Abstract
Three Gorges Reservoir (TGR) in the upper stream of Yangtze River in China is a reservoir with the largest and the longest yearly water-level drop. Considering the fact that most of safety assessments of water samples collected from TGR region were based on chemical analysis, we here employed Caenorhabditis elegans to perform in vivo safety assessment of original surface water samples collected from TGR region in the flood season in Wanzhou, Chongqing. Among the examined five original surface water samples, only exposure to original surface water sample collected from backwater area could induce the significant intestinal ROS production, enhance the intestinal permeability, and decrease the locomotion behavior. Additionally, exposure to original surface water sample collected from backwater area altered the expressions of sod-2, sod-5, clk-1, and mev-1. Moreover, mutation of sod-2 or sod-5 was susceptible to the potential toxicity of original surface water sample collected from backwater area on nematodes. Together, our results imply that exposure to surface water sample from the backwater area may at least cause the adverse effects on intestinal function and locomotion behavior in nematodes.
Similar content being viewed by others
Introduction
Construction of Three Gorges Dam created a novel ecosystem in the upper stream of the Yangtze River in China. Three Gorges Reservoir (TGR) is the world’s largest reservoir with the largest and the longest yearly water-level drop. This reservoir is from Chongqing (west) to Yichang of Hubei province (east), and the distance is approximately 662.9 km1. In the winter, water level of the TGR is 175 m, whereas water level of the TGR is only 145 m in the summer for the aim of flood control2. This water fluctuation every year produces an area of 400 km2 along the upper stream of Yangtze River called water-level fluctuating zone (WLFZ). Meanwhile, it has been gradually recognized that the water fluctuation in TGR region may potentially affect the environmental and the ecological processes, i.e., sediment deposition, ecological sensitivity, soil erosion, and nutrient release3,4. More importantly, the potential pollution from both organic and inorganic pollutants may also be formed in the TGR region5,6,7.
With the rapid development of industrialization and urbanization, the TGR region along Yangtze River would receive a large amount of industrial and residential wastewater and release the large sources of water pollutants into the Yangtze River8. In freshwater ecosystems in TGR region, dissolved heavy metals, organic pollutants, surface-suspended particulate matter, and microplastic pollutant have been detected5,7,9,10,11. Bacterioplankton assemblage analysis in the TGR region further revealed the ecological and the spatial-temporal variations in bacterioplankton community composition12,13. Several reports have implied that the water fluctuation in TGR region may cause the disruptions on algal biomass, species composition, diversity, and richness14,15,16.
Nematodes are the most abundant metazoan in the soil environment. A free-living nematode, Caenorhabditis elegans, is a simple multicellular eukaryote17. Meanwhile, its sensitivity to different toxicants or stresses makes it suitable to offer a model for asking in vivo toxicological questions18,19,20,21,22,23,24,25,26,27. Besides the endpoint of lethality, some useful sublethal endpoints, such as lifespan, development, reproduction, locomotion behavior, and oxidative stress, have been employed to assess the toxicity from different environmental toxicants28,29,30,31,32. More importantly, several reports have demonstrated that C. elegans can be used for ecological risk assessment in water or in sediments33,34,35,36,37,38,39,40,41,42.
So far, most of the data on safety assessment of water samples from TGR region has been analyzed based on chemical analysis5,11,43. In contrast, the exposomics is urgently used to effectively assess the safety issue in the environment in the TGR region44. In China, Chongqing is a city with heavy industry in the central urban area. Wanzhou is further one of the main urban areas in Chongqing in the TGR region. In the present study, we employed the C. elegans to perform the in vivo ecotoxicological assessment of Wanzhou watershed in the flood season in the TGR region. Our study provides an important highlight for our understanding the ecotoxicological risk of watershed in the flood season in the TGR region.
Results
Effects of different surface water samples in the TGR region on survival and body length
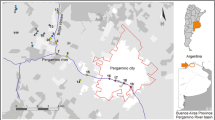
In this study, the surface water samples were from 5 sampling sites (W1, shore water; W2, upstream; W3, shore water; W4, downstream; and W5, backwater area) in Wanzhou, Chongqing (Fig. 1). We first investigated the effects of different surface water samples from TGR region on survival and development of wild-type nematodes. Exposures were performed from L4-larvae for 24-h (acute exposure) in liquid solutions in the presence of food (OP50). After exposure, we found that all the examined surface water samples from TGR region did not affect the survival of wild-type nematodes (Fig. 2a). Similarly, all the examined surface water samples from TGR region could not significantly influence the body length of wild-type nematodes (Fig. 2b). Therefore, all the examined surface water samples from TGR region may be not able to affect the survival and body length in wild-type nematodes.
Effects of different surface water samples in the TGR region on survival and development of wild-type nematodes. (a) Effects of different surface water samples on survival. (b) Effects of different surface water samples on development. Development was assessed by the endpoint of body length. Exposures were performed from L4-larvae for 24-h. Bars represent means ± SD.
Effect of different surface water samples in the TGR region on induction of oxidative stress
Oxidative stress is normally considered as a common mechanism for the toxicity formation from environmental toxicants or stresses in nematodes18,45,46,47. No obvious production of intestinal reactive oxygen species (ROS) was detected in wild-type nematodes after exposure to surface water sample of W1, W2, W3, or W4 (Fig. 3a). In contrast, we found the severe intestinal ROS production after exposure to the surface water sample W5 (Fig. 3a).
