Abstract
Sepsis is a common and often deadly systemic response to an infection, usually caused by bacteria. The gold standard for finding the causing pathogen in a blood sample is blood culture, which may take hours to days. Shortening the time to diagnosis would significantly reduce mortality. To replace the time-consuming blood culture we are developing a method to directly separate bacteria from red and white blood cells to enable faster bacteria identification. The blood cells are moved from the sample flow into a parallel stream using acoustophoresis. Due to their smaller size, the bacteria are not affected by the acoustic field and therefore remain in the blood plasma flow and can be directed to a separate outlet. When optimizing for sample throughput, 1 ml of undiluted whole blood equivalent can be processed within 12.5 min, while maintaining the bacteria recovery at 90% and the blood cell removal above 99%. That makes this the fastest label-free microfluidic continuous flow method per channel to separate bacteria from blood with high bacteria recovery (>80%). The high throughput was achieved by matching the acoustic impedance of the parallel stream to that of the blood sample, to avoid that acoustic forces relocate the fluid streams.
Similar content being viewed by others
Introduction
Sepsis is a systemic response to an infection, and the severe form is a common condition with a yearly incidence rate of 0.3% in the US, and mortality as high as 29%1. This makes sepsis one of the most common reasons for being admitted to intensive care2 and the 10th most common cause of death3. Sepsis is most often caused by bacteria, making fast and appropriate antibiotics treatment crucial for patient survival4,5.
Treatment is ideally guided by identification of the pathogen and its antibiotic resistance pattern. The gold standard for detection of bacteria in blood samples is blood culture, where the sample commonly is cultured in enriched broth until growth is detected. Bacterial strain and antibiotic resistance is then identified by Gram staining and sub culturing, polymerase chain reaction (PCR)6 or mass spectrometry (MS)7. Unfortunately the initial culture step may take from hours to days8, which is too long since mortality increases with 8% per hour of delay of efficient antimicrobial treatment during the first 6 hours of septic shock4. Furthermore, all culture-based detection methods have limited sensitivity, as they may miss slow-growing or fastidious bacteria and bacteria in samples containing antibiotics9.
While awaiting identification of the sepsis causing pathogen, the patient is treated with broad-spectrum antibiotics. This initial treatment is often not sufficient and in 47% of the cases the patient does not respond to this treatment at all10. Broad-spectrum antibiotics do not only act on the disease-causing bacteria, but also on the patient’s normal bacteria flora, causing additional harm. Furthermore, extensive use of broad spectrum antibiotics drives the increasing antibiotics resistance11. A more recently established and fast alternative to blood culture is direct PCR detection without previous culture, typically preceded by lysis of the blood and nucleic acid purification12. However, current PCR methods suffer from limited sensitivity and specificity, and susceptibility to contamination12,13.
Consequently, there is an urgent need for faster and more sensitive procedures for pathogen identification. An alternative would be to separate the bacteria from the red and white blood cells (henceforth collectively called blood cells) based on differences in physical properties prior to PCR detection. Such a separation is challenging, since samples from septic patients typically contain as little as 1–100 colony forming units (cfu) of bacteria or less14 among typically 3∙109 red blood cells (RBCs), 1∙107 white blood cells (WBCs) and 2∙108 platelets per ml of blood15,16. To find the few bacteria that are present in a blood sample, it is critical that the separation method has a very high bacteria recovery, efficient blood cell removal and sufficiently high throughput to process a clinical sample within reasonable time. Additionally, if such methods were available, bacteria detection or removal in blood components for transfusion could potentially prevent sepsis caused by contaminated blood components17.
Several microfluidic separation methods have been applied to separate bacteria from blood cells18,19, including dielectrophoresis20,21,22, inertial effects23,24,25,26, cell margination27,28, surface acoustic waves29,30, bead-based extraction31,32,33, filtering34, centrifugal microfluidics19 and lysis-based methods35,36. The suitability of these methods for sample preparation in sepsis diagnosis is, however, limited, either by a too costly labelling, low sample throughput, limited removal efficiency of cells or lysis debris, or too low bacteria recovery.
In this study we address this challenge by separating bacteria from blood cells through bulk acoustophoresis. We separate the smaller bacteria from the larger blood cells based on the principle that the acoustically induced velocity of a suspended object scales with the square of its radius37,38. Previously we have shown that it is possible to separate bacteria from patient blood samples using bulk acoustophoresis, followed by acoustic enrichment and PCR detection of the separated bacteria39,40,41,42. The sensitivity of the method was, however, limited by that only 11% of the bacteria were recovered in the separation step. In this study we have addressed the modest bacteria recovery reported by our group43 and later others44,45. Using media with adjusted acoustic impedance46,47,48,49, we can now recover 99.7% of the bacteria while removing more than 99.9% of the blood cells. At higher blood concentrations, 20% whole blood, we can still recover 90% of the bacteria while removing more than 99% of the blood cells enabling 1 ml undiluted whole blood to be processed within 12.5 min. To the best of our knowledge that makes this, the fastest label-free microfluidic continuous flow method per channel to separate bacteria from blood with high bacteria recovery (>80%). Combined with downstream enrichment and detection methods in a similar fashion to what we have previously reported, this paves the way for faster and more automated sepsis diagnostics, which potentially could reduce mortality.
Separation Considerations
Separation principle
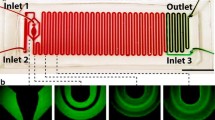
The principle for acoustic separation of bacteria from blood cells is illustrated in Fig. 1. The blood sample, containing bacteria, is laminated on both sides of a central cell-free medium (Fig. 1a). This central flow stream is important to achieve high recovery of bacteria and to enable separation at high blood cell concentrations, because it allocates space for the blood cells to migrate into.
