Abstract
We study the morphology of lipid membranes subjected to intra-membrane viscous flows and interactions with elliptical cylinder substrates. From the non-linear theory of elastic surfaces, a linearized shape equation and admissible boundary conditions are formulated in elliptical coordinates via the Monge representation of a surface. In particular, the intra-membrane viscosity terms are linearized and mapped into elliptic coordinates in order to accommodate more general forms of viscous flow. The assimilated viscous flow is characterized by potential functions which satisfies the continuity condition. A complete solution in terms of Mathieu function is then obtained within the prescription of incremental deformations superposed on large. The results describe smooth morphological transitions over the domain of interest and, more importantly, predicts wrinkle formations in the presence of intra-membrane viscous flow in the surface. Lastly, the obtained solution accommodates the results from the circular cases in the limit of vanishing eccentricity and intra-membrane viscous flow.
Similar content being viewed by others
Introduction
The mechanics of lipid membranes has consistently been the subject of intense research for its importance in the understanding of a wide variety of essential cellular processes1,2,3,4,5. Traditionally, it was believed that cells are surrounded by a thin oil-based barrier, yet the structure of this membrane was not well understood. In 1920s, E. Gorter and F. Grendel.6 found that the cell membrane is composed of lipid molecules (phospholipids) which are generally divided into two important groups: the hydrophilic head parts and hydrophobic tail groups. When dispersed into aqueous solutions, lipid molecules are driven by hydrophobic effects to form a unique bilayer structure (a lipid bilayer) with opposing orientations, that maintain symmetry about a mid-surface. In fact, with the advances in electron microscopy, the bilayer structure was identified as a characteristic of all biological membranes (biomembranes)7. Since they are negligibly thin (typically 5–10 nm), and fragile, the study of various aspects of lipid bilayers is often achieved by employing mathematical models in order to overcome the formidable difficulties of experimental studies. Also, from a mechanical perspective, the responses of a lipid membrane can be idealized as a thin elastic film. Within this context, the development of theoretical models describing the behavior of lipid bilayers has greatly benefited from the differential geometry of a surface and the theory of an elastic surface such that the deformation of energy of a thin membrane can be expressed by the mean and Gaussian curvatures of a surface8,9. In particular, Helfrich proposed a well-known Helfrich energy potential1 which addresses the symmetry of lipid bilayers and further ensures the resulting equilibrium state to be energy minimizing10. This, together with the use of variational principle, and the virtual-work statement, furnish the Euler-Lagrange equations also known as “membrane shape equations”, which have been successfully adopted in a wide range of problems (see, for example11,12,13). The variations of the classical Helfrich model have been continuously investigated in order to provide more efficient descriptions of lipid membranes’ morphology induce by various cellular activities, such as distensions12, tilts14, buddings15,16, spontaneous curvatures11 and substrate interactions17,18,19.
The study of the mechanical responses of membranes under the influences of intra-membrane viscous flow are of particular mechanical interest due to its importance in the explanation of essential cellular functions including budding, fission and vesicle formations20,21,22,23. The theoretical frame work accounting for the effects of intra-membrane viscosity into the model of membrane deformations has been established in24. In there, authors reveal that the dynamics of the membrane system is notably influenced by the presence of intra-membrane viscous flow. The authors in25 developed the comprehensive non-linear model of membranes incorporating the effects of intra-membrane viscosity from the elastic model of surfaces26,27. To this end, authors in28 discussed a compatible linear model within the setting of superposed incremental deformations. However, the analysis presented in28 is limited to certain types of problems where viscous flow is characterized as either constant or simple linear functions, and the interaction occurs through a circular contact region to obtain a mathematically tractable system. In a typical environment, a lipid membrane system is involved in more complex processes4,5 (e.g. interactions through a non-circular domain and the influences from multi-source viscous flows). Therefore, the development of a more comprehensive model may be necessary to promote researches on the related subjects.
In the present work, we study the deformations of lipid membranes interacting with intra-membrane viscous flow and an elliptical cylinder substrate. Utilizing the Monge parameterization of a surface and general curvilinear coordinates, the expressions of linearized shape equation and associated boundary conditions are obtained from the non-liner theory25. The intra-membrane viscosity terms are formulated by means of ‘admissible linearization’ and successively transformed into elliptical coordinates to assimilate more general types of viscous flow. More importantly, we obtained a complete analytic solution by employing adapted iterative reduction and the method of eigenfunction expansion29,30,31, which describes the deformations of lipid membranes when interacting with intra-membrane viscous flow and an elliptical-cross-section substrate. It is found that intra-membrane viscosity induces wrinkle formations of the lipid membrane and the corresponding number of wrinkles exhibits sensitivity to both the radius of the ellipse and the intensity of viscous flow. Comparisons with phenomenologically compatible cases such as a circular substrate -membrane interactions and capillary wrinkle of polymer films, are made where the proposed model successfully reproduces the results from28,32 in the limit of vanishing eccentricity of an ellipse. Further, we obtain solutions corresponding to the case of a lipid membrane subjected to non-uniform viscous flows and dual source flows. This is facilitated by the relaxed form of the prescribed tangential and normal force, and the condition of continuity along and within the elliptical boundaries, unlike those arising in circular boundaries where the admissible set of viscous flows are strictly uniform in one of the coordinate directions28. The resulting deformation fields show clear signs of dual source interference in that both the radial and circumferential wave forms are simultaneously predicted. Case study vis a vis morphologically similar results of shape memory films33 are presented to investigate the potential applicability of the proposed model in the analysis of different types of membrane. In particular, it is found that the principles of superposition from linear elasticity remains valid, even in the presence of general forms of dual source viscous potentials. That is the solution of a dual source problem can be directly obtained by adding solutions of two single source problems. The solutions presented here are of more practical interest in that, essentially, they lead to solutions of problems in which the viscosity effects are characterized by a wide class of potential functions and so can accommodate a correspondingly large set of physically relevant problems. For example, potential applications may be expected in the study of wrinkle-caused disease (e.g. a macular epiretinal membrane34) and the influences of membrane viscosity on various cellular functions such as fusion, fission and budding35. Further, the presented solution reproduces the existing results17 when viscosity effects are removed, and does incorporate the solution of the classical membrane-substrate interaction problem11 in the limit of vanishing eccentricity. In fact, the classical solution obtained directly from the proposed model produces more accurate predictions by identifying the additional Bessel functions, which is reduced from the Mathieu potentials.