Effect of different surface water samples in the TGR region on induction of oxidative stress in wild-type nematodes. (a) Effect of different surface water samples on induction of intestinal ROS production. The left shows the pictures of intestinal ROS production, and the right indicates the comparison of relative fluorescence intensity for signals labeling ROS production in intestine. (b) Effect of different surface water samples on GST-4::GFP expression. Thirty animals were analyzed per treatment. (c) Comparison of expression patterns for genes required for oxidative stress. Exposures were performed from L4-larvae for 24-h. Bars represent means ± SD. **P < 0.01 vs control.
GST-4, a putative glutathione-requiring prostaglandin D synthase, can be increased by exposure to paraquat, a ROS generator, and act as an oxidative stress-response protein in C. elegans48. We further used the transgenic strain of GST-4::GFP to determine the role of examined surface water samples in inducing ROS production. We did not found the obvious alteration in GST-4::GFP expression after exposure to the surface water sample of W1, W2, W3, or W4 (Fig. 3b). Different from this, we observed the expressional increase in the GST-4::GFP after exposure to surface water sample W5 (Fig. 3b).
In nematodes, genes encoding superoxide dismutases (SODs) (sod-1-5), subunits of mitochondrial complex or components in electron transfer chain (isp-1, mev-1, clk-1, and gas-1), insulin signaling (daf-16), and p38 mitogen-activated protein kinase (p38 MAPK) signaling (pmk-1) are involved in the regulation of oxidative stress49,50,51,52,53,54,55,56. After exposure to the surface water sample of W5, no alteration in transcriptional expressions of isp-1, gas-1, sod-1, sod-3, sod-4, daf-16, and pmk-1 was detected (Fig. 3c). In contrast, exposure to the surface water sample of W5 increased expressions of sod-2, sod-5, and clk-1, and decreased mev-1 expression (Fig. 3c). sod-2 encodes a manganese SOD, and sod-5 encodes a copper/zinc SOD.
The insulin signaling pathway can normally regulate the biological processes, such as longevity, through limiting the DAF-16 nuclear localization57. Nevertheless, with the aid of transgenic strain of zIs356, we found that exposure to all the examined surface water samples could not induce the obvious increase in DAF-16:GFP expression in the nuclei (data not shown).
Effect of different surface water samples in the TGR region on intestinal permeability
We further investigated the possible effect of surface water samples in the TGR region on the function of intestine based on the assay of the state of intestinal permeability. Using a non-absorbable blue food dye58, we measured the intestinal barrier function after exposure to the examined surface water samples. In control nematodes, the dye was mainly distributed within the intestinal lumen (Fig. 4a). Similarly, after exposure to the surface water sample of W1, W2, W3, or W4, the dye was also mainly accumulated within the intestinal lumen (Fig. 4). Different from this, exposure to the surface water sample of W5 caused the obvious dye leakage from the intestinal lumen into the body cavity (Fig. 4a), suggesting the potential decline in intestinal integrity in wild-type nematodes exposed to the surface water sample of W5.
Effect of different surface water samples in the TGR region on intestinal permeability in wild-type nematodes. (a) Analysis on the dye distribution and leakage from the intestinal lumen into the body cavity. Arrowheads indicate the dye leakage from the intestinal lumen into the body cavity. (b) Effect of exposure to the sample of W5 on expressions of genes required for the control of intestinal development. Exposures were performed from L4-larvae for 24-h. Bars represent means ± SD. **P < 0.01 vs control.
Moreover, exposure to the surface water sample of W1, W2, W3, or W4 did not significantly affect the transcriptional expressions of egl-8, eps-8, act-5, opt-2, nhx-2, pgp-1, let-413, pkc-3, erm-1, and pgp-3, which are required for the control of intestinal development25 (data not shown). Meanwhile, exposure to the surface water sample of W5 also did not significantly affect the transcriptional expressions of egl-8, eps-8, nhx-2, pgp-1, let-413, pkc-3, and pgp-3 (Fig. 4b). In contrast, exposure to the surface water sample of W5 significantly decreased the transcriptional expressions of erm-1 and opt-2, and increased the transcriptional expression of act-5 (Fig. 4b). In nematodes, ACT-5, ERM-1, EPS-8, PKC-3, OPT-2, NHX-2, PGP-1, and PGP-3 are required for the control of development of intestinal apical domain, LET-413 is required for the control of development of intestinal basolateral domain, and EGL-8 is required for the control of development of intestinal apical junctions. These data implies the possible deficit in development of intestinal apical domain, which further provides an important molecular basis for the intestinal dysfunction in nematodes exposed to the sample of W5.
Effect of different surface water samples in the TGR region on reproduction and locomotion behaviour
In nematodes, neurons and reproductive organs are important secondary targeted organs, once the environmental toxicants can be translocated through the intestinal barrier59. After exposure, surface water sample of W1, W2, W3, or W4 could not significantly alter both the head thrash and the body bend (Fig. 5a). In contrast, exposure to surface water sample of W5 significantly decreased both the head thrash and the body bend (Fig. 5a). Therefore, among the examined five surface water samples in the TGR region, exposure to W5 may cause the alteration in function of motor neurons.
Effects of different surface water samples in the TGR region on locomotion behavior and reproduction in wild-type nematodes. (a) Effect of different surface water samples on locomotion behavior. Locomotion behavior was assessed by the endpoints of head thrash and body bend. (b) Effect of different surface water samples on reproduction. Reproduction was assessed by the endpoint of brood size. Exposures were performed from L4-larvae for 24-h. Bars represent means ± SD. **P < 0.01 vs control.