Acoustic separation principle. (a) Acoustic field off; sample with blood cells (red) and bacteria (green) in plasma (yellow) laminated along sidewalls by a central cell-free medium (blue). (b) Acoustic separation; acoustic field applied, focusing blood cells but not bacteria. (c) Acoustic relocation of fluid streams due to acoustic impedance mismatch; the fluid with the highest acoustic impedance is relocated to the central pressure node. (d) Hydrodynamic interaction; blood cells pull neighbouring bacteria with them through hydrodynamic drag.
When the ultrasound is turned on, a standing acoustic wave is formed between the side walls of the separation channel, creating a primary radiation force on the blood cells directed towards the pressure node located along the centreline of the channel (Fig. 1b). While flowing through the channel, the blood cells are acoustophoretically transported into the cell-free medium and removed through the centre outlet.
The bacteria, which are an order of magnitude smaller compared to the blood cells, are much less affected by the acoustic radiation force, since the acoustophoretic velocity is proportional to the square of the particle radius37,38. In comparison to the blood cells the bacteria are in principle unaffected by the sound field and hence remain in the blood plasma that flow along the side walls to the side channel exits. Any free DNA in the blood plasma is also likely to stay in the plasma and exit with the bacteria. To create some margin to also collect those bacteria that exist at or cross the interface between the laminated liquids, the flow rate of the side outlet was set 50% higher than the side inlet.
Acoustic field acting on inhomogeneous fluids
The acoustic field does not only create a force on the blood cells but the laminated fluid streams can themselves be affected depending on their relative acoustic impedance (i.e. density times speed of sound). This is analogous to how liquids can be layered in a gravity field based on differences in mass density. In this configuration a fluid with high acoustic impedance laminated along the side walls by a fluid with lower acoustic impedance will relocate to the centre of the channel when exposed to the acoustic field50,51.
In the proposed separation the presence of blood cells and plasma proteins in the sample increases the acoustic impedance of the sample stream relative to a regular buffer and hence the sample will relocate to the centre outlet without any separation (Fig. 1c)49,52. In the current study the relocation of the sample stream was prevented by increasing the acoustic impedance of the central medium to match the acoustic impedance of 20% whole blood. Supplementary Figure S1 shows the acoustic impedance for blood at different dilution factors ranging from pure buffer to 20% whole blood, in relation to the acoustic impedance of the unmatched and matched buffer.
Hydrodynamic interactions
Another effect that can hamper the separation is hydrodynamic interaction between the blood cells and the bacteria. Blood cells that are focused to the centre drag along a thin layer of surrounding liquid, including any therein suspended bacteria, to the central stream which interferes with the separation (Fig. 1d). This effect is stronger the higher the blood cell concentration is. Theoretical estimates yield that the hydrodynamic interaction is starting to impact separations at particle volume ratios above 0.001 and at ratios above 0.01 the hydrodynamic particle interaction becomes prominent53. A rule of thumb is that as long as the mean particle distance is more than ten times the particle diameter the hydrodynamic interaction can be neglected.
Acoustic streaming
Apart from creating a force on particles and liquids, the acoustic field can also induce acoustic streaming54 in the fluid and this can lead to a transfer of bacteria or molecules across the streamlines55. For a given acoustic field this effect is more prominent at low flow velocities due to the longer retention time in the channel. For larger objects such as blood cells, the effect of the radiation force is, however, much larger than the effect of acoustic streaming in our system.
Materials and Methods
Microfluidic chip
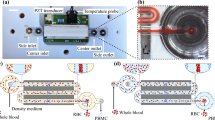
The separation method was tested using a silicon chip with anisotropically wet etched channels sealed by a glass lid, shown in Fig. 2. The chip has a common inlet that branches into a trifurcation with a central buffer inlet that laminates the sample along the separation channel side walls. The separation channel is 29 mm long, 375 µm wide and 150 µm deep and ends with a trifurcation having a central outlet and the side branches connected to a common side outlet. The chip50 and fabrication procedure56 have been described in more detail previously.
Acoustic actuation
The chip was acoustically actuated by a piezoelectric transducer glued underneath the separation channel and connected to a waveform generator (Tektronix AFG3022B, UK Ltd., Bracknell, UK) through an in-house built amplifier. The driving voltage was monitored with an oscilloscope (Tektronix TDS2002C, UK Ltd., Bracknell, UK) and held at 15 V peak-to-peak through an in-house written software controlling the waveform generator. The actuation voltage was chosen to be the highest possible where temperature feed-back loop could still retain the chip at 25 °C. The actuation frequency was tuned by visual optimization of the acoustic focusing and was set to 1.99 MHz unless otherwise stated.
Temperature control
To reach high separation stability over extended time periods, the temperature of the chip was controlled at 25 °C by a Peltier element in contact with the aluminium chip holder. The Peltier element was regulated using a PID regulator (TC0806-RS232 CoolTronic GmBH, Beinwill am See, Switzerland) connected to a Pt1000 temperature sensor glued to the piezoelectric transducer.
Flow control
All flows through the chip were regulated by controlling the pressure in sample tubes connected to the side inlet, side outlet and centre outlet as well as a buffer bottle connected to the centre inlet using a pressure terminal (Fig. 3; VEMA, FESTO, Germany). The pressures were PID regulated through in-house developed LabView software (National Instruments Corporation, Austin, Texas, USA) based on flow rates measured by thermal flow sensors (SLI-1000, Sensirion AG, Staefa ZH, Switzerland). The flow control system has been further described previously50.
Schematic of the system. The acoustophoresis chip (a) is actuated by the piezoelectric transducer (b) and the temperature is controlled by a Peltier element (c). Sample containing blood and bacteria (d) enters the chip through the first inlet. Impedance matched buffer (e) enters through the centre inlet. Blood cells focused into the centre buffer exit the chip through the centre outlet and into the waste container (f) while bacteria exit through the last outlet and end up in a collection tube (g). All flows are controlled by a pressure terminal and flow sensors.