Throughout the paper, we make use of a number of well-established symbols and conventions. Thus, unless otherwise stated, Greek indices take the values in {\(1,2\)} and, when repeated, are summed over their ranges. Lastly, \({(\ast )}_{,\alpha }\) denotes the derivative of ‘\(\ast \)’ with respect to a coordinate \({\theta }^{\alpha }\) and \({W}_{K}\) stands for the derivatives of a scalar-valued function \(W(K)\) with respect to the parameter \(k\).
Viscous Lipid Membranes
In the study of the mechanics of lipid bilayer membranes, it is widely accepted that lipid membranes can be regarded as a continuous elastic surface. Within this idealization, the mechanical responses of a lipid membrane can be modeled via the theory of an elastic surface. In this section, we reformulate the results in the present context for the sake of clarity and completeness, and for the use in the derivation of the compatible linear model. The original derivation concerning the viscous elastic membranes can be found in25.
The equilibrium state of a purely elastic surface, in the presence of normal pressure \(p\), is given by26
Here, \({{\bf{T}}}^{\alpha }\) and \({\bf{n}}\) are the stress vectors and the local surface unit normal, respectively, and the semi-colon denotes the surface covariant differentiation in the sense of the Levi-Civita connection on a surface. The associated surface metric is defined as
where \({{\bf{a}}}_{\alpha }={{\bf{r}}}_{,\alpha }({\theta }^{\alpha },t)\) are the tangent vectors to the surface \(\omega \) induced by the parameterization \({\bf{r}}({\theta }^{\alpha },t),\) the position in \({{\mathbb{R}}}^{3}\) of a point on the surface with coordinate \({\theta }^{\alpha }\). The unit vector field n, which serves as the local surface orientation, is then computed as \({\bf{n}}{\boldsymbol{(}}{\theta }^{\alpha })=\tfrac{1}{2}{\varepsilon }^{\alpha \beta }{{\bf{a}}}_{\alpha }\times {{\bf{a}}}_{\beta }\), where \({\varepsilon }^{\alpha \beta }={e}^{\alpha \beta }/\sqrt{a}\) refers to the permutation tensor density with \(a={\rm{\det }}({a}_{\alpha \beta })\) and \({e}^{11}={e}^{22}=0,{e}^{12}=-\,{e}^{21}=1\). The matrix of the surface metric is a positive-definite (i.e. \(a > 0\)), which further suggests the existence of dual metric \({a}^{\alpha \beta }\), the inverse of the metric \({a}_{\alpha \beta }\) (i.e. \({a}^{\alpha \beta }={({a}_{\alpha \beta })}^{-1}\)). Thus, the dual basis (contravariant basis) can defined as \({{\bf{a}}}^{\alpha }={a}^{\alpha \beta }{{\bf{a}}}_{\beta }\). Combining the above results, the covariant differentiation of the surface covariant is then computed as36
where \({\Gamma }_{\alpha \beta }^{\lambda }={{\bf{a}}}_{\alpha ,\beta }\cdot {{\bf{a}}}^{\lambda }\) are the Christoffel symbols induced by the local surface coordinate. These results furnish the well-known Gauss and Weingarten equations:
where \({b}_{\alpha \beta }\) are the coefficients of the second fundamental form of surface and its covariant cofactor is defined by
The deformation energy of an elastic surface can be expressed via the above two primary parameters: the coefficient of the first fundamental form \({a}_{\alpha \beta }\) and the second fundamental form \({b}_{\alpha \beta }\)8,9,26. Thus, for example, an elastic surface whose free-energy density is expressed by the mean and Gaussian curvatures through \({a}_{\alpha \beta }\) and \({b}_{\alpha \beta }\) (i.e. \(W=W(H,K,\rho ;{a}_{\alpha \beta },{b}_{\alpha \beta })\)), \({{\bf{T}}}^{\alpha }\) takes the following compact form26:
where
and \(\lambda \) is the constitutively-indeterminate Lagrange-multiplier field. The corresponding mean and Gaussian curvatures are computed as36
which also satisfy the following equalities
Now, the viscous stress induced by the straining effects of the fluid is given by37
where \(\nu \) is the intra-membrane shear viscosity and
is the time derivative of the evolving surface metric. Thus, in order to compute viscous stress, it is required to compute \({\dot{{\bf{a}}}}_{\mu }\), which can be obtained via the material time derivative of a position vector \({\bf{r}}\)26:
Accordingly, it is found that
and
where \({v}^{\alpha }=\partial {\theta }^{\alpha }/\partial t,\) and \({{\bf{r}}}_{t}=|{{\bf{r}}}_{t}|{\bf{n}}=w{\bf{n}}\) are respectively the tangential and normal velocities of a material point on the initial surface26,37,38.
It is now straightforward to show from Eqs. (8), (12) and (16) that,
which is the expression of the viscous stress.
