Abstract
We present a linear theory of lipid membranes which accommodates the effects of intra-membrane viscosity into the model of deformations. Within the Monge parameterization, a linearized version of the shape equation describing membrane morphology is derived. Admissible boundary conditions are taken from the existing non-linear model but reformulated and adopted to the present framework. We obtain a complete analytical expression illustrating the deformations of lipid membrane subjected to the influences of intra-membrane viscosity. The result predicts wrinkle phenomena in the event of membrane-substrate interactions. Finally, we mention that the obtained solutions reduce to those from the classical shape equation when the viscosity effects are removed.
Similar content being viewed by others
Introduction
Lipid membranes are composed of transversely oriented lipid molecules (phospholipids) containing hydrophilic head groups and hydrophobic tails. These phospholipids arrange themselves into a two-layered sheet (a bilayer) with opposing orientations that effectively shield the tail groups from the surrounding aqueous solution. It was found by Evert Gorter and F. Grendel1 that a lipid bilayer constitutes cell membrane and later, in 1959, David Robertson2 justified that the bilayer structure is characteristic of all biological membranes (biomembranes). They are quite fragile and negligibly thin (typically 512610 nm) but represent a critically important interface within biological cells mediating interactions between cells and their surrounding environment through cellular processes such as fission, fusion and budding3,4. These cellular functions are facilitated by the membranes morphological transition which is also dependent on the membrane forces and surrounding viscous flows5,6. Therefore, the study of the morphological aspects of membranes is crucial to the understanding of a wide range of essential cellular functions.
Due to the delicate and complex nature of lipid membranes, the study of the various mechanical responses of lipid bilayers can be, most often, practiced with the use of an artificial ‘model’. This also includes the development of continuum-based models in the description of the behavior of lipid bilayers, typically based on the Cosserat theory of elastic surfaces (see7, and the reference therein). Within this context, the work in8 reveals that intra-membrane viscosity has considerable effects on the deformation of lipid membranes. The authors in9 develop the non-linear models of membranes which incorporates intra-membrane viscosity through the adjustment of the equilibrium and boundary conditions based on the conventional theory of elastic surfaces. There the authors further corroborate the relations between lipid flow and membrane shape via the numerical analysis of the resulting coupled PDEs. However, the analysis presented in9 is limited to the case of a rectangular portion of lipid membranes whereas membrane morphology is oftentimes affected by external forces such as interactions between membranes and substrates. In addition, it seems necessary to develop a linear theory in order to facilitate further researches on the related subjects.
In the present work, we reformulate the non-linear governing equations of membranes directly from the membrane free-energy density function within the frame work of tensor analysis of surfaces. A compatible linear theory is then developed by employing the principles of superposed incremental deformations. More importantly, we obtained complete analytical solutions which describes the deformations of lipid membranes subjected to intra-membrane viscous flow. Emphasis is placed on the assimilation of the complex nature of boundary forces and the film/substrate interaction conditions, while maintaining the rigor and sufficient generality in the derivation of linearized shape equations and boundary conditions. The obtained linear model predicts the straining effects when a rectangular portion of membranes is subjected to intra-membrane viscous flow. The result is also aligned with the numerical study conducted under the compatible settings9. In particular, we find that the viscous flows give rise to wrinkle phenomena when the membrane makes contact with a circular substrate. Quantitative comparison is made by assimilating the experimental results reported in10 where we find that the number of wrinkles is sensitive to the thickness of membranes and the radius of interacting substrates. A phenomenologically compatible result is also reported in the work of11,12. We believe that the results may provide an important breakthrough in the study of relevant problems such as the effects of wrinkles on the vesicle fusion/diffusion processes13 and a wrinkle-caused disease of human eyes (e.g. a macular pucker/epiretinal membrane)14. Lastly, we note that our solution also accommodates the scenarios presented in15,16 in the limit of vanishing viscous flow.
Throughout the paper, we make use of a number of well-established symbols and conventions. Thus, unless otherwise stated, Greek indices take the values 1 and 2. Einstein summation is applied for the repeated indices.
