Abstract
SQUAMOSA promoter-binding protein (SBP)-box genes encode a family of plant-specific transcription factors that play roles in plant growth and development. The characteristics of SBP-box genes in rice (Oryza sativa) and Arabidopsis have been reported, but their potential roles in wheat (Triticum aestivum) are not fully understood. In this study, 48 SBP-box genes (TaSBPs) were identified; they were located in all wheat chromosomes except for 4B and 4D. Six TaSBPs were identified as tandem duplication genes that formed three tandem duplication pairs, while 22 were segmentally duplicated genes that formed 16 segmental duplication pairs. Subcellular localization prediction showed TaSBPs were located in nucleus. Among the 48 TaSBPs, 24 were predicted to be putative targets of TamiR156. Phylogenetic analysis showed that TaSBPs, AtSBPs, and OsSBPs that shared similar functions were clustered into the same subgroups. The phylogenetic relationships between the TaSBPs were supported by the identification of highly conserved motifs and gene structures. Four types of cis-elements––transcription-related, development-related, hormone-related, and abiotic stress-related elements––were found in the TaSBP promoters. Expression profiles indicated most TaSBPs participate in flower development and abiotic stress responses. This study establishes a foundation for further investigation of TaSBP genes and provides novel insights into their biological functions.
Similar content being viewed by others
Introduction
The SQUAMOSA promoter-binding protein (SBP)-box gene family belongs to a group of plant-specific genes that encode zinc (Zn) finger proteins. SBP-box genes were first discovered in Antirrhinum majus based on the ability of their protein products to bind to the promoter region of SQUAMOSA, a floral meristem identity gene1. Since then, most SBP-box genes have been identified in different plant species and their functions have been extensively investigated2. SBP-box transcription factors (TFs) are characterized by a highly conserved SBP domain, which contains approximately 75–79 amino acid residues and consists of two Zn finger structures along with a highly conserved nuclear localization signal region (NLS)2.It has been shown that the SBP domain of SBP-box TFs is necessary for binding to a palindromic GTAC core motif3.
SBP-box genes are known to participate in many physiological and molecular processes, including plant architecture, grain quality and yield, flowering, floral induction, and so on4,5,6,7. In rice, OsSPL13 can increase grain yield by regulating the cell size in the grain hull4, OsSPL14 is related to plant architecture and substantially enhances grain yield8, OsSPL16 promotes grain quality and yield9. In Arabidopsis, SPL3 is known to be highly expressed in vegetative and inflorescence apices, floral meristems, leaves, and floral primordials7; SPL3, SPL4, and SPL5 are dramatically up-regulated in response to long-day floral induction10; SPL3/4/5 induce flowering by binding to AP1, LFY, and FUL promoters11. In wheat, TaSPL20 and TaSPL21 govern yield-related traits in hexaploid wheat12 and TaSPL8 modulates leaf angle through auxin and brassinosteroid signaling13, while ectopic expression of TaSPL16 in Arabidopsis delays the emergence of vegetative leaves, increases organ size, and affects yield-related traits14. These studies have indicated that SBP-box genes function in regulation of plant development and growth.
SBP-box genes are also among the conserved plant TFs that are targeted by microRNAs (miRNAs), especially miR156/157 family members15. For example, 10 AtSBP genes are predicted or verified to be targeted by miR156 in Arabidopsis8. In rapeseed (Brassica napus), 44 BnSBPs were predicted to be targeted by miR15616. In rice, there are 11 SBP-box genes that are targets of OsmiR156, and tissue-specific interactions have been revealed between OsmiR156 and OsSBP genes17. However, there are few reports on whether miRNA regulation is conserved in wheat SBP-box genes with miRNA-binding sites.
To date, a large number of SBP-box genes have been identified in different plants: for example, there are 16 in Arabidopsis, 18 in rice18, 29 in maize19, and 19 in grapevines20. Wheat is one of the most important food crops worldwide. Compared with other plant species, the identification and functional analysis of the SBP-box gene family in wheat is not so advanced. In this study, we conducted genome-wide identification of SBP-box genes in wheat and performed a phylogenetic analysis and classified the genes into subgroups to explore the evolution of the SBP-box gene family. The exon–intron structure, the conserved motifs, and expression patterns were also analyzed. This study establishes a foundation for further analysis of SBP-box genes in wheat and other plants species.
Results
Identification of SBP-box genes in wheat
To identify the SBP genes in wheat, we performed a Hidden Markov Model (HMM) search and 48 non-redundant SBP genes were identified in the wheat genome (Supplementary Table S1). The number of hexaploid wheat SBP (TaSBP) genes in wheat (48) was much higher than those in rice (18), maize (31), and Arabidopsis (16)18,19. The 48 TaSBP genes were named TaSBP1A to TaSBP19D according to their distribution on chromosomes and genomic homology. All chromosomes contained at least one TaSBP gene, except for chromosomes 4B and 4D (Fig. 1). As shown in Fig. 1 and Supplementary Table S2, 22 segmentally duplicated genes were identified; these formed 16 segmental duplication pairs. Meanwhile, three tandem duplication pairs were derived from chromosomal tandem duplication. The 48 genes––with the exception of TaSBP11A, TaSBP11B, and TaSBP11D––were verified by expressed sequence tags (ESTs) deposited in the National Center for Biotechnology Information (NCBI) database, and 36 TaSBP genes constituted 12 sets, with every set including three homologous genes in the A, B, and D sub-genomes, respectively.
