Abstract
Sympatric lineages of inbreeding species provide an excellent opportunity to investigate species divergence patterns and processes. Many ambrosia beetle lineages (Curculionidae: Scolytinae) reproduce by predominant inbreeding through sib mating in nests excavated in woody plant parts wherein they cultivate symbiotic ambrosia fungi as their sole source of nutrition. The Xyleborini ambrosia beetle species Cnestus solidus and Cnestus pseudosolidus are sympatrically distributed across eastern Australia and have overlapping morphological variation. Using multilocus sequencing analysis of individuals collected from 19 sites spanning their sympatric distribution, we assessed their phylogenetic relationships, taxonomic status and microbial symbionts. We found no genetic differentiation between individuals morphologically identified as C. solidus and C. pseudosolidus confirming previous suggestions that C. pseudosolidus is synonymous to C. solidus. However, within C. solidus we unexpectedly discovered the sympatric coexistence of two morphologically indistinguishable but genetically distinct lineages with small nuclear yet large mitochondrial divergence. At all sites except one, individuals of both lineages carried the same primary fungal symbiont, a new Ambrosiella species, indicating that fungal symbiont differentiation may not be involved in lineage divergence. One strain of the maternally inherited bacterial endosymbiont Wolbachia was found at high prevalence in individuals of the more common lineage but not in the other, suggesting that it may influence host fitness. Our data suggest that the two Australian Cnestus lineages diverged allopatrically, and one lineage then acquired Wolbachia. Predominant inbreeding and Wolbachia infection may have reinforced reproductive barriers between these two lineages after their secondary contact contributing to their current sympatric distribution.
Similar content being viewed by others
Introduction
Inbreeding through mating with siblings (sib mating) or close relatives can be a successful evolutionary strategy. By reducing genetic recombination, inbreeding can lead to the fixation of advantageous genotypes in populations (Whitlock 2003). Sib mating also effectively removes the constraints of mate finding (Andersen et al. 2012) allowing for efficient reproduction and establishment of propagules in novel environments (Jordal et al. 2001). In insects with haplodiploid sex determination systems, such as in the Hymenoptera or in some lineages of the weevil subfamily Scolytinae, negative impacts commonly associated with inbreeding, such as the accumulation of deleterious alleles, are purged in haploid males (Smith 2000; Peer and Taborsky 2005). Despite the apparent evolutionary advantage of inbreeding, few studies have explored whether and how inbreeding affects species diversification rates. In haplodiploid Scolytinae, regular or predominant inbreeding favours dispersal (Jordal et al. 2000, 2001; Gohli et al. 2016) and, ultimately, promotes allopatric divergence and reinforces reproductive isolation after secondary contact of diverged lineages (Gohli et al. 2017). Recent phylogenetic modelling has furthermore identified predominant inbreeding in Scolytinae as a primary factor responsible for increased diversification rates when compared to outbred Scolytinae lineages (Gohli et al. 2017).
Interactions with microbial endosymbionts such as the common endosymbiont Wolbachia (Alphaproteobacteria), which infects 50 to 60% of arthropods species, can contribute to reproductive isolation between arthropod lineages (Brucker and Bordenstein 2012; Weinert et al. 2015; Detcharoen et al. 2019). These intracellular endosymbionts are maternally inherited and can affect host reproduction by inducing a wide array of reproductive manipulations, such as cytoplasmic incompatibility (CI), thelytokous parthenogenesis, male killing and feminisation (Werren et al. 2008; Shropshire and Bordenstein 2016; Kaur et al. 2021). Many of these manipulations promote the production of infected females thereby increasing Wolbachia prevalence, and can also influence host population dynamics, sex ratios, sex determination systems and/or genetic diversity. In particular, the intraspecific genetic diversity of other maternally inherited elements, such as mitochondria, can be skewed when a Wolbachia strain invades host populations (Hurst and Jiggins 2005; Avtzis et al. 2008; Arthofer et al. 2010; Cariou et al. 2017; Arif et al. 2021; Morrow and Riegler 2021). The reproductive and behavioural host manipulations of Wolbachia can also become coupled with other reproductive barriers, promoting reinforcement of reproductive isolation (Telschow et al. 2005; Shropshire and Bordenstein 2016; Cruz et al. 2021; Kaur et al. 2021; Bruzzese et al. 2021). Furthermore, Wolbachia can confer fitness benefits in some host species, including increased fecundity, nutrient provisioning and pathogen defence (Zug and Hammerstein 2015).
Many lineages of Scolytinae ambrosia beetles engage in predominant inbreeding by sib mating (Keller et al. 2011; Storer et al. 2017; Johnson et al. 2018). Recent phylogenetic analyses have identified at least eight independent origins of inbreeding in Scolytinae (Jordal and Cognato 2012; Pistone et al. 2018), illustrating the evolutionary success of predominant inbreeding in these beetles (Kirkendall et al. 2015). This mating system has mostly been explored in Xyleborini species which construct nests in the xylem tissue of woody plants and cultivate symbiotic ambrosia fungi from propagules or spores stored in the beetles’ mycetangia (also commonly referred to as mycangia) (Francke-Grosmann 1956; Hulcr and Stelinski 2017; Mayers et al. 2022). All studied Xyleborini species are haplodiploid, wherein mated diploid foundress females produce diploid daughters and haploid sons. These siblings can mate with one another, and fertilised females then leave the natal nest to locate new host substrates and start new families (Takenouchi and Takagi 1967; Kirkendall 1983; Jordal et al. 2000, 2002). Males of Xyleborini species are typically dwarfed, almost blind and flightless, and commonly mate with their sisters; however, in some species, such as Xylosandrus germanus and Euwallacea fornicatus, they have been observed to leave their natal nest and wander on the bark of the host plant, presumably to mate with females in other nests in the same host tree (Bright 1968; Peer and Taborsky 2004; Keller et al. 2011; Cooperband et al. 2016). Although additional outbred offspring will add to the fitness of the male, such outbred offspring can suffer outbreeding depression, as experimentally observed in Xylosandrus germanus, thereby reducing the fitness of the female (Peer and Taborsky 2005).
The ecology and distribution of Xyleborini species in Australia has been scarcely studied despite their diversity, which include 58 species from 20 genera (Pullen et al. 2014). Of these, two Cnestus species are commonly collected and widely distributed throughout eastern Australia, from Far North Queensland to Tasmania, Cnestus solidus (Eichhoff 1868) and Cnestus pseudosolidus (Schedl 1936). Froggatt (1926) stated that C. solidus also occurs in Western Australia, and C. pseudosolidus has been intercepted on the Noises Islands, New Zealand, in 1978 (Brockerhoff et al. 2003). Furthermore, there are questions regarding the taxonomy of C. pseudosolidus. In both its initial description by Schedl (1936) and subsequent taxonomic revision by Dole and Beaver (2008) it was highlighted that it intergrades morphologically with C. solidus, with suggestions that the two species names may be synonymous.
Similar to many other xyleborines (Gohli et al. 2017; Johnson et al. 2018) Cnestus species are host generalists: C. solidus has been recorded from Diploglottis australis (Sapindaceae), Eucalyptus spp., in particular smooth-barked eucalypts (Myrtaceae), Macadamia sp. (Proteceae), Malus sylvestris and Prunus armeniaca (Rosaceae), and C. pseudosolidus from Araucaria cunninghamii (Araucariaceae), M. sylvestris and Prunus spp. (Rosaceae), and Mangifera indica (Anacardiaceae) (Froggatt 1926; Wood and Bright 1992; Mitchell and Maddox 2010). In general, Cnestus species are associated with fungi of the genus Ambrosiella (Microascales: Ceratocystidaceae) with which they maintain a coarse co-phylogenetic symbiotic relationship (Skelton et al. 2019; Mayers et al. 2020). However, the fungal symbionts of the two Australian Cnestus species and their phylogenetic relationships with Ambrosiella are still unknown.
In our study, we explored the genetic diversity of Australian Cnestus using mitochondrial and nuclear genome markers and assessed whether they form two distinct sympatric species or their two names should be considered synonymous. Furthermore, we cultured and characterised the diversity of their associated ambrosia fungi. Lastly, because we inadvertently detected Wolbachia in some Cnestus individuals using universal mitochondrial cytochrome oxidase I (COI) barcoding primers, we explored the identity, distribution and prevalence of this endosymbiont in host beetle populations and tested whether this could be indicative of any potential contributions to the divergence of Australian Cnestus species.