Thus, by means of Eqs. (9), (11)2 and (17), and applying the conventional Euclidean dot product in normal \({\bf{n}}\) direction, Eq. (1) becomes25
which serves as the equation of motion (normal direction) of the lipid membrane in the presence of intra-membrane viscosity effects. Further, Δ is the Laplace-Beltrami operator (i.e. \(\Delta \phi ={\phi }_{;\alpha \beta }{a}^{\alpha \beta }\)) on the surface \(\Omega \). Consequently, by projecting Eq. (1) onto the basis coordinate plane of \({{\bf{a}}}_{\alpha }\), the following tangential equations of motion can be obtained:
Much of literature on the mechanics of lipid membranes has revealed that a bilayer membrane can be regarded as a continuous two-dimensional elastic surface where the response functions are governed by the well-known Helfrich energy potential1. The model has been widely adopted in various subjects within bilayer membrane mechanics (see, for example11,12,15, and the references therein). Following the work of25, in this paper, we consider a symmetric membrane of Helfich type (i.e. \(W(H,K)=W(\,-\,H,K)\)), subjected to the membrane-substrate interactions and the effects of intra-membrane viscosity. The corresponding free-energy density function is defined by
where \(k\) and \(\bar{k}\) are empirical bending constants, which pertain to lipid membranes with uniform properties. Thus, from Eqs. (18) and (20), becomes
while the tangential equations (Eq. (19)) remain intact.
Lastly, by invoking Eq. (16), the condition of an incompressible fluid \(\dot{J}/J=\frac{1}{2}{a}^{\alpha \beta }{\dot{a}}_{\alpha \beta }=0\) can be obtained as39
where \(H=\tfrac{1}{2}{a}^{\alpha \beta }{b}_{\alpha \beta }\).
Incremental Deformations of Lipid Membranes
The use of Monge parameterization and admissible linearization is a widely adopted methodology for lipid membrane analysis, and the associated procedures are well documented in the literature (see, for example11,15,18). Here, we reformulate the results for the sake of completeness. Under the Monge parameterization, material points on the membrane surface \(\Omega \) is defined by
where \({\boldsymbol{\theta }}({\theta }^{\alpha })\) is position on a plane \(p\) with unit normal k. The problem of determining the membranes’ deformed configuration is then reduced to solving a single function \(z({\boldsymbol{\theta }},t)\). In the cases of Cartesian coordinates, we have
where \(\{{{\bf{e}}}_{\alpha }\}\) is an orthonormal basis for the plane and, the subscripts of the surface coordinates are dropped and replaced by \(1=x,2=y\), unless otherwise specified. Accordingly, we compute
Here, \(\nabla z={z}_{,\alpha }{{\bf{e}}}_{\alpha }\) is surface gradient, \({\delta }_{\alpha \beta }\) is Kronecker delta and \({\bf{b}}\) is the curvature tensor. Further, the expressions of the dual basis and the Christoffel symbols are obtained as
In the incremental deformation analysis, it is assumed that the gradient of \(z({\theta }^{\alpha },t)\) of all orders are ‘small’ so that their products can be neglected. The procedure is commonly referred to as admissible linearization through which the geometrical and kinematical quantities associated with the surface (Eq. (19)) can be approximated as
where the subscript \({(\ast )}_{p}\) refers to the projected counterparts of \((\ast )\) on the coordinate plane \({\omega }_{p}\), \({\nabla }_{p}^{2}z={z}_{,\alpha \beta }\) \({{\bf{e}}}_{\alpha }\otimes {{\bf{e}}}_{\beta }\) is the second gradient, and \({\Delta }_{p}z=tr({\nabla }_{p}^{2}z)\) is the corresponding Laplacian, respectively.
Linearization of the intra-membrane viscosity terms
In the forthcoming derivations, we present the linearization procedures for the terms associated with the intra-surface viscous flow, which arise in the formulation of membrane equilibrium equations. To proceed, we express the surface gradient of the viscous flow fields and the curvature tensor as
We then compute their traces to obtain
where \({{\bf{a}}}_{\alpha }\cdot {{\bf{a}}}^{\beta }={\delta }_{\alpha }^{\beta }\). Also, from the results in Eqs. (27) and (28) can be approximated, up to the leading order, to
Now, combining Eqs. (29) and (30), we find that,
Thus, Eq. (29) simplifies to
However, since \({z}_{,\alpha \beta }={z}_{,\beta \alpha }\), the above can be re-written as
To obtain the simplified expression of incompressibility condition (22), we evaluate the surface divergence of the viscous flow field as
where \({{\bf{a}}}^{\alpha }\cdot {{\bf{a}}}^{\beta }={{\bf{a}}}_{\lambda }{a}^{\lambda \alpha }\cdot {{\bf{a}}}_{\gamma }{a}^{\lambda \alpha }={a}_{\lambda \gamma }{a}^{\lambda \alpha }{a}^{\lambda \alpha }={a}^{\alpha \beta }\). Substituting the above into Eq. (22), and further invoking Eq. (14), we arrive at
Thus, from Eq. (27), the leading order approximation of the above can be found as
where \({({\rm{div}}{\bf{v}})}_{P}\) is the divergence of the projected coordinate plane \({\Omega }_{p}\).
Consequently, substitution of these linearized expressions, (Eqs. (27), (33) and (36)), into Eqs. (19, 21 and 22) delivers the following normal and tangential equations, and incompressibility conditions:
Here, \(P=P({\theta }^{\alpha },t)\) is understood as a sequence of prescribed surface pressure (see25) from the admissible set of boundary forces which satisfy
where \({\boldsymbol{\gamma }}\times {\boldsymbol{\tau }}={\bf{n}}\). In particular, the compatible linear forms for the moments and normal interaction forces are given by11
where \(\tau ^{\prime} (S)=\frac{d\tau }{dS}=\frac{d\tau }{d{\boldsymbol{\theta }}}\cdot \frac{d{\boldsymbol{\theta }}}{dS}={\nabla }_{p}\tau \cdot {{\boldsymbol{\tau }}}_{p}\) and σ are the arc length derivative on the projected curve, and the empirical constant accounting for the wetting of the interacting boundary, respectively. Hence, the solution of Eq. (37) can be uniquely determined by imposing the admissible boundary conditions, Eqs. (38) and (39).