Prerequisite
The theoretical formulation of a lipid membrane which accounts for the effects of intra-membrane viscosity is presented in9. There the authors obtain the constitutive relation from the theory of an elastic surface via the relation W = ρF and later by substituting viscosity terms in the resulting formulae. In this section, we reformulate the results directly from the membrane free-energy density W = W(H, K, ρ) for the sake of consistency and completeness.
Inviscid lipid membranes
The equilibrium state of a purely elastic surface, subjected to normal pressure p, is given by17
where Tα and n are the stress vectors and the local surface unit normal, respectively. The semi-colon denotes the surface covariant differentiation associated with the Levi-Civita contraction of the surface metric aαβ = aα · aβ, where aα = r,α(θα, t) = ∂r/∂θα. For instance, aα;β is defined by18
Here \({{\rm{\Gamma }}}_{\alpha \beta }^{\lambda }={{\bf{a}}}_{\alpha ,\beta }\cdot {{\bf{a}}}^{\lambda }\) are the Christoffel symbols induced by the local surface coordinate \({\bf{n}}({\theta }^{\alpha })=\tfrac{1}{2}{\varepsilon }^{\alpha \beta }{{\bf{a}}}_{\alpha }\times {{\bf{a}}}_{\beta }\) such that n is a unit-vector field and aα and aβ are the tangent vectors on the deformed surface ω. \({\varepsilon }^{\alpha \beta }={e}^{\alpha \beta }/\sqrt{a}\) refers to the permutation tensor with a = det(aαβ). Thus, for example, we evaluate eαβ as e11 = e22 = 0, e12 = −e21 = 1. The matrix aαβ of the surface metric is a positive-definite, with a > 0, leading to the existence of dual metric aαβ which is the inverse of the surface metric (i.e. aαβ = (aαβ)−1). Hence the to the dual basis is given as aα = aαβaβ. The energy induced on the membranes’ deformations can be expressed via the two primary parameters: the coefficient of the first fundamental form aαβ (the surface metric); and the second fundamental form bαβ (the curvature) defined by bαβ = n · aα,β. For example, in the case of the surface with energy density W = W(aαβ, bαβ), Tα take the following compact form17
where
In the above, ρ and γ are the surface mass density and the constitutively-indeterminate Lagrange-multiplier field, respectively. For lipid membranes whose free-energy density is expressed by the mean and Gaussian curvatures through aαβ and bαβ (i.e. W = W(H, K, ρ;aαβ, bαβ)), the expressions of σβα and Mβα can be obtained by using chain rules \(({\rm{e}}.\,{\rm{g}}.\,\frac{\partial W}{\partial {a}_{\alpha \beta }}=\frac{\partial W}{\partial \rho }\frac{\partial \rho }{\partial {a}_{\alpha \beta }}+\frac{\partial W}{\partial H}\frac{\partial H}{\partial {a}_{\alpha \beta }}+\frac{\partial W}{\partial K}\frac{\partial K}{\partial {a}_{\alpha \beta }})\). To see this, we evaluate
where the expressions of H and K are given explicitly as18
The derivatives of ρ, H and K with respect to aαβ can be evaluated as17
Accordingly, Eq. (5) reduces to
Now using Fρ = (W/ρ − γ/ρ),ρ \(({\rm{i}}.\,{\rm{e}}.\,{W}_{\rho }=\rho {F}_{\rho }+\frac{W}{\rho }+{\gamma }_{\rho }-\frac{\gamma }{\rho })\), we obtain
where Fρ is defined via the relation ρ2Fρ = γ17 and \({b}^{\alpha \beta }=2H{a}^{\alpha \beta }-{\tilde{b}}^{\alpha \beta }\). Thus,
and
Similarly, by using
Eq. (4)2 yields
Consequently, by substituting Eq. (3) into (1), we have
Applying Euclidean dot products in normal n and tangential aα directions and invoking Gauss and Weingarten equations18 aβ;α = bβαn and n,α = −bαβaβ, Eq. (14) becomes
and
Eq. (15) is often referred to as a membrane shape equation when used in conjunction with Helfrich potential19.