Different from the effect of surface water samples in the TGR region on locomotion behavior, we found that all the examined surface water samples (W1, W2, W3, W4, and W5) could not significantly affect the brood size of wild-type nematodes (Fig. 5b).
Effect of different surface water samples in the TGR region on lifespan
We also selected the endpoint of lifespan to investigated the long-term effect of examine surface water samples in the TGR region on wild-type nematodes. After acute exposure, we found that all the examined five original surface water samples did not alter the lifespan (Fig. 6). Therefore, short-term exposure to the examined surface water samples in the TGR region may be not able to alter the lifespan.
Toxicity assessment of surface water samples in sod-2 or sod-5 mutant nematodes
Exposure to the surface water sample of W5 could induce the significant increase in sod-2 and sod-5 as indicated above. In C. elegans, mutation of sod-2 or sod-5 could cause the susceptibility to toxicity of environmental toxicants60,61. In sod-2 or sod-5 mutant nematodes exposed to the surface water sample of W1, W2, W3, or W4, we still did not detect their adverse effects in inducing intestinal ROS production, and on locomotion behavior or brood size (Fig. 7). In sod-2 or sod-5 mutant nematodes, all the examined five surface water samples still could not affect the survival, the body length, and the lifespan (data not shown). In contrast, the sod-2 or sod-5 mutant nematodes were susceptible to toxicity of surface water sample of W5 in the induction of ROS production and in the decrease of locomotion behavior (Fig. 7a and b). In sod-2 or sod-5 mutant nematodes exposed to the surface water sample of W5, we further found the significant reduction in brood size (Fig. 7c).
Effect of sod-2 or sod-5 mutation on the induction of ROS production (a), the locomotion behavior (b), and the brood size (c) in nematodes exposed to the examined different surface water samples in the TGR region. Exposures were performed from L4-larvae for 24-h. Bars represent means ± SD. **P < 0.01 vs control (if not specially indicated).
Analysis on the chemical pollutants from the sample W5
To determining the chemical pollutants from the sample of W5 in inducing toxicity on nematodes, we isolated the liquid phase and the solid phase from the sample of W5 by centrifugation at 10000 g for 10-min. After centrifugation, the liquid phase existed in the supernatant. The pellet was re-suspended with the equal volume of K medium to obtain the solution for the solid phase. After exposure, we found that both the liquid phase and the solid phase could cause the toxicity in inducing intestinal ROS production and on the locomotion behavior (Fig. 8).
In the liquid phase of sample of W5, the elemental analysis of surface water samples further showed Mg as the most abundant element, followed by K and Fe (Table S1). The examined surface water samples also contained moderate amounts of As, Hg, and Pb (Table S1). The content of Mg, K, As, Hg, or Pb in the surface water sample of W5 did not show the obvious difference from that in the other examined surface water samples (Table S1). We only found the moderate increase in the Fe content in the surface water sample of W5 compared with that in the other examined surface water samples (Table S1). However, acute exposure to Fe (0.242 mg/L) did not result in the obvious toxicity on nematodes using lethality, body length, intestinal ROS production, brood size, locomotion behavior, and lifespan as the endpoints (data not shown).
Discussion
In this study, we performed the safety assessment on the effect of five original surface water samples from the TGR region using the model animal of C. elegans. The collected surface water samples are from upstream, shore water, downstream, and backwater area in Yangtze River in Wanzhou, Chongqing (Fig. 1). We selected the flood season (September) to collect the water samples in order to assess the possible effects of some chemicals released from the sediment or surrounding mountains. We performed the acute exposure in this study, because we noticed the existence of certain amount of bacterial community in the water in Yangtze River in TGR region12,13. Meanwhile, all the exposures were performed in the liquid solutions for nematodes at 20 °C.
Based on the toxicity assessment performed in this study, we found that short-term exposure to all the examined five original surface water samples from the TGR region did not significantly affect the survival, the body length, the brood size, and the lifespan wild-type nematodes (Figs 2, 5b and 6). These observations suggest that short-term exposure to all the examined original surface water samples from the TGR region may be not able to influence the survival, the development, the reproduction, and the longevity. Meanwhile, we also did not detect the obvious effect from acute exposure to surface water sample of W1, W2, W3, or W4 in inducing intestinal ROS production (Fig. 3a), in increasing GST-4::GFP expression (Fig. 3b), in enhancing intestinal permeability (Fig. 4), and in decreasing locomotion behavior as reflected by the endpoints of head thrash and body bend (Fig. 5a), which imply that the surface water samples collected from the upstream, shore water, or downstream may also be not able to influence the intestinal function, and the functions of motor neurons in nematodes after short-term exposure. Considering the existence of certain amount of bacterial community in the examined original surface water samples from the TGR region, so far it is difficult to determine the potential effects of long-term exposure to these examined original surface water samples on survival, development, reproduction, and longevity, as well as the effects on intestinal function and locomotion behavior.
We here detected the obvious intestinal ROS production (Fig. 3a), GST-4::GFP increase (Fig. 3b), enhanced intestinal permeability (Fig. 4), and decrease in locomotion behavior (Fig. 5a) in wild-type nematodes acutely exposed to the surface water sample of W5. The surface water sample of W5 was collected from the backwater area in Yangtze River in Wanzhou, Chongqing. Therefore, our results imply that short-term exposure to the surface water sample from the backwater area in Yangtze River in Wanzhou may potentially affect both the intestinal function as reflected by the enhanced intestinal permeability and the function of motor neurons as reflected by the decreased locomotion behavior in nematodes. One possibility is that the disrupted intestinal barrier may allow the entering of a large amount of pollutants from the sample of W5 into the targeted organs, such as the neurons.
