (Total Fledglings) had the strongest direct positive effect on apparent LRS (Fig. 2a). Lifetime number of eggs laid (Total Eggs) and Total Breeding Attempts (adult female survival) also had strong positive effects on LRS indirectly through lifetime fledgling production (Total Fledglings; Table 1, Fig. 2a). Lifetime clutch initiation date of females (Relative Initiation Date) had a direct negative effect on apparent LRS (Table 1, Fig. 2a) and was further strengthened by an indirect negative effect of lifetime clutch initiation date (Relative Initiation Date) on lifetime egg production (Total Eggs) (Table 1). Lastly, the abundance of ponds present in the landscape in the year(s) females bred (Relative Ponds) had a direct positive effect on LRS (Table 1, Fig. 2a). This effect was strengthened by two indirect pathways from Relative Ponds → Total Eggs and Relative Ponds → Total Fledglings, all leading to higher apparent LRS (Table 1). Additionally, the negative effects of breeding later (Relative Initiation Date) on LRS (− 0.01) and egg production (Total Eggs) (− 0.004) appeared to be dampened by pond density (Relative Ponds) because later breeding females tended to lay more eggs and produce more recruits when ponds were more abundant.
Reproductive age (Age of First Breeding) of SK females had a relatively weak positive effect on LRS, but only indirectly via lifetime egg production (0.012), suggesting that delayed breeding had little influence on LRS. A strong negative path between lifetime clutch initiation date (Relative Initiation Date) and body condition (Relative Condition) was detected, indicating that late-nesting females were light-weight for their body size (Fig. 2a); however, female body condition had no effect on apparent LRS.
Total number of breeding attempts (Total Breeding Attempts) contributed indirectly to higher LRS (see above), and not directly as hypothesized. None of the individual trait variables we initially hypothesized as important drivers of lifetime fitness (i.e., Relative Condition, Relative Initiation Date and Age of First Breeding) had a significant relationship with overall number of breeding attempts (Total Breeding Attempts) and were not retained in the final LRS model (Fig. 2a).
British Columbia LRS path model
The original hypothesized path model for BC also had poor fit to the data (Fisher’s C38 = 87.27, p < 0.0001, n = 466). Three significant paths (Average PDSI (soil moisture conditions) → Relative Initiation Date; Relative Condition → Total Fledglings; and Total Breeding Attempts → Total Fledglings) were added to the model. Several non-significant paths (PDSI → Total Fledglings; Age of First Breeding → Total Breeding Attempts; Relative Initiation Date → Total Breeding Attempts; Relative Condition → Total Breeding Attempts; and Total Breeding Attempts → LRS) were removed (all p > 0.50). Overall, the final path model had a good fit to the data (Fisher’s C40 = 39.38, p = 0.50, Fig. 2b).
Lifetime production of fledglings (Total Fledglings) was the strongest direct predictor of female LRS (Table 1). As expected, lifetime number of eggs laid (Total Eggs) and adult female survival (Total Breeding Attempts) were also important contributors to LRS by increasing the total number of fledglings produced by females (Table 1, Fig. 2b). Lifetime clutch initiation date (Relative Initiation Date) had a direct negative effect on LRS (Fig. 2b), and the effect was strengthened by an indirect pathway through lifetime egg production (Total Eggs), suggesting that late breeding females had lower LRS (Table 1, Fig. 2b). Age (Age of First Breeding) and body condition (Relative Condition) of females only weakly influenced LRS through indirect paths. Females that delayed breeding until two or three years old tended to lay more eggs (Fig. 2b), but this had no effect on LRS (Table 1). Females in relatively higher body condition over their lifetime generally produced more fledglings and more recruits (Fig. 2b), although this effect was relatively weak (Table 1).
The number of breeding attempts made by females did not increase apparent LRS directly, as hypothesized (Fig. 2b), although a weak direct path from Total Breeding Attempts → Total Fledglings was detected (Fig. 2b), strengthening the effect of number of breeding attempts on overall LRS (see above). Again, none of the trait variables hypothesized to influence a female’s number of breeding attempts (Total Breeding Attempts) were retained in the final model (Fig. 2b).
Several indirect pathways including soil moisture conditions (Average PDSI) yielded a weak positive effect on LRS (Table 1), suggesting that wetter soil conditions in BC tended to increase LRS (Fig. 2b). Soil moisture had a positive effect on LRS via earlier clutch initiation date (Relative Initiation Date), but this was countered by early breeding females being more likely to experience cold weather (Relative Severity), which had a negative effect on fledgling production (Total Fledglings) (Fig. 2b).
Heritability of life-history traits: sister and mother-daughter pairs
Across sites, there was no significant correlation in total fledglings, eggs or breeding attempts or relative initiation date between mothers and their daughters (p-values > 0.48) nor with sister pairs (p-values > 0.29) (Table 2).
Discussion
Our results for LRS in two geographically distinct populations using hundreds of known-age female tree swallows monitored for > 15 years were consistent with four important generalizations: (1) most females are short-lived and breed only once in their lifetime; (2) early annual onset of breeding is key to their long-term reproductive success; (3) age of first breeding, individual quality and genetics were not strong predictors of traits related to LRS; and (4) environmental conditions experienced in the year(s) females bred generated large variation in LRS. These results have important conservation implications as climate change can alter local environmental conditions on the breeding grounds that can directly affect population demographics in passerines with short lifespans. Overall, consistent with other animal models, the dominant drivers of tree swallow LRS are timing of initiating breeding, longevity and environmental conditions that influence production of offspring17,21,34. Additionally, higher lifetime production of fledglings was the strongest correlate of female apparent LRS, lending support for the common use and assumption of fledging success as a proxy for LRS in short-lived passerines35.