Viscous lipid membranes
Viscous stress arises due to the straining effects of the fluid and can be accommodated by the time derivative of the evolving surface metric20. In a typical environment, where lipid membranes are subjected to morphological transitions, the reference velocity of the system is low and therefore the corresponding Reynolds numbers are sufficiently small21,22. Further, it is widely accepted that lipid membranes are relatively stiff against areal dilation in comparison with bending or shear motions23,24. Thus, in the forthcoming derivations, we adopt the assumption of incompressible fluid and thereby find the expression of the corresponding stress as
where ν is the intra-membrane shear viscosity and the superscript dot \((\dot{\ast })\) refers to the time derivative. Since aαβ = aα · aβ, we find
In convected coordinates, \({\dot{{\bf{a}}}}_{\lambda }\) is defined as17 \({\dot{{\bf{a}}}}_{\lambda }=\partial {\bf{u}}{\boldsymbol{/}}\partial {\theta }^{\lambda }={{\bf{u}}}_{,\lambda }\) where \({\bf{u}}=\dot{{\bf{r}}}={v}^{\alpha }{{\bf{a}}}_{\alpha }+w{\bf{n}}\) is the velocity of a material point on the initial surface. It is now trivial to show that
Now, in view of Eqs (18 and 19), we find
Further, from Eqs (10), (11), (17) and (20), the expression of the viscous stress can be derived as
where we also use the relations: aαλaβμbλμ = bαβ and \({b}^{\alpha \beta }=2H{a}^{\alpha \beta }-{\tilde{b}}^{\alpha \beta }\). The equation of motion (normal direction) of the lipid membrane in the presence of intra-membrane viscosity effects is therefore obtianed from Eqs (13), (15) and (21)
Utilizing the identities \(H=\tfrac{1}{2}{a}^{\alpha \beta }{b}_{\alpha \beta }\), \({b}_{\beta \alpha }={b}_{\alpha }^{\mu }{a}_{\mu \beta }\), \({a}^{\alpha \lambda }{a}_{\lambda \beta }={\delta }_{\beta }^{\alpha }\) and \({a}^{\beta \alpha }K={b}_{\mu }^{\beta }{\tilde{b}}^{\mu \alpha }\) and knowing the fact that covariant derivatives of the dual metric and the covariant cofactor identically vanish \(({\rm{i}}.\,{\rm{e}}{\rm{.}}\,{a}_{;\beta }^{\alpha \beta }=0,\,{\tilde{b}}_{;\beta }^{\alpha \beta }=0)\), the above equation further reduces to
where Δ is the Laplace-Beltrami operator (i.e. Δϕ = ϕ;αβaαβ), defined on the surface.
Similarly, by substituting Eqs (13) and (21) into Eq. (16), we obtain
where \({({v}_{\mu ;\lambda }+{v}_{\lambda ;\mu })}_{;\alpha }={({v}_{\mu ;\lambda }+{v}_{\lambda ;\mu })}_{,\alpha }-({v}_{\beta ;\lambda }+{v}_{\lambda ;\beta }){{\rm{\Gamma }}}_{\mu \alpha }^{\beta }-({v}_{\beta ;\mu }+{v}_{\mu ;\beta }){{\rm{\Gamma }}}_{\lambda \alpha }^{\beta }\). Invoking \({b}^{\beta \alpha }={b}_{\lambda }^{\alpha }{a}^{\lambda \beta }\) and γ,α = −λ,α − WKK,α − WHH,α, Eq. (24) further reduces to
Since aβα ≠ 0, the above becomes
which serves as the tangential equations of motion.
In the case of uniform membranes of the Helfrich type, the energy density W is defined by
where k and \(\bar{k}\) are empirical constants (the bending moduli). It is noted here that, within the framework of the forgoing model, membranes with continuously distributed proteins can be accommodated through the energy density function: \(W=k(\sigma ){H}^{2}+\bar{k}(\sigma )K;\sigma ({\theta }^{\alpha },t)\), where σ(θα, t) describes the areal concentration of proteins on the membrane surface. However, the case is excluded from the present study in an effort to obtain mathematically tractable equations. Now, Eqs (23) and (26) yield
while Eq. (25) remains intact. From the incompressibility condition \(\dot{J}/J=\frac{1}{2}{a}^{\alpha \beta }{a}_{\alpha \beta }=0\)25 together with Eqs (6) and (20), we also find
Finally, admissible boundary conditions (e.g., boundary forces f and moments M on ∂ω) of Eq. (27) are derived in detail in9 and15. In the case of uniform viscous Helfrich membranes, these are given by
and corner forces
where
are the normal curvatures of ω in the direction of ν and τ and τ is the twist of w on the (ν, τ) axes with τ = n × ν. We note here that the normal force remains intact in the presence of intra-membrane viscosity effects.