Methods
Cnestus collections and Ambrosiella culturing
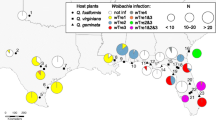
We analysed a total of 96 individuals of Cnestus from 21 localities throughout New South Wales (NSW), Queensland and Tasmania (Fig. 1; Table S1). At 14 sites beetles were collected in flight using cross-vane panel traps (three traps per site, each up to 5 km apart from one another), baited with 70% ethanol as lure, and a 50/50 blend of propylene glycol and pure ethanol in the collection container at the base. In NSW, traps were deployed between September to March throughout 2018–2020, and the propylene glycol/ethanol mix was replaced monthly (weekly at Cedar Ridge Road, Kurrajong, NSW). In Queensland, samples were collected in April 2019 and March 2020. Cnestus individuals, once collected, were stored in pure ethanol at −20 °C.
For the remaining seven sites, we used previously collected specimens. Specimens from Lamington National Park were sourced from the Australian National Insect Collection (ANIC); specimens from Brisbane, Barakula State Forest and Gladstone were sourced from the Queensland Department of Agriculture and Fisheries Insect Collection (QDAF), and specimens from Bracknell, Musselroe and Cape Portland were sourced from the Tasmanian Museum and Art Gallery Invertebrate Zoology Collection (TMAG). All these collection specimens had been stored in pure ethanol and kept at −20 °C, except for those from Tasmania, which had been point mounted. As per Schedl (1936) and Dole and Beaver (2008), and by comparisons with the paratype of C. pseudosolidus, individuals that were uniformly black and had an abrupt and flattened elytral declivity were identified as C. pseudosolidus while those that were red or not uniformly coloured and had a more sloping elytral declivity were identified as C. solidus.
Ambrosiella was cultured from five live Cnestus individuals collected in flight from Cedar Ridge Road (Fig. 1). For this, traps were checked twice daily throughout September and October in 2020 and any newly arrived beetles were removed from the surface of the alcohol mix when still alive. Beetles were split between the pronotum and mesonotum, and the anterior (head and prothorax, approximate location of mycetangia) and posterior (mesothorax, metathorax and abdomen) parts were placed on separate streptomycin malt agar plates (SMA: 1% malt extract, 1.5% agar, 100 ppm streptomycin sulfate added after autoclaving) and incubated at room temperature (Mayers et al. 2018). Fungal colonies were subcultured on SMA every 4-5 days until only one fungal morphotype grew per plate. These morphotypes were then subcultured on plates of malt yeast extract agar (MYEA: 2% malt extract, 0.2% yeast extract, 1.5% agar) to further assess growth and morphology.
DNA extraction and sequencing
All 96 individuals in this study were used for DNA extraction. Of these, 92 individuals underwent dissections to separate the head, elytra, mycetangia, thorax and abdomen. The head and elytra were stored as tissue vouchers (vouchered at ANIC, with accession numbers listed in Table S1). Cnestus beetle DNA was extracted from the thorax and abdomen using the Qiagen PowerSoil kit following a modified protocol; tissues were homogenised in 60 µL of C1 lysis buffer, then 20 µL of proteinase K (20 mg/ml) was added and homogenates were incubated for three hours at 56 °C with gentle shaking. For the mycetangia dissections, the prothorax and the mesothorax were separated with sterilised forceps, and the scutellum was removed from underneath the posterior margin of the pronotal disc. In Cnestus, the mesonotal mycetangia are connected to the scutellum (which also harbours mycetangial pits), so both the mycetangia and scutellum were used for symbiont fungal DNA extraction. Mycetangial fungal DNA was extracted using the Gentra Puregene kit following manufacturers protocols optimised for 5–10 mg of tissue. For the four dry specimens (two from Bracknell, one each from Musselroe and Cape Portland), a modified Qiagen DNEasy kit protocol was followed, where the whole specimen was non-destructively lysed in 180 µL of Buffer ATL, then then 20 µL of proteinase K (20 mg/ml) was added and samples were incubated overnight at 56 °C with gentle shaking. Lastly, DNA from the fungal cultures were extracted using the PowerSoil kit protocol described as above.
To assess the genetic diversity of C. solidus and C. pseudosolidus we amplified fragments of the mitochondrial cytochrome oxidase I (COI) (n = 52) and nuclear arginine kinase (ArgK) (n = 49) genes from the thorax and abdomen tissue DNA extracts. Initially the LCO1480 and HCO2198 (Folmer et al. 1994) universal COI barcoding primers were used; however, in initial tests seven out of 20 individuals preferentially amplified coxA gene sequences of Wolbachia, as previously observed in other insect COI barcoding studies (e.g., Mathenge et al. 2015). Therefore, we used the S1718/A2411 primer pair for amplification and sequencing of COI (Simon et al. 1994). Additionally for systematic placement of C. solidus and C. pseudosolidus, the carbamoyl-phosphate synthetase 2 (CAD) (n = 5) and elongation factor 1-alpha (EF1α) (n = 5) genes as well as the 2D-3D segment of the nuclear 28S rRNA (n = 7) gene were amplified. To assess the presence of Ambrosiella associated with individual specimens, we amplified the internal transcribed spacer ITS1 and ITS2 regions of the fungal nuclear rRNA gene cassette from all 92 mycetangia DNA extracts using two Ceratocystidaceae-specific primer pairs with overlapping regions for sequence assembly (Harrington et al. 2014). Additionally, the full length ITS region (ITS1F/ITS4) was amplified and sequenced from fungal culture morphotypes. For three of the culture morphotypes identified as Ambrosiella (through NCBI BLAST), the 28S rRNA (large subunit; LSU), 18S rRNA (small subunit; SSU) and translation elongation factor 1-alpha (tef1-α) genes were amplified for phylogenetic analyses. Lastly, we screened 92 Cnestus individuals (excluding the dried Tasmanian specimens) for Wolbachia using primers for the Wolbachia surface protein (wsp) gene from the thorax and abdomen tissue DNA extracts. For a subset of wsp-positive individuals, the multilocus sequence typing (MLST) genes gatB (n = 7), coxA (n = 6), hcpA (n = 7), ftsZ (n = 3) and fbpA (n = 8) (Baldo et al. 2006) were sequenced for sequence type identification and phylogenetic analysis of the Wolbachia strain. All PCR reactions were performed in 15 µL containing 0.3 µL (20 nM) of each primer (Table S2), 3 µL of 5x MyTaq Red Buffer, 0.3 µL of MyTaq DNA polymerase (Bioline), 2 µL of template DNA (1 µL was used for COI) and PCR water (Qiagen). All successfully amplified products were Sanger sequenced in both directions.
Sequence chromatograms were visually assessed for quality. Sequences of protein coding genes were assessed for open reading frames to exclude potential pseudogenes (e.g., nuclear mitochondrial gene copies or NUMTs) in Geneious v10.2.6 (Kearse et al. 2012). For each locus and specimen, forward and reverse complementary sequences were assembled using the de-novo assembly function in Geneious, followed by the removal of primer sequences. Additional sequences were downloaded from GenBank (NCBI), including LSU, SSU and tef1-α of representative fungal species of the Ceratocystidaceae (Table S3), and COI, ArgK, CAD and EF1α of Cnestus and other scolytine species (Table S4). For comparisons of the fungal ITS an aligned reference dataset (Mayers et al. 2020) was downloaded (http://purl.org/phylo/treebase/phylows/study/TB2:S22560). Lastly, wsp, gatB, coxA, hcpA, ftsZ and fbpA sequences of Wolbachia strains characterised in previous scolytine studies (Kawasaki et al. 2010, 2016), and sequences for the Wolbachia strains wHa, wNo and wRi were downloaded from PubMLST (Tables S5, 6).