Life-history theory predicts that individuals making greater investment in current reproduction should suffer lower subsequent survival or future reproduction36. This fitness trade-off has been observed in LRS studies in some species (e.g., eastern kingbirds (Tyrannus tyrannus)37, song sparrows (Melospiza melodia)38, and northern elephant seals (Mirounga angustirostris)21). In contrast, our results suggest that survival and reproduction may positively covary in tree swallows. We speculated that this pattern is indicative of individual quality where some individuals are better able to invest in self-maintenance and still achieve higher LRS39. Interestingly, none of the individual trait characteristics we measured, aside from timing of breeding, were related to longevity or LRS. Although stochasticity can generate a positive correlation between survival and reproduction22, our results are consistent with previous experimental research in tree swallows failing to detect direct costs of reproduction40. For example, females raising experimentally enlarged broods fledged more offspring with no apparent cost to their return rates or future breeding success41,42. Instead, Shutler et al.42 hypothesized that fitness in tree swallows is constrained more by timing of initiating breeding, since females breeding later in the season produced fewer recruits in their lifetime. This phenomenon was confirmed in our study systems, each with much larger samples of known-age birds.
In seasonal environments, one of the most prominent determinants of reproductive success is timing of initiating breeding43. Females that breed late in the season generally lay fewer eggs in their lifetime and have lower apparent LRS18,20. That we detected a stronger effect of later-season breeding on LRS than on the lifetime number of eggs supports previous studies demonstrating a benefit of early-season breeding on post-fledging survival20,44. The relative number of recruits decreased with lifetime clutch initiation date, suggesting that females who generally breed early in the season produce the majority of recruits, while those that are relatively late-season breeders produce only a few locally recruited offspring (Fig. S1). While we cannot rule out the possibly that offspring hatched later in the season permanently dispersed outside the study sites, previous research in diverse animal species has shown that later hatched (or born) offspring are generally lower quality and have lower apparent return rates than offspring produced early in the season45,46.
Declines in reproductive success associated with breeding later in the season are hypothesized to be the consequence of differences in individual quality or environmental conditions between early- and late-season breeders47. Females that bred early in the season had higher body condition compared to later-season breeders, consistent with the individual quality hypothesis, yet condition had virtually no effect on LRS. While we recognize that we were unable to control for potential effects on the body condition of females such as time of day that females were captured or weather48, links between adult female body condition and annual reproductive success are seldom detected in tree swallows29,49,50. Additionally, female body condition was less influential for recruitment or LRS in other species, such as the great tit (Parus major)51, common pochard (Aythya ferina)52, and northern wheatear (Oenanthe oenanthe)53. Instead, our results are consistent with experimental studies showing that the date in the season when a female breeds is causally related to the number of fledging or recruiting offspring29,54. Seasonally declining food supply is one mechanism hypothesized to underlie differences in the number of fledglings or recruits produced by early- and late-season breeding females47,50,55. While we lack long-term data on aerial insect abundance, Harriman et al.50 previously showed that insect biomass is generally lower at the BC site, which may explain the higher number of females producing no recruits and the stronger negative effect of timing of breeding on LRS at BC. Additionally, abundant ponds reflecting higher food supply32 likely dampened the negative effect of late season breeding on LRS at SK.
Similar with other studies that have attempted to simultanously disentangle effects of individual quality versus environmental factors for offspring recruitment or LRS20,52, we found that body condition and reproductive age of females had relatively weak effects on LRS. Although Winkler et al.18 recently reported that clutch initiation date was correlated for mothers and their daughters that began breeding for the first time as yearlings, we found no significant correlation among mothers and their daughters, even when we considered only those breeding as yearlings (p = 0.44, n = 50). The lack of correlation between sisters and mother-daughter pairs in clutch initiation date and other life-history components further indicates that genetic variation has little influence on LRS, although we recognize that our sample sizes are small, and some sisters may not share a genetic father due to extra-pair paternity. In other species, age of first breeding had positive effects on LRS, such as goshawks (Accipiter gentilis)19 and blue-footed boobies (Sula nebouxii)56; however, benefits of this strategy to LRS may be limited to species with a long lifespan and early costs of reproduction, or when delaying breeding to an older age does not result in lower LRS57, as in tree swallows. Overall, our conclusions are similar to work in other species reporting little effect of female traits on LRS20,58 and a lack of heritability of LRS27,59. This may reflect the larger effect of environmental conditions relative to genetic variation on lifetime fitness of individuals in short-lived species60,61.
Environmental factors have been reported as important drivers of variation in LRS among individuals, but these studies are generally limited to a single population15,62 or populations within close proximity58. Our results show that environmental drivers causing variation in LRS can vary across a species’ range and may explain geographic differences in population changes. In SK, females that bred when ponds were abundant were able to make greater investments in reproduction, suggesting that aquatic insect food supply or quality can offset reproductive costs32,63. Although we were unable to directly assess links between pond abundance and survival because our measure of Relative Ponds was standardized to be independent of lifetime breeding attempts, higher apparent survival of females in years following higher pond abundance has been previously reported64. Thus, ponds are critically important for terrestrial species that rely on aquatic food resources, highlighting the urgent need to protect prairie wetland basins from further loss and degradation.
At the BC site where wetter environmental conditions contrast those in more arid SK, we observed that wetter soil moisture conditions did not lead to higher LRS, consistent with Harriman’s33 report that PDSI was not an important predictor of juvenile apparent survival. PDSI had a weak positive effect on lifetime relative initiation date; however, early-season breeding females were more likely to encounter cold weather while raising nestlings, as reported for an Ontario population of tree swallows65. Although unpredictable, cold weather events can have devastating effects on nestling survival5, our measure of cold severity did not fully account for the low total number of fledglings produced by some females at BC. Weegman et al.11 also found that low fledgling success at this BC site was unrelated to local weather. Previous work in tree swallows suggests that the production of fledglings is an important driver of population growth11,66, and is the strongest determinant of LRS; therefore, mechanisms underlying lower fledging success at the BC site warrant further investigation.