Monge Representation and Superposed Incremental Deformations
In order to study the responses of the membrane, we use the Monge representation with space vector r(θα, t) representing material points on the membrane surface w, which is given by
where θ(θα) is position on a plane defined by the unit normal k and z(θ, t) is height function that describes the bilayer membrane mid-plane shape. The Monge representation is an approximation of out-of-plane deformations in which no folds of the membrane are allowed, and hence, z(θ, t) is restricted to a single-valued function. For instance, the membrane surface can be represented by orthonormal Cartesian basis θ = θαeα and, unless otherwise specified, the subscripts of the surface coordinates are dropped and replaced by 1 = x, 2 = y for convenience. Within this setting, we compute
where is δαβ Kronecker delta, \(\nabla z={z}_{,\alpha }{{\bf{e}}}_{\alpha }\) is the gradient evaluated on the surface and is b curvature tensor with components \({b}_{\alpha \beta }={z}_{,\alpha \beta }/\sqrt{a}\),
Further, the normal velocity and the Christoffel symbols are computed as
and
respectively.
The evaluation of the resulting shape equation Eq. (27) in terms of Eqs (33–36) furnishes a highly nonlinear PDE system, which most often requires heavy computational resources. Instead, a means of ‘admissible linearization’ can be employed to make the system mathematically tractable with minimum loss of generality. The concept of the superposed incremental deformations has been widely and successfully adopted in the relevant subject of studies (see, for example15,26,27,28). Within this prescription, the derivatives of z(θ, t) of all orders are considered to be ‘small’ (e.g. \({z}_{,\alpha }\ll 1.\)), and therefore, their products can be neglected. Accordingly, using the notation ‘\(\simeq \)’ to identify equations to the leading order in z, we have
where the subscript \({(\ast )}_{p}\) refers to the projected counterparts of \((\,\ast \,)\) on the coordinate plane ωp, \({\nabla }_{p}^{2}z={z}_{,\alpha \beta }\) \({{\bf{e}}}_{\alpha }\otimes {{\bf{e}}}_{\beta }\) is the second gradient and \({{\rm{\Delta }}}_{p}z=tr({\nabla }_{p}^{2}z)\) is the corresponding Laplacian, respectively. In particular, the straining (20), viscous stress (17) and incompressibility condition (28) can be approximated as:
and
Further, the equations of motion in normal and tangential directions (Eqs (25) and (27)) can be approximated as
In view of, Eqs (38) and (40) (e.g. z,αβ = z,βα, \({v}_{\lambda ,\alpha \lambda }={v}_{\lambda ,\lambda \alpha }\ldots \)), the above are equivalent to
To obtain simplified edge conditions, let r(S, t) = r(θ(S), t), where θ(S) is the arc length parameterization of the projected curve ∂ωp on the plane ωp. Thus, under the Monge representation, we obtain
Eq. (43) is equivalent to
Accordingly, Eqs (43 and 44). yield
Comparing the right sides of Eqs (45 and 46) and referring Eq. (37), we have
where νp = τp × k is the unit normal to the projected curve. Consequently,
where (να)p = eα · vp and (να)p = eα · τp and eαβ is the unit alternator defined by e12 = −e21 = 1 and e11 = e22 = 0. Also, invoking Eqs (31), (37), (47) and (48), becomes
and similarly for
Therefore, it follows from Eqs (29)1–3 and (49–51) that
and
Now, the normal force is given by
where \({(\ast )}_{,\nu }\) is the normal derivatives on ∂ω. By Eq. (46), the arclength derivatives satisfies the approximation
Here \(\tau ^{\prime} (S)=\frac{d\tau }{dS}=\frac{d\tau }{d{\boldsymbol{\theta }}}\cdot \frac{d{\boldsymbol{\theta }}}{dS}={\nabla }_{p}\tau \cdot {\tau }_{p}\) is the arclength derivative on the projected curve. In addition, H,ν can be re-written as
Consequently, by substituting Eqs (37) and (48) into Eq. (56), the leading order expression of H,α can be evaluated on the projected plane as
