Abstract
Staphylococcus aureus causing persistent, recurrent bovine intramammary infections are still a major challenge to dairy farming. Generally, one or a few clonal lineages are predominant in dairy herds, indicating animal-to-animal transfers and the existence of distinct pathotypic traits. The aim of this study was to determine if long term persistence and spreading of S. aureus are associated with specific phenotypic traits, including cellular invasion, cytotoxicity and biofilm formation. Mastitis isolates were collected over a 3-years period from a single dairy herd, resulting in two persistent subtypes, the high within-herd prevalent subtype ST9 (CC9)-methicillin-susceptible S. aureus (MSSA), designated HP/ST9, and the low within-herd prevalent subtype ST504 (CC705)-MSSA, designated LP/ST504. Characterization of the two different coexisting persistent subtypes showed that the following phenotypic traits are particularly associated with high within-herd prevalence: lack of capsular polysaccharide expression, high cellular invasiveness, low cytotoxicity and high biofilm/ poly-N-acetylglucosamine (PNAG) production, which may concomitantly contribute to the spreading of HP/ST9 within the herd. By contrast to HP/ST9, LP/ST504 is characterized by the formation of colony dendrites, which may help the bacteria to access deeper tissues as niches for persistence in single animals. Thus, within a single herd, two different types of persistence can be found in parallel, allowing longtime persistence of S. aureus in dairy cattle. Furthermore, this study indicates that ST9 (CC9)-MSSA strains, which are currently thought to have their primary reservoir in swine and humans, can also successfully spread to new hosts and persist in dairy herds for years.
Similar content being viewed by others
Introduction
Staphylococcus aureus frequently causes intramammary infections (IMI) in dairy cattle. Most cases are chronic, persistent IMI, which are difficult to treat and prone to resurgence, and thus often accompanied by long-lasting cost intensive antibiotic treatment and premature culling1,2. Besides negative consequences for animal welfare and milk quality, S. aureus presents also a significant public health threat due to its zoonotic potential and risk of foodborne intoxications in humans3,4.
One of the most important aggravating factors of bovine S. aureus IMI is the capability of the bacteria to evade clearing by antibiotics and by the host immune system resulting in long-lasting persistent infections. Several phenotypic strain characteristics have been suggested to be linked to S. aureus long-term persistence in the mammary gland, including the capacity to form biofilms and to invade and/or survive intracellularly. Bacterial biofilms are recalcitrant to antibiotic treatment and host immune defense mechanisms5,6. The biofilm matrix comprises a complex mixture of several extracellular polymeric substances, such as polysaccharides, proteins and DNA. The polymeric N-acetylglucosamine (PNAG) is the main exapolysaccharide of the S. aureus biofilm matrix, which mediates the bacteria-to-bacteria interactions after primary attachment7. S. aureus is commonly recognized as an extracellular pathogen, but it is becoming increasingly evident that it can survive and even replicate inside non-professional as well as professional phagocytes8,9. The isolation of viable intracellular S. aureus from alveolar cells and macrophages derived from milk of chronically infected cows underscored the in vivo relevance of this niche10. Since several studies have shown a high prevalence of non-encapsulated strains in persistent bovine IMI (up to 86%)4,11 as well as human chronic infections12, loss of capsular polysaccharide (CP) expression may be a key feature associated with chronicity of S. aureus. It was reported that capsule loss promotes adherence and internalization of S. aureus in vitro13,14. A recent study of Bardiau et al. revealed a correlation of biofilm formation, low CP expression and cellular invasion, fostering the hypothesis that certain S. aureus strains might be associated with specific intracellular and extracellular niches within the host15. However, hitherto knowledge on particular strain characteristics allowing S. aureus to persist and spread within herds remains largely unknown.
S. aureus IMI often results in one or a few dominant clones within a herd indicating transmissibility between animals and the preference of particular pathotypic traits16,17. Recent studies indicate that high within-herd prevalence of S. aureus is linked to specific genotypes18. To gain further insights into the association of S. aureus within-herd prevalence with phenotypic and genotypic strain properties, we monitored S. aureus in a dairy herd for the period of three years by means of Fourier transform infrared (FTIR) spectroscopy. FTIR spectroscopy was employed since it is a biophotonic based method of high discriminatory power19, which was shown to a be a very suitable method for S. aureus subtyping20, allowing to track specific S. aureus biotypes within the dairy chain4. For our current study, two different S. aureus subtypes isolated from the same dairy herd, which were able to persist for years but differed in their within-herd prevalence, were selected. Genotypic and phenotypic properties associated with persistence, including cellular invasion, biofilm formation and cytotoxicity of these two S. aureus subtypes were investigated to unravel the mechanisms contributing to persistence and spread of S. aureus in herds of dairy cattle.
Results
S. aureus persistence and spreading in a single dairy herd was monitored over a period of three years. Twelve cows out of in average 25 lactating cows were at least once positively tested for S. aureus. A total of 58 S. aureus isolates were submitted to FTIR spectroscopic and genotypic subtyping.