Short-lived species generally breed only once in their lifetime62, which is consistent with our finding of female tree swallows making only a single breeding attempt regardless of their age of first breeding (Fig. 3). While environmental factors, such as pond abundance and winter conditions can influence female survival64, we examined whether female quality influenced number of breeding attempts (Supplemental Analysis 1). Similar to LRS, we found no strong effects of female body condition or age of first breeding on number of breeding attempts, but females that were relatively earlier season breeders made more breeding attempts (Table S1 and Supporting Supplementary Text). Nevertheless, given the importance of survival to LRS, we encourage further studies to conduct formal survival analyses incorporating measurements of individual quality and environmental factors across the annual cycle. We also recognize the possibility that we may have detected only a single breeding attempt for most females because they dispersed, especially at the BC site where fledgling success was low11, or bred elsewhere or in natural cavities before acquiring a nest box at the SK site given nest box occupancy was high67. Breeding site dispersal for adult tree swallows is low68, but fewer yearling females were detected breeding at the SK site compared to BC. We cannot fully account for permanent dispersal of juveniles69 or the fact that female tree swallows could breed as yearlings elsewhere, leading to underestimates of the number of females making more than one breeding attempt. Therefore, while our results for tree swallows are consistent with the general patterns of LRS for short-lived species9,10, we report ‘apparent’ estimates of LRS and urge other studies to do so as well.
Our results suggest that climate change projections of increased frequency of extreme climate events may generate large, and perhaps distinct, effects on apparent LRS of species across their range. In the prairies where current warmer, wetter conditions associated with climate change appear to be benefiting some species70, climate scenarios are forecasting more frequent and severe drought conditions and widespread drying of ponds71,72, which our results suggest could negatively affect the fitness of terrestrial species. Such effects will not only occur via habitat loss, but also through the loss of aquatic food resources for a variety of species. In the interior of BC, predictions of increased precipitation73 combined with unpredictable cold weather events experienced by early-season breeders may create poor breeding conditions. Precipitation reduces the availability of aerial insect food resources, with negative consequences on offspring quality and population growth74. Although the effects we detected were weak, increased frequency of inclement weather has the potential to exacerbate these effects in the future. In contrast, increased precipitation leading to wetter conditions and earlier breeding combined with warmer spring temperatures have the potential to create more favorable conditions during breeding.
By evaluating the effects of individual characteristics and weather fluctuations and climate extremes using a lifetime perspective in a short-lived avian species, we discovered that most individuals bred only once and environmental conditions can generate large variation in lifetime fitness. Given that changes in climate are affecting local environments producing wetter or drier, and possibly warmer conditions, we predict that geographic differences in LRS may be a strong determinant of population change caused by climate variation.
Materials and methods
Study species
Tree swallows are migratory, aerial insectivores that breed throughout the central and northern parts of North America30. In most of its range, this species produces a single brood per season with clutch sizes ranging from 1–9 eggs, although clutches containing 4–7 eggs are most common30. Extra-pair paternity in this species is high, with up to 85% of broods containing at least one extra-pair offspring75, but egg dumping by other females is rare76. The average lifespan of tree swallows is approximately 2.7 years30. Female apparent survival probability is approximately 50%64, while juvenile apparent survival probability is 6% at the SK site and 5% in BC11. Female tree swallows show high breeding site fidelity68 and do not generally disperse after a season with poor breeding success77. Female tree swallows intensively compete for nest sites78, and because nest sites may be limited, females in their second-year (SY) of life (i.e., their first breeding season) are sometimes competitively excluded from breeding in their first season79.
Study areas and general field methods
Tree swallows were studied in Canada at a Saskatchewan (SK) site, 1990–2017, and at a site in British Columbia (BC), 2001–2017. The SK site is located in the prairie pothole region, at the St. Denis National Research Area, 40 km east of Saskatoon (52oN, 106oW); the area contains > 100 wetland basins of varying permanency (when flooded, basins contain “ponds”), as well as grassland, cultivated land, and groves of aspen (Populus tremuloides)80. The BC site, located within 30 km of Prince George (53° N, 123° W), is in the sub-boreal forest and has cooler temperatures, and higher spring and summer precipitation (Table S1). The BC site consists of hayfields intermixed with stands of deciduous trees and few small permanent wetlands81. At both sites, wooden nest boxes are mounted on metal or wooden posts spaced approximately 20–30 m apart. Further details regarding these sites are described in Shutler and Clark77, Dawson et al.82, Bitton and Dawson83, and Harriman et al.50.
Reproductive data from breeding tree swallows were collected from May to August. Nest boxes were visited regularly to record clutch initiation date (i.e., day the first egg was laid, where 1 January = 1), clutch size, hatching date, and number of nestlings hatching. Most adult tree swallows were captured within a few days after the last egg in a nest hatched; in a few instances, females were captured prior to the hatching of eggs64. Captured birds were identified as female or male by the presence of a brood patch or cloacal protuberance, respectively, and unbanded adults were banded with an individually numbered aluminum leg band. Because of consistent capture and banding efforts for tree swallows at our sites over the study period, nearly every breeding adult and all nestling tree swallows were banded, and recapture probabilities were high11. Upon capture, most adults were weighed using a spring balance (± 0.5 g) and wing length was measured using a ruler (± 0.5 mm). Nestling were banded either when they were 12 or 16 days old. Nests were visited 20 days after hatching to record the number of nestlings that successfully fledged.
Path model hypotheses and data analysis
Data on annual reproductive success for known-age adult female tree swallows breeding in SK (1990–2015) and BC (2001–2015) were summarized over all breeding attempts made by each female. We included females that bred up to 2015 and detected recruits up to 2017. Analyses were based on 496 (SK) and 466 (BC) known-age adult female tree swallows with complete information for all explanatory variables (see below). Exact age was known with certainty for females banded as nestlings or birds identified as first-year breeders (i.e., SY) according to their dorsal plumage color84. Females involved in studies that could influence survival and hence LRS (e.g., those deployed with geolocators85) were excluded. Data for males were not analyzed; unlike females, estimates of apparent LRS would be unreliable given frequent extra-pair paternity75.