Bacteria culture and staining
Pseudomonas putida KT2440 (Trevisan) Migula (ATCC® 47054™) was chosen as a biosafety level 1 model for the common sepsis pathogen Pseudomonas aeruginosa14. P. putida were cultured overnight on a shaker at 30 °C in medium mixed from tryptone (30 g/l), yeast extract (20 g/l) and 3-(N-Morpholino)propanesulfonic acid (MOPS, 10 g/l), adjusted to pH 7 and sterilized by autoclaving. Before addition to the blood sample they were washed by centrifugation and stained with SYTO® 9 green fluorescent nucleic acid stain (Life Technologies, Grand Island, NY, USA).
Blood collection and dilution
Venous peripheral blood samples were collected from healthy volunteers in Vacutainer® tubes either containing sodium citrate solution (10% of final volume) or spray-coated with ethylenediaminetetraacetic acid potassium salt (K2EDTA) as anticoagulant (BD Biosciences, Franklin Lakes, NJ, USA). All samples were diluted in 1× phosphate-buffered saline (PBS) containing 0.05% sodium azide (NaN3) to prevent the added bacteria from multiplying and 2 g/l bovine serum albumin (BSA) to avoid cell adhesion to the channel walls (further on referred to as buffer). The study protocol was approved by The Regional Ethical Review Board in Lund (Dnr 2011/400) and informed consent was obtained from all donors. All methods were carried out in accordance with relevant guidelines and regulations.
Cell counting
All cells were counted through flow cytometry (FACSCanto II cytometer and FACSDiva software, BD Biosciences, Franklin Lakes, NJ, USA), as exemplified in Fig. 4. The analyzed sample volume was measured using counting beads (Trucount™ tubes, BD Biosciences, Franklin Lakes, NJ, USA; Bacteria Counting Kit, Life Technologies, Grand Island, NY, USA). The RBC, WBC and platelet populations were initially identified through CD235a, CD45 and CD41a antibody labelling respectively (BD Biosciences, Franklin Lakes, NJ, USA) and subsequently gated through forward (FSC) and side scatter (SSC) (Fig. 4). The RBC and WBC populations overlap in forward and side scatter and were therefore counted together as blood cells. Most of the cells that are counted as blood cells are RBCs since they are typically 2-3 orders of magnitude more abundant than the WBCs, therefore this number is also a good estimate of the number of RBCs. The bacteria were gated on fluorescence, since they otherwise would overlap with background noise and the platelet population in forward and side scatter.
The bacteria recovery is here estimated as the fraction of the bacteria exiting the system through the side outlet relative all bacteria exiting side and centre outlet. The blood cell removal efficiency is the number of blood cells exiting through the centre outlet relative the total number of blood cells exiting the system. All reported numbers are average steady state separation performance corrected for the volume of buffer present in the system at the start of a run and the volume of sample left in the system at the end of a run.
Visual confirmation of the bacteria separation principle
The separation principle was confirmed by visual inspection of the inlet and outlet trifurcations through a microscope. Citrated blood was diluted to 1% (0.9% blood when the citrate solution is accounted for) in buffer and injected at 200 µl/min through the sample inlet. To visualize the plasma flow in the chip the sample was spiked with 1 g/l fluorescein sodium (The British Drug Houses LTD., Poole, England) instead of bacteria. The plasma was laminated along the side walls by a central buffer flow (300 µl/min) containing 1% Percoll (Sigma-Aldrich, Schnelldorf, Germany) to increase the acoustic impedance of the central flow stream. The outlet flow rates were set to 300 µl/min for the side outlet and 200 µl/min at the centre outlet and the chip was actuated in a half wavelength resonance mode at 1.989 MHz.
Optimization and evaluation of the bacteria separation
The separation performance was optimized and evaluated using diluted whole blood spiked with P. putida bacteria. Whole blood with EDTA as anticoagulant was diluted in buffer and spiked with 5∙105 P. putida bacteria per µl whole blood. This high concentration of bacteria was used to enable evaluation of the acoustic separation process by counting individual bacteria in the respective outlets using flow cytometry. The bacteria concentration was still considered low enough to prevent the bacteria concentration from affecting the separation.
To test the influence of blood concentration on the bacteria separation, blood samples were diluted to 1, 2, 5, 10 and 20% whole blood. Flow rates were 200 µl/min sample input, 300 µl/min centre buffer, 300 µl/min exiting through the joint side outlet and 200 µl/min exiting through the centre outlet. To demonstrate the importance of acoustic impedance matching of the laminated fluids, either a pure buffer or an impedance matched buffer (70% buffer and 30% Histopaque 1077; Sigma-Aldrich, Schnelldorf, Germany) was used as centre buffer.
The effect of varying the flow rate was subsequently tested by performing the same experiment at sample flow rates of 50, 100, 200 and 400 µl/min for 1 and 20% whole blood. The ratios between all flow rates were kept the same as in the previous measurements. In these experiments an impedance matched centre buffer consisting of 70% buffer and 30% Histopaque 1077 was used.
Data availability
All data generated during the current study is available from the corresponding author on request.
Results and Discussion
Visual confirmation of the bacteria separation principle
The separation principle was confirmed visually through microscopy of the inlet and outlet trifurcations with the ultrasound turned off and on (Fig. 5). A blood sample diluted to 0.9% was injected through the sample inlet and was laminated on either side of an impedance matched cell free medium injected in the centre inlet. When the ultrasound was turned on, the blood cells were tightly focused by the primary acoustic radiation force to a narrow dark line in the centre buffer and removed through the centre outlet. The plasma, here visualized with dye, remained near the side walls and exited through the side channels to the bacteria outlet. A narrow band of clean buffer was seen in the outlet side branches, creating some margin for recovering also slightly displaced bacteria. This margin was intentionally created by setting the centre inlet buffer flow rate higher than the centre blood outlet flow rate. The slight broadening of the plasma stream at the outlet (Fig. 5 lower right) when the ultrasound is active as compared to without ultrasound is attributed to the effect of acoustic streaming.