Sequence alignment and phylogenetic analysis
All sequences were aligned with MUSCLE using default parameters and then edited manually. To assess the genetic diversity of Australian Cnestus species, we analysed COI (555 bp) and ArgK (750 bp) independently and as a concatenated dataset. For systematic placement of our samples within the Cnestus genus, we analysed a combined dataset of the COI, ArgK, CAD (462 bp), EF1α (813 bp) and 28S rRNA (741 bp) genes. To assess the phylogenetic placement of Ambrosiella sequences obtained from mycetangia DNA extracts and fungal cultures of Cnestus, we aligned their ITS sequences (555 bp) with the previously aligned ITS dataset from Mayers et al. (2020) to preserve diagnostic indel regions. We also used LSU (582 bp), SSU (1692 bp) and tef1-α (1250 bp) to explore the systematic identity of the Ambrosiella cultures obtained from Cedar Ridge Road. All Wolbachia wsp and MLST gene sequences from Cnestus beetles collected throughout the range were identical, so a single representative sequence for each marker was used for phylogenetic analyses of Wolbachia. We then aligned the wsp sequence (603 bp) of this strain with those downloaded from PubMLST, and indels were retained. We also aligned the MLST sequences of this strain with those downloaded from PubMLST, generating a final matrix consisting of gatB (369 bp), coxA (402 bp), hcpA (444 bp), ftsZ (435 bp) and fbpA (429 bp).
Bayesian and maximum likelihood (ML) analyses were performed on all alignments (except for the multilocus Ambrosiella dataset for which only Bayesian analyses were conducted), with 1st and 2nd codon positions of protein coding sequences partitioned differently from 3rd codon positions. Bayesian analyses were performed with BEAST2 v2.6.5 (Bouckaert et al. 2014), using an uncorrelated relaxed log-normal clock and a birth-death tree prior, and nucleotide substitution models of partitioned loci were inferred using the bModelTest package (Bouckaert and Drummond 2017) with up to three independent MCMC runs for 100 million generations sampled every 10,000 generations. Independent runs were combined using LogCombiner v2.6.5 (Bouckaert et al. 2014). Convergence to stationarity and effective sample size values of all model parameters were assessed with TRACER v1.7.1 (Rambaut et al. 2018). A maximum clade credibility tree with a 10% burn-in for all analysed datasets was inferred using TreeAnnotator v2.6.0 (Bouckaert et al. 2014). ML analyses were conducted in IQ-Tree 1.6.12 (Minh et al. 2020) with concatenated alignments. Substitution models were estimated with ModelFinder Plus and bootstrap support was calculated with UFBoot (Hoang et al. 2018) with 1000 replicates.
For comparisons with a previous phylogenetic study of Ambrosiella (Mayers et al. 2020), maximum parsimony (MP) analysis was used to explore the phylogenetic relationships of ITS sequences obtained in our study with those of this previous study using the previously published analytical pipeline (Mayers et al. 2020). Accordingly, analyses were performed in PAUP v4.0a168 (Swofford 2003) as this software allows gaps to be treated as a fifth character, preserving diagnostic indels present in ITS (Mayers et al. 2020). A heuristic search was used, starting trees were sampled via stepwise addition and bootstrap support was calculated with 1000 replicates. This alignment contained 555 characters, of which 385 were constant, 34 were variable and parsimony uninformative, and 136 were parsimony-informative. All trees were visualised with FigTree v1.4.4 (Rambaut 2014) and then edited in Inkscape 1.0.
Following phylogenetic analyses, we tested whether Cnestus individuals of the two lineages found in our study (lineage 1 n = 40, lineage 2 n = 12) constituted distinct species with distance-based and tree-based species delimitation methods. We first calculated between group average nucleotide p-distances for COI and ArgK between Cnestus lineages using MEGA 11 (Tamura et al. 2021). For the distance-based analysis, the online implementation of Automatic Barcode Gap Discovery (ABGD) (Puillandre et al. 2012) with the Kimura 2-P model was used to assess barcoding gap distances in both the COI and ArgK alignments. We then performed a Bayesian implementation of the Poisson tree processes (bPTP) (Zhang et al. 2013) on the ultrametric trees of COI and ArgK produced with BEAST2 (Figures S1 and S2, respectively). The bPTP analyses run for 100 million generations and sampled every 10,000 generations. The first 10% of trees were discarded as burn-in for both analyses. Following this, intraspecific mitochondrial haplotype diversity of Cnestus populations were inferred with a minimum spanning network analysis implemented in PopART (Leigh and Bryant 2015). In order to further analyse patterns of molecular diversity within and between lineages for COI and ArgK we applied an AMOVA using the pegas package (Paradis 2010) in R. Three non-synonymous nucleotide substitutions were observed between the two lineages in COI while all nucleotide polymorphisms in the ArgK alignment were synonymous. Therefore, we further explored whether COI violated assumptions of neutral evolution with Tajima’s neutrality test in Mega XI (Tamura et al. 2021).
Results
Genetic diversity of Australian Cnestus
We sequenced a fragment of the mitochondrial COI gene from 52, and a fragment of the nuclear ArgK gene from 49 Australian Cnestus females (Table S4). Alignments of these genes highlight that two main sequence variants are present throughout populations of Australian Cnestus. For both genes we did not find any ambiguous sites in sequence chromatograms that could have been indicative of NUMTs or heterozygosity. Furthermore, all COI sequences coded for two similar amino acid sequences (however with three non-synonymous substitutions) which were similar to closely related species further indicating that no pseudogenes had been sequenced. Similarly for the ArgK nine non-synonymous substitutions were present in the alignment.
Bayesian and ML phylogenetic analyses of the concatenated COI and ArgK gene sequence data sets together with sequences of other xyleborine species (Fig. 2), and of these two genes individually (Figures S1 and S2) produced identical topologies for Australian Cnestus indicating no mitochondrial–nuclear discordance. Two distinct sympatric lineages of Cnestus were identified and were strongly supported in all analyses (Fig. 2, Figures S1 and S2). We found both lineages distributed from southern NSW to central Queensland (Fig. 1 and Table 1), with a higher abundance of individuals of lineage 1 (n = 40) throughout the range compared to lineage 2 (n = 12). Furthermore, three specimens of lineage 1 were also found in Far North Queensland, and one in Tasmania. At several sites, individuals of both lineages were collected from the same trap and at the same collection times (Table S1). Between these two lineages we found an average p-distance of 0.00872 for ArgK and a substantially higher average p-distance of 0.0912 for COI. According to COI sequence data, both species delimitation methods identified the two lineages as distinct species; however, based on ArgK both lineages were not distinct species.
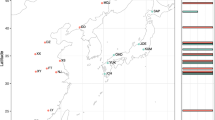
a Minimum spanning network analysis of the cytochrome oxidase I gene fragments of Cnestus individuals collected throughout eastern Australia. Dashes on network branches indicate individual nucleotide substitutions between haplotypes and the size of the circles is proportional to the number of samples within each haplotype. Lineage 1 haplotypes are on top and lineage 2 haplotypes on the bottom of the network. Relative proportions of Wolbachia infections identified using wsp are given next to the circles. b Phylogenetic tree of the concatenated cytochrome oxidase I (COI) and arginine kinase (ArgK) gene fragments of Cnestus individuals collected across eastern Australia. Samples identified as C. solidus are labelled Cso, while those identified as C. pseudosolidus are labelled Cps. Wolbachia-positive individuals are represented in blue. Support is given at the nodes with black dots indicating >0.95 posterior probability and >95% bootstrap. Lineage identity of Cnestus solidus is provided on the branches. Scale bar indicates 0.02 substitutions per nucleotide position. To the right of the tips are the results of ABGD and bPTP species delimitation analyses of the COI and ArgK genes where gaps in the bars represent observed species delimitation splits for COI but not ArgK.
Mitochondrial haplotype network analysis identified two primary haplotypes in each of the two lineages, with a higher haplotype diversity in lineage 1 than lineage 2 (Fig. 2A). Additionally, individuals with unique COI haplotypes (singletons) that were one base pair different to more prevalent haplotypes were identified. It is unclear whether these are attributable to small population sample sizes (e.g., Girringun n = 1) or whether they represent true mitochondrial diversity within sample sites (e.g., Cedar Ridge n = 6 and Bulburin n = 2). No significant differences of molecular diversity were identified within lineages for both COI (σ = −9.4836e-05, p = 0.901) and ArgK (σ = −1.7781e-06, p = 0.9901). Additionally, COI was found to violate assumptions of neutral evolution from Tajima’s Neutrality Test, D = 1.296194 because of the three non-synonymous substitutions. Of these three COI non-synonymous substitutions between the two Cnestus solidus lineages, only lineage 2 possessed a synapomorphic amino acid with outgroup Cnestus species. In addition to the COI and ArgK analyses of 52 and 49 individuals, respectively, there was an average pairwise similarity between the two lineages for the three nuclear marker genes that were chosen from the overall sample set: 99.6% for CAD (n = 20), 99.4% for EF1α (n = 4) and 100% for the 28S rRNA gene (n = 6).