We included nine explanatory variables for the breeding lifespan of each female that we hypothesized would influence LRS (Table 3). We expected that the age of a female’s first breeding attempt, ‘Age of First Breeding’, would lead to higher LRS for females that begin breeding as yearlings because they would make more breeding attempts than females that delayed breeding until two years of age or older86 (Table 3). We also expected that females making more breeding attempts in their lifetime, ‘Total Breeding Attempts’, would produce more recruits directly20 or indirectly because they laid more eggs, ‘Total Eggs’, and produced more fledgling, ‘Total Fledglings’, in their lifetime37 (Table 3). Minimum clutch size or fledged offspring were used in rare instances when exact values were unknown (n = 10 [SK] and n = 3 [BC]). In a few cases (n = 9 for SK, n = 6 for BC), we excluded a breeding attempt if that attempt resulted in no fledged offspring (e.g., human-caused nest failure). Few females re-nested after brood failure (n = 5 at each site) and we only counted such cases as a single breeding attempt, but we used the clutch initiation date and clutch size from the first breeding attempt and number of fledged offspring from the re-nesting attempt, following Murphy37, when averaging or summing values over all breeding attempts.
Female tree swallows that breed earlier in the season are higher quality individuals18 so we anticipated that females breeding relatively early in the season throughout their lifetime would have higher LRS because they lay more eggs42 and raise nestlings when insect food resources are more plentiful50 (Table 3). This measure of ‘Relative Initiation Date’ is the average standardized clutch initiation date for all breeding attempts, calculated by subtracting the annual 5% clutch initiation date for all nests from each individual female’s initiation date following Clark et al.89. Initiation date was standardized separately for after-second-year (ASY) and SY females because SY females generally breed later than ASY females90.
Early season breeding females are heavier29, better able to acquire insect food resources87,88 and can invest in self-maintenance while breeding91. Therefore, we expected females that were relatively early season breeders during their life to be in higher body condition, ‘Relative Condition’, when captured while rearing nestlings and make more lifetime breeding attempts (Table 3). Relative Condition was the residual from a linear model that included body mass as the response variable and female age category (SY or ASY), wing length, year, and the number of days from when females initiated their clutch to being captured as explanatory variables. Residuals were averaged over all breeding attempts, except for 27 (SK) and 41 (BC) females where mass or wing measurements were missing and average values were calculated with the available data. Time of capture may influence the body mass of females48. Although females at the Saskatchewan site were mainly captured in the morning, exact capture times are unknown, so we explored how capture time influenced body mass for 451 of 466 females with data for capture time at the British Columbia site. While the body mass of known-age adult females was positively related to time of capture (standardized estimate (SE), β = 0.24 ± 0.05), body condition residuals for these females computed with and without capture time were nearly identical (linear regression estimate (SE), β = 1.00 ± 0.01).
Cold, inclement weather can lower insect availability31 and result in nestling mortality5; therefore, we hypothesized that the lifetime relative number of cold days experienced by females when their nestlings were 4–12 days old, ‘Relative Severity’, would have negative effects on lifetime fledgling production (Table 3). Relative Severity was calculated only for BC because 67% of females experienced severe cold weather during breeding, whereas 65% of females in SK experienced no cold snaps in their lifetime. Cold days were calculated as the number of cold snaps, i.e., days when maximum temperature was below 18.5 °C, identified by Winkler et al.31 as the critical temperature for insect flight, that spanned between two to seven days, multiplied by the number of days offspring were in the nest. We did not use cold snaps consisting of only one day because a daily fluctuation below 18.5 °C during a period of favorable weather is unlikely to affect nestling survival. Additionally, since longer cold snaps may be more severe than shorter ones, those spanning three or more days were weighted by 2, whereas cold snaps of 2 days were weighted by 1. Relative severity is the number of cold days, summed over all of a female’s breeding attempts, and because females making more breeding attempts have higher cumulative number of cold days, we centered to the mean cold severity value for the number of breeding attempts made (i.e., 1, 2, 3, and ≥ 4). Daily maximum temperature data were retrieved from Environment and Climate Change Canada (https://climate.weather.gc.ca/index_e.html).
Aerial insects, the main food of swallows30, can influence the timing of clutch initiation92 and investment in reproduction88, and because insect abundance is greater in years with abundant ponds32 we expected lifetime abundance of ponds, ‘Relative Ponds’, experienced by females to influence fledgling production (Table 3). Relative ponds is the number of wetland basins containing water in May at the SK site (Fig. S2), summed over all of a female’s breeding attempts. Although higher May pond counts influence survival of adult females at Saskatchewan64, we separated total breeding attempts from pond abundance by mean-centering the cumulative pond value for the number of breeding attempts made (i.e., 1, 2, 3, and ≥ 4) because females making more total breeding attempts have a higher cumulative total pond count. Given that wetlands are generally permanent in BC, we used the average Palmer Drought Severity Index (PDSI) for June and July, which has been shown previously to provide a suitable measure of local moisture conditions at our study sites33. For instance, at the SK site, PDSI is positively correlated both with number of ponds (this study: r = 0.59, p = 0.001, n = 26) and their depth33 in May. Site-specific PDSI values were obtained from Agriculture and Agri-Food Canada.
We performed confirmatory path analysis26 using the package PIECEWISESEM93 in R (version 3.6.1)94 to test causal hypotheses explaining how individual traits and environmental factors directly and indirectly affect apparent LRS of female tree swallows. Extreme values of some variables were truncated89 and log-transformed when they improved the fit of data to linear models testing claims of conditional independence between explanatory variables (see below). We simultaneously performed analyses using SAS (PROC CALIS)95 and the standardized residuals and goodness of fit tests additionally confirmed that final models provided a very good fit to the data.
Our hypothesized path model was evaluated using Shipley’s goodness of fit test, the Fisher’s C statistic, provided by the PIECEWISESEM package93. Path models that fail the goodness of fit test have a significant p-value, indicating that there are associations between variables (i.e., paths) missing from the model26. These missing paths are identified by performing simultaneous tests of conditional independence between variables not included in the path model and significant p-values indicate missing paths26,93, which were then added to the path model. Non-significant paths were removed from the model until only significant pathways remained58. A final model with a non-significant p-value indicated good fit to the data26. Standardized path coefficients for direct effects between variables were extracted from the final path model; total effects of variables were calculated by multiplying standardized path coefficients for each indirect path, and then summing all indirect and direct pathways20.
For our path analyses, Total Fledglings and apparent LRS (= total recruits) reflect offspring raised in the nest of a female because 28% of females at SK and 21% of females at BC were involved in experiments where eggs or nestlings were swapped, added or removed from nests50,77. Our main results and conclusions were unchanged when we limited analyses to fledglings and recruits genetically related to the female (n = 449 [SK] and n = 448 [BC]).