The physical features of the TaSBP genes were predicted. The protein length varied from 192 (TaSBP2A) to 1124 (TaSBP16D) amino acids; the isoelectric point varied from 5.73 (TaSBP6A) to 9.87 (TaSBP2D); and the molecular weight varied from 20.117 kDa (TaSBP2B) to 123.141 kDa (TaSBP16D). Detailed information is presented in Supplementary Table S1.
Subcellular localization of TaSBP proteins
The results of protein subcellular localization showed that all TaSBP proteins were located in the nucleus (Supplementary Table S1). To determine if wheat TaSBPs localize to nucleus, we cloned three TaSBPs from Chinese Spring and assessed the subcellular localization of the encoded TaSBPs by transient expression assays in wheat protoplasts, using translational fusions to GFP. As shown in Fig. 2, all three proteins (TaSBP4B, TaSBP9B, and TaSBP10A) expressed TaSBP-GFP fusion proteins in transformed wheat protoplasts, TaSBP4B, TaSBP9B, and TaSBP10A localized on the nucleus.
Subcellular localization of three TaSBPs. The selected TaSBP genes were cloned from Chinese Spring and used to construct CaMV35S::TaSBPs–GFP vectors in which GFP was fused at the C-terminus. The merged pictures include the green fluorescence channel and the chloroplast autofluorescence channel. Bar = 20 μm.
Multiple alignment and phylogenetic analysis of TaSBPs
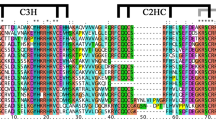
All the TaSBP proteins were aligned using ClustalW. As shown in Fig. 3, each SBP domain contained a complete SBP domain with two Zn finger motifs and one nuclear localization signal region (NLS). The first Zn finger motif was a CCCH type motif, and the second was a CCHC type motif.
To further evaluate the phylogenetic relationships of TaSBPs and other plant SBPs, we selected 263 SBP sequences from ten species according to previous studies16,18,19,21,22, including six monocot species (48 Triticum aestivum, 18 Oryza sativa, 29 Zea mays, 17 Sorghum bicolor, 18 Setaria italica, and 16 Brachypodium distachyon) and four dicot species (16 Arabidopsis thaliana, 28 Brassica rapa, 59 Brassica napus, and 15 Solanum tuberosum), and constructed a phylogenetic tree based on the full-length protein sequences alignment. According to the phylogenetic analysis (Fig. 4), SBPs from these ten plant species could be classified into night subgroups. The largest subgroup (I) contained 48 SBP members. The smallest subgroup (VIII) contained 14 members. Except for subgroup II and VIII, the other subgroups contained at least three TaSBPs. As shown in Fig. 4, subgroup II and VIII only contained dicot SBPs, while subgroups IV and VI only had monocot SBP members. Based on the phylogenetic analysis, all TaSBP members were classified into seven subgroups (Fig. 5A).
Phylogenetic relationship of SBPs. An un-rooted phylogenic tree was constructed in MEGA 7 on the basis of multiple alignment of full-sequences from six monocot species (T. aestivum, O. sativa, Z. mays, S. bicolor, S. italica, and B. distachyon) and four dicot species (A. thaliana, B. napus, B. rapa, and S. tuberosum).
Phylogenetic relationships, conserved motifs, and gene structures of TaSBPs. (A) Wheat TaSBPs were classified into seven groups according to bootstrap values and the phylogenetic analysis of wheat and other plant species; (B) ten conserved motifs were identified in protein sequences of TaSBPs; (C) Gene structures. Exons and introns were indicated by boxes and lines respectively.
Conserved motifs, gene structure, and sites targeted by miR156
The SBP domain forms the core of SBP transcription factors and binds to the promoter of their downstream genes. In total, 10 conserved motifs were identified and designated motif 1 to 10 (Fig. 5B). Among them, motifs 1, 2, and 4 were in the basic region and the hinge region of the SBP domain. Motifs 3, 6, 7, and 10 were only found in subgroup V; motif 5 was only present in subgroups VI and VIII; and motif 9 was only found in subgroup VIII. The structure of the TaSBP genes was also examined to elucidate the gene function (Fig. 5C). The number of exons ranged from 2 to 11; subgroups I, II, III, IV, V, and VII only contained 2–4 exons, while subgroups VI and VIII had more than 10 exons. The TaSBP genes in the same subgroup shared similar gene structures.