and therefore, we obtain
Solutions to the Linearized Systems
Consider the case where the membrane flows over a rectangular portion of the plane \((\frac{-\,a}{2}\le x\le \frac{a}{2},\frac{-\,b}{2}\le y\le \frac{b}{2})\). Driven by the controlled force, lipid molecules are flowing in through the boundary, while the boundary remains clamped. The corresponding kinematic edge conditions are
In view of Eq. (37), the later implies that \(\nabla z=0\) and thus both z,α and the normal derivative z,ν = ναz,α vanish on the boundary. Accordingly, the edge moment acting on the boundary Eq. (52)1 becomes
It is suggested that the kinetic conditions lead to the particular set of the tangential and normal force (i.e. fτ = 0, fν = −q), where q is prescribed surface pressure (see, for example9 and29). Thus, Eq. (52)2–3 furnishes
where the approximations have been made via Eq. (53). We note here that, up to leading order, membrane shape has negligible effects on the surface flow, whereas shape is influenced by the flow via the viscosity term in the shape equation (41). Admissible linearization thus reduces the non-linear, fully coupled equations to a system of PDEs with one-way coupling. Further, the present model incorporates the purely elastic theory of lipid membranes in the limit of vanishing viscosity \(\nu \simeq 0\);
while the kinetic boundary conditions remain intact (i.e. z = 0 and \({\nabla }_{p}z=0\)). This observation, in turn, suggests that the decay of a dynamic solution to a purely elastic solution can be investigated by assigning q(t) on the boundary in a way that
where q(θα, t) is understood as a uniform function assigned in the interior (i.e. q(θα, t) = q(θα)) at each sequential step. Then the equations of motion Eq. (42) and boundary conditions Eq. (61) become
and
respectively, where q is prescribed.
Within the domain of interest (a rectangular portion), we now create a particular set of intra-surface viscous flow as
Since vx,x + vy,y = A − A = 0 and either τxνy or τyνx vanishes on the boundary (e.g. for \(x=\frac{a}{2}\) boundary, νp = −vxe1 and τp = −τye2 so that vy = τx = 0), Eqs (64)3 and (65)1 are automatically satisfied. Similarly, it follows from Eqs (65 and 66) that
which agree with the elastic boundary condition in the limit as discussed in25,26,27,28,29 (i.e. λ = −q). In addition, Eq. (64)2 is also satisfied because both \({\bar{\lambda }}_{,\gamma }\) and Δvγ vanish identically (e.g. Δvx = Δ(Ax) = 0 and \({\bar{\lambda }}_{,x}=0.\)). Consequently, the systems of coupled PDEs (64-65) now reduces to
In the case of vanishing p, the solution of the above is obtained by30
where
By imposing boundary conditions (68)2–4, the unknown constants (e.g. An, Bn etc…) can be completely determined. Here, we omit details for the sake of conciseness which can be found in16. The value of intramembrane surface viscosity ν = 10−4 pN · s/nm and the bending modulus of the membrane k = 82 pN · nm are adopted from the work of31 and32, respectively. We also note that the data are obtained under the normalized setting unless otherwise specified. It is clear from Fig. 1 that membrane shape is influenced by the viscous flow via the viscosity term in the shape equation (64)1. More precisely, the applied flow gives rise to straining effects on the membrane shape (Fig. 1) in lateral direction. The corresponding transverse deflections (Fig. 2) decrease with the increasing velocity field of viscous flow. In addition, Fig. 2a illustrates that the obtained solutions accommodate those presented in16 in the limit of vanishing viscous flow. This can also be seen by the reduction of Eq. (64)1 to the classical shape equation (see, for example15,16) in the absence of viscous flow (i.e. vα = 0) and so too the corresponding solutions.