Microscope images of the chip inlet and outlet trifurcations with ultrasound off and on. The blood sample enters through the side channels and is laminated along the side walls. When the ultrasound is turned on, blood cells are acoustically focused from the plasma to the centre buffer and removed through the centre channel. The plasma, spiked with yellow dye for visualization, remains laminated along the side walls and is collected through the side channels.
Acoustic impedance matching and hydrodynamic interaction
Performing the separation at high cell concentrations increases throughput and reduces separation time. However, elevated cell concentrations can compromise separation performance due to increased hydrodynamic cell-bacteria interactions. Increasing the blood concentration also increases the acoustic impedance of the sample, which may cause sample relocation to the centre hindering separation if the acoustic impedance of the sample increases above that of the centre buffer.
To investigate critical boundaries with respect to blood concentration the recovery of bacteria and the removal efficiency of blood cells were measured for whole blood concentrations ranging from 1–20% at a sample flow rate of 200 µl/min. The separations were performed with (Fig. 6b) and without (Fig. 6a) acoustic impedance matched medium as the central buffer. Higher whole blood concentrations than 20% were not possible since that did compromise flow sensor accuracy.
Effect of impedance matching and blood concentration on separation performance. Fraction of total output of each particle type in the bacteria outlet for 1, 2, 5, 10 and 20% blood without (a) and with (b) acoustic impedance matching of the centre buffer at a sample flow rate of 200 µl/min. The plots show mean values ± standard deviation of three repeats.
The blood cells were efficiently removed in all experiments, with less than 0.2% exiting through the bacteria outlet for all experiments (Fig. 6a,b, red lines). The bacteria recovery, on the other hand, was highly dependent on acoustic impedance matched centre buffer (green lines). With standard cell-free medium (Fig. 6a) a bacteria recovery of 71% could be achieved for 1% blood, but this quickly dropped to below 20% for blood concentrations of 5% and above. The reduced bacteria recovery at higher whole blood concentrations is due to the increased acoustic impedance caused by the high cell and plasma concentration of the sample stream in relation to the cell-free central medium. This causes the whole sample liquid, including both bacteria and blood cells, to relocate to the centre of the channel and exit through the blood outlet.
On the contrary, Fig. 6b demonstrates that the sample relocation does not occur when increasing the acoustic impedance of the central cell-free medium. With high impedance cell-free medium, the bacteria recovery was as high as 99% for 1% whole blood and dropped to 89% for 20% whole blood. The drop in recovery can be explained by an increased hydrodynamic interaction causing the blood cells to drag bacteria into the central stream. At 20% whole blood the cell volume fraction is approximately 0.08–0.1 (assuming an haematocrit of 40–50%), which vastly supersedes the critical level where hydrodynamic interaction starts to impact the acoustophoretic separation53.
The data underpins the importance of acoustic impedance matching of the central cell-free medium to achieve efficient separation, but also demonstrates that in spite of the negative influence of the hydrodynamic interaction at high blood concentrations (20% whole blood) a relatively high bacteria recovery of 89% can be accomplished, which may be sufficient when high throughput is priority and larger sample volumes are available.
Optimization of flow rates
To maximize throughput, sample flow rates of 50–400 µl/min were tested for 1% and 20% whole blood (Fig. 7a,b, respectively) using a central impedance matched buffer. These flow rates were chosen to stay within the range of the flow sensors. Even at high flow rates, the passage time for blood cells through the separation channel was sufficient for efficient focusing and removal. The blood cell contamination in the bacteria outlet remained below 0.1% for all settings except at the highest flow rate for 20% whole blood, where it was 0.87%.
Effect of blood concentration and flow rate on separation performance. Fraction of total output of each particle type in the bacteria outlet for different flow rates when running (a) 1% whole blood and (b) 20% whole blood. The plots show mean values ± standard deviation of three repeats. The centre buffer was impedance matched using 30% Histopaque.
For both blood concentrations, the bacteria recovery was improved at higher flow rates, reaching 99.7% for the 1% blood processed at maximum flow rate. This is because the bacteria spend less time in the separation channel at higher flow rates and are thus less affected by the acoustic radiation force and streaming that may otherwise mix them into the central fraction. The bacteria recovery is higher for the lower blood concentration due to lower hydrodynamic interaction, which is consistent with the data in Fig. 6.
Platelet separation
Platelets are only slightly larger than the bacteria that were used in these experiments and as expected they behaved similarly in the separations (blue lines in Figs 6–7). The presented separation method can therefore also be used to separate platelets from blood cells with 95% of the platelets exiting through the side outlet and 99.9% of the blood cells exiting through the centre outlet (Fig. 7a). This can be compared to earlier data from Dykes et al.57 for removal of platelets from peripheral blood progenitor cell products in a similar setup, where 89% of the platelets exited through the side outlet and 97.8% of white blood cells exited through a central outlet. More recent data from Chen et al.49 demonstrates separation of platelets from blood at an impressive throughput of 5 ml/min, but at a modest separation performance of 88.4% RBC removal and a platelet recovery of 86.2% in a large 17 mm wide layered acoustic resonator with impedance matched buffers.
Since the platelets are slightly larger than the bacteria, they display somewhat higher acoustophoretic mobility. The difference in focusing between platelets and bacteria is most apparent at low flow rates for 1% blood (Fig. 7a), and the gap indicates that also the platelets can be acoustically separated from the bacteria if desired by optimizing the separation further.
Optimized separation settings
Based on the results above, various separation settings can be chosen depending on if bacteria recovery, blood cell removal efficiency or sample throughput is prioritized (Table 1, priority in bold face). The highest bacteria recovery that was achieved was 99.7%, while the highest blood cell removal was 99.99%. The shortest time to process 1 ml of whole blood (before dilution 5×) was 12.5 min, while achieving a bacteria recovery of 90% and a blood cell removal efficiency above 99%.