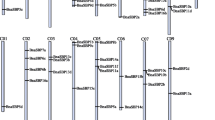
To identify the miR156-mediated post-transcriptional regulation of TaSBP genes, we searched the coding sequences (CDSs) and 3′-untranslated region (UTR) sequences of all TaSBPs for miR156-binding sites. The results showed that 24 TaSBPs (half of the TaSBPs) had miR156-binding sites (sequences that were complementary to the mature TamiR156 sequences), with 19 in the CDSs and 5 in the 3′-UTR regions (Fig. 6).
Cis-acting elements in the promoters of TaSBP genes
Cis-acting elements in gene promoters are crucial regions involved in transcription factor binding for the initiation of transcription. To further explore the possible biological functions of TaSBP genes, the 2-kb upstream promoter regions of all TaSBP genes were used to predict cis-acting elements using the PlantCARE database. Four types of cis-acting elements––transcription-related, development-related, hormone-related, and abiotic stress-related elements––were identified (Fig. 7). Transcription-related cis-elements–including the TATA-box and CAAT-box––were found in all the TaSBP genes. Development-related cis-elements included meristem-specific regulatory elements (CCGTCC-box and CAT-box). Hormone-related cis-elements included the methyl jasmonate (MeJA)-responsive element (CGTC) and the abscisic acid (ABA)-responsive element (ABRE; ACGTG). Abiotic stress-related cis-elements included the drought response element (MYB-binding site [MBS]), low-temperature response element (LTR; CCGAAA), and anoxic specific inducibility element GC-motif (A/CGCCGCGCA). The findings indicated that the phylogenetically similar genes shared identical cis-elements. For example, group IV members TaSBP4A, TaSBP4B, and TaSBP4D had the similar proportion of cis-elements, group VIII members TaSBP1A, TaSBP1B, and TaSBP1D harbored the similar proportion of cis-elements.
Analysis of the expression patterns of the TaSBP genes
To obtain the temporal and spatial expression patterns of TaSBP genes, the expression profiles were analyzed using high-throughput data from previous research18. As shown in Fig. 8, 95.83% (46/48) of TaSBP genes were detected in at least one tissue. Further, it can be seen that 89.58% were highly expressed in the inflorescence, especially when two nodes or internodes were visible and when the stem reached its maximum length.
Expression profiles of TaSBP genes in different tissues and under different abiotic stresses based on transcriptome data. The 15 tissues were as follows: the root when the cotyledon emerged (a), three leaves were visible (b), and the stem reached its maximum length (c); the stem when two nodes or internodes were visible (d), half of the flowers were open (e), and elongation had begun (f); the leaf when the main shoot and axillary shoots were visible (with three nodes) (g), the cotyledon emerged (h), and the whole plant grain had formed (i); the inflorescence when the flowers opened (j), two nodes or internodes were visible (k), and the stem reached its maximum length (l); the grain when 30–50% of the whole plant grain had formed (m), 70–100% of the whole grain had formed (n), and the whole plant grain had ripened (o). The abiotic stresses were as follows: (p) normal condition, (q) heat stress for 1 h, (r) heat stress for 6 h, (s) drought stress for 1 h, (t) drought stress for 6 h, (u) heat and drought stress combination for 1 h, (v) heat and drought stress combination for 6 h.
To elucidate the roles of these TaSBP genes in response to abiotic stresses, expression profiles of TaSBP genes under different abiotic stresses were also examined. The results showed that the expression of 79.17% (38/48) of the TaSBP genes was detected and some of them were highly expressed in response to heat and drought stresses. The phylogenetically similar genes shared similar expression patterns. For example, the subgroup I genes had similar expression patterns, and the subgroup VIII genes were expressed in all tissues.
To elucidate the roles of TaSBP genes in wheat growth and development, we examined the relative expression levels of 10 TaSBP genes (each group choose at least one TaSBP gene) in four tissues (roots, stems, leaves, and inflorescences, collected at the heading stage) (Fig. 9A) and under different abiotic stresses (Fig. 9B). All the TaSBP genes were detected in at least one of the tissues examined, but different expression levels were observed. All of them were highly expressed in inflorescences. In addition, TaSBP10A and TaSBP12B/D were highly expressed in stems, and TaSBP19B/D was mainly expressed in leaves. These results suggested that these genes may play different roles in wheat growth and development.
Results of quantitative RT-PCR of 10 TaSBP genes (A) in different tissues and (B) under different abiotic stresses. The horizontal and vertical co-ordinates represent four different tissues/abiotic stresses and the relative expression, respectively. Statistically significant differences are indicated: *P < 0.05; **P < 0.01 (Student’s t-test).
To explore the potential roles of TaSBP genes under different abiotic stresses, seedling plants were subjected to heat, cold, ABA, salt, and drought. The changes in the transcript levels of the genes were analyzed using quantitative real-time (RT)-PCR. Results showed that all of them were induced by different abiotic stresses (Fig. 9B). The expression level of TaSBP9B and TaSBP19B/D were significantly down-regulated under different abiotic stresses when compared to control. Under heat treatment, the expression levels of TaSBP1A/B/D and 16A were significantly up-regulated. Under cold stress, the expression levels of TaSBP6A, TaSBP16A, and TaSBP17B/D was significantly up-regulated, while TaSBP9A/D, TaSBP9B, TaSBP10A, and TaSBP19B/D were significantly down-regulated.