We tested for correlations in lifetime number of breeding attempts, eggs laid, fledglings produced, and relative initiation date between 26 sister (n = 16 SK, n = 10 BC) and 140 mother-daughter (n = 71 SK, n = 69 BC) pairings. Variables were standardized (mean = 0, standard deviation = 1) separately by site, and data were then pooled prior to analysis using Spearman’s correlations. We were unable to test whether apparent LRS or age of first breeding were positively related between sisters and mother and their daughters because there was limited variation in these variables due to the majority of females producing no recruits or breeding at 1 or 2 years old.
Animal handling and ethics
All handling and research on tree swallows were approved by institutional animal care committees, the University of Saskatchewan and University of Northern BC, and carried out in accordance with the Canadian Council on Animal Care guidelines.
Data availability
Data available upon reasonable request from L.L.B. and R.G.C.
References
White, T. C. R. The role of food, weather and climate in limiting the abundance of animals. Biol. Rev. 83, 227–248. https://doi.org/10.1111/j.1469-185X.2008.00041.x (2008).
Moreno, J. & Møller, A. P. Extreme climatic events in relation to global change and their impact on life histories. Curr. Zool. 57, 375–389. https://doi.org/10.1093/czoolo/57.3.375 (2011).
Briedis, M., Hahn, S. & Adamík, P. Cold spell en route delays spring arrival and decreases apparent survival in a long-distance migratory songbird. BMC Ecol. 17, 11. https://doi.org/10.1186/s12898-017-0121-4 (2017).
Welbergen, J. A., Klose, S. M., Markus, N. & Eby, P. Climate change and the effects of temperature extremes on Australian flying-foxes. Proc. R. Soc. B Biol. Sci. 275, 419–425. https://doi.org/10.1098/rspb.2007.1385 (2008).
Griebel, I. & Dawson, R. D. Predictors of nestling survival during harsh weather events in an aerial insectivore, the tree swallow (Tachycineta bicolor). Can. J. Zool. 97, 81–90. https://doi.org/10.1139/cjz-2018-0070 (2019).
Intergovernmental Pannel on Climate Change (IPCC). Global warming of 1.5°C. An IPCC Special Report on the impacts of global warming of 1.5°C above pre-industrial levels and related global greenhouse gas emission pathways, in the context of strengthening the global response to the threat of climate change. (eds. Masson-Delmotte, V. et al.) (World Meteorological Organization, 2018).
Intergovernmental Pannel on Climate Change (IPCC). Climate change 2007: Impacts, adaptation and vulnerability. Contribution of working group II to the fourth assessment report of the Intergovernmental Panel on Climate Change. (eds. Parry, M. L., O. F. Canziani, J. P. Palutikof, P. J. van der Linden and Hanson, C.E.) 976 (Cambridge University Press, Cambridge, 2007).
Bailey, L. D. & van de Pol, M. Tackling extremes: challenges for ecological and evolutionary research on extreme climatic events. J. Anim. Ecol. 85, 85–96. https://doi.org/10.1111/1365-2656.12451 (2016).
Clutton-Brock, T. H. Reproductive Success: Studies of Individual Variation in Contrasting Breeding Systems (The University of Chicago Press, Chicago, 1988).
Newton, I. Lifetime Reproductive Success in Birds (Academic Press, Cambridge, 1989).
Weegman, M. D., Arnold, T. W., Dawson, R. D., Winkler, D. W. & Clark, R. G. Integrated population models reveal local weather conditions are the key drivers of population dynamics in an aerial insectivore. Oecologia 185, 119–130. https://doi.org/10.1007/s00442-017-3890-8 (2017).
Cam, E., Link, W. A., Cooch, E. G., Monnat, J.-Y. & Danchin, E. Individual covariation in life-history traits: seeing the trees despite the forest. Am. Nat. 159, 96–105. https://doi.org/10.1086/324126 (2002).
Chambert, T., Rotella, J. J., Higgs, M. D. & Garrott, R. A. Individual heterogeneity in reproductive rates and cost of reproduction in a long-lived vertebrate. Ecol. Evol. 3, 2047–2060. https://doi.org/10.1002/ece3.615 (2013).
Cam, E. et al. Looking for a needle in a haystack: inference about individual fitness components in a heterogeneous population. Oikos 122, 739–753. https://doi.org/10.1111/j.1600-0706.2012.20532.x (2013).
Jenouvrier, S., Péron, C. & Weimerskirch, H. Extreme climate events and individual heterogeneity shape life-history traits and population dynamics. Ecol. Monogr. 85, 605–624 (2015).
Jensen, H. et al. Lifetime reproductive success in relation to morphology in the house sparrow Passer domesticus. J. Anim. Ecol. 73, 599–611. https://doi.org/10.1111/j.0021-8790.2004.00837.x (2004).
Saino, N. et al. Longevity and lifetime reproductive success of barn swallow offspring are predicted by their hatching date and phenotypic quality. J. Anim. Ecol. 81, 1004–1012. https://doi.org/10.1111/j.1365-2656.2012.01989.x (2012).
Winkler, D. W. et al. Full lifetime perspectives on the costs and benefits of lay date variation in tree swallows. Ecology 101, e03109. https://doi.org/10.1002/ecy.3109 (2020).
Krüger, O. Age at first breeding and fitness in goshawk Accipiter gentilis. J. Anim. Ecol. 74, 266–273. https://doi.org/10.1111/j.1365-2656.2005.00920.x (2005).
Blums, P. & Clark, R. G. Correlates of lifetime reproductive success in three species of European ducks. Oecologia 140, 61–67. https://doi.org/10.1007/s00442-004-1573-8 (2004).
Le Boeuf, B., Condit, R. & Reiter, J. Lifetime reproductive success of northern elephant seals (Mirounga angustirostris). Can. J. Zool. 97, 1203–1217. https://doi.org/10.1139/cjz-2019-0104 (2019).