Furthermore, we found no support for Schedl’s (1936) description of C. pseudosolidus as a distinct species because morphological species identity did not correlate with genetic lineage identity (Fig. 2B), and, therefore, C. pseudosolidus should be considered a synonym of the senior name C. solidus. We also inferred the phylogenetic placement of the two C. solidus lineages using COI, ArgK, CAD, EF1a and 28S rRNA genes of five individuals relative to five other species within the Cnestus genus (Figure S3). All Cnestus species formed a strongly supported monophyletic group. Both Bayesian and ML phylogenies identified two distinct and strongly supported lineages of C. solidus.
Isolation of Ambrosiella and phylogenetic identification
We cultured three fungal morphospecies from five live C. solidus individuals collected at Cedar Ridge. These were identified as an Ambrosiella sp., a Penicillium sp. and a Pichia sp. by ITS sequencing and comparison with ITS reference sequences using BLAST (NCBI). Ambrosiella was cultured from all five plates with the posterior part of beetle specimens (mesothorax, metathorax and abdomen), and from three of the five plates with the anterior part of beetle specimens (head and prothorax), all of which had identical ITS sequences. Colonies on MYEA grew to 30 mm in diameter after 14 days at 20 °C and were light green in the centre and darker green toward the edges. Clear hyphae surrounded the fungal mass, and clumped aerial tufts also grew toward the edges of the fungal mass. Cultures smelled sweet and similar to overripe bananas. Amplification and sequence analysis of ITS revealed that all cultures had the same ITS sequence. Furthermore, we obtained from the cultured Ambrosiella isolates the LSU, SSU and tef1-α sequences which were all identical.
Of the 91 dissected mycetangia DNA extracts, 81 successfully amplified ITS using the Ceratocystidaceae primers. A subset of 31 ITS PCR amplicons that derived from individuals throughout the range of C. solidus were sequenced. All sequences were identified as Ambrosiella using BLAST (NCBI), and consisted of two distinct ITS sequence variants, representing two different isolates. The most prolific Ambrosiella isolate, herein referred to as Amb1, had an ITS sequence identical to that of the cultures (Fig. 3), and was found in all populations of both lineages of C. solidus except for one population. All individuals collected in South East Forests (SEF), all of lineage 1, were associated with a second Ambrosiella isolate with a different ITS sequence, herein referred to as Amb2 (Table 1). An attempt was made to collect beetles from South East Forest to culture Amb2, however none were found as most of the habitat was burned during the catastrophic bushfires of 2019–2020.
Sequences generated from this study are in bold; fungal cultures from Cnestus solidus are listed with DAR collection accession codes and original isolation numbers, and sequences directly amplified from beetle mycangia are labelled with an asterisk (*). Bootstrap support is provided at the nodes with black dots indicating >95% support while the grey dot indicates >80% support. Scale bar indicates number of bp changes.
According to the phylogenetic analysis of ITS, Amb1 was placed as a weakly supported sister taxon to A. aff. grossmanniae in the A. xylebori clade (Fig. 3). Amb2 formed a strongly supported group with fungi in the A. beaveri clade but was highly diverged from all included species. In the Bayesian multilocus phylogeny, the cultured Ambrosiella sp. 1 (Amb1) was placed as a new sister lineage to A. catenulata, but with little support (Figure S4). Attempts were made to also obtain LSU, SSU and tef1-α for Amb2 from mycetangia DNA extracts of South East Forest individuals, however, these PCR reactions failed to amplify.
Wolbachia infection prevalence and strain identification
Of the 93 tested C. solidus individuals, 58 were PCR positive for the Wolbachia gene wsp (Table 1). All Wolbachia-positive individuals belonged to lineage 1 (36 Wolbachia positive out of 40 individuals), whereas all lineage 2 individuals (12) tested negative. We sequenced wsp from 17 individuals and Wolbachia MLST genes from five individuals throughout the range of C. solidus, and all sequences for each of the genes were identical. Additionally, sequence chromatograms of wsp and all MLST genes had no ambiguous sites, suggesting individuals are infected by the same strain belonging to the Wolbachia supergroup A, herein referred to as wCsolid. Bayesian and ML analyses of all sequence sets identified wHa as the closest relative (Fig. 4a and b), with just one divergent nucleotide between the wsp sequences of the two strains, whereas all MLST loci were identical between wCsolid and wHa (ST-19).
Phylogenies are based on a wsp gene sequences (603 bp) and b Wolbachia MLST gene sequences (2079 bp). For all analyses support is provided at the nodes; black dots indicate both >0.95 posterior probability and >95% bootstrap support, dark grey dots indicate only >0.95 posterior probability, and light grey dots indicate only >95% bootstrap support. The Wolbachia strain infecting Cnestus solidus lineage 1 (wCsolid) is in bold. Wolbachia supergroup B sequences (wNo and the strains in the bottom clade in the MLST gene tree) were used as outgroups. Scale bars indicate 0.02 substitutions per nucleotide position.
Discussion
Lineage divergence in Cnestus solidus
We discovered two genetically distinct yet morphologically indistinguishable lineages of C. solidus that occur in sympatry throughout eastern Australia. Both lineages are monophyletic and apparently share the same ecological niche (Froggatt 1926; Wood and Bright 1992; Mitchell and Maddox 2010) which could allow occasional cross-fertilization between family units. Yet, we did not detect any nuclear–mitochondrial discordance between the two lineages. Therefore, these two lineages could perhaps be considered incipient species or even distinct cryptic species which may be unable to hybridize. Furthermore, we detected substantially greater variation in the mitochondrial gene COI (9%) than in nuclear markers such as ArgK (0.9%), including no substitutions in the 28S rRNA gene. Comprehensive analyses conducted across most of the genera of Xyleborini by Cognato et al. (2020) identified that a 10–12% sequence nucleotide divergence for the mitochondrial COI gene and a 2–3% sequence nucleotide divergence for the nuclear CAD gene can confidently delimit species boundaries. In contrast to the mitochondrial COI nucleotide sequence divergence, the observed nuclear gene sequence divergence between the two Cnestus lineages and the results of species delimitation analyses of ArgK indicate that they do not constitute distinct species. Furthermore, these patterns are in line with intraspecific divergence within other xyleborine species (Jordal and Kambestad 2014; Gohli et al. 2016; Cognato et al. 2020; Jordal and Tischer 2020; Smith and Cognato 2022). For mitochondrial markers such as COI, barcoding sequence divergence of 2–3% (Ashfaq et al. 2016; Hebert et al. 2003) may indicate species boundaries in some insect groups, however, this is not the case for most beetles, and particularly not for inbreeding xyleborine ambrosia beetles (Cognato 2006; Cognato et al. 2020; Smith and Cognato 2022). As discussed by Cognato et al. (2020), haplodiploid female sex ratio biases and inbreeding can greatly amplify the rate at which COI nucleotide substitutions occur. While species delimitation analyses of COI suggested that the two lineages of Cnestus could be separate species, we also found that the COI sequence divergence violates assumptions of neutral evolution. This could be due to a selective sweep or population bottleneck in association with Wolbachia infection in lineage 1. Wolbachia and other maternally inherited factors that are coinherited with mitochondria, influence host fitness and hence prevalence in hosts can cause impact such divergence patterns due to their maternal coinheritance (Cariou et al. 2017; Arif et al. 2021). Therefore, we do not consider these two lineages as distinct species (Templeton 1989; De Queiroz 2007; Freudenstein et al. 2016). Our results further indicate that C. pseudosolidus is synonymous with C. solidus which is the senior species name. Our finding provides genetic support for a previous study which suggested that the two species appear to intergrade morphologically (Dole and Beaver 2008). We present their synonymy after the concluding remarks.