Discussion
Characteristics of SBP-box genes in wheat
The SBP-box proteins are characterized by a conserved SBP domain containing about 75 amino acids and they constitute a large family of transcription factors in plants. It has been proposed that SBP-box genes are plant specific3. In the present study, 48 wheat SBP-box genes were identified, accounting for 0.63% of all wheat annotation genes, which is more than that in rice (0.45%), Arabidopsis (0.58%), maize (0.57%), and B. distachyon (0.51%)18,19,23. In terms of sub-genomes, there are 16, 16, and 16 members in wheat A, B, and D sub-genomes respectively, this number in each sub-genome is similar with the result in rice (18), S. bicolor (17), S. italica (18), and B. distachyon (16)18,23,24. Duplication analysis showed that there were 16 segmental duplication pairs were formed by 22 TaSBPs, and three tandem duplication pairs were constructed by 6 TaSBPs. Duplication promoted TaSBPs gene expansion, maybe is the reason why the number of TaSBPs is more than other plant species.
Multiple sequence analysis showed that each of TaSBPs contained two Zn finger motifs, CCCH and CCHC type motifs, and one NLS region in the SBP domain, they constitute the main identifying characteristics of SBP-box proteins. Genes within the same phylogenetic subgroup shared a similar length, gene structure, and motif composition. For example, all subgroup I, II, IV, and VII members had three exons, motifs 3, 6, 7, and 10 were only found in subgroup V. Therefore, the similar gene structure and motif composition of SBP-box genes in wheat might reflect their evolutionary relationships. In the promoters of these TaSBP-box genes, four kinds of cis-acting elements––transcription-related, development-related, hormone-related, and abiotic stress-related elements––were detected, and phylogenetically similar genes shared the same cis-elements.
In addition, phylogenetic analysis revealed that the TaSBP-box proteins had a close evolutionary relationship with other plant SBP-box genes, especially with monocot plants. SBPs from ten plant species could be classified into night subgroups (Fig. 4), and there are two subgroups, II and VIII, only contained dicot plant SBPs, while IV and VI only had monocot SBP members, indicated that the closer relationship between monocot plant SBPs.
SBP-box genes play important roles in plant development and growth
SBP-box genes control a large range of processes underlying flower growth and development. For example, in Arabidopsis, constitutive expression of AtSPL3 results in very early flowering and frequent changes in morphology25; AtSPL2, AtSPL10, and AtSPL11 in Arabidopsis control morphological changes associated with shoot maturation in the reproductive phase26; AtSPL8 affects pollen sac development and also controls gynoecium development27; the miR156-SPL3 module controls FLOWERING LOCUST expression to regulate ambient temperature-responsive flowering28. In rice, OsSPL14 is highly expressed in the reproductive stage and promotes flower development; it also affects panicle branching29; OsSPL8 (OsLG1) controls ligule development and inflorescence architecture30,31. In the tomato plant, LeSPL/CNR is crucial for normal fruit development and ripening32. In wheat, TaSPL20 and TaSPL21, corresponding to TaSBP13D and TaSBP11D in this study, respectively, were highly expressed in the lemma and palea12. Ectopic expression of TaSPL20 or TaSPL21 in rice revealed that these genes have similar functions in regard to increasing the number of primary branches, secondary branches, grain number, and panicle length12. Ectopic expression of TaSPL16 (TaSBP15B in this study) in Arabidopsis delays the emergence of vegetative leaves, promotes early flowering, increases organ size, and affects yield-related traits14. TaSPL8 (TaSBP4D in this study) in wheat affects lamina joint development and plant architecture13. In this study, we found that 89.58% of TaSBP genes were highly expressed in the inflorescence according to ArrayExpress data, especially when two nodes or internodes were visible and when the stem reached its maximum length. Additionally, in our quantitative RT-PCR analysis of different tissues, all of the 10 selected TaSBP genes were highly expressed in flowers. These results suggest that TaSBP genes might play important roles in plant development and growth.
Conservation of miR156-binding sites in SBP-box genes
miRNAs play key roles in regulating the transcription of target genes. Most studies show that overexpression of miR164, miR159a, miR319, miR319, and miR399 affect members of the NAC, MYB, TCP, GAMYB, and WRKY transcription factor families, respectively13. Regarding the SBP-box gene family, tissue-specific interactions between OsmiR156 and OsSBP genes were found in rice17. Previous studies indicated that the miR156 function important in plant development and growth. For example, miR156 directly repressed the expression of SBP-box genes that function in juvenile-to-adult transition in wheat and Arabidopsis33,34. miR156 also play important roles in controlling flowering, leaf development, plant architecture by targeting SBP-box genes. For example, overexpression of miR156 delays Arabidopsis flowering and decreases apical dominance by regulating SBP-box genes35. In wheat, overexpression of tae-miR156 leads to increased tiller number and severe defects in spikelet formation36.