Comparison to other methods
In Table 1 the results presented in this article are compared to other label-free microfluidic continuous flow methods to separate bacteria from blood. It should be noted that not all methods presented in Table 1 are aiming towards sepsis diagnostics, but should rather be seen as typical data for various microfluidic separation methods.
If the intention is to use the method for sample preparation for sepsis diagnostics, the challenge is typically to find as few as 1–100 cfu, or less, of bacteria14 among approximately 3∙109 RBCs, 1∙107 WBCs and 2∙108 platelets per ml of blood15,16. To be able to detect any of the few bacteria present in the sample, it is crucial to minimize the bacteria loss in the separation. Inertial, margination and other acoustic methods typically report 20–90% loss of bacteria, which significantly affects sensitivity. Impedance matched acoustophoresis is the method that demonstrates the highest bacteria recovery with down to 0.3% bacteria loss, compared to SAW and dielectrophoresis, the second and third best methods in Table 1, with 12 and 13% bacteria loss respectively.
To be able to concentrate the few bacteria from a large sample to a small detection volume it is necessary to remove the RBCs that typically constitute almost half of the blood volume. Every percent of RBCs that remains add 3∙107 RBCs per cfu of bacteria in a typical sample (1 cfu of bacteria and 3∙109 RBCs per ml), meaning that the removal efficiency should ideally be extremely high. Inertial and margination methods leave 10–29% of the RBCs in the sample, taking up a large volume hindering enrichment and hiding the bacteria among a vast number of cells. In this regard, dielectrophoresis, SAW and acoustophoresis stand out from the other methods in Table 1 as the only methods reaching above 99% RBC removal.
Another key factor for sample preparation is throughput. The low bacteria concentration means that a relatively large sample, often 1–10 ml, has to be processed to enable detection of bacteria. Since every hour is critical in sepsis diagnostics, such a volume should ideally be processed within minutes.
The throughputs of most microfluidic methods such as dielectrophoresis, inertial separation and SAW are hampered by low sample flow rates or high sample dilution factors. The throughput of margination is relatively high since whole blood can be processed, but comes at the cost of low bacteria recovery and RBC removal. However, by using a moderate dilution (5×) and relatively high flow rate (400 µl/min), 1 ml whole blood equivalent can be processed by impedance matched acoustophoresis within 12.5 minutes (~7∙106 RBCs/s), making this the fastest label-free microfluidic continuous flow method per channel achieving high bacteria recovery (>80%).
Outlook
Although the presented method yields high bacteria recovery, blood cell removal efficiency and throughput, there is still room for further improvement. The currently used setup was limited in flow rate and blood concentration by the flow sensors. Further increase in throughput may be achieved by using higher blood cell concentrations, larger channels58 or multiple parallel separation channels22. Using higher blood cell concentrations would also reduce the need for subsequent sample concentration, but would demand alternative flow control methods and likely reduces bacteria recovery.
It will be important to confirm this proof-of-concept with a larger number of samples from healthy volunteers as well as septic patients to ensure statistical certainty and robustness to variations in the sample composition. Sepsis may for instance affect RBC size15, WBC count16 and plasma viscosity59, which may in turn affect the separation. Our previous study where 57 blood samples from sepsis patients were processed by acoustophoresis indicated increased RBC sedimentation rate as well as increased platelet activation in some patient samples compared to blood from healthy donors, but without any evidence of detrimental impact on the acoustophoretic separation42.
Additionally, the bacteria concentrations are much lower in samples from septic patients than in the current experiments with spiked blood14. This is not expected to have affected the separations, since the bacteria were spiked into the blood at approximately ten times lower concentration than the much larger RBCs16. The RBCs should consequently dominate any particle-particle interaction, but this should be confirmed. Our previous study did, however, show that acoustic separation, acoustic enrichment and bacteria detection is possible at clinically relevant bacteria concentrations42.
The method also needs to be validated for the wide range of bacteria species and strains that may be found in samples from septic patients14. They are all similar in size to the model strain and single bacteria are therefore likely to behave similarly in the acoustic separation. A common challenge for all separation based methods is, however, that many pathogenic bacteria are known to form clusters60, reside inside WBCs, or adhere to blood cells61 or platelets62,63. They may then be removed with the larger cells and lost. This problem is shared with all other methods in Table 1 where separation is to some degree based on cell size. Adhesion of bacteria to cells may at least to some extent be remedied by chemically preventing adhesion64,65.
The presented acoustophoretic bacteria separation method opens up a route for development of rapid sepsis diagnostics by integrating the acoustic separation with downstream enrichment, purification and detection of the bacteria. Apart from bulk enrichment methods such as centrifugation, more integration-friendly label-free microfluidic enrichment methods including dielectrophoresis20, acoustic focusing55 and acoustic trapping66 have shown promising results. When choosing from these, high bacteria recovery and throughput matched to the separation are key requirements. There are also several possible detection methods, such as microscopy, flow cytometry, mass spectrometry and PCR, with PCR being a particularly strong candidate for detection of low numbers of bacteria including determination of species and antibiotic resistance pattern.
We have earlier investigated the possibility to integrate an early version of the acoustic separation with bacteria enrichment through acoustic trapping66 followed by chip-based PCR detection as schematically shown in Fig. 8a 42. With that prototype system Escherichia coli could be detected in the clinical sepsis samples with the highest bacteria concentrations within two hours, underlining both the potential of acoustic sample preparation for sepsis diagnostics as well as the necessity of high bacteria recovery. Replacing the initial acoustophoretic blood cell removal unit with the herein proposed method would increase the demands on subsequent bacteria enrichment because of higher dilution and flow rates, but the lower blood cell contamination and 7–9 times higher bacteria recovery may enable faster sepsis diagnostics also for patients with lower grade bacteremia.
Suggested application of impedance matched acoustic bacteria separation for sepsis diagnostics. (a) Previously presented method to acoustically remove red blood cells, acoustically trap bacteria from remaining plasma and finally detect bacteria by PCR42 (b) Suggested replacement of the initial acoustophoretic blood cell separation with the impedance matched buffer method proposed herein, improving bacteria recovery from 11% to >99%.