Based on the analyzed physicochemical properties of collected five original surface water samples from the TGR region, we did not find the obvious difference in water level, longitude for sampling site, latitude for sampling site, altitude, water temperature, turbidity, pH value, total dissolved solids, biochemical oxygen demand, and chemical oxygen demand for the surface water sample of W5 from those for the surface water sample of W1, W2, W3, or W4 (Table S2). Therefore, the detected adverse effects of original surface water sample of W5 on intestinal function, locomotion behavior, and brood size may be not due to the difference in physicochemical properties from the examined other original surface water samples.
In the freshwater in the TGR region, dissolved heavy metals, and organic pollutants, such as endocrine-disrupting compounds, have been detected7,9,11. In the soil or the sediment in the TGR region, inorganic compounds, such as herbicide, and heavy metals, such as Hg and Cd, have also been detected62,63,64. Moreover, in the fresh water in the TGR region, two dominated bacterioplankton communities, Proteobacteria (39.26%) and Actinobacteria phyla (37.14%), were detected13. Recently, the evidence has been raised that the microbial structure and function can be affected by the environmental changes from the Three Gorges Dam65,66. In this study, we found that both the liquid phase and the solid phase contributed to the toxicity of the sample of W5 on nematodes (Fig. 8). Additionally, the observed acute toxicity on nematodes from the liquid phase of sample of W5 may not mainly due to the existence of heavy metals (Table S1), which implied that the potential pollutants in the liquid phase of sample of W5 may be the organic pollutants. The further identification of organic pollutants in the liquid phase and pollutants of bacterial community in the solid phase for the collected original surface water sample of W5 may be helpful for answering the reasons for the observed adverse effects of W5 on nematodes.
In this study, we further found that exposure to the surface water sample of W5 could significantly increase the expressions of sod-2, sod-5, and clk-1, and decrease the expression of mev-1 (Fig. 3b). In C. elegans, mev-1 and clk-1 belong to the components constituting the molecular machinery in controlling the activation of oxidative stress50,53. This observation suggests that exposure to the surface water sample of W5 may result in intestinal ROS production by altering the molecular basis of oxidative stress. In C. elegans, increase in sod-2 and sod-5 may mediate a protection response for animals against the toxic effect from environmental toxicants or stresses45,49. Therefore, the surface water sample of W5 may also potentially activate a protection response in nematodes after exposure. Nevertheless, the activated protection response may be not able to counteract the adverse effect from the exposure to surface water sample of W5 in nematodes.
The sod-2 or sod-5 mutant was susceptible to the toxicity of environmental toxicants60,61. Moreover, we found that exposure to the surface water sample of W1, W2, W3, or W4 still could not cause the adverse effects in sod-2 or sod-5 mutant nematodes (Fig. 7). These results confirm the safe property for short-term exposure to the surface water sample of W1, W2, W3, or W4 on environmental organisms. Additionally, we observed that exposure to the surface water sample of W5 could result in the more severe toxicity in sod-2 or sod-5 mutant compared with wild-type nematodes (Fig. 7a and b), and mutation of sod-2 or sod-5 even caused the significant reduction in brood size (Fig. 7c). These observations demonstrate that the potential adverse effects of surface water sample of W5 can be further strengthened in environmental organisms with the susceptible property.
In conclusion, we employed the model animal of C. elegans to perform the safety assessment of the collected original surface water samples in the TGR region. Using lethal and some sublethal endpoints, we found that all the examined original surface water samples from the TGR region did not significantly affect the survival, development, reproduction, and longevity in wild-type nematodes. Exposure to the surface water sample of W1, W2, W3, or W4 also did not obviously induce the intestinal ROS production, alter the intestinal permeability, and affect the locomotion behavior. Additionally, exposure to the surface water sample of W1, W2, W3, or W4 could not cause the adverse effects on sod-2 or sod-5 mutant nematodes. In contrast, only exposure to the surface water sample of W5 could significantly induce the intestinal ROS production, enhance the intestinal permeability, and decrease the locomotion behavior. Moreover, mutation of sod-2 or sod-5 was susceptible to the adverse effects of the surface water sample of W5 on nematodes. The further analysis on the pollutants from the sample of W5 implies that both the liquid phase and the solid phase contributed to the observed toxicity in nematodes. Therefore, our results suggest that the surface water sample collected from the backwater area in Yangtze River in Wanzhou, Chongqing may have adverse effects on environmental organisms. Our data provide the important basis for the further systematic safety evaluation of the surface water samples collected in Yangtze River in the TGR region.
Methods
Study sites and sample collection
The surface water samples were collected from 5 sampling sites (W1, shore water; W2, upstream; W3, shore water; W4, downstream; and W5, backwater area) in Wanzhou, Chongqing. The sampling season was selected in the flood season (September 18, 2017). At this season, the bacterioplankton community was generally higher than that in the impoundment season13. Samples were stored in a car refrigerator with about 0 °C after collection. Water samples were collected according to the standard method67, and analyzed after being transported back to the laboratory in a cooler.
Water temperature and pH were measured in situ using a portable SG2-ELK pH meter (Mettler-Toledo Co., Shanghai, China). Total dissolved solids were measured in situ using a portable SG3 conductivity meter (Mettler-Toledo Co., Shanghai, China). Turbidity was measured using a 1900C turbidimeter (Hach Co., Colorado, USA). Biochemical oxygen demand was analyzed according to the Chinese standard examination method for water quality (HJ 505–2009). Chemical oxygen demand was analyzed according to Chinese standard examination method for water quality (GB 11914–89). The daily flow and water level data of sampling sites were obtained from Yangtze River Hydrological Network (http://www.cjh.com.cn). The related information for the collected surface water samples in TGR region is shown in Table S2. Major elements of surface water samples were determined by inductively coupled plasma atomic emission spectroscopy (ICP, GE Co., USA).