Microbial symbionts associated with Australian Cnestus
Few studies have detected more than one species, and often just one strain of ambrosia fungi associated with individual ambrosia beetle species. The large, well-developed mycetangia of Xyleborini beetles have been proposed to maintain species-specific symbioses with individual Ambrosiella species (Harrington et al. 2014; Mayers et al. 2015). For example, phylogeographic analyses of the Ambrosiella associated with X. germanus and Anisandrus dispar found no genetic diversity in their fungal associates in Europe, supporting the idea of strict clonal transmission in their host beetle species (van de Peppel et al. 2018). Recently, however, multiple closely related Ambrosiella species were found in various populations of X. germanus throughout its native range in Japan (Ito and Kajimura 2017). In contrast, we detected two phylogenetically distinct Ambrosiella isolates that were placed in two separate Ambrosiella clades in association with C. solidus throughout the range of this beetle species (albeit one of these two Ambrosiella isolates was only found in one of the 19 analysed sites). This suggests that symbioses between ambrosia beetle species and fungi may be more flexible than previously anticipated.
The variation that we found in the Ambrosiella associated with an ambrosia beetle species is, to our knowledge, the first detection of a clade level symbiont switch in a xyleborine ambrosia beetle species with respect to Ambrosiella fungi. This suggests that tight specificity between ambrosia partners may not always be the rule (Harrington et al. 2014; Mayers et al. 2015). Previous co-phylogenetic analyses by Skelton et al. (2019) have identified a rather coarse relationship between Cnestus and Ambrosiella fungi in the beaveri clade. Despite a co-phylogenetic relationship, it was nevertheless demonstrated experimentally that ambrosia beetles can survive on ambrosia fungi of the alternate (xylebori) clade and acquire their spores in their mycetangia, even though the probability for spore transfer was low (Skelton et al. 2019). Our data identified that C. solidus is predominantly associated with a different (xylebori) clade of Ambrosiella, suggesting that the symbiosis between ambrosia beetles and fungi can be flexible. It is interesting that we have only identified one population in association with an Ambrosiella species of the beaveri clade which is usually the main associate with Cnestus (Skelton et al. 2019). Future studies should therefore explore in more detail the population genetics and evolutionary history of ambrosia fungi associated with the Australasian species of Cnestus. This could yield interesting insights into the observed asymmetric frequency of Ambrosiella species associations and reveal any potential selective sweeps that may have generated the current affiliations. For this, however, the culturing and sequencing of other marker genes of Amb2 are needed as currently our results are based on the phylogenetic analysis of ITS, a multicopy marker in the fungal genome (Yang et al. 2018). The level of divergence observed in this study could be a result of ITS marker divergence and may not reflect the true evolutionary divergence of the isolate. Therefore, further sequence data for the phylogenetic placement of Amb2 are needed.
We also discovered that lineage 1 of C. solidus was associated with a new Wolbachia strain, wCsolid. According to wsp gene and MLST analyses, this strain is closely related to wHa, first described from a Hawaiian population of Drosophila simulans (Poinsot and Mercot 1997, Ellegaard et al. 2013), and unrelated to Wolbachia strains infecting other scolytine beetles (Kawasaki et al. 2016). wCsolid was highly prevalent in lineage 1 of C. solidus, infecting 34 out of 40 beetles (85%), but entirely absent from lineage 2. Wolbachia has been found to infect a wide range of scolytines (Vega et al. 2002; Zchori-Fein et al. 2006; Arthofer et al. 2009; Schebeck et al. 2018; Bykov et al. 2020), and, specifically, haplodiploid ambrosia beetles (Kawasaki et al. 2010, 2016). Kawasaki et al. (2016) suggested that regular inbreeding and haplodiploidy may promote more efficient maternal transmission of this endosymbiont, and this could also contribute to the observed high prevalence of Wolbachia in lineage 1 of C. solidus.
Potential involvement of symbionts in Cnestus lineage divergence
Allopatric divergence coupled with predominant inbreeding are important factors in scolytine species diversification, while host-tree switching has been found to have little impact on diversification rates (Gohli et al. 2017). According to available information and findings of our study, there appears to be no ecological niche differentiation between the two sympatric lineages of C. solidus. First, it has been noted that C. solidus is a tree generalist (Froggatt 1926; Wood and Bright 1992; Mitchell and Maddox 2010), however this does not preclude the potential existence of two lineages with some specialisation because we were not able to test host plant associations of our genotyped specimens. Secondly, and more importantly, both lineages were associated with the same Ambrosiella fungus. These symbionts may allow an ambrosia beetle to be flexible with respect to host tree selection (Kirkendall et al. 2015; Raffa et al. 2015; Gohli et al. 2017). Additionally, as both lineages were collected in the same traps at the same time at several sites, there appears to be no phenological differentiation in adult emergence and dispersal. Therefore, given that the two C. solidus lineages have a likely broad host tree selection, a similar distribution and phenology, as well as the same Ambrosiella symbiont, we do not expect niche specialisation or displacement to have generated sympatric divergence between lineages.
Given that Wolbachia occurred only in lineage 1, and at high prevalence, this suggests that it may affect the host’s reproduction and/or fitness, and as a consequence possibly also the divergence of the two lineages. Cytoplasmic incompatibility is one of the most frequently reported host reproductive manipulations by Wolbachia (Werren et al. 2008; Brucker and Bordenstein 2012; Shropshire and Bordenstein 2016; Kaur et al. 2021). In the case of CI, should an infected male of lineage 1 attempt to outbreed with an uninfected female of lineage 2, this would result in embryonic mortality. However, infected lineage 1 females could potentially still successfully mate with lineage 2 males, leading to a disruption of the current linkage of nuclear genes with mitochondrial genes and Wolbachia infections, yet we did not observe this. Therefore, it is unlikely that CI is involved as a mechanism of reproductive isolation between the two lineages unless there is another barrier which prevents successful pairings of lineage 1 females with lineage 2 males. It is also unlikely that Wolbachia causes thelytokous parthenogenesis in the infected lineage as we would expect all individuals of the infected lineage to carry Wolbachia, which we have not observed. Furthermore, Wolbachia-induced thelytoky has so far not yet been demonstrated in any beetle species. We also do not expect that Wolbachia-induced male killing or feminisation occurs in C. solidus. This is because Wolbachia-induced male killing could be disastrous for inbreeding lineages; furthermore, all known and rare examples of Wolbachia-induced feminisation are restricted to terrestrial isopods as well as two butterfly and one hemipteran species (Kageyama et al. 2012).
In the absence of any reproductive manipulation, Wolbachia could still provide a fitness benefit to lineage 1, and consequently occur at high prevalence in the more commonly found lineage and influence mitochondrial diversity patterns of this species. In some host species Wolbachia can provide fitness benefits such as an increase in fecundity, nutrient provisioning and pathogen defence (Zug and Hammerstein 2015). In a haplodiploid scolytine, the date-seed beetle Coccotrypes dactyliperdia, Wolbachia was found associated with increased dispersal of infected beetles (Tremmel et al. 2020). Future research should further investigate whether Wolbachia manipulates reproduction and/or improves fitness in C. solidus.
Furthermore, we have also identified haplotype diversity within the infected lineage of C. solidus, suggesting that no recent mtDNA sweeps associated with Wolbachia had occurred and Wolbachia had been present for long enough for the lineage to acquire new haplotype diversity. Beyond this we found patterns for non-neutral molecular evolution in COI that correlates with Wolbachia infections in lineage 1. Interestingly, while the infection of Wolbachia in lineage 1 did not impact population genetic diversity, we found that the sequence divergence between lineage 2 and outgroup Cnestus taxa was smaller than that between lineage 1 individuals and outgroup Cnestus taxa. This could indicate independent historical selective sweeps or, alternatively, population bottle necks in both lineages.
In the absence of factors that indicate sympatric speciation processes for the two C. solidus lineages, it is likely that they diverged in allopatry followed by secondary contact leading to their contemporary sympatric distribution. It has previously been discussed that sib mating promotes dispersal of propagules into novel environments and subsequent allopatric divergence of lineages (Eliassen and Jordal 2021; Jordal et al. 2000, 2001, 2006). Range expansions and contractions of the Australian wet forests occurred during the Pliocene and this resulted in allopatric separations of many species (Gallagher et al. 2003; Martin 2006; Byrne et al. 2011). It is likely that beetles of lineage 1 became infected with Wolbachia when populations were allopatrically separated. Then, after secondary contact, the reproductive isolation of the two lineages has been maintained through sib mating and, possibly, Wolbachia-induced host effects.