Conclusions
In this study we have demonstrated that bulk acoustophoresis can be used to separate bacteria from blood cells with, to the best of our knowledge, higher bacteria recovery and throughput per channel than any other label-free microfluidic continuous flow method with high bacteria recovery (>80%) and throughput relevant for diagnostic purposes. This was enabled primarily by matching the acoustic impedance of the centre sheath fluid to the sample, as well as by optimizing the blood concentration and sample flow rate. In combination with downstream bacteria enrichment and detection, the proposed acoustophoresis based bacteria separation method opens up for potential development of new and faster methods to diagnose sepsis.
References
Angus, D. C. et al. Epidemiology of severe sepsis in the United States: analysis of incidence, outcome, and associated costs of care. Critical care medicine 29, 1303–1310 (2001).
Vincent, J. L. et al. Sepsis in European intensive care units: results of the SOAP study. Critical care medicine 34, 344–353 (2006).
Hoyert, D. L., Arias, E., Smith, B. L., Murphy, S. & Kochanek, K. Deaths: final data for 1999. National vital statistics report. Hyattsville, Maryland: National Center for Health Statistics 49 (1999).
Kumar, A. et al. Initiation of inappropriate antimicrobial therapy results in a fivefold reduction of survival in human septic shock. Chest 136, 1237–1248, https://doi.org/10.1378/chest.09-0087 (2009).
Kumar, A. et al. Duration of hypotension before initiation of effective antimicrobial therapy is the critical determinant of survival in human septic shock. Critical care medicine 34, 1589–1596, https://doi.org/10.1097/01.ccm.0000217961.75225.e9 (2006).
Hirvonen, J., von Lode, P., Nevalainen, M., Rantakokko-Jalava, K. & Kaukoranta, S.-S. One-step sample preparation of positive blood cultures for the direct detection of methicillin-sensitive and-resistant Staphylococcus aureus and methicillin-resistant coagulase-negative staphylococci within one hour using the automated GenomEra CDX™ PCR system. European journal of clinical microbiology & infectious diseases 31, 2835–2842 (2012).
Carbonnelle, E. et al. MALDI-TOF mass spectrometry tools for bacterial identification in clinical microbiology laboratory. Clin Biochem 44, 104–109, https://doi.org/10.1016/j.clinbiochem.2010.06.017 (2011).
Biondi, E. A. et al. Blood culture time to positivity in febrile infants with bacteremia. JAMA pediatrics 168, 844–849, https://doi.org/10.1001/jamapediatrics.2014.895 (2014).
Lehmann, L. E. et al. Improved detection of blood stream pathogens by real-time PCR in severe sepsis. Intensive care medicine 36, 49–56, https://doi.org/10.1007/s00134-009-1608-z (2010).
Tabah, A. et al. Characteristics and determinants of outcome of hospital-acquired bloodstream infections in intensive care units: the EUROBACT International Cohort Study. Intensive care medicine 38, 1930–1945, https://doi.org/10.1007/s00134-012-2695-9 (2012).
Martin, G. S. Sepsis, severe sepsis and septic shock: changes in incidence, pathogens and outcomes. Expert Review of Anti-Infective Therapy 10, 701–706 (2012).
Stefani, S. Diagnostic techniques in bloodstream infections: where are we going? International journal of antimicrobial agents 34, S9–S12 (2009).
Chun, K. et al. Sepsis Pathogen Identification. J Lab Autom 20, 539–561, https://doi.org/10.1177/2211068214567345 (2015).
Dorn, G. L., Land, G. A. & Wilson, G. E. Improved Blood Culture Technique Based on Centrifugation: Clinical Evaluation. Journal of Clinical Microbiology 9, 391–396 (1979).
Jo, Y. H. et al. Red cell distribution width is a prognostic factor in severe sepsis and septic shock. Am J Emerg Med 31, 545–548, https://doi.org/10.1016/j.ajem.2012.10.017 (2013).
Piagnerelli, M., Cotton, F., Van Nuffelen, M., Vincent, J. L. & Gulbis, B. Modifications in erythrocyte membrane protein content are not responsible for the alterations in rheology seen in sepsis. Shock 37, 17–21, https://doi.org/10.1097/SHK.0b013e318237d55a (2012).
Brecher, M. E. & Hay, S. N. Bacterial contamination of blood components. Clin Microbiol Rev 18, 195–204, https://doi.org/10.1128/CMR.18.1.195-204.2005 (2005).
Foudeh, A. M., Didar, T. F., Veres, T. & Tabrizian, M. Microfluidic designs and techniques using lab-on-a-chip devices for pathogen detection for point-of-care diagnostics. Lab on a chip 12, 3249–3266 (2012).
Pitt, W. G. et al. Rapid separation of bacteria from blood-review and outlook. Biotechnol Prog 32, 823–839, https://doi.org/10.1002/btpr.2299 (2016).
Park, S., Zhang, Y., Wang, T. H. & Yang, S. Continuous dielectrophoretic bacterial separation and concentration from physiological media of high conductivity. Lab on a chip 11, 2893–2900, https://doi.org/10.1039/c1lc20307j (2011).
Kuczenski, R. S., Chang, H. C. & Revzin, A. Dielectrophoretic microfluidic device for the continuous sorting of Escherichia coli from blood cells. Biomicrofluidics 5, 32005–3200515, https://doi.org/10.1063/1.3608135 (2011).
Bisceglia, E. et al. A generic and label free method based on dielectrophoresis for the continuous separation of microorganism from whole blood samples. Sensors and Actuators B: Chemical 212, 335–343, https://doi.org/10.1016/j.snb.2015.02.024 (2015).
Mach, A. J. & Di Carlo, D. Continuous scalable blood filtration device using inertial microfluidics. Biotechnol Bioeng 107, 302–311, https://doi.org/10.1002/bit.22833 (2010).