Tuljapurkar, S., Steiner, U. K. & Orzack, S. H. Dynamic heterogeneity in life histories. Ecol. Lett. 12, 93–106. https://doi.org/10.1111/j.1461-0248.2008.01262.x (2009).
Snyder, R. E. & Ellner, S. P. Pluck or luck: does trait variation or chance drive variation in lifetime reproductive success?. Am. Nat. 191, E90–E107. https://doi.org/10.1086/696125 (2018).
Cam, E., Aubry, L. M. & Authier, M. The conundrum of heterogeneities in life history studies. Trends Ecol. Evol. 31, 872–886. https://doi.org/10.1016/j.tree.2016.08.002 (2016).
Jenouvrier, S. et al. When the going gets tough, the tough get going: effect of extreme climate on an Antarctic seabird’s life history. bioRxiv https://doi.org/10.1101/791855 (2019).
Shipley, B. Confirmatory path analysis in a generalized multilevel context. Ecology 90, 363–368. https://doi.org/10.1890/08-1034.1 (2009).
Grant, P. R. & Grant, B. R. Non-random fitness variation in two populations of Darwin’s finches. Proc. R. Soc. B. Biol. Sci. 267, 131–138. https://doi.org/10.1098/rspb.2000.0977 (2000).
Johnson, W. C., Boettcher, S. E., Poiani, K. A. & Guntenspergen, G. Influence of weather extremes on the water levels of glaciated prairie wetlands. Wetlands 24, 385–398. https://doi.org/10.1672/0277-5212(2004)024[0385:IOWEOT]2.0.CO;2 (2004).
Dawson, R. D. Timing of breeding and environmental factors as determinants of reproductive performance of tree swallows. Can. J. Zool. 86, 843–850. https://doi.org/10.1139/Z08-065 (2008).
Winkler, D. W. et al. Tree swallow (Tachycineta bicolor). In The Birds of North America Online (ed. Poole, A.) (Cornell Lab of Ornithology, Cornell, 2011).
Winkler, D. W., Luo, M. K. & Rakhimberdiev, E. Temperature effects on food supply and chick mortality in tree swallows (Tachycineta bicolor). Oecologia 173, 129–138. https://doi.org/10.1007/s00442-013-2605-z (2013).
Fast, M. Climate Variability, Timing of Nesting and Breeding Success of Tree Swallows (Tachycineta bicolor) (University of Saskatchewan, Saskatchewan, 2007).
Harriman, V. B. Seasonal Variation in Quality and Survival of Nestlings Tree Swallows (Tachycineta bicolor): Tests of Alternative Hypotheses (University of Saskatchewan, Saskatchewan, 2014).
Martínez-Padilla, J., Vergara, P. & Fargallo, J. A. Increased lifetime reproductive success of first-hatched siblings in common kestrels Falco tinnunculus. Ibis 159, 803–811. https://doi.org/10.1111/ibi.12494 (2017).
Weatherhead, P. J. & Dufour, K. W. Fledging success as an index of recruitment in red-winged blackbirds. Auk 117, 627–633. https://doi.org/10.1093/auk/117.3.627 (2000).
Williams, G. C. Natural selection, the costs of reproduction, and a refinement of Lack’s principle. Am. Nat. 100, 687–690 (1966).
Murphy, M. T. Lifetime reproductive success of female eastern kingbirds (Tyrannus tyrannus): influence of lifespan, nest predation, and body size. Auk 124, 1010–1022. https://doi.org/10.1093/auk/124.3.1010 (2007).
Tarwater, C. E. & Arcese, P. Young females pay higher costs of reproduction in a short-lived bird. Behav. Ecol. Sociobiol. 71, 84. https://doi.org/10.1007/s00265-017-2309-1 (2017).
Vedder, O. & Bouwhuis, S. Heterogeneity in individual quality in birds: overall patterns and insights from a study on common terns. Oikos 127, 719–727. https://doi.org/10.1111/oik.04273 (2018).
Murphy, M. T., Armbrecth, B., Vlamis, E. & Pierce, A. Is reproduction by tree swallows cost free?. Auk 117, 902–912. https://doi.org/10.1093/auk/117.4.902 (2000).
Wheelwright, N. T., Leary, J. & Fitzgerald, C. The costs of reproduction in tree swallows (Tachycineta bicolor). Can. J. Zool. 69, 2540–2547. https://doi.org/10.1139/z91-358 (1991).
Shutler, D., Clark, R. G., Fehr, C. & Diamond, A. W. Time and recruitment costs as currencies in manipulation studies on the costs of reproduction. Ecology 87, 2938–2946. https://doi.org/10.1890/0012-9658(2006)87[2938:TARCAC]2.0.CO;2 (2006).
Verhulst, S. & Nilsson, J. Å. The timing of birds’ breeding seasons: a review of experiments that manipulated timing of breeding. Philos. Trans. R. Soc. B. 363, 399–410. https://doi.org/10.1098/rstb.2007.2146 (2008).
Grüebler, M. U. & Naef-Daenzer, B. Fitness consequences of timing of breeding in birds: date effects in the course of a reproductive episode. J. Avian Biol. 41, 282–291. https://doi.org/10.1111/j.1600-048X.2009.04865.x (2010).
Plard, F. et al. The influence of birth date via body mass on individual fitness in a long-lived mammal. Ecology 96, 1516–1528. https://doi.org/10.1890/14-0106.1 (2015).
Raja-aho, S., Eeva, T., Suorsa, P., Valkama, J. & Lehikoinen, E. Juvenile barn swallows Hirundo rustica L. from late broods start autumn migration younger, fuel less effectively and show lower return rates than juveniles from early broods. Ibis 159, 892–901. https://doi.org/10.1111/ibi.12492 (2018).
Svensson, E. Natural selection on avian breeding time: causality, fecundity-dependent, and fecundity-independent selection. Evolution 51, 1276–1283. https://doi.org/10.1111/j.1558-5646.1997.tb03974.x (1997).
Rioux Paquette, S., Pelletier, F., Garant, D. & Bélisle, M. Severe recent decrease of adult body mass in a declining insectivorous bird population. Proc. R. Soc. B. Biol. Sci. 281, 20140649. https://doi.org/10.1098/rspb.2014.0649 (2014).