Conclusions
Our study represents a comprehensive study of the evolutionary ecology of an Australian scolytine ambrosia beetle species and its associated microbial symbionts. Molecular phylogenetics of the Cnestus specimens sampled throughout their range in Australia supported the findings by Dole and Beaver (2008) that C. solidus and C. pseudosolidus intergrade morphologically and, therefore, constitute only one species. Interestingly, however, we identified two sympatric cryptic lineages with no indication of hybridisation between the two. In the more prevalent beetle lineage 1, we also discovered a new strain of Wolbachia that may be associated with the different relative abundance and divergence between the two lineages. Lastly, we also identified two divergent isolates of Ambrosiella associated with C. solidus, one of which was prevalent throughout their geographical range, and divergent from the Ambrosiella symbionts of other Cnestus species across the world. These two Ambrosiella isolates associated with C. solidus highlight a potentially relaxed selectivity of mycetangia towards fungal symbionts. Further investigation of the mitochondrial and nuclear population genetics of C. solidus should explore the historical demography of both lineages and look for signatures of effective population size expansion or contraction associated with selective sweeps either caused by, or correlated with, Wolbachia.
Taxonomy
Cnestus solidus (Eichhoff 1868), comb. nov.
Xyleborus solidus Eichhoff 1868: 151
Xylosandrus solidus (Eichhoff 1868): combination by Wood and Bright 1992: 800
Xyleborus pseudosolidus Schedl 1936: 530, syn. nov.
Xylosandrus pseudosolidus (Schedl): combination by Wood and Bright, 1992
Cnestus pseudosolidus (Schedl): combination by Dole and Beaver 2008
Our phylogenetic analysis of Australian Cnestus species did not support Schedl’s (1936) description of C. pseudosolidus but provided genetic evidence for the findings by Dole and Beaver (2008) that C. solidus and C. pseudosolidus intergrade morphologically. Furthermore, we detected both C. solidus and C. pseudosolidus morphotypes in both genetic lineages. Given the morphological overlap between C. solidus and C. pseudosolidus (Dole and Beaver 2008; Schedl 1936) and the lack of genetic divergence between these two morphospecies we synonymise the species name C. pseudosolidus (Schedl 1936) with the senior C. solidus (Eichhoff 1868).
Data availability
Isolates of Ambrosiella sp. 1 were deposited with the Plant Pathology Herbarium, Department of Primary Industries NSW (accession codes DAR85400-DAR85403). All Cnestus voucher specimens, stored in ethanol and pinned, have been deposited in the ANIC, CSIRO, under accession numbers 25-086124 - 25-085827. All sequence data generated in this study are available from NCBI GenBank with accession codes provided in the supplementary information.
References
Andersen HF, Jordal BH, Kambestad M, Kirkendall LR (2012) Improbable but true: the invasive inbreeding ambrosia beetle Xylosandrus morigerus has generalist genotypes. Ecol Evol 2:247–257
Arif S, Gerth M, Hone‐Millard WG, Nunes MDS, Dapporto L, Shreeve TG (2021) Evidence for multiple colonisations and Wolbachia infections shaping the genetic structure of the widespread butterfly Polyommatus icarus in the British Isles. Mol Ecol 30:5196–5213
Arthofer W, Riegler M, Avtzis DN, Stauffer C (2009) Evidence for low-titre infections in insect symbiosis: Wolbachia in the bark beetle Pityogenes chalcographus (Coleoptera, Scolytinae). Environ Microbiol 11:1923–1933
Arthofer W, Avtzis DN, Riegler M, Stauffer C (2010) Mitochondrial phylogenies in the light of pseudogenes and Wolbachia: re-assessment of a bark beetle dataset. ZooKeys 56:269–280
Ashfaq M, Hebert P, Naaum A (2016) DNA barcodes for bio-surveillance: Regulated and economically important arthropod plant pests. Genome 59:933–954
Avtzis DN, Arthofer W, Stauffer C (2008) Sympatric occurrence of diverged mtDNA lineages of Pityogenes chalcographus (Coleoptera, Scolytinae) in Europe. Biol J Linn Soc 94:331–340
Baldo L, Hotopp JCD, Jolley KA, Bordenstein SR, Biber SA, Rhitoban R, et al. (2006) Multilocus sequence typing system for the endosymbiont Wolbachia pipientis. AEM 72(11):7098–7110
Bouckaert RR, Drummond AJ (2017) bModelTest: Bayesian phylogenetic site model averaging and model comparison. BMC Evol Biol 17:42
Bouckaert R, Heled J, Kühnert D, Vaughan T, Wu C-H, Xie D et al. (2014) BEAST 2: A Software Platform for Bayesian Evolutionary Analysis. In: Prlic A, (ed). PLoS Comput Biol 10:e1003537
Bright DE (1968) Review of the tribe xyleborini in America north of Mexico (Coleoptera: Scolytidae). Can Entomol 100:1288–1323
Brockerhoff EG, Knížek M, Bain J (2003) Checklist of indigenous and adventive bark and ambrosia beetles (Curculionidae: Scolytinae and Platypodinae) of New Zealand and interceptions of exotic species (1952–2000). N Zeal. Entomol 26:29–44
Brucker RM, Bordenstein SR (2012) Speciation by symbiosis. Trends Ecol Evol 27:443–451
Bruzzese DJ, Schuler H, Wolfe TM, Glover MM, Mastroni JV, Doellman MM et al. (2021) Testing the potential contribution of Wolbachia to speciation when cytoplasmic incompatibility becomes associated with host‐related reproductive isolation. Mol Ecol 31:2935–2950
Bykov R, Kerchev I, Demenkova M, Ryabinin A, Ilinsky Y (2020) Sex-specific Wolbachia infection patterns in populations of Polygraphus proximus Blandford (Coleoptera; curculionidae: Scolytinae). Insects 11:547
Byrne M, Steane DA, Joseph L, Yeates DK, Jordan GJ, Crayn D et al. (2011) Decline of a biome: Evolution, contraction, fragmentation, extinction and invasion of the Australian mesic zone biota. J Biogeogr 38:1635–1656
Cariou M, Duret L, Charlat S (2017) The global impact of Wolbachia on mitochondrial diversity and evolution. J Evol Biol 30:2204–2210
Cognato AI (2006) Standard percent DNA sequence difference for insects does not predict species boundaries. J Econ Entomol 99:1037–1045
Cognato AI, Sari G, Smith SM, Beaver RA, Li Y, Hulcr J et al. (2020) The essential role of taxonomic expertise in the creation of DNA databases for the identification and delimitation of Southeast Asian ambrosia beetle species (Curculionidae: Scolytinae: Xyleborini). Front Ecol Evolution 8:27
Cooperband MF, Stouthamer R, Carrillo D, Eskalen A, Thibault T, Cossé AA et al. (2016) Biology of two members of the Euwallacea fornicatus species complex (Coleoptera: Curculionidae: Scolytinae), recently invasive in the U.S.A., reared on an ambrosia beetle artificial diet. Agric Entomol 18:223–237