Wu, Z., Willing, B., Bjerketorp, J., Jansson, J. K. & Hjort, K. Soft inertial microfluidics for high throughput separation of bacteria from human blood cells. Lab on a chip 9, 1193–1199, https://doi.org/10.1039/b817611f (2009).
Hou, H. W., Bhattacharyya, R. P., Hung, D. T. & Han, J. Direct detection and drug-resistance profiling of bacteremias using inertial microfluidics. Lab on a chip 15, 2297–2307, https://doi.org/10.1039/c5lc00311c (2015).
Faridi, M. A. et al. Elasto-inertial microfluidics for bacteria separation from whole blood for sepsis diagnostics. J Nanobiotechnology 15, 3, https://doi.org/10.1186/s12951-016-0235-4 (2017).
Wei Hou, H. et al. A microfluidics approach towards high-throughput pathogen removal from blood using margination. Biomicrofluidics 6, 24115–2411513, https://doi.org/10.1063/1.4710992 (2012).
Hou, H. W. et al. Broad spectrum immunomodulation using biomimetic blood cell margination for sepsis therapy. Lab on a chip 16, 688–699, https://doi.org/10.1039/c5lc01110h (2016).
Ai, Y., Sanders, C. K. & Marrone, B. L. Separation of Escherichia coli bacteria from peripheral blood mononuclear cells using standing surface acoustic waves. Analytical chemistry 85, 9126–9134, https://doi.org/10.1021/ac4017715 (2013).
Li, S. et al. Acoustofluidic bacteria separation. J Micromech Microeng 27, https://doi.org/10.1088/1361-6439/27/1/015031 (2017).
Xia, N. et al. Combined microfluidic-micromagnetic separation of living cells in continuous flow. Biomed Microdevices 8, 299–308, https://doi.org/10.1007/s10544-006-0033-0 (2006).
Chung, H. J., Castro, C. M., Im, H., Lee, H. & Weissleder, R. A magneto-DNA nanoparticle system for rapid detection and phenotyping of bacteria. Nat Nanotechnol 8, 369–375, https://doi.org/10.1038/nnano.2013.70 (2013).
Kang, J. H. et al. An extracorporeal blood-cleansing device for sepsis therapy. Nat Med 20, 1211–1216, https://doi.org/10.1038/nm.3640 (2014).
Hwang, K.-Y. et al. Rapid detection of bacterial cell from whole blood: Integration of DNA sample preparation into single micro-PCR chip. Sensors and Actuators B: Chemical 154, 46–51, https://doi.org/10.1016/j.snb.2009.11.005 (2011).
Mahalanabis, M., Al-Muayad, H., Kulinski, M. D., Altman, D. & Klapperich, C. M. Cell lysis and DNA extraction of gram-positive and gram-negative bacteria from whole blood in a disposable microfluidic chip. Lab on a chip 9, 2811–2817, https://doi.org/10.1039/b905065p (2009).
Zelenin, S. et al. Microfluidic-based isolation of bacteria from whole blood for sepsis diagnostics. Biotechnology letters, 1–6 (2014).
Lenshof, A., Magnusson, C. & Laurell, T. Acoustofluidics 8: Applications of acoustophoresis in continuous flow microsystems. Lab on a chip 12, 1210–1223 (2012).
Bruus, H. Acoustofluidics 7: The acoustic radiation force on small particles. Lab on a chip 12, 1014–1021, https://doi.org/10.1039/c2lc21068a (2012).
Petersson, K. et al. In The 18th International Conference on Miniaturized Systems for Chemistry and Life Sciences (µTAS 2014).
Mellhammar, L. et al. In The annual meeting of NSCMID (the Nordic Society of Clinical Microbiology and Infectious Diseases).
Evander, M. et al. In 2015 International Congress on Ultrasonics. (ed. N. F. Declercq) 102 (2015).
Ohlsson, P. et al. Integrated Acoustic Separation, Enrichment, and Microchip Polymerase Chain Reaction Detection of Bacteria from Blood for Rapid Sepsis Diagnostics. Analytical chemistry 88, 9403–9411, https://doi.org/10.1021/acs.analchem.6b00323 (2016).
Ohlsson, P., Petersson, K., Lenshof, A., Åstrand-Grundström, I. & Laurell, T. In The 10th Ultrasonic Standing Network Conference, USWNet 2012. 52–53.
Ngamsom, B. et al. On-chip acoustophoretic isolation of microflora including S. typhimurium from raw chicken, beef and blood samples. J Microbiol Methods 123, 79–86, https://doi.org/10.1016/j.mimet.2016.01.016 (2016).
Silva, R. et al. Rapid prototyping and parametric optimization of plastic acoustofluidic devices for blood-bacteria separation. Biomed Microdevices 19, 70, https://doi.org/10.1007/s10544-017-0210-3 (2017).
Ohlsson, P., Petersson, K., Augustsson, P. & Laurell, T. In The 17th International Conference on Miniaturized Systems for Chemistry and Life Sciences (µTAS 2013). 1320–1322.
Ohlsson, P., Petersson, K. & Laurell, T. In The 18th International Conference on Miniaturized Systems for Chemistry and Life Sciences (µTAS 2014).
Ohlsson, P., Petersson, K. & Laurell, T. in 2015 International Congress on Ultrasonics. (ed. N. F. Declercq) 103 (2015).
Chen, Y. et al. High-throughput acoustic separation of platelets from whole blood. Lab on a chip 16, 3466–3472, https://doi.org/10.1039/c6lc00682e (2016).
Deshmukh, S., Brzozka, Z., Laurell, T. & Augustsson, P. Acoustic radiation forces at liquid interfaces impact the performance of acoustophoresis. Lab on a chip 14, 3394–3400, https://doi.org/10.1039/c4lc00572d (2014).
Karlsen, J. T., Augustsson, P. & Bruus, H. Acoustic Force Density Acting on Inhomogeneous Fluids in Acoustic Fields. Phys Rev Lett 117, 114504, https://doi.org/10.1103/PhysRevLett.117.114504 (2016).