Formulations in the elliptical coordinates
We consider the cases when lipid membranes interact through the elliptical contact domain of a transmembrane substrate, and are subjected a general class of intra-membrane viscous flow (see, Fig. 1). The deformations of lipid membranes defined on an elliptical domain can be examined by using the mapping,
such that
through which the rectangular Cartesian coordinates \((x,y)\) are mapped to the elliptical coordinates \((\xi ,\eta )\). The semi focal length \(c\) is defined by \(c=\sqrt{{a}^{2}-{b}^{2}}\) and \(\xi \in [0,\infty )\), and \(\eta \in [0,2\pi ]\) are respectively the radial and angular coordinates. Accordingly, Eqs. (40 and 41) furnish the gradient and Laplacian in elliptical coordinates as
The condition of incompressibility (i.e. \({v}_{\alpha ,\alpha }=0\)) then yields
from which the admissible set of viscous flow field is found to be
so that Eq. (44) is satisfied (i.e. \({v}_{\xi ,\xi }+{v}_{\eta ,\eta }=w(\xi ,\eta )-w(\xi ,\eta )=0\)). In the analysis, we assume \({C}_{1}={C}_{2}=0\) for the sake of simplicity. The cases of non-zero coefficients can be easily accommodated via the principles of superposition which will be discussed in the later section.
The membrane-substrate interaction occurs through the wall of the elliptical substrate where the corresponding domain of interest, \(\Omega \), and interacting boundary, \(\partial \Omega \), are defined respectively as
Using the mapping functions in Eqs. (40 and 41), the associated boundary conditions can be obtained from Eq. (38) such that
Here, the repeated indices, \(\alpha \) and \(\beta \), when summed over their ranges {\(1,2\)}, refer to \(\xi \) and \(\eta \) in elliptic coordinates. On the boundaries (i.e. \(\xi ={\xi }_{o}\) and \(\xi ={\xi }_{i}\)), we find
and thereby reduce Eq. (47) to
In particular, since the membrane-substrate interaction condition (i.e. \({\bf{n}}-\nabla z={\bf{k}}\), see11,13) requires \(\nabla z=0\) at the inner boundary (\(\xi ={\xi }_{i}\)), the normal force (Eq. (39)) becomes
We continue by rewriting \({\nabla }_{p}H\) using Eq. (42) and subsequently reduce Eq. (50) to
where,
Further, applying the similar schemes as in the above, it is not difficult to show
Consequently, by combining the above results, we reformulate Eq. (37) and the associated boundary conditions as
subjected to
Remark 1.
It should be noted that the restrictions on the continuity conditions (\({\rm{div}}({\bf{v}})=0\)) and the prescribed tangential (\({f}_{\tau }=0\)) force can be relaxed along and within the elliptical boundaries unlike those arising in circular cases where the admissible set of viscous flows are required to be strictly uniform in one of the coordinate directions (i.e. either \({v}_{r}=const\) or \({v}_{\theta }=const\)) to satisfy the constraints28. This is mainly due to the confined descriptions of the circular interaction boundary where the rate of change in the unit normal and tangent on the circular boundary remains constant so that the associated normal velocity fields \({v}_{r}\) always points to the center of a circular substrate. Thus, \({v}_{r}\) is required to be vanished by its gradient \({v}_{r,r}\) or gradient of tangential velocity \({v}_{\theta ,\theta }\) to satisfy the continuity condition; i.e.,
Such restriction can be relaxed in the case of the elliptic interaction boundary, since the rate of change in local coordinate is not necessarily constant, yet they vary with respect to the coordinates \(\xi \) and \(\eta \) (see, Eqs. (42 and 43)). This further suggests that the normal velocity field \({v}_{\xi }\) does not necessarily points to the center of an elliptical substrate (see, Fig. 1) and therefore no restrictions are necessary for \({v}_{\xi }\). In results, the associated flow fields (\({v}_{\xi }\) and \({v}_{\eta }\)) can accommodate more general forms such as non-uniform viscous flows and periodic wave form of viscous flows (no need to be strictly constant) without violating the aforementioned constitutive restrictions. Examples regarding these cases will be discussed in the following section.
Lastly, Eqs. (54 and 55) serve as the linearized shape equation system which describes the morphology of lipid membranes under the influences of membrane-substrate interactions and general forms of viscous flows. In the analysis, we also impose \(z({\xi }_{i},\eta )=0\) for the purpose of comparison with the existing literature.
Solutions to the Linearized Systems
It can be seen from Eqs. (42), (43) and (54) that the gradient, Laplacian and the resulting PDEs in elliptical curvilinear coordinate continuously vary with respect to the material points \((\xi ={\xi }_{o},\eta ={\eta }_{o})\) on the membrane surface, where \({\xi }_{o}\) and \({\eta }_{o}\) denote a particular configuration of the surface. In other words, the associated tangential and normal velocities are simultaneously updated as material points move over the membrane surface. Therefore, the solution of the Eq. (54), which is coupled with the viscous velocity fields, cannot be accommodated by the conventional separation variable method of modified Helmholtz equation. In this section, we combine the method of adoptive iteration and the principle of eigenfunction expansions29,30,31, and obtained the complete expression of the membrane’s shape function \(z(\xi ,\eta )\).
To proceed, we assume the solution of the form
Here, \(\varphi (\xi ,\eta )\) is the plane harmonic function, which is chosen as
to accommodate the desired behavior (i.e. \(|\nabla z|\to 0\)) as approaching the boundary. In particular, the unknown potential \(H(\xi ,\eta ,q)\) can be expressed as40
where \(c{e}_{n}(\eta ,q)\) and \(K{e}_{m}(\xi ,q)\) are the modified Mathieu functions of the first and second kind, respectively, and \(q\) (\(q > 0\)) is the associated parameter (see, for example41).
Now, by substituting Eqs. (45) and (59) into Eq. (54), and invoking the orthogonal properties of the Mathieu function,
we obtain the following expressions for \({T}_{mn}\):
The detailed procedures which can be found in29,30,31 are omitted here for the sake of brevity.