The high IMI prevalent and persistent S. aureus isolates belong to ST9 (CC9)
FTIR spectroscopy as a fast, high-throughput screening tool was initially used to subtype the herd-specific diversity of S. aureus strains and to follow potential spreading between cows4,20. Using hierarchical cluster analysis (HCA) of FTIR spectroscopic data we were able to discriminate two distinct FTIR biotypes (Fig. 1). The high IMI prevalent biotype I (n = 50; 86.2%) successfully persisted for years in several cows and spread at the herd level over the whole period infecting ten cows in total. Using molecular subtyping methods, this biotype could be assigned to spa-type t1939, ST9 [clonal-complex (CC) 9] and agr-II (hereinafter referred as high-prevalent/ST9, HP/ST9). The strains belonging to biotype II (n = 8, 13.8%) showed consistently lower within-herd prevalence. The latter biotype, which was isolated from the udder of two cows over a period of more than two years was assigned to spa-type t529, ST504 (CC705, former CC151) and agr-II (hereinafter referred as low-prevalent/ST504, LP/ST504). A summary and a detailed schema of isolates are provided in Table 1 and Suppl. 1, respectively.
HCA dendrogam of FTIR biotyping. FTIR spectroscopy was employed to follow S. aureus persistence and spreading at herd level. In total two different FTIR biotypes were identified (I, II). The HP/ST9 isolates persisted and successively spread within the herd and did not express CP phenotypically, as they were defined as non-typeable (NT), whereas the LP/ST504 isolates expressed CP serotype 8 (CP8).
Based on FTIR spectroscopic and genotypic subtyping, persistent mastitis isolates were divided into two groups related to either high- or low within-herd IMI prevalence. From each group, three S. aureus mastitis isolates derived from the last sampling time point of different animals were selected and further investigated regarding their properties associated with persistence, including cellular invasion, cytotoxicity and biofilm formation. The well documented and characterized bovine strain RF122 (ST504/ CC705)21, capable to reproducibly induce severe mastitis, was included as reference strain in this study (hereinafter referred RF122/ST504).
HP/ST9 does not express a capsule
Since loss of capsular polysaccharide expression is thought to be associated with a chronic state of mastitis14, we utilized the recently developed CP typing system based on artificial neuronal network-assisted FTIR spectroscopy to identify capsule expressing (CP5, CP8) and non-expressing (non-typeable, NT) strains22, which was accompanied by cap specific PCR determination. All HP/ST9 isolates expressed no CP and were found to be positive for the cap5 specific allele, whereas all LP/ST504 isolates expressed CP serotype 8 (CP8/ cap8). These results underpin that lack of CP expression is associated with high within-herd prevalence.
HP/ST9 exhibits high internalization capacity on MAC-T cells
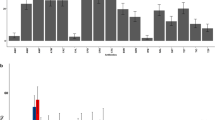
Staphylococcal host cell invasion is considered to be one mechanism to establish a long-lasting, chronic infection23. Thus, we investigated the capacity of HP/ST9 and LP/ST504 isolates to internalize bovine epithelial cells in vitro, using bovine mammary epithelial (MAC-T) cells. MAC-T cells were incubated with S. aureus at a multiplicity of infection (MOI) of 10 to 50 for 1 h at 37 °C, and cells were subsequently washed and antibiotic-containing medium was added to eradicate the extracellular staphylococci. The high prevalent HP/ST9 showed a significant higher capacity to internalize epithelial cells compared to LP/ST504 and the bovine reference strain RF122/ST504. The internalization capacities of LP/ST504 isolates and strain RF122/ST504 showed no significant differences (Fig. 2A). Therefore, it is tempting to speculate that a high internalization capacity promotes staphylococcal high within-herd prevalence.
Bacterial internalization capacity and cellular cytotoxicity. From each group with either high or low within-herd IMI prevalence, HP/ST9 and LP/ST504, three different S. aureus mastitis isolates and the mastitis reference strain RF122 were comparatively tested. (A) S. aureus internalization into mammary epithelial (MAC-T) cells measured as CFU remaining after 2 h of incubation and killing of extracellular bacteria. (B) Cytotoxicity of S. aureus supernatants on MAC-T cells. The relative viability was expressed based on RPMI diluted in DMEM (ratio 1:1)-treated cells (=100%). Each bar represents the arithmetic mean ± standard deviation (SD) from three independent experiments. (*p < 0.05, **p < 0.01; ***p < 0.001; ****p < 0.0001).
HP/ST9 exhibits low cytotoxicity
The cytotoxic capacity of the isolates was examined in order to assess a possible correlation to within-herd prevalence. S. aureus cytotoxic effect was investigated by measuring the viability of MAC-T cells. Mitochondrial activity of RPMI-treated cells (100% viability) was used for normalization. HP/ST9 isolates showed only very low in vitro cytotoxicity. In contrast, LP/ST504 isolates and RF122/ST504 yielded a significantly stronger cytotoxicity than HP/ST9 (Fig. 2B).
HP/ST9 produces more biofilm and higher amounts of PNAG than LP/ST504
Bacterial biofilms have been described to be associated with chronic bovine udder infections5, thus we next assessed whether high within-herd prevalent strains might be associated with an increased capacity to form biofilms in vitro, using a 96-well assay. In addition, the production of a major component of S. aureus biofilm the poly-N-acetyl-β-(1–6)-glucosamine (PNAG), also referred as extracellular matrix polysaccharide intercellular adhesin (PIA), was quantified using a lectin-based ELISA. HP/ST9 isolates turned out to produce moderate amounts of biofilm, but significantly more biofilm compared to LP/ST504 isolates, which are weak biofilm producers (Fig. 3A). Furthermore, the quantitation of PNAG expressed in biofilms revealed that HP/ST9 isolates are strong PNAG producers while LP/ST504 isolates are weak PNAG producers. The reference strain RF122/ST504 showed a biofilm/ PNAG phenotype similar to that of HP/ST9 isolates (Fig. 3B). These results indicate that biofilm production and particularly a high PNAG content in biofilms correlate with high within-herd prevalence.