Winkler, D. W. & Allen, P. E. The seasonal decline in tree swallow clutch size: physiological constraint or strategic adjustment?. Ecology 77, 922–932 (1996).
Harriman, V. B., Dawson, R. D., Bortolotti, L. E. & Clark, R. G. Seasonal patterns in reproductive success of temperate-breeding birds: experimental tests of the date and quality hypotheses. Ecol. Evol. 7, 2122–2132. https://doi.org/10.1002/ece3.2815 (2017).
Verhulst, S., van Balen, J. H. & Tinbergen, J. M. Seasonal decline in reproductive success of the great tit: variation in time or quality?. Ecology 76, 2392–2403 (1995).
Blums, P., Clark, R. G. & Mednis, A. Patterns of reproductive effort and success in birds: path analyses of long-term data from European ducks. J. Anim. Ecol. 71, 280–295. https://doi.org/10.1046/j.1365-2656.2002.00598.x (2002).
Pärt, T., Knape, J., Low, M., Öberg, M. & Arlt, D. Disentangling the effects of date, individual, and territory quality on the seasonal decline in fitness. Ecology 98, 2102–2110. https://doi.org/10.1002/ecy.1891 (2017).
Brinkhof, M. W. G. & Cavé, A. J. Food supply and seasonal variation in breeding success: an experiment in the European coot. Proc. R. Soc. B Biol. Sci. 264, 291–296. https://doi.org/10.1098/rspb.1997.0041 (1997).
Rossmanith, E., Höntsch, K., Blaum, N. & Jeltsch, F. Reproductive success and nestling diet in the lesser spotted woodpecker (Picoides minor): the early bird gets the caterpillar. J. Ornithol. 148, 323–332. https://doi.org/10.1007/s10336-007-0134-4 (2007).
Kim, S. Y., Velando, A., Torres, R. & Drummond, H. Effects of recruiting age on senescence, lifespan and lifetime reproductive success in a long-lived seabird. Oecologia 166, 615–626. https://doi.org/10.1007/s00442-011-1914-3 (2011).
Mourocq, E. et al. Life span and reproductive cost explain interspecific variation in the optimal onset of reproduction. Evolution 70, 296–313. https://doi.org/10.1111/evo.12853 (2016).
Hoset, K. S., Villers, A., Wistbacka, R. & Selonen, V. Pulsed food resources, but not forest cover, determine lifetime reproductive success in a forest-dwelling rodent. J. Anim. Ecol. 86, 1235–1245. https://doi.org/10.1111/1365-2656.12715 (2017).
Teplitsky, C., Mills, J. A., Yarrall, J. W. & Merilä, J. Heritability of fitness components in a wild bird population. Evolution 63, 716–726. https://doi.org/10.1111/j.1558-5646.2008.00581.x (2009).
McCleery, R. H. et al. Components of variance underlying fitness in a natural population of the great tit Parus major. Am. Nat. 164, E62–E72. https://doi.org/10.1086/422660 (2004).
Salles, O. C. et al. Strong habitat and weak genetic effects shape the lifetime reproductive success in a wild clownfish population. Ecol. Lett. 23, 265–273. https://doi.org/10.1111/ele.13428 (2020).
McCleery, R. H. & Perrins, C. M. Lifetime reproductive success of the great tit, Parus major in Reproductive success 136–153 (The University of Chicago Press, Chicago, 1988).
Twining, C. W., Shipley, J. R. & Winkler, D. W. Aquatic insects rich in omega-3 fatty acids drive breeding success in a widespread bird. Ecol. Lett. 21, 1812–1820. https://doi.org/10.1111/ele.13156 (2018).
Clark, R. G. et al. Geographic variation and environmental correlates of apparent survival rates in adult tree swallows Tachycineta bicolor. J. Avian Biol. 49, 012514. https://doi.org/10.1111/jav.01659 (2018).
Cox, A. R., Robertson, R. J., Rendell, W. B. & Bonier, F. Population decline in tree swallows (Tachycineta bicolor) linked to climate change and inclement weather on the breeding ground. Oecologia 192, 713–722. https://doi.org/10.1007/s00442-020-04618-8 (2020).
Cox, A. R., Robertson, R. J., Fedy, B. C., Rendell, W. B. & Bonier, F. Demographic drivers of local population decline in tree swallows (Tachycineta bicolor) in Ontario, Canada. Condor 120, 842–851. https://doi.org/10.1650/CONDOR-18-42.1 (2018).
Shutler, D. et al. Spatiotemporal patterns in nest box occupancy by tree swallows across North America. Avian Conserv. Ecol. https://doi.org/10.5751/ACE-00517-070103 (2012).
Winkler, D. W. et al. Breeding dispersal and philopatry in the tree swallow. Condor 106, 768–776. https://doi.org/10.1093/condor/106.4.768 (2004).
Lambrechts, M. M. et al. Will estimates of lifetime recruitment of breeding offspring on small-scale study plots help us to quantify processes underlying adaptation?. Oikos 86, 147–151. https://doi.org/10.2307/3546579 (1999).
Mantyka-Pringle, C. et al. Antagonistic, synergistic and direct effects of land use and climate on Prairie wetland ecosystems: ghosts of the past or present?. Divers. Distrib. 25, 1924–1940. https://doi.org/10.1111/ddi.12990 (2019).
Johnson, W. C. et al. Prairie wetland complexes as landscape functional units in a changing climate. Bioscience 60, 128–140. https://doi.org/10.1525/bio.2010.60.2.7 (2010).
Zhao, Q., Silverman, E., Fleming, K. & Boomer, G. S. Forecasting waterfowl population dynamics under climate change—does the spatial variation of density dependence and environmental effects matter?. Biol. Conserv. 194, 80–88. https://doi.org/10.1016/j.biocon.2015.12.006 (2016).
British Columbia Ministry of Environment. Indicators of climate change for British Columbia update. (Province of British Columbia, 2016).
Cox, A. R., Robertson, R. J., Lendvai, Á. Z., Everitt, K. & Bonier, F. Rainy springs linked to poor nestling growth in a declining avian aerial insectivore (Tachycineta bicolor). Proc. R. Soc. B Biol. Sci. 286, 20190018. https://doi.org/10.1098/rspb.2019.0018 (2019).