In the present study, target prediction showed that 24 TaSBPs have miR156-binding sites and that phylogenetically similar genes shared the same miR156-binding site. SBP-box genes with a miRNA-binding site existed across many subgroups (I, III, IV, VI, and VII) in wheat, suggesting conservation of miRNA-binding sites because of their functional importance. Previous study showed that tae-miR156-TaSPL3/17 interact with DWARF53 to regulate TEOSINTE BRANCHED1 (TaTB1) and BARREN STALK1 (TaBA1) expression, thus regulated wheat tillering and spikelet development36. Wheat TaSPL16 (TaSBP15B in this study) gene have miR156-binding sites in their terminal exons14. It has been reported that miR156 is responsible for the temporal expression of SPL13 during vegetative development35. As in Arabidopsis and Brassica napus, a previous study reported that the homologous genes in wheat are predicted to be targets of miR15616. Moreover, in the present study, the sites complementary to miR156 were located in the CDS of 19 TaSBP genes and in the 3′UTR regions of five TaSBP genes. These results showed that the miR156-binding site in SBP-box genes is conserved across plant species.
Materials and methods
Data retrieval and identification of SBP-box genes
To identify the SBP-box genes in wheat, the HMMER profile of the SBP-box-binding domain (PF03110) was obtained from the Pfam database (https://pfam.xfam.org/) and searched against the protein sequences of wheat with a threshold of e < 1e−5. The SBP-box protein sequences of 16 Arabidopsis and 19 rice SBP-box genes18 were retrieved from the Ensembl Plants database, and used to conduct a BLASTP search against the protein sequences of wheat with the threshold of e < 1e−5 and identity of 50%. After BLASTP, a self-blast and manual correction was performed to remove the alternative splicing events and redundancy. Finally, the NCBI-Conserved Domains Database (CDD; https://www.ncbi.nlm.nih.gov/cdd) and Simple Modular Architecture Research Tool (SMART) program (https://smart.embl.de/) were used to confirm the putative SBP-box proteins. The subcellular location of SBP-box genes was predicted using the CELLO web tool (https://cello.life.nctu.edu.tw/). The theoretical isoelectric point and molecular weight of SBP-box genes were predicted using the ExPASy tool (https://www.expasy.org/). To further verify the existence of TaSBP genes in wheat, we performed BLASTN37 to search for expressed sequence tags (ESTs) using the CDSs of TaSBP genes.
The protein sequences, cDNA sequences, DNA sequences, upstream 2-kb genomic DNA sequences, and CDSs of SBP-box genes used in this study were downloaded from the Ensembl Plants database (https://plants.ensembl.org/index.html) for further analysis.
Subcellular localization of TaSBPs
To check the subcellular localization of TaSBP4B, TaSBP9B, and TaSBP10A protein in wheat protoplast, a number of GFP fusion proteins were constructed. The cDNAs of full-length TaSBP4B, TaSBP9B, and TaSBP10A were cloned in frame with GFP to generate the constructs, respectively. These recombinant plasmids were transformed into wheat protoplasts using a polyethylene glycol (PEG)-mediated transient transformation system38. Visualization of the fluorescent proteins was performed using an Olympus IX83-FV1200 confocol microscope with excitation wavelengths of 460/480 nm for GFP and 633 nm for chloroplast.
Chromosomal location of TaSBP genes and gene duplication
The chromosome distribution information of TaSBP genes was obtained from the Ensembl Plants database (https://plants.ensembl.org/index.html). Duplications of TaSBP genes were analyzed using MCScanx39 with E-value < 1 × 10−10. Tandem duplication events were defined as adjacent homologous TaSBP genes on the same chromosome with no more than one intervening gene, while the segmental duplication events were generated through polyploidy and chromosome rearrangements40. The chromosome locations and gene duplications were visualized using Circos41.
Multiple sequence alignment and phylogenetic tree construction
The full-length amino acid sequences of the TaSBPs were used for multiple sequence alignment and phylogenetic analysis. Multiple sequence alignments of amino acid sequences were performed using the ClustalW program42 with default parameters. An un-rooted neighbor-joining tree was constructed with 1000 bootstrap replications using MEGA 7.0 software based on the full-length protein sequence alignment43.
Gene structure and conserved motif analyses
The exon–intron structure of TaSBP genes was graphically displayed using the Gene Structure Display Server44 using the CDSs and DNA sequences of TaSBPs. The amino acid sequences of TaSBPs were used to predict the conserved motifs using the MEME Suite web server45 with the maximum number of motifs set at 10 and the optimum width of motifs from 5 to 200 amino acids.
Cis-element analyses and miR156-binding site prediction
The upstream 2-kb genomic DNA sequences of TaSBP genes were submitted to the PlantCARE database (https://bioinformatics.psb.ugent.be/webtools/plantcare/html/) to identify the cis-elements in the TaSBP gene promoters. To predict the putative miR156-binding sites, the full lengths of TaSBP genes including CDS and UTR sequences were analyzed using the psRNATarget tool (https://plantgrn.noble.org/psRNATarget/?function).