Biofilm production and PNAG content in biofilm. Biofilm (A) and PNAG in biofilm (B) was measured after 24 h growth in TSB media in a 96-well plate static biofilm assay. Un-inoculated medium served as a cut-off value (ODc) for the quantitative classification of biofilm and PNAG production in no, weak, moderate and strong producers based on their average OD values. Each bar represents the arithmetic mean ± standard deviation (SD) from three independent experiments. (*p < 0.05, **p < 0.01; ***p < 0.001; ****p < 0.0001).
HP/ST9 and LP/ST504 isolates show no differences in their virulence gene and antibiotic resistance profiles
The presence of S. aureus adhesion (fnbpA, fnbpB, clfA, clfB), biofilm-associated (icaA, icaD) and S. aureus enterotoxin (SEs; sea, seb, sec, sed, see, seg, seh, sei, sej) genes was analyzed, using the panel of representative HP/ST9 and LP/ST504 strains, but no differences in virulence genes nor in the antibiotic resistance genes were observed between the high and low prevalent isolates. Both, high and low within-herd prevalent strains, were positive for fnbpA, clfA, clfB, icaA, icaD and seg, sei, (see also Table 1). In addition, antibiotic resistance was tested and revealed a broad susceptibility to the majority of tested antibiotics, but no distinct differences between the antibiotic resistant profiles between high and low prevalent isolates were found. Resistance against pirlimicin, erythromycin and marbofloxacin, but not against oxacillin (methicillin) for the high and low within-herd prevalent isolates was found. Antimicrobial resistance profiles are summarized in Table 2.
HP/ST9 and LP/ST504 colonies show differences in formation of dendrites
Furthermore, the formation of spreading dendrites has been linked to survival and spreading of S. aureus within host24,25. We therefore assessed the capacity of the high and low-prevalent isolates to spread on agar media. As depicted in Fig. 4, colonies of the low prevalent LP/ST504 showed formation of dendrites while no dendrite formation was observed in HP/ST9.
Expansion of bacteria on agar media. Bacterial cultures of HP/ST9 and LP/ST504 isolates were inoculated in the middle of the agar plate and subsequently incubated overnight at 37 °C. LP/ST504 formed colony dendrites on the agar media, whereas the HP/ST9 subtype not. Images are representatives from three independent experiments.
Discussion
Several studies reported links between biofilm formation, CP expression and internalization capacity to chronicity in bovine IMI. However, these studies are based on well-established mastitis reference strains, such as RF122 and Newbould 30526, or on strains derived from different local or clonal (CC) origin15,27, while data from studies addressing differences of specific phenotypic and virulence traits of persistent strains in the context of within-herd prevalence are still lacking. To gain insights into the mechanism allowing S. aureus to spread and successfully persist within a herd, this study focuses on isolates collected over a 3-years period from a single dairy herd, which differed in their within-herd prevalence. Notable, all isolates could be assigned to two distinct biotypes and sequence types/ clonal complexes, namely HP/ST9 (CC9) and LP/ST504 (CC705), which were associated with high and low within-herd prevalence, respectively. Further analyses of representative strains revealed distinct phenotypic characteristics for both subtypes, which might at least partially explain their differences found for their prevalence on herd level.
Our study demonstrates that non-encapsulation and cellular invasion on bovine epithelial cells is associated with high within-herd prevalence of S. aureus. This is in line with previous findings that loss of CP expression is one key S. aureus feature associated with persistence23. It has been suggested that non-encapsulated S. aureus isolates might more effectively enter epithelial cells than encapsulated strains. Internalization might protect bacteria from clearance by the immune system and allow longtime persistence in chronically infected hosts23. Moreover, capsule-negative S. aureus strains have also been shown to induce chronic mastitis in a mouse model14 and the prevalence of non-encapsulated strains was reported to be higher in chronic than in acute infections in bovine mastitis as well as in human osteomyelitis and cystic fibrosis11,28,29.
Furthermore, reduced cytotoxicity was proposed to represent an additional mechanism contributing to persistence of staphylococcal infections30. This hypothesis is fostered by the results of our current study, showing that high within-herd prevalence is associated with a low cytotoxic potential. Contradictory, a higher potential cytotoxicity for high within-herd prevalent ST8 strains was proposed by a recent study of Capra et al.31. Thus, further studies will be necessary, including different ST types, before firm conclusion can be drawn on the importance and role of strain cytotoxicity for S. aureus persistence in dairy cattle.