Consequently, the general solution in Eq. (57) can be found in the form
where \(\mu =\sqrt{2\lambda /k}\) is the natural length scale which is commonly adopted in the membrane studies (see, for example11,14,25). Details regarding the dimensionless variables adopted in the present work will be discussed in later section. The unknown constants \({A}_{m},{B}_{m},{C}_{m}\) and \({D}_{m}\) can be completely determined by imposing the admissible boundary conditions. For instance, the substrate-membrane interaction conditions (55) require
on the boundary from which we find that,
and
where
Constant viscous flow potential: w(ξ, η) = A
The membrane systems may be exposed to the constant viscous flow within a cell such as the selective transport of molecules inside the lipid bilayers where the molecules travel with the constant flow from one side of the membrane to the objective protein42. To assimilate such constant viscous flow, we consider the case when \(w(\xi ,\eta )=A\), and thereby reduce Eq. (62) to
Equations (63), (65) and (67) then deliver
where
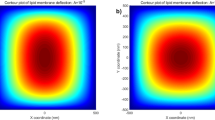
The resulting function \(z(\xi ,\eta )\) in Eq. (68) describes the morphology of a lipid membrane when subjected to membrane-substrate interactions and the effects of uniform intra-membrane viscous flow. The associated results are presented in Figs. 1, 2 and 3. In the assimilation, we adopt the value of intra-membrane surface viscosity \(\nu ={10}^{-4}\,pN\cdot s/nm\) and the flexural modulus of the membrane \(k=82\,pN\cdot nm\) from the work of43,44. The Lagrange multiplier \(\lambda \) is dependent on membrane systems in consideration and usually do not have definite range of values. The values of \(\lambda \) commonly used in the literatures is about \(\lambda \propto {10}^{-4}\,pN/nm\). In the present study, we assimilate data under the normalized setting using the aforementioned values. The dimensionless parameters used in the simulations are adopted from the works11,14,25 as;
Comparisons: Number of wrinkles on thin polymer films32.
We have found that the viscous flow gives rise to wrinkle phenomena, when the normalized magnitude of viscous flow is greater than the critical number (i.e. \(\tfrac{A\nu }{\lambda }\ge {10}^{-15}\)). Especially, the number of radial wrinkles with respect to the intra-membrane viscous flows (A) and the radius of the inner ellipse (μc) are illustrated in Fig. 2. The top two figures indicate that, with the same inner ellipse, the number of wrinkles reduce as the magnitude of viscous flow decreases from \({10}^{-5}\) to \({10}^{-7}\). Further, the right and/or left two figures shows that the number of wrinkles increase as the inner radius of an ellipse increase from 0.3 to 0.6 while the magnitude of the viscous flow remains the same (i.e. \(A={10}^{-5}\) (left) and \(A={10}^{-7}\) (right)). Phenomenologically compatible results can be found in the relevant works such as circular substrate-membrane interactions28, capillary wrinkles on thin polymer films32 and theoretical study on an elastic surface45, where the number of radial wrinkles depends upon the size of the inner radius and membrane thickness. The proposed model successfully reproduces the reported results under the physically similar/compatible settings (see Figs. 3 and 4). In fact, the solutions presented in28 are the special case of the presented solution (see, Figs. 3 and 4) in the limit of vanishing eccentricity of elliptical domains (i.e. \(h({\xi }_{i},\eta )=a{(1-{e}^{2}{\cos }^{2}\eta )}^{1/2}=a\) for \(e\to 0\)). Lastly, we note here that the predicted wrinkle states are unique and stable, since the proposed model satisfies strict quasi-convexity via the minimization of membranes’ strain-energy potentials (see, for example45,46).
Non-uniform viscous flow potential: w(ξ, η) = A sin ξ cos η (waveform)
In this section, we consider membrane systems with non-uniform viscous flow. The non-uniform cases can be observed in various cellular activities such as the transportation of the intracellular membrane and the transmembrane proteins induced by the viscous flow with tension gradient47. In this case, the viscose flow field becomes non-uniform due to the interactions with tension gradient field.
Membranes subjected to the waveform of non-uniform viscous flows can be examined by introducing the following potential function,
where the intensity of wavy flow can be controlled by the parameters \(E\) and \(F\). In the assimilation, we set \(E=F=1\) for simplicity. Accordingly, from Eq. (62), we obtain the following expression of \({T}_{mn}(\xi ,\eta )\), addressing the viscous effects,
Combining (63), (65) and (72), the complete solution describing the membranes’ morphology can then be found as
where \({\sigma }_{1}\) is defined in Eq. (69). Similar to the constant viscous cases, the resulting deformation fields (radial wave deformations) are sensitive to both the dimension of an inner ellipse and the intensity of viscous flow; i.e., the number of waves reduces as A decreases from \({10}^{-5}\) to \({10}^{-7}\) (See. Fig. 5). But, more importantly, the transverse wave deformations of the membrane and the corresponding vertical deflections die out as they approach the remote boundary. As a result, the corresponding boundary remains intact and stable (See. Fig. 6). In the event of vanishing \(A\), the wave deformations are completely removed from the entire domain of interest so that the vertical deformation profile reduces to the results in17, where the authors present the analysis of elliptical substrate-membrane interaction problems without the considerations of viscosity effects (See. Fig. 6). Also, Fig. 7. shows that the obtained solution accommodates the results of circular substrate-membrane interaction problems in28 when the eccentricity converges to zero (i.e. \(e\to 0\)). In fact, the solutions in Figs. 6 and 7 become essentially identical for sufficiently small value of \(A\); i.e., \(A\le {10}^{-16}\) for case in Fig. 6 and \(A\le {10}^{-8}\) and \(A\le {10}^{-10}\) for the cases in Fig. 7. In the assimilations, the classical solutions obtained from the proposed model are intentionally reproduced at \(A={10}^{-15}\), \(A={10}^{-7}\) and \(A={10}^{-9}\) for the purpose of visual demonstration.
Dual source problems: w(ξ, η) = A + A sin ξ cos η
The proposed model is sufficiently general in that the viscous effects from both radial and circumferential directions can be simultaneously considered. To demonstrate this, we introduce the following dual source potential
and subsequently obtain from Eq. (62) that
Thus, from Eqs. (63) and (65), the deformation mapping function \(z(\xi ,\eta )\) can be obtained in the same manner as in the single source cases.