Expression patterns of TaSBP genes
High-throughput sequencing data for wheat were obtained from the ArrayExpress database (https://www.ebi.ac.uk/arrayexpress) under accession number E-MTAB-448446. These data were used to analyze the expression profiles of TaSBP genes in 15 tissues, i.e. the root when the cotyledon emerged, three leaves were visible, and the stem reached its maximum length; the stem when the nodes or internodes were visible, half of the flowers were open, and elongation had begun; the leaf when the main shoot and axillary shoots were visible (with three nodes), the cotyledon emerged, and the whole plant fruit had formed; the inflorescence when the flowers opened, two nodes or internodes were visible, and the stem reached maximum length; and the grain when 30–50% of the whole plant grain had formed, 70–100% of the whole plant grain had formed, and the whole plant grain had ripened.
Plant growth and stress treatment
The wheat cultivar Chinese Spring was planted in an artificial climate chamber at 24/22 °C (day/night) with a photoperiod of 16/8 h (day/night). Roots, stems, leaves, and inflorescences were collected at the heading stage and used for tissue analyses. For abiotic stress, 10-day-old seedlings were subjected to heat (42 °C), cold (4 °C), drought (20% polyethylene glycol [PEG] 6000), salt (200 mM NaCl), and ABA (100 μmol ABA). Subsequently, whole seedlings (including root and leaves) were collected for RNA isolation.
RNA extraction, cDNA synthesis, and quantitative RT-PCR
Total RNA was extracted using a TIANGEN RNA Extraction Kit (Beijing, China) according to the manufacturer’s protocol. First-strand cDNA was synthesized using FastKing gDNA Dispelling RT SuperMix (Beijing, China). To assess gene expression, quantitative RT-PCR was performed on a Thermal Cycler Dice Real Time System III (ThermoFisher Scientific, China). Three biological replications were performed for each sample and 15-μL reaction systems containing 7.5 μL SYBR Premix Ex Taq (TAKARA), 0.75 μL (10 pmol/μL) each of forward and reverse primers, 0.5 μL of cDNA (200 ng/μL), and 5.5 μL of H2O. The quantitative RT-PCR was performed according to our previous study47. To normalize the total amount of cDNA present in each reaction, the wheat ACTIN gene was co-amplified as an endogenous control for calibration of the relative expression48. The relative expression level was calculated using the 2−△△CT method49. The primers for ten sets of TaSBP genes (TaSBP1A/B/D, TaSBP4B/4D, TaSBP6A, TaSBP9A/9D, TaSBP9B, TaSBP10A, TaSBP16A, TaSBP17B/17D, TaSBP18B/D, and TaSBP19B/19D) were universal in each set, because of the highly conserved sequences in the A, B, and D sub-genomes. Thus, the detected expression represented a combination of up to three homologous genes. The primers are listed in Supplementary Table S3.
Conclusions
In this study, we systematically identified TaSBP genes in the wheat genome. Forty-eight TaSBPs were identified and each contained a conserved SBP-box domain. The chromosome locations, gene and protein structures, subcellular localization, phylogenetic relationships, miR156-binding sites, and cis-elements were also characterized. The TaSBP expression levels in different tissues indicated that they were responsible for flower development. Quantitative RT-PCR analysis showed that the tested TaSBP genes were highly expressed in inflorescences and in response to abiotic stressors. This study establishes a foundation for further investigation of TaSBP genes and provides novel insights into their biological functions.
References
Klein, J. & Saedler, H. P. A new family of DNA binding proteins includes putative transcriptional regulators of the Antirrhinum majus floral meristem identity gene SQUAMOSA. Mol. Gen. Genet. 250, 7–16 (1996).
Birkenbihl, R., Jach, G. H. & Huijser, P. Functional dissection of the plant-specific SBP-domain: Overlap of the DNA-binding and nuclear localization domains. J. Mol. Biol. 352, 585–596 (2005).
Cardon, G. et al. Molecular characterisation of the Arabidopsis SBP-box genes. Gene 237, 91–104 (1999).
Si, L. Z. et al. OsSPL13 controls grain size in cultivated rice. Nat. Genet. 48, 447–456 (2016).
Jiao, Y. Q. et al. Regulation of OsSPL14 by OsmiR156 defines ideal plant architecture in rice. Nat. Genet. 42, 541–544 (2010).
Luo, L., Li, W. Q., Miura, K., Ashikari, M. & Kyozuka, J. Control of tiller growth of rice by OsSPL14 and Strigolactones, which work in two independent pathways. Plant Cell Physiol. 53, 1793–1801 (2012).
Cardon, G. H., Höhmann, S., Nettesheim, K., Saedler, H. & Huijser, P. Functional analysis of the Arabidopsis thaliana SBP-box gene SPL3: A novel gene involved in the floral transition. Plant J. 12, 367–377 (2012).
Kazuhiko, Y. et al. A novel zinc-binding motif revealed by solution structures of DNA-binding domains of Arabidopsis SBP-family transcription factors. J. Mol. Biol. 337, 49–63 (2004).