Increased biofilm formation has been associated with chronic bovine udder infections and in line with this, we found higher levels of biofilm formation by strains with high within-herd prevalence. The same applies for the biofilm PNAG content, which mediates the bacterial intercellular connection and is produced by the icaABCD operon encoded enzymes. In the present study, all biotypes were positively tested for icaA and icaD genes. These results support previous studies, showing a high prevalence of genes of the icaA and icaD loci among S. aureus mastitis isolates32,33. As reported for the clumping dispersal in S. aureus bloodstream infections34, high levels of PNAG in biofilm could be an advantageous dissemination mechanism by facilitating staphylococcal bacteria-to-bacteria interaction, which mediates the dispersal ability of the pathogen in the bovine mammary gland.
Based on the results of an in vitro agar plate assay, it has been discussed by Pollitt et al.25,35, that the formation of spreading dendrites might contribute to the establishment of S. aureus infections and translocation of the bacteria within the host. Since the low prevalent subtype LP/ST504 but not the high prevalent HP/ST9 subtype showed the formation of spreading dendrites in the agar plate assay, it is tempting to speculate that dendrite formation of the low prevalent S. aureus bovine mastitis isolates might be an adaptive feature to facilitate dispersal deep into the alveolar units of the mammary gland tissue fostering persistence within single animals rather than spreading within a herd.
Recent epidemiological studies showed that only the three clonal complexes CC8, CC97, and CC705, are largely dominating bovine IMI associated strains36. Isolates of ST504 (CC705) are known to frequently cause mastitis in cattle worldwide and these strains are described as archetypical bovine strains. In our study, LP/ST504 isolates were capable to persist for more than two years, but showed only limited success in spreading at herd level. This finding agrees with previous reports that CC705 strains exhibit only low contagiousness and are restricted to very few cows per herd resulting in low within-herd prevalence16. Notably, the isolates showing high within herd prevalence and persistence for years of our current study belong to ST9 (CC9). S. aureus ST9 methicillin- susceptible S. aureus (MSSA) and methicillin- resistant S. aureus (MRSA) strains are generally considered to exhibit their main reservoir in swine and also capable to colonize and infect humans37 and have so far only reported from some rare cases of bovine mastitis in Europe and China36,38.
Conclusion
This study provides evidence for the existence of specific phenotypic traits linked to persistence of S. aureus in dairy cattle. Two persistent subtypes were found differing in their within-herd prevalence. In particular, the importance of lack of CP expression and high cellular invasiveness for S. aureus spreading within chronically infected dairy cattle herds is highlighted. Additionally, low level of cellular cytotoxicity and high biofilm/ PNAG production might support the capacity of strains to successfully persist and spread in bovines. Furthermore, first evidence is presented that the swine- and human-associated ST9 (CC9)-MSSA can also cause long-term persistent bovine IMI and is able to successfully spread between cows at the herd level, resulting in high within herd prevalence. This high dissemination potential of HP/ST9 discovered in our work renders dairy herds as a possible underestimated reservoir for human associated ST9 (MSSA/ MRSA) strains and thus, might represent a serious threat to public health, which requires further attention.
Materials and Methods
Sampling, isolation and identification of bovine isolates
S. aureus isolates were recovered from milk samples in frame of routine microbial mastitis diagnostics derived from cows (Simmental breed) with subclinical mastitis from a single dairy herd with 25 animals in average (tethered housing, manual milking stanchion) located at the provincial state Upper Austria over a period of more than three years (2010–2013). There was neither a study-related interference in any way with the herd management by the professional Veterinarian nor were bacteria experimentally administered to the cows. Subclinical IMI was characterized by milk somatic cell counts >200.000 cells/mL with no macroscopic changes following the guidelines of the National Mastitis Councils Laboratory Handbook on Bovine Mastitis39. Cases of subclinical, chronic, persistent S. aureus mastitis were regularly treated by antibiotics during dry-off. Milk samples were collected aseptically from foremilk and stored at −80 °C until bacteriological analysis39. Milk samples (approx. 10 ml) were centrifuged and a loop of sediment (10 μl) was streaked onto on Columbia agar supplemented with 5% sheep blood (CBA) (Thermofisher Scientific Inc., Oxoid Ltd., Hampshire, UK). CBA plates were incubated at 37 °C and examined after 24–48 hours for bacterial growth. Presumptive S. aureus isolates were confirmed by S. aureus species specific nuc-PCR40 and FTIR spectroscopy as mentioned below.
Reference strains
Bovine strain RF122 (ST504/ CC705, former CC151), capable to reproducibly induce severe mastitis, was included as reference strain in this study21. S. aureus Reynolds prototype strain CP5 and its isogenic mutants Reynolds CP8 and Reynolds CP- (non-encapsulated)41 were used as controls for cap genotyping as well as for FTIR spectroscopic serotyping.
Identification, subtyping and capsular serotyping by FTIR spectroscopy
Sample preparation and FTIR measurement was performed as described previously22. In brief, isolates were grown on tryptone soy agar (TSA) plates (Thermo Fisher Scientific, Oxoid) at 30 °C for 24 h. FTIR spectra of dried intact bacteria were recorded in transmission mode with an HTS-XT microplate adapter coupled to a Tensor 27 FTIR spectrometer (Bruker Optics GmbH, Ettlingen, Germany). Species identification was performed using a FTIR reference spectral library and confirmed by nuc-PCR40. S. aureus high-resolution strain typing by FTIR spectroscopy was performed as previously reported using average spectra of measurements performed on three different days20. Using the same spectra, capsular serotypes (CP5, CP8, NT) were determined by artificial neuronal network (ANN) analysis as previously established in our lab22.