Membrane-Substrate Interactions in the Presence of Viscous Flow
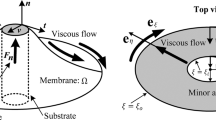
In light of the foregoing discussions, we now investigate a membrane-substrate interaction problem under the influence of intra-membrane viscosity. Driven by the bi-normal (transverse direction) force fn, the interaction occurs on the boundary r = a of an annular portion of the membrane, while it remains clamped (i.e. z = 0 and n = k). Under a polar-coordinate parametrization of the Monge plane, Eq. (32) is replaced by
where r is the radius and θ is the azimuth; er is the usual radial unit vector at azimuth θ. We take {θ1, θ2} = {r, θ} and recast Eq. (33) as
Further, the normal velocity and the curvature tensor are given by
where
By extracting leading order terms, Eqs (72–74) yield
and the incompressibility condition Eq. (28) becomes
The equations of motion (42) and boundary conditions (61) are approximated as
and
Lastly, on the interacting boundary, we have from15 and28 that
where σ is an empirical wetting constant. In the present case (i.e. νp = −er and τp = eθ), the above condition reduces to
Example 1: Circumferentially dominant viscous flow
We consider a case in which
so that (77)2–3 are met and thus yield \(\bar{\lambda }\simeq 0\) and \(\lambda \simeq -\,q\). Accordingly, in the case of vanishing p, Eqs (77 and 78) become
subjected to
where \({\mu }^{2}=\frac{2\lambda }{k}\). A complete analytical solution of the above PDEs is available via the method presented in33,34,35,36 and is obtained by
and
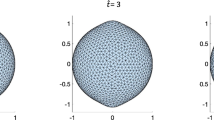
where \({n}^{2}=\frac{4A\nu }{k}+\frac{{m}^{2}}{2}\) and \({q}^{4}=\frac{8{m}^{2}A\nu }{k}\). The solutions are the first and second kinds of the modified Bessel functions and usual Bessel functions of the order n and q with parameter μ, denoted conventionally by, In, Kn, Jn, and Yn, respectively37. Similarly as in the rectangular case, it is found that the intra-membrane viscosity, in the case of circumferentially dominant flow, leads to straining effects on the membrane (Fig. 3) and the resulting transverse deflection is reduced with increasing velocity of viscous flow (Fig. 4a). More importantly, the viscous flow gives rise to wrinkling phenomena when \(\frac{A\nu }{\lambda }\ge {10}^{-9}\). Based on our analysis of a rectangular portion of membranes and the observations from9 where no interactions are considered, we infer that the interaction forces between the membrane and the substrate give rise to wrinkling phenomena. Further clarification of such phenomena is, however, beyond the scope of present study due to the paucity of available data. It is shown in Fig. 4(a) that both the amplitude of wrinkles and the magnitude of transverse deflections decay away as they approach to far field boundary. Also, Fig. 4(b) illustrates that the solutions from the proposed model (dot lines) reduce to those predicted by the existing model (solid lines)15 in the limit of vanishing viscous flow (i.e. \(A\simeq 0\)). In other words, the presented solution is general enough to accommodate the particular solution where the viscous effects are not integrated.
Example 2: Radially dominant flow
In this section, radially dominant flow is considered where we have
Similar to the previous case, Eqs (77 and 78), together with the above yield
Again, using the methods in33,34,35,36, the solution of Eq. (88) is obtained by the following explicit form;
and
where and \({s}^{2}=\frac{{m}^{2}}{2}\) and t2 = mμ. Similar to the circular case, radially arranged wrinkles begin to form as the parameter exceeds the critical value of \(\frac{A\nu }{\lambda }\ge 3\times {10}^{-10}\) and they vanish at the remote boundary. The number of radial wrinkles increases with the growing effects of viscous flow (Fig. 5) and with larger radius of an inner circle. A similar tendency can be found in the work of10 (See, Fig. 6) where the authors measure the number of wrinkles on thin polymer films under the compatible settings as considered in the present work. In addition, we assimilate the experiments in10 by using the proposed analytical model and present the results in Fig. 7. Although, the obtained model is not intended for the analysis of thin polymer films, it still provides reasonable agreement with the results in10 (see, Fig. 7) that the number of wrinkles is sensitive to both the thickness ‘t’ (inversely proportional) and the inner radius ‘a’ (proportional). The results are also align with the theoretical developments regarding to finely wrinkled states of the membrane via the minimization of the strain-energy function and by its quasi-convexification11,12. Potential applications to biomechanics may include monitoring vesicle thickness and enhancing vesicle fusion processes. However, prior to these applications, it would be necessary to further clarify and/or justify the obtained results by employing the aforementioned theory.