Tenje, M. et al. Acoustophoretic removal of proteins from blood components. Biomed Microdevices 17, 95, https://doi.org/10.1007/s10544-015-0003-5 (2015).
Ley, M. W. & Bruus, H. Continuum modeling of hydrodynamic particle-particle interactions in microfluidic high-concentration suspensions. Lab on a chip 16, 1178–1188, https://doi.org/10.1039/c6lc00150e (2016).
Rayleigh, L. On the Circulation of Air Observed in Kundt’s Tubes, and on Some Allied Acoustical Problems. Philosophical Transactions of the Royal Society of London 175, 1–21 (1884).
Antfolk, M., Muller, P. B., Augustsson, P., Bruus, H. & Laurell, T. Focusing of sub-micrometer particles and bacteria enabled by two-dimensional acoustophoresis. Lab on a chip 14, 2791–2799, https://doi.org/10.1039/c4lc00202d (2014).
Augustsson, P., Magnusson, C., Nordin, M., Lilja, H. & Laurell, T. Microfluidic, Label-Free Enrichment of Prostate Cancer Cells in Blood Based on Acoustophoresis. Analytical chemistry 84, 7954–7962, https://doi.org/10.1021/ac301723s (2012).
Dykes, J., Lenshof, A., Astrand-Grundstrom, I. B., Laurell, T. & Scheding, S. Efficient removal of platelets from peripheral blood progenitor cell products using a novel micro-chip based acoustophoretic platform. PloS one 6, e23074, https://doi.org/10.1371/journal.pone.0023074 (2011).
Hawkes, J. J., Barber, R. W., Emerson, D. R. & Coakley, W. T. Continuous cell washing and mixing driven by an ultrasound standing wave within a microfluidic channel. Lab on a chip 4, 446–452, https://doi.org/10.1039/b408045a (2004).
Késmárky, G., Kenyeres, P., Rábai, M. & Tóth, K. Plasma viscosity: a forgotten variable. Clinical hemorheology and microcirculation 39, 243–246 (2008).
Thornton, M. M. et al. Multicellularity and antibiotic resistance in Klebsiella pneumoniae grown under bloodstream-mimicking fluid dynamic conditions. The Journal of infectious diseases 206, 588–595, https://doi.org/10.1093/infdis/jis397 (2012).
Brekke, O. L. et al. Neisseria meningitidis and Escherichia coli are protected from leukocyte phagocytosis by binding to erythrocyte complement receptor 1 in human blood. Molecular immunology 48, 2159–2169, https://doi.org/10.1016/j.molimm.2011.07.011 (2011).
Siboo, I. R., Chambers, H. F. & Sullam, P. M. Role of SraP, a Serine-Rich Surface Protein of Staphylococcus aureus, in binding to human platelets. Infect Immun 73, 2273–2280, https://doi.org/10.1128/IAI.73.4.2273-2280.2005 (2005).
Kline, K. A., Falker, S., Dahlberg, S., Normark, S. & Henriques-Normark, B. Bacterial adhesins in host-microbe interactions. Cell host & microbe 5, 580–592, https://doi.org/10.1016/j.chom.2009.05.011 (2009).
Aberg, V. & Almqvist, F. Pilicides-small molecules targeting bacterial virulence. Org Biomol Chem 5, 1827–1834, https://doi.org/10.1039/b702397a (2007).
Salit, I. & Gotschlich, E. Hemagglutination by purified type I Escherichia coli pili. Journal of Experimental Medicine 146, 1169–1181 (1977).
Hammarstrom, B., Laurell, T. & Nilsson, J. Seed particle-enabled acoustic trapping of bacteria and nanoparticles in continuous flow systems. Lab on a chip 12, 4296–4304, https://doi.org/10.1039/c2lc40697g (2012).
Acknowledgements
We would like to thank all the donors who have kindly donated blood to this study, as well as Ingbritt Åstrand-Grundström, Helen Larsson and Stefan Scheding for organizing the collection of blood samples. The research leading to these results has received funding from the European Union Seventh Framework Programme (FP7/2007-2013) under grant agreement n° 259848, as well as from Vinnova: Innovationer för Framtidens Hälsa—CellCARE (grant No.: 2009-00236), Knut and Alice Wallenberg Foundation (grant no. KAW 2012.0023), the Swedish Research Council Formas “Strong research Environments” project No. 2011-1692 BioBridges, the Swedish Research Council Grant No.: 2014-6273 and the Sten K Johnson Foundation.
Author information
Authors and Affiliations
Contributions
All authors contributed to the planning and analysis of the experiments, as well as the writing of the manuscript. P.O. and K.P. performed the experiments.
Corresponding authors
Ethics declarations
Competing Interests
P.O., P.A. and T.L. are shareholders, T.L. is a board member, P.O. and K.P. are part time employees and P.A. is part time consultant for AcouSort A.B., a Lund University spin-off company commercializing acoustofluidic technology.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ohlsson, P., Petersson, K., Augustsson, P. et al. Acoustic impedance matched buffers enable separation of bacteria from blood cells at high cell concentrations. Sci Rep 8, 9156 (2018). https://doi.org/10.1038/s41598-018-25551-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-25551-0
- Springer Nature Limited
This article is cited by
-
Label-free separation of peripheral blood mononuclear cells from whole blood by gradient acoustic focusing
Scientific Reports (2024)
-
Acoustofluidic Interfaces for the Mechanobiological Secretome of MSCs
Nature Communications (2023)
-
Fabrication of spectroscopic microfluidic chips for mastitis detection in raw milk
Scientific Reports (2023)
-
Gradient acoustic focusing of sub-micron particles for separation of bacteria from blood lysate
Scientific Reports (2020)
-
Investigation of effect of design and operating parameters on acoustophoretic particle separation via 3D device-level simulations
Microfluidics and Nanofluidics (2020)