Wang, S. K. et al. Control of grain size, shape and quality by OsSPL16 in rice. Nat. Genet. 44, 950–954 (2012).
Jung, J. H., Lee, H. J., Ryu, J. Y. & Park, C. M. SPL3/4/5 Integrate developmental aging and photoperiodic signals into the FT-FD module in Arabidopsis flowering. Mol. Plant. 9, 1647–1659 (2016).
Wang, J. W., Czech, B. & Weigel, D. miR156-Regulated SPL transcription factors define an endogenous flowering pathway in Arabidopsis thaliana. Cell 138, 738–749 (2009).
Zhang, B. et al. Functional conservation and divergence among homoeologs of TaSPL20 and TaSPL21, two SBP-box genes governing yield-related traits in hexaploid wheat. Plant Physiol. 174, 1177–1191 (2017).
Liu, K. Y. et al. Wheat TaSPL8 modulates leaf angle through auxin and brassinosteroid signaling. Plant Physiol. 181, 179–194 (2019).
Cao, R. F. et al. Identification and functional characterization of squamosa promoter binding protein-like gene TaSPL16 in wheat (Triticum aestivum L.). Front. Plant Sci. 10, 212 (2019).
Lakhotia, N. et al. Identification and characterization of miRNAome in root, stem, leaf and tuber developmental stages of potato (Solanum tuberosum L.) by high-throughput sequencing. BMC Plant Biol. 14, 6–6 (2014).
Cheng, H. T. et al. Genomic identification, characterization and differential expression analysis of SBP-box gene family in Brassica napus. BMC Plant Biol. 16, 196 (2016).
Xie, K. B., Wu, C. Q. & Xiong, L. Z. Genomic organization, differential expression, and interaction of SQUAMOSA promoter-binding-like transcription factors and microRNA156 in rice. Plant Physiol. 142, 280–293 (2006).
Yang, Z. F. et al. Comparative study of SBP-box gene family in Arabidopsis and rice. Gene 407, 1–11 (2008).
Zhang, W., Bei, L. I. & Bin, Y. U. Genome-wide identification, phylogeny and expression analysis of the SBP-box gene family in maize (Zea mays). J. Integr. Agric. 15, 29–41 (2016).
Wang, Y., Hu, Z., Yang, Y., Chen, X. & Chen, G. Genome-wide identification, phylogeny, and expression analysis of the SBP-box gene family in grapevine. Russ. J. Plant Physiol. 57, 273–282 (2010).
Kavas, M., Kızıldoğan, A. K. & Abanoz, B. Comparative genome-wide phylogenetic and expression analysis of SBP genes from potato (Solanum tuberosum). Comput. Biol. Chem. 67, 131–140 (2017).
Song, S. et al. Genome-wide organization and expression profiling of the SBP-box gene family in Chinese Jujube (Ziziphus jujuba Mill.). Int. J. Mol. Sci. 18, 1734 (2017).
Davidson, R. M. et al. Comparative transcriptomics of three Poaceae species reveals patterns of gene expression evolution. Plant J. 71, 492–502 (2012).
Chang, J. Z. et al. Genome-wide identification and expression analysis of SBP-box gene family in Sorghum bicolor L.. Hereditas. 38, 569–580 (2016).
Gandikota, M. et al. The miRNA156/157 recognition element in the 3′ UTR of the Arabidopsis SBP box gene SPL3 prevents early flowering by translational inhibition in seedlings. Plant J. 49, 683–693 (2010).
Masahito, S., Tomotsugu, K., Nobutaka, M. & Masaru, O. T. Arabidopsis SBP-box genes SPL10, SPL11 and SPL2 control morphological change in association with shoot maturation in the reproductive phase. Plant Cell Physiol. 50, 2133–2145 (2009).
Unte, U. S. et al. SPL8, an SBP-box gene that affects pollen sac development in Arabidopsis. Plant Cell. 15, 1009–1019 (2003).
Kim, J. J. et al. The microRNA156-SQUAMOSA PROMOTER BINDING PROTEIN-LIKE3 module regulates ambient temperature-responsive flowering via FLOWERING LOCUS T in Arabidopsis. Plant Physiol. 159, 461–478 (2012).
Miura, K. et al. OsSPL14 promotes panicle branching and higher grain productivity in rice. Nat. Genet. 42, 545–549 (2010).
Lee, J., Park, J. J., Song, L. K., Yim, J. & An, G. Mutations in the rice liguleless gene result in a complete loss of the auricle, ligule, and laminar joint. Plant Mol. Biol. 65, 487–499 (2007).
Ishii, T. et al. OsLG1 regulates a closed panicle trait in domesticated rice. Nat. Genet. 45, 462–465 (2013).
Kenneth, M. et al. A naturally occurring epigenetic mutation in a gene encoding an SBP-box transcription factor inhibits tomato fruit ripening. Nat. Genet. 38, 948–952 (2006).