Number of wrinkles on thin polymer films10. From [Huang, J. et al. 50–653 (2007)]. Reprinted with permission from AAAS.
Comparison with the experimental result in10.
Data acquisition
Figures in the manuscript are prepared by visualizing analytical solutions presented in the manuscript. For the purpose, a commercial software (MATLAB) is used.
Ethics statement
This work did not involve any collection of human data.
Data accessibility statement
This work does not have any experimental data.
References
Gorter, E. & Grendel, F. On Bimolecular Layers of Lipoids on the Chromocytes of the Blood. J. Exp. Med. 41(4), 439–443 (1925).
Robertson, J. D. The Ultrastructure of Cell Membranes and Their Derivatives. Biochem. Soc. Symp. 16, 3–43 (1959).
Chernomrdik, L. & Kozlov, M. Mechanics of membrane fusion. Nat. Struct. Mol. Biol. 15, 675–683 (2008).
Lenz, M., Morlot, S. & Roux, A. Mechanical requirements for membrane fission: common facts from various examples. FEBS Lett. 583, 3839–3846 (2009).
Bruinsma, R. & Pincus, P. Protein Aggregation in Membranes. Curr. Opin. Solid State Mater. Sci. 1(3), 401–406 (1996).
Benedict, J. R. et al. Aggregation and Vesiculation of Membrane Proteins by Curvature-Mediated Interactions. Nature 447(7143), 461–464 (2007).
Steigmann, D. J. On the relationship between the Cosserat and Kirchhoff–Love theories of elastic shells. Math Mech Solids 4, 275–288 (1999b).
Arroyo, M. & DeSimone, A. Relaxation dynamics of fluid membranes. Phys. Rev. E 79(031915), 1–17 (2009).
Rangamani, P., Agrawal, A., Mandadapu, K. K., Oster, G. & Steigmann, D. J. Interaction between surface shape and intra-surface viscous flow on lipid membranes. Biomech Model Mechanobiol. 12(4), 833–845 (2013).
Huang, J. et al. Capillary wrinkling of floating thin polymer films. Science 317(5838), 650–653 (2007).
Steigmann, D. J. Tension-field theory. Proc. Roy. Soc. Lond. A 429, 141–73 (1990).
Goodbrake, C., Steigmann, D. J. Mechanics of an elastic membrane infused with a liquid. Int. J. Mech. Sci. https://doi.org/10.1016/j.ijmecsci.2017.07.062 (2017).
Cerda, E. & Mahadevan, L. Geometry and physics of wrinkling. Phys. Rev. lett. 90(7), 074302 (2003).
Joshi, M., Agrawal, S. & Christoforidis, J. B. Inflammatory mechanisms of idiopathic epiretinal membrane formation. Mediators Inflamm. https://doi.org/10.1155/2013/192582 (2013).
Agrawal, A. & Steigmann, D. J. Boundary-Value Problems in the Theory of Lipid Membranes. Continuum Mech. Therm. 21(1), 57–82 (2009).
Belay, T., Kim, C. I. & Schiavone, P. Analytical Solution of Lipid Membrane Morphology Subjected to Boundary Forces on the Edges of Rectangular Membrane. Continuum Mech. Therm. 28(1–2), 305–315 (2015).
Steigmann, D. J. Fluid films with curvature elasticity. Arch. Ration. Mech. Anal. 150, 127–52 (1999).