C. elegans maintenance
The used nematode strain was wild-type N2, mutants of sod-2(gk257) and sod-5(tm1146), and transgenic strains of zIs356[Pdaf-16::daf-16a/b::GFP], and CL2166/dvIs19[gst-4::GFP]. Gravid nematodes were transferred into centrifuge tubes, and lysed with bleaching solution to separate the eggs and the animals. Age synchronous population of L4-larvae was obtained as described68.
Exposure
Exposures were performed from L4-larvae for 24-h (acute exposure) in liquid solutions in the presence of food (OP50) at 20 °C. After exposure, nematodes were used for toxicity assessment with lethality, body length, intestinal ROS production, brood size, locomotion behavior, and lifespan as the endpoints. Control exposure was the treatment with liquid K medium. The exposure was performed in triple replicates.
Toxicity assessment
For the lethality assay, the nematodes are judged to be dead if they can not respond to the stimulus using a small metal wire. Fifty nematodes were examined per treatment.
Body length was used to assess the growth of nematodes. Body length was determined by measuring the flat surface length of nematodes using Image-Pro® Express software. Thirty nematodes were examined per treatment.
Intestinal ROS production was used to assess the oxidative stress in intestine69. The method was performed as described previously70. The 5′,6′-chloromethyl-2′,7′dichlorodihydro-fluorescein diacetate (CM-H2DCFDA) can detect the presence of intracellular produced ROS species. The examined nematodes were incubated with 1 μM CM-H2DCFDA solution for 3 h in the dark. The examined nematodes were analyzed at 488 nm of excitation wavelength and at 510 nm of emission filter under a laser scanning confocal microscope. Relative fluorescence intensity of ROS signals was semi-quantified in comparison to the intestinal autofluorescence. Thirty nematodes were examined per treatment.
The method for analysis for head thrash and body bend was performed as described previously71,72. The behaviors were analyzed under the dissecting microscope by eyes. A head thrash is defined as a change in the direction of bending at the mid body, and a body bend is defined as a change in the direction of the part of the nematodes corresponding to the posterior bulb of the pharynx along the y axis, assuming that nematode was traveling along the x axis. Fifty nematodes were examined per treatment.
The method for brood size was performed as described previously73,74. The number of offspring at all stages beyond the egg was counted. Thirty nematodes were examined per treatment.
Lifespan was used to reflect the long-term effect of certain toxicants in nematodes. The method was performed as described previously75. After the exposure, we started to record the lifespan of nematodes. During the lifespan recording, the hermaphrodites were transferred daily for the first 7 days of adulthood in order to avoid the possible effect from the progeny. Nematodes were checked every day. Fifty nematodes were examined per treatment. Graphs are representative of at least three trials.
Reverse-transcription and quantitative real-time polymerase chain reaction (qRT-PCR)
After synthesis of the cDNA, relative transcriptional expressions of certain genes were analyzed in an ABI 7500 real-time PCR system (Biotium, USA). Relative quantification of targeted genes in comparison to a reference gene of tba-1 encoding a tubulin was determined. Primer information is provided in Table S3.
Intestinal permeability assay
The examined nematodes were suspended in blue food dye (5.0% wt/vol in water) in the presence of OP50 for 3 h. After that, the nematodes were transferred onto normal NGM plates seeded with OP50 to analyze the blue food dye in the body cavity using a microscope. Twenty nematodes were examined per treatment.
Statistical analysis
Statistical analysis was performed using SPSS 12.0 software. The differences between groups was determined using analysis of variance (ANOVA). Probability level of 0.05 was considered as the statistically significant. The lifespan data were analyzed using a 2-tailed 2 sample t-test (Minitab Ltd, Coventry, UK).
Change history
18 October 2018
A correction to this article has been published and is linked from the HTML and PDF versions of this paper. The error has been fixed in the paper.
References
Ye, L., Han, X., Xu, Y. & Cai, Q. Spatial analysis for spring bloom and nutrient limitation in Xiangxi Bay of Three Gorges Reservoir. Environ. Monitor. Assess. 127, 1351145 (2017).
Li, Z. et al. Soil–air greenhouse gas fluxes influenced by farming practices in reservoir drawdown area: A case at the Three Gorges Reservoir in China. J. Environ. Manag. 181, 64–73 (2016).
Strehmel, A., Schmalz, B. & Fohrer, N. Evaluation of land use, land management and soil conservation strtegies to reduce non-point source pollution loads in the Three Gorges Region, China. Environ. Manag. 58, 906–921 (2016).
Huang, Y. et al. Air-water CO2 and CH4 fluxes along a river-reservior continuum: case study in the Pengxi River, a tributary of the Yangtze River in the Three Gorges Reservior, China. Environ. Monit. Assess. 189, 223 (2017).
Zhao, P., Tang, X., Tang, J. & Wang, C. Assessing water quality of Three Gorges Reservoir, China, over a five-year period from 2006 to 2011. Water. Resour. Manag. 27, 4545–4558 (2013).
Ye, C., Cheng, X., Liu, W. & Zhang, Q. Revegetation impacts soil nitrogen dynamics in the water level fluctuation zone of the Three Gorges Reservoir, China. Sci. Total. Environ. 517, 76–85 (2015).