Molecular subtyping and virulence factor detection
MLST was performed according to Enright et al.42 with the exception of the downstream trimming position used to define gmk alleles, which has been adjusted to 417 bp, compared to the previous 429 bp42. The allelic profiles and ST were assigned based on the MLST website (http://saureus.mlst.net/). For spa typing the sequence of a polymorphic VNTR in the 3’ coding region of the S. aureus–specific staphylococcal protein A (spa) was used43 (www.spaserver.ridom.de). Spa types were assigned according to the repeat succession with the spaTyper free public application software (http://spatyper.fortinbras.us/). Agr-groups were determined by multiples PCR according to Gilot et al.44. The following genes were amplified by PCR: icaA/D45, cap5/846, fnbpA/B47 and clfA/B48. S. aureus enterotoxin (SE) profiles were determined by PCR including sea, seb, sec, sed, seg, seh, sei, and sej49.
Antibiotic resistance profiling
The antimicrobial resistance (AMR) was tested by applying the commercially available Micronaut-S Mastitis 2 MIC microtiter plate assay (Merlin; Sifin diagnostics GmbH, Berlin, Germany) including a panel of 10 antimicrobials: penicillin G, ampicillin, cefazolin, cefaperazon, cefquinom, oxacillin, pirlimycin, erythromycin, amoxicillin-clavulanic acid, and marbofloxacin. The breakpoints for minimum inhibitory concentrations (MICs) were determined according to the actual Eucast (http://www.eucast.org/clinical_breakpoints/) and Clinical and Laboratory Standards Institute (CLSI) standards50.
Invasion assay
Bovine mammary epithelial cell line (MAC-T) was used for S. aureus internalization assay and was performed as previously reported with slight modifications51. Briefly, confluent MAC-T cell monolayers (~2 × 105 cells/well) were inoculated with bacteria to produce multiplicities of infection (MOI) of 10–50. After incubation for 1 h at 37 °C under 5% CO2, the wells were washed with PBS and the invasion medium supplemented with gentamicin (final conc. 400 µg/mL) was added to each well to kill extracellular bacteria. After 1 h the monolayer was lysed by the addition of 0.025% Triton X-100 in sterile distilled water to release intracellular staphylococci. The CFU number was determined by quantitative plating on TSA agar. Three independent experiments on different days were performed each in triplicates per isolate.
Cytotoxicity
S. aureus cytotoxic effect was assessed by measuring the viability of MAC-T cells based on tetrazolium salt WST-1 (Roche, Basel, Switzerland) to detect the mitochondrial activity of viable cells. Strains were grown under iron-depleted RPMI 1640 (Biochrome) to mimic mastitic conditions in vivo52. After 24 h of growth, the cultures were centrifuged and the supernatant was filtered on 0.22μm PVDF membrane units and subsequently diluted in DMEM at ratio 1:1. MAC-T cells were grown in this media for 24 h in 5% CO2 and the absorbance was read at 450 nm after adding WST-1 reagent for 60 min. All values were normalized to the cytotoxicity of cells treated with iron-depleted RPMI diluted in DMEM (ratio 1:1), which was set at 100% viability. Three independent experiments were performed in triplicates.
Biofilm production ability and PNAG content
Biofilm formation was quantitatively assessed in a microtiter well plate assay using the CV staining method53. S. aureus overnight cultures were diluted 1:100 in trypticase soy broth (TSB, Oxoid) supplemented with 0.25% glucose (TSB-glucose) and transferred to 96-well polystyrene microtiter plates. After 24 h of static incubation at 37 °C, wells were washed, dried at 40 °C and the biofilm was fixed with methanol for 15 min prior staining with 0.1% CV. The quantity of biofilm biomass was determined by reading the optical density (OD) at 630 nm.
PNAG in biofilms was quantified according to Cramton et al.54 with slight modifications54. The biofilms for the PNAG assay were grown as described above. Biofilms were fixed by adding 95% ethanol for 15 min and blocked for 1 h at 37 °C in 1% w/v BSA in PBS plus 0.05% Tween 20 (PBST). After removal, wheat germ agglutinin horseradish peroxidase conjugate (WGA–HRP) (Sigma-Aldrich, St. Louis, MO, USA), was incubated for 30 min at 37 °C, stained with the peroxidase chromogenic substrate o-phenylenediamine (OPD) and subsequently measured at OD 490 nm. Un-inoculated medium served as a cut-off value (ODc) and the quantitative classification of biofilm production and PNAG content was based on average OD values according to Stepanovic et al.55. Three independent experiments were performed in at least quadruplicates for both assays.
Motility assay
The motility assay was performed according to the detailed guidelines in Pollitt et al.35. In brief, an always fresh TSB media was prepared containing Bacto agar (0.34% in total), and D-Glucose (Sigma) (50 mM final conc.) was added after autoclave. Poured petri dishes were subsequently dried for 30 min in a safety cabinet and UV sterilized. Five µL of bacterial culture was spotted on the agar plate and plates were incubated overnight in a sealed plastic box at 37 °C.