Figure 8 illustrates the deformation configuration of the membranes under the influence of dual source viscous flow. It is shown that both the radial and circumferential wave patterns are simultaneously observed. Morphologically similar cases are reported in the work of33 where the authors examined the wrinkle phenomena of a thin gold layer (10 nm in thickness) when subjected to thermal stresses from the adjoined polymer substrate. In cases of thin membranes, thermal stresses may be understood as a particular type of the surface stress48,49. Therefore, the results may bear close resemblance with the present case where the membrane’s deformations are induced by the surface interaction forces which are transmitted from the acting viscous flows. The obtained solution assimilates the experimental results in33 when compatible conditions are applied (see, Figs. 9 and 10). This further suggests that the proposed model may be of practical interest in the morphological study of thin film structures. Such investigations are, however, limited in the present study due to the lack of available data.
Remark 2.
The results in Fig. 8 further indicates that the principle of superposition remains valid in the present cases. The principle is widely adopted in various engineering problems with simple initial and/or boundary value problems of either first (Dirichlet) or second (Neumann) type50,51,52. However, such practices are largely absent in the membrane studies due the complexity of mixed boundary conditions (i.e. both the Dirichlet and Neumann boundary conditions are prescribed on the boundaries), and the limited access for the solutions of membrane systems subjected to coupled-physics environment. In the present case, the solutions of single source problems (i.e. \(w=A\) and \(w=A\,\sin \,\xi \,\cos \,\eta \)) can be obtained from Eqs. (67), (68), (72) and (73) that
for uniform flow \(w=A\) and
for non-uniform flow \(w=A\,\sin \,\xi \,\cos \,\eta \). It is clear from Eqs. (77)–(79) that the structure of the solution \(z(\xi ,\eta )\) remains intact. In fact, only \({T}_{mn}(\xi ,\eta )\) part of the solutions (i.e. Eqs. (76) and (78)) are affected with respect to the varying viscous flows. In particular, by adding Eqs. (76) and (78), we find
The above is the same as the solution obtained from the dual source problem (Eq. (75) and Fig. 8). This, in turn, suggests that the solutions of dual source problems can be obtained directly from the solutions of single source problems (see, also, Figs. 8 and 11) via simple summations. In other words, the principle of superposition remains valid even with the presence of intra-membrane viscous flows and interaction forces. The result may further promote the study of various different influences of viscous flows onto membrane-substrate systems by minimizing computational complexities and resources.
Reduction to the circular lipid membrane problems
The solution of a classical membrane-substrate problem11 can also be obtained directly from the present model. To demonstrate this, we evaluate (when \(e=0\))
where a, m, and \({K}_{m}(\mu \rho )\) are the radius of the inner circle, the separation constant and the modified Bessel function of second kind of order \(m\), respectively. Also, \(r=c{e}^{\xi }/2\) and \({G}_{m}\) are arbitrary constants with respect to the order \(m\).
Now, substituting the above into Eq. (62) yields
where we define \({\bf{A}}=\frac{\nu }{{\pi }^{2}\lambda }\frac{1}{{a}^{3}}{K}_{m}(\mu r)[1-\pi ].\) Thus, from Eqs. (68) and (82) we find
But, since \({G}_{m}/{G}_{m}=1\), Eq. (82) further reduces to
Finally, we substitute \({\mu }^{2}=2\lambda /k\) in the above and thereby obtain
The obtained solution in Eq. (85) is the same as in11 (Eq. 135), except the Bessel terms associated with the logarithmic function \(({\rm{i}}{\rm{.e}}\tfrac{{K}_{0}(\mu a)}{\mu {K^{\prime} }_{0}(\mu a)}\,\log (\tfrac{r}{a}))\).
Remark 3.
The method proposed in the present study is unique in that it utilizes both the iterative reduction scheme and the method of eigenfunction expansions while invoking the orthogonal properties of the Mathieu function. This further allows one to identify more wide class of potential functions of Mathieu type that the traditional method is limited in prediction. Further, since the Mathieu potential reduces to the Bessel function at the particular configuration of \(e=0\); i.e.,
the solutions of circular substrate interaction problems11,18 can be accommodated by the proposed model as a special case (i.e. \(e=0\), see, Figs. 7 and 12). In fact, Eq. (85) yields better predictions (slightly more resemble to the non-linear solution) when compared with the existing results (see, Fig. 12). This is due the presence of additionally predicted Bessel terms which cannot be obtained by the classical Helmholtz equations defined in the circular system. This further allows one to consider more general, and perhaps more realistic classes of viscous flows especially those arising in circular boundaries. For example, in circular problems, the generalization of the viscous potential Eq. (45) and the implementation of dual source flow Eq. (74) is no longer possible due to the confined descriptions of the associated circular boundary (see. Remark. 1.). Such difficulties can be overcome by creating desired forms of viscous flows in an elliptic coordinate where the corresponding continuity equation is the same form as in the Cartesian coordinate (i.e. \({v}_{\xi ,\xi }+{v}_{\eta ,\eta }=0\), see, Eq. (44)), and reducing the obtained solutions to the circular cases where the transition is always possible since the conformal mapping of an ellipse to a circle exists53.
Comparison with existing models: membrane-circular substrate interaction problem11.
The results obtained in the present study are of more practical interest in that, when used in conjunction with the principle of superposition (see. Remark. 2.), they essentially lead to the solution of a class of problems in which the viscous effects are characterized by a much wider and more realistic class of functions. Potential applications may be extended to retina clinical study of wrinkle-caused vision impairment34 and the effects of viscous flows on essential cellular functions such as fusion, fission and vesicle formation35,54,55. For example, the idiopathic epiretinal membranes (iERMs) is a common pathology which have been observed in more than 20% of eyes from elderly person34,56. When iERMs are thicker with contractile properties, they cause surface wrinkling of the retina resulting impaired vision (a macular epiretinal membrane)34,57. Such wrinkle formations are most often induced by the interactions between the posterior vitreous cortex and the retina58. Since nearly all the emmetropic retinas are oblate in shape in both transverse axial and sagittal sections59, the wrinkle formations on the retina may share close similarity to the elliptical membrane-substrate systems examined by the proposed model. In addition, wrinkle involved deformations and the directional elongation of the vesicle are often caused by the viscous shear flows and/or directional viscous flows54,55. Therefore, the proposed model may be employed to study the morphological transitions of cell membranes associated with those cellular activities.