Zhang, K. et al. Occurrence and characteristics of microplastic pollution in Xiangxi Bay of Three Gorges Reservior, China. Environ. Sci. Technol. 51, 3794–3801 (2017).
Zhang, J. & Deng, W. Industrial structure change and Iits eco-environmental influence since the establishment of municipality in Chongqing, China. Proc. Environ. Sci. 2, 517–526 (2010).
Wang, W., Ndungu, A. W. & Wang, J. Monitoring of endocrine-disrupting compounds in surface water and sediments of the Three Gorges Reservior region, China. Arch. Environ. Contam. Toxicol. 71, 509–517 (2016).
Gao, M., Zhu, K., Bi, Y. & Hu, Z. Spatiotemporal patterns of surface-suspended particulate matter in the Three Gorges Reservior. Environ. Sci. Pollut. Res. 23, 3569–3577 (2016).
Zhao, X., Li, T., Zhang, T., Luo, W. & Li, J. Distribution and health risk assessment of dissolved heavy metals in the Three Reservior, China (section in the main urban area of Chongqing). Environ. Sci. Pollut. Res. 24, 2697–2710 (2017).
Xiao, G. et al. Occurrence and infection risk of waterborne pathogens in Wanzhou watershed of the Three Gorges Reservior, China. J. Environ. Sci. 25, 1913–1924 (2013).
Li, Z. et al. Responses of spatial-temporal dynamics of bacterioplankton community to large-scale reservoir operation: a case study in the Three Gorges reservoir, China. Sci. Rep. 7, 42469 (2017).
Wu, J. et al. The three gorges dam: an ecological perspective. Front. Ecology Environ. 2, 241–248 (2004).
Floehr, T. et al. Yangtze Three Gorges Reservoir, China: A holistic assessment of organic pollution, mutagenic effects of sediments and genotoxic impacts on fish. J. Environ. Sci. 38, 63–82 (2015).
Nie, Y., Zhang, Z., Shen, Q., Gao, W. & Li, Y. Significance of different carbon forms and carbonic anhydrase activity in monitoring and prediction of algal blooms in the urban section of Jialing River, Chongqing, China. Environ. Sci. Processes & Impacts 18, 600–612 (2016).
Brenner, S. The genetics of Caenorhabditis elegans. Genetics 77, 71–94 (1974).
Leung, M. C. et al. Caenorhabditis elegans: an emerging model in biomedical and environmental toxicology. Toxicol. Sci. 106, 5–28 (2008).
Zhuang, Z.-H. et al. Function of RSKS-1-AAK-2-DAF-16 signaling cascade in enhancing toxicity of multi-walled carbon nanotubes can be suppressed by mir-259 activation in Caenorhabditis elegans. Sci. Rep. 6, 32409 (2016).
Helmcke, K. J., Syversen, T., Miller, D. M. III & Aschner, M. Characterization of the effects of methylmercury on Caenorhabditis elegans. Toxicol. Appl. Pharmacol. 240, 265–272 (2009).
Svendsen, C., Siang, P., Lister, L. J., Rice, A. & Spurgeon, D. J. Similarity, independence, or interaction for binary mixture effects of nerve toxicants for the nematode Caenorhabditis elegans. Environ. Toxicol. Chem. 29, 1182–1191 (2010).
McElwee, M. K., Ho, L. A., Chou, J. W., Smith, M. V. & Freedman, J. H. Comparative toxicogenomic responses of mercuric and methyl-mercury. BMC Genom. 14, 698 (2013).
Zhi, L.-T., Fu, W., Wang, X. & Wang, D.-Y. ACS-22, a protein homologous to mammalian fatty acid transport protein 4, is essential for the control of toxicity and translocation of multi-walled carbon nanotubes in Caenorhabditis elegans. RSC Adv. 6, 4151–4159 (2016).
Du, H. et al. Endosulfan isomers and sulfate metabolite induced reproductive toxicity in Caenorhabditis elegans involves genotoxic response genes. Environ. Sci. Technol. 49, 2460–2468 (2015).
Wu, Q.-L. et al. Contributions of altered permeability of intestinal barrier and defecation behavior to toxicity formation from graphene oxide in nematode Caenorhabditis elegans. Nanoscale 5, 9934–9943 (2013).
Zhao, L., Rui, Q. & Wang, D.-Y. Molecular basis for oxidative stress induced by simulated microgravity in nematode Caenorhabditis elegans. Sci. Total Environ. 607-608, 1381–1390 (2017).
Zhao, L., Wan, H.-X., Liu, Q.-Z. & Wang, D.-Y. Multi-walled carbon nanotubes-induced alterations in microRNA let-7 and its targets activate a protection mechanism by conferring a developmental timing control. Part. Fibre Toxicol. 14, 27 (2017).
Roh, J. et al. Ecotoxicity of silver nanoparticles on the soil nematode Caenorhabditis elegans using functional ecotoxicogenomics. Environ. Sci. Technol. 43, 3933–3940 (2009).
Zhao, L., Qu, M., Wong, G. & Wang, D.-Y. Transgenerational toxicity of nanopolystyrene particles in the range of μg/L in nematode Caenorhabditis elegans. Environ. Sci.: Nano 4, 2356–2366 (2017).
Yu, X.-M., Guan, X.-M., Wu, Q.-L., Zhao, Y.-L. & Wang, D.-Y. Vitamin E ameliorates the neurodegeneration related phenotypes caused by neurotoxicity of Al2O3-nanoparticles in C. elegans. Toxicol. Res. 4, 1269–1281 (2015).