Statistical analysis
Statistical analysis was performed by using Graph-Pad Prism (GraphPad Software, Inc., La Jolla, USA; version 5.00). Statistically significant differences were calculated by using appropriate statistical methods as indicated including 1-way analysis of variance (ANOVA), followed by Dunnett’s multiple comparisons test for comparing the reference strain RF122 to the herd derived strains and the Sidak’s multiple comparisons test for comparing between high and low within-herd prevalent strains. p values less than 0.05 were considered significant and are indicated in the figures as follows: *p < 0.05, **p < 0.01; ***p < 0.001; ****p < 0.0001.
Data Availability
The datasets generated and/or analyzed during the current study are available from the corresponding author upon request.
References
Schukken, Y. H. et al. Host-response patterns of intramammary infections in dairy cows. Vet Immunol Immunopathol. 144, 270–289, https://doi.org/10.1016/j.vetimm.2011.08.022 (2011).
Smith, G. W., Lyman, R. L. & Anderson, K. L. Efficacy of vaccination and antimicrobial treatment to eliminate chronic intramammary Staphylococcus aureus infections in dairy cattle. J Am Vet Med Assoc. 228, 422–425 (2006).
Johler, S. et al. Outbreak of staphylococcal food poisoning among children and staff at a Swiss boarding school due to soft cheese made from raw milk. J Dairy Sci. 98, 2944–2948, https://doi.org/10.3168/jds.2014-9123 (2015).
Kummel, J. et al. Staphylococcus aureus Entrance into the Dairy Chain: Tracking S. aureus from Dairy Cow to Cheese. Front Microbiol. 7, 1603, https://doi.org/10.3389/fmicb.2016.01603 (2016).
Melchior, M. B., Vaarkamp, H. & Fink-Gremmels, J. Biofilms: a role in recurrent mastitis infections? Vet J. 171, 398–407, https://doi.org/10.1016/j.tvjl.2005.01.006 (2006).
Scali, F., Camussone, C., Calvinho, L. F., Cipolla, M. & Zecconi, A. Which are important targets in development of S. aureus mastitis vaccine? Res Vet Sci. 100, 88–99, https://doi.org/10.1016/j.rvsc.2015.03.019 (2015).
O’Gara, J. P. ica and beyond: biofilm mechanisms and regulation in Staphylococcus epidermidis and Staphylococcus aureus. FEMS Microbiol lett. 270, 179–188, https://doi.org/10.1111/j.1574-6968.2007.00688.x (2007).
Almeida, R. A., Matthews, K. R., Cifrian, E., Guidry, A. J. & Oliver, S. P. Staphylococcus aureus invasion of bovine mammary epithelial cells. J Dairy Sci. 79, 1021–1026, https://doi.org/10.3168/jds.S0022-0302(96)76454-8 (1996).
Bayles, K. W. et al. Intracellular Staphylococcus aureus escapes the endosome and induces apoptosis in epithelial cells. Infect Immun. 66, 336–342 (1998).
Hebert, A., Sayasith, K., Senechal, S., Dubreuil, P. & Lagace, J. Demonstration of intracellular Staphylococcus aureus in bovine mastitis alveolar cells and macrophages isolated from naturally infected cow milk. FEMS Microbiol lett. 193, 57–62 (2000).
Sordelli, D. O. et al. Capsule expression by bovine isolates of Staphylococcus aureus from Argentina: genetic and epidemiologic analyses. J Clin Microbiol. 38, 846–850 (2000).
Lattar, S. M. et al. Capsule expression and genotypic differences among Staphylococcus aureus isolates from patients with chronic or acute osteomyelitis. Infect Immun. 77, 1968–1975, https://doi.org/10.1128/IAI.01214-08 (2009).
Pohlmann-Dietze, P. et al. Adherence of Staphylococcus aureus to endothelial cells: influence of capsular polysaccharide, global regulator agr, and bacterial growth phase. Infect Immun. 68, 4865–4871 (2000).
Tuchscherr, L. P. et al. Capsule-negative Staphylococcus aureus induces chronic experimental mastitis in mice. Infect Immun. 73, 7932–7937, https://doi.org/10.1128/IAI.73.12.7932-7937.2005 (2005).
Bardiau, M. et al. Existence of two groups of Staphylococcus aureus strains isolated from bovine mastitis based on biofilm formation, intracellular survival, capsular profile and agr-typing. Vet Microbiol. 185, 1–6, https://doi.org/10.1016/j.vetmic.2016.01.003 (2016).
Graber, H. U. et al. Mastitis-related subtypes of bovine Staphylococcus aureus are characterized by different clinical properties. J Dairy Sci. 92, 1442–1451, https://doi.org/10.3168/jds.2008-1430 (2009).
Haveri, M., Roslöf, A., Rantala, L. & Pyörälä, S. Virulence genes of bovine Staphylococcus aureus from persistent and nonpersistent intramammary infections with different clinical characteristics. J Appl Microbiol. 103, 993–1000, https://doi.org/10.1111/j.1365-2672.2007.03356.x (2007).
Cremonesi, P. et al. Genomic characteristics of Staphylococcus aureus strains associated with high within-herd prevalence of intramammary infections in dairy cows. J Dairy Sci. 98, 6828–6838, https://doi.org/10.3168/jds.2014-9074 (2015).
Wenning, M. & Scherer, S. Identification of microorganisms by FTIR spectroscopy: perspectives and limitations of the method. Appl Microbiol Biotechnol. 97, 7111–7120, https://doi.org/10.1007/s00253-013-5087-3 (2013).