Liu, H. H., Tian, X., Li, Y. J., Wu, C. A. & Zheng, C. C. Microarray-based analysis of stress-regulated microRNAs in Arabidopsis thaliana. RNA 14, 836–843 (2008).
Qin, D. D. et al. Heat stress-responsive transcriptome analysis in heat susceptible and tolerant wheat (Triticum aestivum L.) by using Wheat Genome Array. BMC Genom. 9, 432 (2008).
Schwab, R. et al. Specific effects of MicroRNAs on the Plant Transcriptome. Dev. Cell 8, 517–527 (2005).
Liu, J., Cheng, X. L., Liu, P. & Sun, J. Q. miR156-targeted SBP-box transcription factors interact with DWARF53 to regulate TEOSINTE BRANCHED1 and BARREN STALK1 expression in bread wheat. Plant Physiol. 17, 1931–1948 (2017).
Johnson, M. et al. NCBI BLAST: A better web interface. Nucleic Acids Res. 36, W5-9 (2008).
Shan, Q. W., Wang, Y. P., Li, J. & Gao, C. X. Genome editing in rice and wheat using the CRISPR/Cas system. Nat. Protoc. Erecipes Res. 9, 2395–2410 (2014).
Wang, Y. P. et al. MCScanX: A toolkit for detection and evolutionary analysis of gene synteny and collinearity. Nucleic Acids Res. 40, e49–e49 (2012).
Zhu, Y. et al. Soybean (Glycine max) expansin gene superfamily origins: Segmental and tandem duplication events followed by divergent selection among subfamilies. BMC Plant Biol. 14, 93 (2014).
Krzywinski, M. et al. Circos: An information aesthetic for comparative genomics. Genome Res. 19, 1639–1645 (2009).
Thompson, J.D., Gibson, T.J. & Higgins, D.G. Multiple sequence alignment using ClustalW and ClustalX. Curr. Protoc. Bioinform. Chapter 2, Unit 2.3.1–2.3.22 (2002).
Kumar, S., Stecher, G. & Tamura, K. MEGA7: Molecular evolutionary genetics analysis version 7.0 for bigger datasets. Mol. Biol. Evol. 33, 1870–1874 (2016).
Hu, B. et al. GSDS 2.0: An upgraded gene feature visualization server. Bioinformatics 31, 1296–1297 (2015).
Bailey, T. L., Johnson, J., Grant, C. E. & Noble, W. S. The MEME suite. Nucleic Acids Res. 43, W39-49 (2015).
Pingault, L. et al. Deep transcriptome sequencing provides new insights into the structural and functional organization of the wheat genome. Genome Biol. 16, 29 (2015).
Li, Z. et al. Identification of the WUSCHEL-related homeobox (WOX) gene family, and interaction and functional analysis of TaWOX9 and TaWUS in wheat. Int. J. Mol. Sci. 21, 1581 (2020).
Ramesh, S. A. et al. Aluminum-activated malate transporters can facilitate GABA transport. Plant Cell. 30, 1147–1164 (2018).
Livak, K. J. & Schmittgen, T. D. Analysis of relative gene expression data using real-time quantitative PCR and the 2 −ΔΔ C T method. Methods 25, 402–408 (2001).
Acknowledgements
We thank Charlesworth (https://www.cwauthors.com.cn) for its linguistic assistance during the preparation of this manuscript.
Funding
This research was supported by the National Support Program of China (No. 2015BAD27B01), the Technological Innovation and Over Planning Projects of Shaanxi Province (No. 2014KTZB02-01-02) and the National Natural Science Foundation of China (31771785).
Author information
Authors and Affiliations
Contributions
G.Z. and X.C. supervised the experiments; Y.L. performed most of the experiments, analyzed the data, and prepared the figures and tables; Y.L. wrote the manuscript; Q.S. and Y.Z. performed some analyses and prepared Fig. 1; Z.L. and J.G. helped me prepare Fig. 7. All authors revised the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Li, Y., Song, Q., Zhang, Y. et al. Genome-wide identification, characterization, and expression patterns analysis of the SBP-box gene family in wheat (Triticum aestivum L.). Sci Rep 10, 17250 (2020). https://doi.org/10.1038/s41598-020-74417-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-74417-x
- Springer Nature Limited
This article is cited by
-
TaSPL6B, a member of the Squamosa promoter binding protein-like family, regulates shoot branching and florescence in Arabidopsis thaliana
BMC Plant Biology (2024)
-
Identification and expression analysis of SQUAMOSA promoter-binding protein (SBP) genes in mungbean
Plant Biotechnology Reports (2023)
-
Genome-wide analysis of glutamate receptor gene family in allopolyploid Brassica napus and its diploid progenitors
Genetica (2023)
-
Genome-wide identification and analysis of the evolution and expression pattern of the SBP gene family in two Chimonanthus species
Molecular Biology Reports (2023)
-
Systematic Identification, Evolution and Expression Analysis of the SPL Gene Family in Sugarcane (Saccharum spontaneum)
Tropical Plant Biology (2021)