Qu, M., Li, Y.-H., Wu, Q.-L., Xia, Y.-K. & Wang, D.-Y. Neuronal ERK signaling in response to graphene oxide in nematode Caenorhabditis elegans. Nanotoxicology 11, 520–533 (2017).
Zhi, L.-T. et al. Graphene oxide induces canonical Wnt/β-catenin signaling-dependent toxicity in Caenorhabditis elegans. Carbon 113, 122–131 (2017).
Wolfram, G. et al. Assessing the impact of chemical pollution on benthic invertebrates from three different European rivers using a weight-of-evidence approach. Sci. Total Environ. 438, 498–509 (2012).
Turner, E. A. et al. Assessing different mechanisms of toxicity in mountaintop removal/valley fill coal mining-affected watershed samples using Caenorhabditis elegans. PLoS ONE 8, e75329 (2013).
Höss, S., Henschel, T., Haitzer, M., Traunspurger, W. & Steinberg, C. E. Toxicity of cadmium to Caenorhabditis elegans (Nematoda) in whole sediment and pore water–the ambiguous role of organic matter. Environ. Toxicol. Chem. 20, 2794–801 (2001).
Bragigand, V. et al. Influence of biological and ecological factors on the bioaccumulation of polybrominated diphenyl ethers in aquatic food webs from French estuaries. Sci. Total Environ. 368, 615–626 (2006).
Zhang, H. et al. Ecotoxicological assessment of lanthanum with Caenorhabditis elegans in liquid medium. Metallomics 2, 806–810 (2010).
Tejeda-Benítez, L., Noguera-Oviedo, K., Aga, D. S. & Olivero-Verbel, J. Toxicity profile of organic extracts from Magdalena River sediments. Environ. Sci. Pollut. Res. Int. 25, 1519–1532 (2018).
Haegerbaeumer, A. et al. A comparative approach using ecotoxicological methods from single-species bioassays to model ecosystems. Environ. Toxicol. Chem. 35, 2987–2997 (2016).
Traunspurger, W. et al. Ecotoxicological assessment of aquatic sediments with Caenorhabditis elegans (Nematoda)-A method for testing liquid medium and whole-sediment samples. Environ. Toxicol. Chem. 16, 245–250 (1997).
Höss, S. et al. Nematode species at risk - A metric to assess pollution in soft sediments of freshwaters. Environ. Int. 37, 940–949 (2011).
International Organization for Standardization. Water quality determination of the toxic effect of sediment and soil samples on growth, fertility and reproduction of Caenorhabditis elegans (Nematoda). ISO 10872:2010. Geneva, Switzerland (2010).
Tang, J. et al. Tempo-spatial analysis of water quality in tributary bays of the Three Gorges Reservior region (China). Environ. Sci. Pollut. Res. 22, 16709–16720 (2015).
Schramm, K. et al. Chemical- and effect-oriented exposomics: Three Gorges Reservior (TGR). Environ. Sci. Pollut. Res. 20, 7075–7062 (2013).
Ren, M.-X., Zhao, L., Lv, X. & Wang, D.-Y. Antimicrobial proteins in the response to graphene oxide in Caenorhabditis elegans. Nanotoxicology 11, 578–590 (2017).
Tejeda-Benitez, L. & Olivero-Verbel, J. Caenorhabditis elegans, a biological model for research in toxicology. Rev. Environ. Contam. Toxicol. 237, 1–35 (2016).
Estevez, A. O. et al. Selenium induces cholinergic motor neuron degeneration in Caenorhabditis elegans. Neurotoxicology 33, 1021–1032 (2012).
Tawe, W., Eschbach, M.-L., Walter, R. D. & Henkle-Duehrsen, K. Identification of stress-responsive genes in Caenorhabditis elegans using RT-PCR differential display. Nucleic Acids Res. 26, 1621–1627 (1998).
Hunter, T., Bannister, W. H. & Hunter, G. J. Cloning, expression, and characterization of two manganese superoxide dismutases from Caenorhabditis elegans. J. Biol. Chem. 272, 28652–28659 (1997).
Ishii, N. et al. A mutation in succinate dehydrogenase cytochrome b causes oxidative stress and ageing in nematodes. Nature 394, 694–697 (1998).
Kayser, E. B., Morgan, P. G., Hoppel, C. L. & Sedensky, M. M. Mitochondrial expression and function of GAS-1 in Caenorhabditis elegans. J. Biol. Chem. 276, 20551–20558 (2001).
Feng, J., Bussiere, F. & Hekimi, S. Mitochondrial electron transport is a key determinant of life span in Caenorhabditis elegans. Dev. Cell 1, 633–644 (2001).
Miyadera, H. et al. Altered quinone biosynthesis in the long-lived clk-1 mutants of Caenorhabditis elegans. J. Biol. Chem. 276, 7713–7716 (2001).
Zhao, Y.-L. et al. p38 MAPK-SKN-1/Nrf signaling cascade is required for intestinal barrier against graphene oxide toxicity in Caenorhabditis elegans. Nanotoxicology 10, 1469–1479 (2016).
Hu, Q., D’Amora, D. R., MacNeil, L. T., Walhout, A. J. M. & Kubiseski, T. J. The oxidative stress response in Caenorhabditis elegans requires the GATA transcriptional factor ELT-3 and SKN-1/Nrf2. Genetics 206, 1909–1922 (2017).
Tullet, J. M. A. et al. The SKN-1/Nrf2 transcriptional factor can protect against oxidative stress and increase lifespan in C. elegans by distinct mechanisms. Aging Cell 16, 1191–1194 (2017).