Johler, S., Stephan, R., Althaus, D., Ehling-Schulz, M. & Grunert, T. High-resolution subtyping of Staphylococcus aureus strains by means of Fourier-transform infrared spectroscopy. Syst Appl Microbiol 39, 189–194, https://doi.org/10.1016/j.syapm.2016.03.003 (2016).
Fitzgerald, J. R., Hartigan, P. J., Meaney, W. J. & Smyth, C. J. Molecular population and virulence factor analysis of Staphylococcus aureus from bovine intramammary infection. J Appl Microbiol. 88, 1028–1037 (2000).
Grunert, T. et al. Rapid and reliable identification of Staphylococcus aureus capsular serotypes by means of artificial neural network-assisted Fourier transform infrared spectroscopy. J Clin Microbiol. 51, 2261–2266, https://doi.org/10.1128/JCM.00581-13 (2013).
Tuchscherr, L., Loffler, B., Buzzola, F. R. & Sordelli, D. O. Staphylococcus aureus adaptation to the host and persistence: role of loss of capsular polysaccharide expression. Future Microbiol. 5, 1823–1832, https://doi.org/10.2217/fmb.10.147 (2010).
Kaito, C. & Sekimizu, K. Colony spreading in Staphylococcus aureus. J. Bacteriol. 189, 2553–2557, https://doi.org/10.1128/JB.01635-06 (2007).
Pollitt, E. J. G. & Diggle, S. P. Defining motility in the Staphylococci. Cellular and molecular life sciences: CMLS 74, 2943–2958, https://doi.org/10.1007/s00018-017-2507-z (2017).
Peton, V. et al. Fine-tuned characterization of Staphylococcus aureus Newbould305, a strain associated with mild and chronic mastitis in bovines. BMC Vet Res. 45, 106, https://doi.org/10.1186/PREACCEPT-6950585981338355 (2014).
Budd, K. E., Mitchell, J. & Keane, O. M. Lineage associated expression of virulence traits in bovine-adapted Staphylococcus aureus. Vet Microbiol. 189, 24–31, https://doi.org/10.1016/j.vetmic.2016.04.013 (2016).
Sutter, D. E. et al. Capsular serotype of Staphylococcus aureus in the era of community-acquired MRSA. EMS Immunol Med Microbiol. 63, 16–24, https://doi.org/10.1111/j.1574-695X.2011.00822.x (2011).
von Eiff, C. et al. Distribution of capsular and surface polysaccharide serotypes of Staphylococcus aureus. Diagn Microbiol Infect Dis. 58, 297–302, https://doi.org/10.1016/j.diagmicrobio.2007.01.016 (2007).
Smeltzer, M. S. Staphylococcus aureus Pathogenesis: The Importance of Reduced Cytotoxicity. Trends Microbiol. 24, 681–682, https://doi.org/10.1016/j.tim.2016.07.003 (2016).
Capra, E. et al. Genomic and transcriptomic comparison between Staphylococcus aureus strains associated with high and low within herd prevalence of intra-mammary infection. BMC Microbiol. 17, 21, https://doi.org/10.1186/s12866-017-0931-8 (2017).
Dhanawade, N. B., Kalorey, D. R., Srinivasan, R., Barbuddhe, S. B. & Kurkure, N. V. Detection of intercellular adhesion genes and biofilm production in Staphylococcus aureus isolated from bovine subclinical mastitis. Vet Res Commun. 34, 81–89, https://doi.org/10.1007/s11259-009-9326-0 (2009).
Szweda, P., Schielmann, M., Milewski, S., Frankowska, A. & Jakubczak, A. Biofilm production and presence of ica and bap genes in Staphylococcus aureus strains isolated from cows with mastitis in the eastern Poland. Pol J Microbiol. 61, 65–69 (2012).
Gronnemose, R. B. et al. A novel in vitro model for haematogenous spreading of S. aureus device biofilms demonstrating clumping dispersal as an advantageous dissemination mechanism. Cell Microbiol. 19, https://doi.org/10.1111/cmi.12785 (2017).
Pollitt, E. J., Crusz, S. A. & Diggle, S. P. Staphylococcus aureus forms spreading dendrites that have characteristics of active motility. Sci. Rep. 5, 17698, https://doi.org/10.1038/srep17698 (2015).
Boss, R. et al. Bovine Staphylococcus aureus: Subtyping, evolution, and zoonotic transfer. J Dairy Sci. 99, 515–528, https://doi.org/10.3168/jds.2015-9589 (2016).
Kehrenberg, C., Cuny, C., Strommenger, B., Schwarz, S. & Witte, W. Methicillin-resistant and -susceptible Staphylococcus aureus strains of clonal lineages ST398 and ST9 from swine carry the multidrug resistance gene cfr. Antimicrob Agents Chemother. 53, 779–781, https://doi.org/10.1128/AAC.01376-08 (2009).
Wang, X. M. et al. Methicillin-resistant Staphylococcus aureus ST9 from a case of bovine mastitis carries the genes cfr and erm(A) on a small plasmid. J Antimicrob Chemother. 67, 1287–1289, https://doi.org/10.1093/jac/dks028 (2012).
Hogan, J. et al. Laboratory Handbook on Bovine Mastitis. National Mastitis Council, Madison, WI, ISBN:0932147038 9780932147035 (1999).