Cruz MA, Magalhães S, Sucena É, Zélé F (2021) Wolbachia and host intrinsic reproductive barriers contribute additively to postmating isolation in spider mites. Evolution 75:2085–2101
Detcharoen M, Arthofer W, Schlick-Steiner BC, Steiner FM (2019) Wolbachia megadiversity: 99% of these microorganismic manipulators unknown. FEMS Microbiol Ecol 95:1–5
De Queiroz K (2007) Species concepts and species delimitation. Syst Biol 56:879–886
Dole SA, Beaver RA (2008) A review of the Australian species of Xylosandrus Reitter (Coleoptera: Curculionidae: Scolytinae). Coleopt Bull 62:481–492
Eichhoff WJ (1868) Neue amerikanische Borkenkäfer-Gattungen und Arten. Berl Entomologische Z 11(3-4):145–152
Eliassen JM, Jordal BH (2021) Integrated taxonomic revision of Afrotropical Xyleborinus (Curculionidae: Scolytinae) reveals high diversity after recent colonization of Madagascar. Insect Syst Divers 5:4
Ellegaard KM, Klasson L, Näslund K, Bourtzis K, Andersson SGE (2013) Comparative genomics of Wolbachia and the bacterial species concept. PLoS Genet 9(4):e1003381
Folmer O, Black M, Hoeh W, Lutz R, Vrijenhoek R (1994) DNA primers for amplification of mitochondrial cytochrome c oxidase subunit I from diverse metazoan invertebrates. Mol Mar Biol Biotechnol 3:294–299
Francke-Grosmann H (1956) Hautdrüsen als Träger der Pilzsymbiose bei Ambrosiakäfern. Z für Morphol und Ökologie der Tiere 45:275–308
Freudenstein JV, Broe MB, Folk RA, Sinn BT (2016) Biodiversity and the species concept – lineages are not enough. Syst Biol 66:644–656
Froggatt WW (1926) Forest insects. Aust J 9:256–260
Gallagher SJ, Greenwood DR, Taylor D, Smith AJ, Wallace MW, Holdgate GR (2003) The Pliocene climatic and environmental evolution of southeastern Australia: Evidence from the marine and terrestrial realm. Palaeogeogr Palaeoclimatol Palaeoecol 193:349–382
Gohli J, Selvarajah T, Kirkendall LR, Jordal BH (2016) Globally distributed Xyleborus species reveal recurrent intercontinental dispersal in a landscape of ancient worldwide distributions. BMC Evol Biol 16:37
Gohli J, Kirkendall LR, Smith SM, Cognato AI, Hulcr J, Jordal BH (2017) Biological factors contributing to bark and ambrosia beetle species diversification. Evolution 71:1258–1272
Harrington TC, McNew D, Mayers C, Fraedrich SW, Reed SE (2014) Ambrosiella roeperi sp. nov. is the mycangial symbiont of the granulate ambrosia beetle, Xylosandrus crassiusculus. Mycologia 106:835–845
Hebert P, Ratnasingham S, deWaard J (2003) Barcoding animal life: cytochrome c oxidase subunit 1 divergences among closely related species. Proc R Soc B: Biol Sci 270:96–99
Hoang DT, Chernomor O, von Haeseler A, Minh BQ, Vinh LS (2018) UFBoot2: Improving the ultrafast bootstrap pproximation. Mol Biol Evol 35:518–522
Hulcr J, Stelinski LL (2017) The ambrosia symbiosis: from evolutionary ecology to practical management. Annu Rev Entomol 62:285–303
Hurst GDDD, Jiggins FM (2005) Problems with mitochondrial DNA as a marker in population, phylogeographic and phylogenetic studies: The effects of inherited symbionts. Proc R Soc B Biol Sci 272:1525–1534
Ito M, Kajimura H (2017) Landscape-scale genetic differentiation of a mycangial fungus associated with the ambrosia beetle, Xylosandrus germanus (Blandford) (Curculionidae:Scolytinae) in Japan. Ecol Evol 7:9203–9221
Johnson AJ, McKenna DD, Jordal BH, Cognato AI, Smith SM, Lemmon AR et al. (2018) Phylogenomics clarifies repeated evolutionary origins of inbreeding and fungus farming in bark beetles (Curculionidae, Scolytinae). Mol Phylogenet Evol 127:229–238
Jordal B, Tischer M (2020) Genetic and taxonomic assessment of the widespread Afrotropical ambrosia beetle Xyleborus principalis (Coleoptera, Scolytinae). Int J Trop Insect Sci 40:707–715
Jordal BH, Cognato AI (2012) Molecular phylogeny of bark and ambrosia beetles reveals multiple origins of fungus farming during periods of global warming. BMC Evol Biol 12:133
Jordal BH, Kambestad M (2014) DNA barcoding of bark and ambrosia beetles reveals excessive NUMTs and consistent east-west divergence across Palearctic forests. Mol Ecol Resour 14:7–17
Jordal BH, Normark BB, Farrell BD (2000) Evolutionary radiation of an inbreeding haplodiploid beetle lineage (Curculionidae, Scolytinae). Biol J Linn Soc 71:483–499
Jordal BH, Beaver RA, Kirkendall LR (2001) Breaking taboos in the tropics: Incest promotes colonization by wood-boring beetles. Glob Ecol Biogeogr 10:345–357
Jordal BH, Emerson BC, Hewitt GM (2006) Apparent ‘sympatric’ speciation in ecologically similar herbivorous beetles facilitated by multiple colonizations of an island. Mol Ecol 15:2935–2947
Jordal BH, Normark BB, Farrell BD, Kirkendall LR (2002) Extraordinary haplotype diversity in haplodiploid inbreeders: Phylogenetics and evolution of the bark beetle genus Coccotrypes. Mol Phylogenet Evol 23:171–188
Kageyama D, Narita S, Watanabe M (2012) Insect sex determination manipulated by their endosymbionts: Incidences, mechanisms and implications. Insects 3:161–199
Kaur R, Shropshire JD, Cross KL, Leigh B, Mansueto AJ, Stewart V et al. (2021) Living in the endosymbiotic world of Wolbachia: A centennial review. Cell Host Microbe 29:879–893
Kawasaki Y, Ito M, Miura K, Kajimura H (2010) Superinfection of five Wolbachia in the Alnus ambrosia beetle, Xylosandrus germanus (Blandford) (Coleoptera: Curuculionidae). Bull Entomol Res 100:231–239
Kawasaki Y, Schuler H, Stauffer C, Lakatos F, Kajimura H (2016) Wolbachia endosymbionts in haplodiploid and diploid scolytine beetles (Coleoptera: Curculionidae: Scolytinae). Environ Microbiol Rep. 8:680–688
Kearse M, Moir R, Wilson A, Stones-Havas S, Cheung M, Sturrock S et al. (2012) Geneious Basic: An integrated and extendable desktop software platform for the organization and analysis of sequence data. Bioinformatics 28:1647–1649
Keller L, Peer K, Bernasconi C, Taborsky M, Shuker DM (2011) Inbreeding and selection on sex ratio in the bark beetle Xylosandrus germanus. BMC Evol Biol 11:359
Kirkendall LR (1983) The evolution of mating systems in bark and ambrosia beetles (Coleoptera: Scolytidae and Platypodidae). Zool J Linn Soc 77:293–352
Kirkendall LR, Biedermann PHW, Jordal BH (2015). Evolution and diversity of bark and ambrosia beetles. In: Bark Beetles. In: Vega F, Hofstetter RW (eds) Biology and Ecology of Native and Invasive Species. Academic Press, pp 85–156.