Lin, K., Hsin, H., Libina, N. & Kenyon, C. Regulation of the Caenorhabditis elegans longevity protein DAF-16 by insulin/IGF-1 and germline signaling. Nat. Genet. 28, 139–145 (2001).
Gelino, S. et al. Intestinal autophagy improves healthspan and longevity in C. elegans during dietary restriction. PLoS Genet. 12, e1006135 (2016).
Zhao, Y.-L., Wu, Q.-L., Li, Y.-P. & Wang, D.-Y. Translocation, transfer, and in vivo safety evaluation of engineered nanomaterials in the non-mammalian alternative toxicity assay model of nematode Caenorhabditis elegans. RSC Adv. 3, 5741–5757 (2013).
Wu, Q.-L., Zhao, Y.-L., Li, Y.-P. & Wang, D.-Y. Susceptible genes regulate the adverse effects of TiO2-NPs at predicted environmental relevant concentrations on nematode Caenorhabditis elegans. Nanomedicine: Nanotechnol. Biol. Med. 10, 1263–1271 (2014).
Song, S. et al. Molecular basis for antioxidant enzymes in mediating copper detoxification in the nematode Caenorhabditis elegans. PLoS One 9, e107685 (2014).
Chen, Z., Schmidt, B. & Schaffer, A. Uptake and decomposition of the herbicide propanil in the plant Bidens pilosa L. dominating in the Yanftze three Gorges Reservior (TGR), China. Environ. Sci. Pollut. Res. 24, 11141–11153 (2017).
Du, H. et al. Mercury-methylating genes dsrB and hgcA in soils/sediments of the Three Gorges Reservior. Environ. Sci. Pollut. Res. 24, 5001–5011 (2017).
Wang, T., Pan, J. & Liu, X. Characterization of heavy metal contamination in the soil and sediment of the Three Gorges Reservior, China. J. Environ. Sci. Health Part A 52, 201–209 (2017).
Xiao, G. et al. Occurrence and potential health risk of Cryptosporidium and Giardia in the Three Gorges Reservior, China. Water Res. 47, 2431–2445 (2013).
Yan, Q. et al. Impact of the Three Gorges Dam on microbial structure and potential function. Sci. Rep. 5, 8605 (2015).
Chen, D. Guidelines for the investigation of aquatic organisms in rivers. China Science Publishing & Media Ltd (2014).
Donkin, S. G. & Dusenbery, D. B. A soil toxicity test using the nematode Caenorhabditis elegans and an effective method recovery. Arch. Environ. Contam. Toxicol. 25, 145–151 (1993).
Liu, Z.-F., Zhou, X.-F., Wu, Q.-L., Zhao, Y.-L. & Wang, D.-Y. Crucial role of intestinal barrier in the formation of transgenerational toxicity in quantum dots exposed nematodes Caenorhabditis elegans. RSC Adv. 5, 94257–94266 (2015).
Yang, R.-L. et al. Metallothioneins act downstream of insulin signaling to regulate toxicity of outdoor fine particulate matter (PM2.5) during Spring Festival in Beijing in nematode Caenorhabditis elegans. Toxicol. Res. 5, 1097–1105 (2016).
Chen, H., Li, H.-R. & Wang, D.-Y. Graphene oxide dysregulates Neuroligin/NLG-1-mediated molecular signaling in interneurons in Caenorhabditis elegans. Sci. Rep. 7, 41655 (2017).
Yang, R.-L., Ren, M.-X., Rui, Q. & Wang, D.-Y. A mir-231-regulated protection mechanism against the toxicity of graphene oxide in nematode Caenorhabditis elegans. Sci. Rep. 6, 32214 (2016).
Wu, Q.-L. et al. Genome-wide identification and functional analysis of long noncoding RNAs involved in the response to graphene oxide. Biomaterials 102, 277–291 (2016).
Zhao, Y.-L., Yang, J.-N. & Wang, D.-Y. A microRNA-mediated insulin signaling pathway regulates the toxicity of multi-walled carbon nanotubes in nematode Caenorhabditis elegans. Sci. Rep. 6, 23234 (2016).
Rui, Q., Lu, Q. & Wang, D.-Y. Administration of Bushenkangshuai Tang alleviates the UV irradiation- and oxidative stress-induced lifespan defects in nematode Caenorhabditis elegans. Front. Med. China 3, 76–90 (2009).
Author information
Authors and Affiliations
Contributions
Conceived and designed the experiments: D.W. Performed the experiments and analyzed the data: G.X., L.Z., Q.H., J.Y., H.D., D.G., M.X., G.L. and Z.C. Wrote the paper: D.W.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Xiao, G., Zhao, L., Huang, Q. et al. Toxicity evaluation of Wanzhou watershed of Yangtze Three Gorges Reservoir in the flood season in Caenorhabditis elegans. Sci Rep 8, 6734 (2018). https://doi.org/10.1038/s41598-018-25048-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-25048-w
- Springer Nature Limited
Keywords
This article is cited by
-
Seasonal pollution and surface characteristics of microplastics in surface water in the Wanzhou section of the Three Gorges Reservoir, China
Environmental Science and Pollution Research (2023)
-
Using Caenorhabditis elegans to assess the ecological health risks of heavy metals in soil and sediments around Dabaoshan Mine, China
Environmental Science and Pollution Research (2022)
-
Biosafety assessment of Acinetobacter strains isolated from the Three Gorges Reservoir region in nematode Caenorhabditis elegans
Scientific Reports (2021)