Brakstad, O. G., Aasbakk, K. & Maeland, J. A. Detection of Staphylococcus aureus by polymerase chain reaction amplification of the nuc gene. J Clin Microbiol. 30, 1654–1660 (1992).
Watts, A. et al. Staphylococcus aureus strains that express serotype 5 or serotype 8 capsular polysaccharides differ in virulence. Infect Immun. 73, 3502–3511, https://doi.org/10.1128/IAI.73.6.3502-3511.2005 (2005).
Enright, M. C., Day, N. P., Davies, C. E., Peacock, S. J. & Spratt, B. G. Multilocus sequence typing for characterization of methicillin-resistant and methicillin-susceptible clones of Staphylococcus aureus. J Clin Microbiol. 38, 1008–1015 (2000).
Harmsen, D. et al. Typing of methicillin-resistant Staphylococcus aureus in a university hospital setting by using novel software for spa repeat determination and database management. J Clin Microbiol. 41, 5442–5448 (2003).
Gilot, P., Lina, G., Cochard, T. & Poutrel, B. Analysis of the genetic variability of genes encoding the RNA III-activating components Agr and TRAP in a population of Staphylococcus aureus strains isolated from cows with mastitis. J Clin Microbiol. 40, 4060–4067 (2002).
Cramton, S. E., Gerke, C., Schnell, N. F., Nichols, W. W. & Gotz, F. The intercellular adhesion (ica) locus is present in Staphylococcus aureus and is required for biofilm formation. Infect Immun. 67, 5427–5433 (1999).
Verdier, I. et al. Identification of the capsular polysaccharides in Staphylococcus aureus clinical isolates by PCR and agglutination tests. J Clin Microbiol. 45, 725–729, https://doi.org/10.1128/JCM.01572-06 (2007).
Booth, M. C., Pence, L. M., Mahasreshti, P., Callegan, M. C. & Gilmore, M. S. Clonal associations among Staphylococcus aureus isolates from various sites of infection. Infect Immun. 69, 345–352, https://doi.org/10.1128/iai.69.1.345-352.2001 (2001).
Stutz, K., Stephan, R. & Tasara, T. SpA, ClfA, and FnbA genetic variations lead to Staphaurex test-negative phenotypes in bovine mastitis Staphylococcus aureus isolates. J Clin Microbiol. 49, 638–646, https://doi.org/10.1128/jcm.01148-10 (2011).
Gonano, M., Hein, I., Zangerl, P., Rammelmayr, A. & Wagner, M. Phenotypic and molecular characterization of Staphylococcus aureus strains of veterinary, dairy and human origin. Epidemiol Infect. 137, 688–699, https://doi.org/10.1017/S0950268808001453 (2009).
Performance Standards for Antimicrobial Susceptibility Testing: Twenty-Fourth Informational Supplement. CLSI document M100–S24, Clinical and Laboratory Standards Institute, Wayne, PA (2014).
Buzzola, F. R. et al. Differential abilities of capsulated and noncapsulated Staphylococcus aureus isolates from diverse agr groups to invade mammary epithelial cells. Infect Immun. 75, 886–891, https://doi.org/10.1128/IAI.01215-06 (2007).
Le Marechal, C. et al. Development of serological proteome analysis of mastitis by Staphylococcus aureus in ewes. J Microbiol Methods. 79, 131–136, https://doi.org/10.1016/j.mimet.2009.08.017 (2009).
Trotonda, M. P., Tamber, S., Memmi, G. & Cheung, A. L. MgrA represses biofilm formation in Staphylococcus aureus. Infect Immun. 76, 5645–5654, https://doi.org/10.1128/IAI.00735-08 (2008).
Cramton, S. E., Ulrich, M., Gotz, F. & Doring, G. Anaerobic conditions induce expression of polysaccharide intercellular adhesin in Staphylococcus aureus and Staphylococcus epidermidis. Infect Immun. 69, 4079–4085, https://doi.org/10.1128/IAI.69.6.4079-4085.2001 (2001).
Stepanovic, S. et al. Quantification of biofilm in microtiter plates: overview of testing conditions and practical recommendations for assessment of biofilm production by staphylococci. APMIS 115, 891–899, https://doi.org/10.1111/j.1600-0463.2007.apm_630.x (2007).
Acknowledgements
We thank Monika Deutsch, Karin Siebert-Gulle and Valerie Wagner from our lab for their skillful technical assistance in FTIR spectroscopic measurements, cell culture and internalization assays. We are grateful to Jean C. Lee (Brigham & Women’s Hospital, Harvard Medical School, Boston, MA, USA) for providing us the Reynolds strains and their isogenic mutant strains.
Author information
Authors and Affiliations
Contributions
T.G. and M.E-S. conceived and designed this study. T.G., B.S. and F.W. performed sampling and the experiments. T.G., B.S., D.O.S., F.R.B. and M.E-S. analyzed and interpreted the data. T.G. and M.E-S. wrote the manuscript. All authors reviewed and approved the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Grunert, T., Stessl, B., Wolf, F. et al. Distinct phenotypic traits of Staphylococcus aureus are associated with persistent, contagious bovine intramammary infections. Sci Rep 8, 15968 (2018). https://doi.org/10.1038/s41598-018-34371-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-34371-1
- Springer Nature Limited