Ethics statement
This work did not involve any collection of human data.
Data acquisition
Figures in the manuscript are prepared by visualizing analytical solutions presented in the manuscript. For the purpose, a commercial software (MATLAB) is used.
Data availability
This work does not have any experimental data.
References
Helfrich, W. Elastic properties of lipid bilayers: theory and possible experiments. Z. Naturforsch. 28, 693–703 (1973).
Evance, E. A. & Skalak, R. Mech. Thermodynam. Bio Membran. CRC Press, Boca Raton, FL (1980).
Secomb, T. W. & Skalak, R. Surface flow of viscoelastic membranes in viscous fluids. Q. J. Mech. Appl. Math. 35, 233–247 (1982).
Seifert, U. Configurations of fluid membranes and vesicles. Adv. Phys. 46(1), 13–137 (1997).
Jaric, M., Seifert, U., Wintz, W. & Wortis, M. Vesicular instabilities: the prolate-to-oblate transition and other shape instabilities of fluid bilayer membranes. Phys. Rev. E 52, 6623 (1995).
Gorter, E. & Grendel, F. On Bimolecular Layers of Lipoids on the Chromocytes of the Blood. J. Exp. Med. 41(4), 439–443 (1925).
Robertson, J. D. The Ultrastructure of Cell Membranes and Their Derivatives. Biochem. Soc. Symp. 16, 3–43 (1959).
Naghdi, P. M. On a variational theorem in elasticity and its application to shell theory. J. Appl. Mech. 31(4), 647–653 (1964).
Naghdi, P. M. The Theory of Shells and Plates. In: Truesdell, C. (ed.) Linear Theories of Elasticity and Thermoelasticity. Springer, Berlin, Heidelberg (1973).
Agrawal, A. & Steigmann, D. J. Coexistent fluid-phase equilibria in biomembranes with bending elasticity. J. Elast. 93(1), 63–80 (2008).
Agrawal, A. & Steigmann, D. J. Boundary-Value Problems in the Theory of Lipid Membranes. Contin. Mech. Therm. 21(1), 57–82 (2009).
Kim, C. I. & Steigmann, D. J. Distension-induced gradient capillarity in lipid membranes. Contin. Mech. Thermodyn. 27(4–5), 609–621 (2014).
Belay, T., Kim, C. I. & Schiavone, P. Analytical Solution of Lipid Membrane Morphology Subjected to Boundary Forces on the Edges of Rectangular Membrane. Contin. Mech. Therm. 28(1–2), 305–315 (2015).
Rangamani, P. & Steigmann, D. J. Variable tilt on lipid membranes. Proc. Math. Phys. Eng. Sci. 470(2172), 20140463 (2014).
Belay, T., Kim, C. I. & Schiavone, P. Mechanics of lipid bilayer subjected to thickness distension and membrane budding. Math. Mech. Solids. 23(1), 67–84 (2016).
Belay, T., Kim, C. I. & Schiavone, P. Budding formation of lipid membranes in response to the surface diffusion of transmembrane proteins and line tension. Math. Mech. Solids. 22(11), 2091–2107 (2016).
Belay, T., Kim, C. I. & Schiavone, P. Interaction-induced morphological transitions of lipid membranes in contact with an elliptical cross section of a rigid substrate. J. Appl. Mech. ASME. 83(1), 011001 (2016).
Zeidi, M. & Kim, C. I. Notes on superposed incremental deformations in the mechanics of lipid membranes. Math. Mech. Solids, https://doi.org/10.1177/1081286517734608 (2017).
Kim, C. I. A discussion on the mechanics of lipid membranes: Lagrange multipliers and a singular substrate. Z. Ange. Math. Phys. 68(4), 84 (2017).
Chernomrdik, L. & Kozlov, M. Mechanics of membrane fusion. Nat. Struct. Mol. Biol. 15, 675–683 (2008).
Lenz, M., Morlot, S. & Roux, A. Mechanical requirements for membrane fission: common facts from various examples. FEBS Lett. 583, 3839–3846 (2009).
Bruinsma, R. & Pincus, P. Protein Aggregation in Membranes. Curr. Opin. Solid. State Mater. Sci. 1(3), 401–406 (1996).
Benedict, J. R. et al. Aggregation and Vesiculation of Membrane Proteins by Curvature-Mediated Interactions. Nature 447(7143), 461–464 (2007).
Arroyo, M. & DeSimone, A. Relaxation dynamics of fluid membranes. Phys. Rev. E 79(031915), 1–17 (2009).
Rangamani, P., Agrawal, A., Mandadapu, K. K., Oster, G. & Steigmann, D. J. Interaction between surface shape and intra-surface viscous flow on lipid membranes. Biomech. Model. Mechanobiol. 12(4), 833–845 (2013).
Steigmann, D. J. Fluid films with curvature elasticity. Arch. Ration. Mech. Anal. 150, 127–52 (1999).
Steigmann, D. J. On the relationship between the Cosserat and Kirchhoff–Love theories of elastic shells. Math. Mech. Solids 4, 275–288 (1999b).
Zeidi, M. & Kim, C. I. The effects of intra-membrane viscosity on lipid membrane morphology: complete analytical solution. Sci. Rep. 8(1), 12845 (2018).
Read, W. Series solution for laplace equation with nonhomogenious mixed boundary conditions and irregualr boundaries. Math. Comput. Model. 17, 9–19 (1993).