O’Brien, E. L. & Dawson, R. D. Context-dependent genetic benefits of extra-pair mate choice in a socially monogamous passerine. Behav. Ecol. Sociobiol. 61, 775–782. https://doi.org/10.1007/s00265-006-0308-8 (2007).
Whittingham, L. A. & Dunn, P. O. Female responses to intraspecific brood parasitism in the tree swallow. Condor 103, 166–170. https://doi.org/10.1093/condor/103.1.166 (2001).
Shutler, D. & Clark, R. G. Causes and consequences of tree swallow (Tachycineta bicolor) dispersal in Saskatchewan. Auk 120, 619–631. https://doi.org/10.1093/auk/120.3.619 (2003).
Leffelaar, D. & Robertson, R. J. Nest usurpation and female competition for breeding opportunities by tree swallows. Wilson Bull. 97, 221–224 (1985).
Stutchbury, B. J. & Robertson, R. J. Floating populations of female tree swallows. Auk 102, 651–654 (1985).
Clark, R. G. & Shutler, D. Avian habitat selection: pattern from process in nest-site use by ducks?. Ecology 80, 272–287. https://doi.org/10.1890/0012-9658(1999)080[0272:AHSPFP]2.0.CO;2 (1999).
O’Brien, E. L. & Dawson, R. D. Perceived risk of ectoparasitism reduces primary reproductive investment in tree swallows Tachycineta bicolor. J. Avian Biol. 36, 269–275. https://doi.org/10.1111/j.0908-8857.2005.03562.x (2005).
Dawson, R. D., Lawrie, C. C. & O’Brien, E. L. The importance of microclimate variation in determining size, growth and survival of avian offspring: experimental evidence from a cavity nesting passerine. Oecologia 144, 499–507. https://doi.org/10.1007/s00442-005-0075-7 (2005).
Bitton, P.-P. & Dawson, R. D. Age-related differences in plumage characteristics of male tree swallows Tachycineta bicolor: hue and brightness signal different aspects of individual quality. J. Avian Biol. 39, 446–452. https://doi.org/10.1111/j.0908-8857.2008.04283.x (2008).
Hussell, D. J. T. Age and plumage color in female tree swallows. J. F. Ornithol. 54, 312–318 (1983).
Gómez, J. et al. Effects of geolocators on reproductive performance and annual return rates of a migratory songbird. J. Ornithol. 155, 37–44. https://doi.org/10.1007/s10336-013-0984-x (2014).
Gustafsson, L. & Pärt, T. Acceleration of senescence in the collared flycatcher Ficedula albicollis by reproductive costs. Nature 347, 279–281. https://doi.org/10.1038/347279a0 (1990).
Nooker, J. K., Dunn, P. O. & Whittingham, L. A. Effects of food abundance, weather, and female condition on reproduction in tree swallows (Tachycineta bicolor). Auk 122, 1225–1238. https://doi.org/10.1093/auk/122.4.1225 (2005).
Ardia, D. R., Wasson, M. F. & Winkler, D. W. Individual quality and food availability determine yolk and egg mass and egg composition in tree swallows Tachycineta bicolor. J. Avian Biol. 37, 252–259. https://doi.org/10.1111/l.2006.0908.8857.03624.x (2006).
Clark, R. G., Pöysä, H., Runko, P. & Paasivaara, A. Spring phenology and timing of breeding in short-distance migrant birds: phenotypic responses and offspring recruitment patterns in common goldeneyes. J. Avian Biol. 45, 457–465. https://doi.org/10.1111/jav.00290 (2014).
Robertson, R. J. & Rendell, W. B. A long-term study of reproductive performance in tree swallows: the influence of age and senescence on output. J. Anim. Ecol. 70, 1014–1031. https://doi.org/10.1046/j.0021-8790.2001.00555.x (2001).
Ardia, D. R. Individual quality mediates trade-offs between reproductive effort and immune function in tree swallows. J. Anim. Ecol. 74, 517–524. https://doi.org/10.1111/j.1365-2656.2005.00950.c (2005).
Dunn, P. O., Winkler, D. W., Whittingham, L. A., Hannon, S. J. & Robertson, R. J. A test of the mismatch hypothesis: how is timing of reproduction related to food abundance in an aerial insectivore?. Ecology 92, 450–461. https://doi.org/10.1890/10-0478.1 (2011).
Lefcheck, J. S. PiecewiseSEM: piecewise structural equation modelling in R for ecology, evolution, and systematics. Methods Ecol. Evol. 7, 573–579. https://doi.org/10.1111/2041-210X.12512 (2016).
R Core Team. R: A Language and Environment for Statistical Computing (R Foundation for Statistical Computing, Vienna, 2019).
SAS Institute Incorporated. SAS (Data Analysis Software System), Version 9.4 (SAS Institute Incorporated, 2016).
Acknowledgements
We thank all the volunteer and student field assistants who collected data used in this paper, and we are grateful for the landowners allowing us to work on their property. This work was supported by the Natural Sciences and Engineering Research Council of Canada (NSERC) through grants to R.G.C, C.A.M, and R.R.D and Mathematics of Information Technology and Complex Systems (MITACS) through an Accelerate fellowship to L.L.B. Additional funding was provided by the Environment and Climate Change Canada (R.G.C), and University of Northern British Columbia, Canada Foundation for Innovation, and the British Columbia Knowledge Development Fund (R.D.D).
Author information
Authors and Affiliations
Contributions
R.G.C., L.L.B., and R.D.D. conceived the study, L.L.B., R.D.D., R.G.C. and C.A.M. conducted field work, L.L.B. analyzed the data and wrote the manuscript, and L.L.B., R.D.D., C.A.M., and R.G.C. revised the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Berzins, L.L., Dawson, R.D., Morrissey, C.A. et al. The relative contribution of individual quality and changing climate as drivers of lifetime reproductive success in a short-lived avian species. Sci Rep 10, 19766 (2020). https://doi.org/10.1038/s41598-020-75557-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-75557-w
- Springer Nature Limited