Sokolnikoff, I. S. Tensor Analysis: Theory and Applications. (Wiley, New York, 1951).
Helfrich, W. Elastic properties of lipid bilayers: theory and possible experiments. Z. Naturforsch. 28, 693–703 (1973).
Aris, R. Vectors, Tensors and the Basic Equations of Fluid Mechanics. (Dover, N.Y., 1989).
Seifert, U. Configurations of fluid membranes and vesicles. Adv Phys. 46(1), 13–137 (1997).
Jaric, M., Seifert, U., Wintz, W. & Wortis, M. Vesicular instabilities: the prolate-to-oblate transition and other shape instabilities of fluid bilayer membranes. Phys. Rev. E 52, 6623 (1995).
Evance, E. A. & Skalak, R. Mechanics and thermodynamics of bio membranes. CRC Press, Boca Raton, FL (1980)
Secomb, T. W. & Skalak, R. Surface flow of viscoelastic membranes in viscous fluids. Q. J. Mech. Appl. Math. 35, 233–247 (1982).
Steigmann, D. J., Baesu, E., Rudd, R. E., Belak, J. & McElfresh, M. On the variational theory of cell-membrane equilibria. Interface Free Bound. 5, 357–366 (2003).
Rangamani, P. & Steigmann, D. J. Variable tilt on lipid membranes. Proc. Math. Phys. Eng. Sci. 470(2172), 20140463 (2014).
Belay, T., Kim, C. I. & Schiavone, P. Interaction-induced morphological transitions of lipid membranes in contact with an elliptical cross section of a rigid substrate. J. Appl. Mech. ASME. 83(1), 011001 (2016).
Zeidi, M. & Kim, C. I. Notes on superposed incremental deformations in the mechanics of lipid membranes. Math. Mech. Solids. https://doi.org/10.1177/1081286517734608 (2017).
Kim, C. I. & Steigmann, D. J. Distension-induced gradient capillarity in lipid membranes. Contin. Mech. Thermodyn. 27(4–5), 609–621 (2014).
Imark, C. E. & Gerdemeli, I. An exact solution for the deflection of a clamped rectangular plate under uniform load. Appl. Math. Sci. 1, 2129–2137, https://doi.org/10.1007/s00161-014-0333-1 (2007).
Hochmuth, R. M. & Waugh, R. E. Erythrocyte membrane elasticity and viscosity. Ann. Rev. Physiol. 49, 209–19 (1987).
Derenyi, I., Julicher, F. & Prost, J. Formation and interaction of membrane tubes. Phys. Rev. Lett. 88(23), 238101 (2002).
Read, W. W. Analytical solutions for a helmholtz equation with dirichlet boundary conditions and arbitrary boundaries. Math. Comput. Model. 24(2), 23–34 (1996).
Huang, Yan & Zhang, Xiao-Jin General analytical solution of transverse vibration for orthotropic rectangular thin plates. J. Marine. Sci. and Appl. 1(2), 78–82 (2002).
Read, W. Series solution for laplace equation with nonhomogenious mixed boundary conditions and irregualr boundaries. Math. Comput. Model. 17, 9–19 (1993).
Attarnejad, R. & Ghorbani A. A general fourth order ordinary diffrential equation withj solution in terms of bessel functions: theory and engnering application. Structural Eng. Mech (2007).
Lebedev, N. N. Special functions and their applications. (Dover, New York, 1972).
Acknowledgements
This work was supported by the Natural Sciences and Engineering Research Council of Canada via Grant #RGPIN 04742 and the University of Alberta through a start-up grant. Kim would like to thank Professor David Steigmann for his invaluable and inspirational advice in the subject through his unpublished manuscript.
Author information
Authors and Affiliations
Contributions
The corresponding author (Kim) formulate theory (including mathematical models) and interpreted the results. The first author (Mahdi) solved the resulting equations and prepared the figures.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zeidi, M., Kim, C.I. The Effects of Intra-membrane Viscosity on Lipid Membrane Morphology: Complete Analytical Solution. Sci Rep 8, 12845 (2018). https://doi.org/10.1038/s41598-018-31251-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-31251-6
- Springer Nature Limited