Read, W. W. Analytical solutions for a helmholtz equation with dirichlet boundary conditions and arbitrary boundaries. Math. Comput. Model. 24(2), 23–34 (1996).
Huang, Yan & Zhang, X.-J. General analytical solution of transverse vibration for orthotropic rectangular thin plates. J. Marine. Sci. Appl. 1(2), 78–82 (2002).
Huang, J. et al. Capillary wrinkling of floating thin polymer films. Science 317(5838), 650–653 (2007).
Zhao, Y., Huang, W. M. & Fu, Y. Q. Formation of micro/nano-scale wrinkling patterns atop shape memory polymers. J. Micromech. Microeng. 21(6), 067007 (2011).
Joshi, M., Agrawal, S. & Christoforidis, J. B. Inflammatory mechanisms of idiopathic epiretinal membrane formation. Mediators Inflamm., https://doi.org/10.1155/2013/192582 (2013).
Cerda, E. & Mahadevan, L. Geometry and physics of wrinkling. Phys. Rev. lett. 90(7), 074302 (2003).
Sokolnikoff, I. S. Tensor Analysis: Theory and Applications. (Wiley, New York, 1951).
Aris, R. Vectors, Tensors and the Basic Equations of Fluid Mechanics. (Dover, N.Y., 1989).
Scriven, L. E. Dynamics of a fluid interface equation of motion for Newtonian surface fluids. Chem. Eng. Sci. 12(2), 98–108 (1960).
Steigmann, D. J., Baesu, E., Rudd, R. E., Belak, J. & McElfresh, M. On the variational theory of cell-membrane equilibria. Interface Free. Bound. 5, 357–366 (2003).
Kukla, S. Green’s function for vibration problems of an elliptical membrane. J. Appl. Math. Comput. Mech. 10(2), 129–134 (2011).
McLachlan, N. W. Theory and Application of Mathieu Functions. Clarendon Press, Oxford (1947).
Cooper GM. The Cell: A Molecular Approach. 2nd edition. Sunderland (MA): Sinauer Associates (2000).
Hochmuth, R. M. & Waugh, R. E. Erythrocyte membrane elasticity and viscosity. Ann. Rev. Physiol. 49, 209–19 (1987).
Derenyi, I., Julicher, F. & Prost, J. Formation and interaction of membrane tubes. Phys. Rev. Lett. 88(23), 238101 (2002).
Steigmann, D. J. Tension-field theory. Proc. Roy. Soc. Lond. A 429, 141–73 (1990).
Goodbrake, C. & Steigmann, D. J. Mechanics of an elastic membrane infused with a liquid. Int. J. Mech. Sci., https://doi.org/10.1016/j.ijmecsci.2017.07.062 (2017).
Fogelson, B. & Mogilner, A. Computational estimation of membrane flow and tension gradient in motile cell. PLoS One 9(1), e84524 (2014).
Gurtin, M. E. & Murdoch, A. I. A Continuum Theory of Elastic Material Surfaces. Arch. Ration. Mech. Anal. 57(4), 291–323 (1975).
Gurtin, M. E., Weissmuller, J. & Larche, F. A General Theory of Curved Deformable Interface in Solids at Equilibrium. Philos. Mag. A 78(5), 1093–1109 (1998).
Ferreira, E. R. & Boulanger, P. Superposition of transverse and longitudinal finite-amplitude waves in a deformed Blatz—Ko Material. Math. Mech. Solids. 12(5), 543–558 (2007).
Karayaka, M. & Kurath, P. Deformation and failure behavior of woven composite laminates. J. Eng. Mater-T ASME 116(2), 222–232 (1994).
Delale, F. & Erdogan, F. Transverse shear effect in a circumferentially cracked cylindrical shell. Q. Appl. Math. 37(3), 239–258 (1979).
Muskhelishvili N. I. Some Basic Problems of the Mathematical Theory of Elasticity, Noordhoff, P. Groningen, The Netherlands (1953).
Walter, A., Heinz, R. & Herbert. Shear induced deformation of microcapsules: shape oscillations and membrane folding. Colloids Surf. A: Physicochem. Eng. Asp. 183, 123–132 (2001).
Kantsler, V., Enrico, S. & Victor, S. Vesicle dynamics in time-dependent elongation flow: Wrinkling instability. Phys. Rev. Lett. 99(17), 178102 (2007).
Roth, A. M. Surface wrinkling retinopathy in eyes enucleated at autopsy. Trans. Am. Acad. Ophthalmol. Otolaryngol. 75, 1047–1058 (1971).
Kampik, A. N. S. E. L. M. et al. Ultrastructural features of progressive idiopathic epiretinal membrane removed by vitreous surgery. Am. J. Ophthalmol. 90(6), 797–809 (1980).
Sebag, J. Anomalous posterior vitreous detachment: a unifying concept in vitreo-retinal disease. Graefe’s archive Clin. Exp. Ophthalmol. 242(8), 690–698 (2004).
Atchison, D. A. et al. Shape of the retinal surface in emmetropia and myopia. Investig. Ophthalmol. & Vis. Sci. 46(8), 2698–2707 (2005).
Acknowledgements
This work was supported by the Natural Sciences and Engineering Research Council of Canada via Grant #RGPIN 04742 and the University of Alberta through a start-up grant. Kim would like to thank Professor David Steigmann for his invaluable and inspirational advice in the subject through his unpublished manuscript. Stated in the manuscript. This work was supported by the Natural Science and Engineering Research Council of Canada via Grat #RGPIN 04742.
Author information
Authors and Affiliations
Contributions
The corresponding author (Kim) formulate theory (including mathematical models) and interpreted the results. The first author (Liu) solved the resulting equations and prepared the figures.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Liu, Z., Kim, Ci. Deformation analysis of lipid membranes subjected to general forms of intra-membrane viscous flow and interactions with an elliptical-cross-section substrate. Sci Rep 10, 478 (2020). https://doi.org/10.1038/s41598-019-57179-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-57179-z
- Springer Nature Limited