Leigh JW, Bryant D (2015) POPART: Full-feature software for haplotype network construction. Methods. Ecol Evol 6:1110–1116
Martin HA (2006) Cenozoic climatic change and the development of the arid vegetation in Australia. J Arid Environ 66:533–563
Mathenge CW, Riegler M, Beattie GAC, Spooner-Hart RN, Holford P (2015) Genetic variation amongst biotypes of Dactylopius tomentosus. Insect Sci 22:360–374
Mayers CG, Bateman CC, Harrington TC(2018) New Meredithiella species from mycangia of Corthylus ambrosia beetles suggest genus-level coadaptation but not species-level coevolution. Mycologia 110:63–78
Mayers CG, McNew DL, Harrington TC, Roeper RA, Fraedrich SW, Biedermann PHW et al. (2015) Three genera in the Ceratocystidaceae are the respective symbionts of three independent lineages of ambrosia beetles with large, complex mycangia. Fungal. Biol 119:1075–1092
Mayers CG, Harrington TC, Masuya H, Jordal BH, McNew DL, Shih H-H et al. (2020) Patterns of coevolution between ambrosia beetle mycangia and the Ceratocystidaceae, with five new fungal genera and seven new species. Persoonia - Mol Phylogeny Evol Fungi 44:41–66
Mayers CG, Harrington TC, Biedermann PHW (2022) Mycangia Define the Diverse Ambrosia Beetle–Fungus Symbioses. In: Schultz TR, Gawne R, Peregrine PN (eds) The Convergent Evolution of Agriculture in Humans and Insects. The MIT Press, pp. 105–142
Minh BQ, Schmidt HA, Chernomor O, Schrempf D, Woodhams MD, Von Haeseler A et al. (2020) IQ-TREE 2: New models and efficient methods for phylogenetic inference in the genomic era. Mol Biol Evol 37:1530–1534
Mitchell A, Maddox C (2010) Bark beetles (Coleoptera: Curculionidae: Scolytinae) of importance to the Australian macadamia industry: an integrative taxonomic approach to species diagnostics. Aust J Entomol 49:104–113
Morrow J, Riegler M (2021) Genome analyses of four Wolbachia strains and associated mitochondria of Rhagoletis cerasi expose cumulative modularity of cytoplasmic incompatability and cytoplasmic hitchhiking across host populations. BMC Genom 22:616
Paradis E (2010) Pegas: An R package for population genetics with an integrated-modular approach. Bioinformatics 26:419–420
Peer K, Taborsky M (2004) Female ambrosia beetles adjust their offspring sex ratio according to outbreeding opportunities for their sons. J Evol Biol 17:257–264
Peer K, Taborsky M (2005) Outbreeding depression, but no inbreeding depression in haplodiploid ambrosia beetles with regular sibling mating. Evolution 59:317–323
Pistone D, Gohli J, Jordal BH (2018) Molecular phylogeny of bark and ambrosia beetles (Curculionidae: Scolytinae) based on 18 molecular markers. Syst Entomol 43:387–406
Poinsot D, Merçot H (1997) Wolbachia infection in Drosophila simulans: does the female host bear a physiological cost? Evolution 51(1):180–186
Puillandre N, Lambert A, Brouillet S, Achaz G (2012) ABGD, Automatic Barcode Gap Discovery for primary species delimitation. Mol Ecol 21:1864–1877
Pullen KR, Jennings D, Oberprieler RG (2014) Annotated catalogue of Australian weevils (Coleoptera: Curculionoidea). Zootaxa 3896:1–481
Raffa KF, Grégoire J-C, Staffan Lindgren B (2015). Natural History and Ecology of Bark Beetles. In: Bark Beetles. Biology and Ecology of Native and Invasive Species. Vega F., Hofstetter RW Ed. Academic Press, pp 1–40
Rambaut A, Drummond AJ, Xie D, Baele G, Suchard MA (2018) Posterior summarization in bayesian phylogenetics using tracer 1.7. Syst Biol 67:901–904
Rambaut A (2014). Figtree v1.4.2.
Schebeck M, Feldkirchner L, Marín B, Krumböck S, Schuler H, Stauffer C (2018) Reproductive manipulators in the bark beetle Pityogenes chalcographus (Coleoptera: Curculionidae)—The role of Cardinium, Rickettsia, Spiroplasma, and Wolbachia. J Insect Sci 18:4
Schedl K (1936) Scolytidae and Platypodidae. Contribution 35. The collection of the South Australian Museum. Rec South Aust Mus 5:513–535
Shropshire JD, Bordenstein SR (2016) Speciation by symbiosis: the microbiome and behavior. MBio 7:e01785–15
Simon C, Frati F, Beckenbach A, Crespi B, Liu H, Flook P (1994) Evolution, weighting, and phylogenetic utility of mitochondrial gene sequences and a compilation of conserved Polymerase Chain Reaction primers. Ann Entomol Soc Am 87:651–701
Skelton J, Johnson AJ, Jusino MA, Bateman CC, Li Y, Hulcr J (2019) A selective fungal transport organ (mycangium) maintains coarse phylogenetic congruence between fungus-farming ambrosia beetles and their symbionts. Proc R Soc B Biol Sci 286:20182127
Smith NGC (2000) The evolution of haplodiploidy under inbreeding. Heredity 84:186–192
Smith S, Cognato I (2022) New non-native pseudocryptic Cyclorhipidion species (Coleoptera: Scolytinae: Xyleborini) found in the United States as revealed in a multigene phylogeny. Insect Syst Divers 6:2
Storer C, Payton A, McDaniel S, Jordal B, Hulcr J (2017) Cryptic genetic variation in an inbreeding and cosmopolitan pest, Xylosandrus crassiusculus, revealed using dd RAD seq. Ecol Evol 7:10974–10986
Swofford DL (2003) PAUP. Phylogenetic Analysis Using Parsimony (and Other Methods), Version 4a168. Sinauer Associates, Sunderland, Massachusetts
Takenouchi Y, Takagi K (1967) A chromosome study of two parthenogenetic scolytid beetles. Annot Zool Jpn 40:105–110
Tamura K, Stecher G, Kumar S (2021) MEGA11: Molecular Evolutionary Genetics Analysis Version 11. Mol Biol Evol 38:3022–3027
Telschow A, Yamamura N, Werren JH (2005) Bidirectional cytoplasmic incompatibility and the stable coexistence of two Wolbachia strains in parapatric host populations. J Theor Biol 235:265–274
Templeton AR (1989) The meaning of species and speciation: A genetic perspective. In: Otte D, Endler JA Eds Speciation and its consequences. Sinauer Associates, Sunderland, Massachusetts, p 3–27
Tremmel M, Steinitz H, Kliot A, Harari A, Lubin Y (2020) Dispersal, endosymbiont abundance and fitness-related consequences of inbreeding and outbreeding in a social beetle. Biol J Linn Soc 129:717–727
van de Peppel LJJ, Aanen DK, Biedermann PHW (2018) Low intraspecific genetic diversity indicates asexuality and vertical transmission in the fungal cultivars of ambrosia beetles. Fungal Ecol 32:57–64
Vega FE, Benavides P, Stuart JA, O’Neill SL (2002) Wolbachia infection in the coffee berry borer (Coleoptera: Scolytidae). Ann Entomol Soc Am 95:374–378
Weinert LA, Araujo-Jnr EV, Ahmed MZ, Welch JJ (2015) The incidence of bacterial endosymbionts in terrestrial arthropods. Proc R Soc B Biol Sci 282:20150249
Werren JH, Baldo L, Clark ME (2008) Wolbachia: master manipulators of invertebrate biology. Nat Rev Microbiol 6:741–751
Whitlock MC (2003) Fixation probability and time in subdivided populations. Genetics 164:767–779
Wood SL, Bright DE (1992) A catalog of Scolytidae and Platypodidae (Coleoptera), part 2: Taxonomic index volume A. Gt Basin Nat Mem 13:1–1310
Yang RH, Su JH, Shang JJ, Wu YY, Li Y, Bao DP et al. (2018) Evaluation of the ribosomal DNA internal transcribed spacer (ITS), specifically ITS1 and ITS2, for the analyses of fungal diversity by deep sequencing. PLoS ONE 13:e0206428
Zchori-Fein E, Borad C, Harari AR (2006) Oogenesis in the date stone beetle, Coccotrypes dactyliperda, depends on symbiotic bacteria. Physiol Entomol 31:164–169
Zhang J, Kapli P, Pavlidis P, Stamatakis A (2013) A general species delimitation method with applications to phylogenetic placements. Bioinformatics 29:2869–2876
Zug R, Hammerstein P (2015) Bad guys turned nice? A critical assessment of Wolbachia mutualisms in arthropod hosts. Biol Rev 90:89–111
Acknowledgements
We thank Rolf Oberprieler, Debbie Jennings and Hermes Escalona from ANIC, Simon Grove from TMAG and Justin Bartlett from QDAF for access to Cnestus specimens. We thank Chase Mayers for suggestions for the isolation of the Ambrosiella fungi and for advice on phylogenetic analyses. This research was supported by a Western Sydney University Postgraduate scholarship awarded to JRMB and a Holsworth Wildlife Research Endowment (Equity Trustees Charitable Foundation & the Ecological Society of Australia) awarded to JRMB and MR.
Funding
Open Access funding enabled and organized by CAUL and its Member Institutions.
Author information
Authors and Affiliations
Contributions
JRMB, BHJ and MR conceptualised the research and designed the experiments. JRMB and MR collected the data, JRMB, BHJ and MR analysed and interpreted the data. JRMB wrote the manuscript with input of BHJ and MR. All authors agreed to the submitted manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Associate editor Darren Obbard.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Bickerstaff, J.R.M., Jordal, B.H. & Riegler, M. Two sympatric lineages of Australian Cnestus solidus share Ambrosiella symbionts but not Wolbachia. Heredity 132, 43–53 (2024). https://doi.org/10.1038/s41437-023-00659-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41437-023-00659-w
- Springer Nature Switzerland AG