Abstract
The Tetraspanins (Tspan) protein family, also known as the tetraspanin family, contains 33 family members that interact with other protein molecules such as integrins, adhesion molecules, and T cell receptors by forming dimers or heterodimers. The Tspan protein family regulates cell proliferation, cell cycle, invasion, migration, apoptosis, autophagy, tissue differentiation, and immune response. More and more studies have shown that Tspan proteins are involved in tumorigenesis, epithelial-mesenchymal transition, thrombosis, tumor stem cell, and exosome signaling. Some drugs and microRNAs can inhibit Tspan proteins, thus providing new strategies for tumor therapy. An in-depth understanding of the functions and regulatory mechanisms of the Tspan protein family, which can promote or inhibit tumor development, will provide new strategies for targeted interventions in the future.
Similar content being viewed by others
Facts
-
The Tspan protein family is strongly associated with the development and progression of cancer.
-
Members of the Tspan family of proteins serve as biomarkers of cancer progression.
-
The search for effective drugs that induce cancer death and inhibit the death of immune cells and normal tissue cells is a promising direction for future cancer therapy.
Open Questions
-
Which structural elements contribute to the dual role of Tspan protein family members in cancer development and progression?
-
How does the Tspan protein family play a role in the biological processes of cancer? What are the primary mechanisms?
-
Can drugs targeting Tspan proteins, either directly or indirectly, be effective in cancer treatment?
Introduction
Tetraspanins (Tspans), first discovered in 1990, constitute a superfamily of 20–50 kDa transmembrane proteins that are widely expressed and distributed in multicellular organisms. They play crucial roles in both physiological and pathological processes, and exhibit highly conserved protein sequences within the family and across distantly related species [1]. Studies have demonstrated the widespread expression of Tspans across various tissues and cell types, with many cells expressing multiple Tspan members [2]. The Tspan protein family in humans consists of 33 members, ranging from Tspan1 to Tspan33, with specific tissue-specific expression patterns [3]. For instance, Tspan5 exhibits high expression in cortical regions and Purkinje cells [4], while Tspan7 is highly expressed in brain tissue and islets [5, 6], Moreover, Tspan26 represents a B-cell-surface antigen that demonstrates widespread expression in mature B cells [7]. Tspans can interact with their own or cell-surface molecules, including integrins, immunoglobulin superfamily proteins, proteases, growth factor receptors, and intracellular signaling molecules, forming complexes known as Tetraspanin-enriched microdomains (TEMs) [8]. TEMs regulate essential physiological processes, including cell signaling, adhesion, migration, proliferation, and differentiation [9].
Depending on the tumor type and the specific member of the Tspan protein family, Tspans can have either a promoting or a suppressing role in cancer. For example, Tspan1 and Tspan8 are upregulated in pancreatic cancer and colorectal cancer, respectively, and promote tumor cell invasion, migration, and autophagy [10, 11]. Tspan15 plays a pivotal role in promoting tumor cell proliferation by enhancing ERK phosphorylation, which in turn drives the expression and secretion of connective tissue growth factor (CTGF). This signaling cascade ultimately drives the proliferation of hepatocellular carcinoma (HCC) [12]. However, Tspan32 stands as a notable exception to the overall pro-tumorigenic role of Tspans. Its overexpression effectively impedes leukemia progression and impairs the proliferation of leukemia stem cells [13]. This highlights the complex and context-dependent regulation of Tspans in tumor development. Apart from these examples, other members of the Tspan protein family also influence tumor cell behavior and response to therapy, making them potential markers and targets of tumor development. It can offer novel strategies for targeted drug therapy. The majority of Tspan proteins play a promoting role in tumor occurrence and development. Consequently, the judicious utilization of Tspan inhibitors can yield improved strategies for clinical treatment and tumor prognosis.
The molecular structure of the Tspan protein family
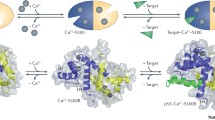
Tspans consist of four highly hydrophobic and conserved transmembrane structural domains: TM1, TM2, TM3, and TM4. TM1 and TM2 are linked by a small extracellular loop (EC1) containing 20–28 amino acids, while TM3 and TM4 are connected by a larger extracellular loop (EC2) comprising 76–131 amino acids [14, 15]. TM3 are connected by a very short intracellular loop (IL), and Tspans include short cytoplasmic N-terminal and C-terminal ends [16]. Tspans have several distinct features from other proteins with four TMs. Firstly, the TM1, TM3, and TM4 structural domains feature polar residues of unknown function, which include an absolutely conserved CCG (Cys-Cys-Gly sequence) motif and two additional cysteine residues. These highly conserved amino acid residues constitute one of the most distinctive features that set Tspans apart from other proteins with four transmembrane domains [17]. In addition, more than 50% of the four-span proteins carry the PxxCC (Pro-x-x-Cys-Cys) motif, where x can be any amino acid [15, 18]. Cysteine pairs in EC2 play an important role in the correct folding of the structural domain by forming disulfide bridges (Fig. 1).
Each Tspans has specific partners, including integrins and histocompatibility antigens, etc. These form TEMs through protein-protein interactions, which enable the transmission of signals from extracellular, intermembrane, and intracellular proteins, directly or indirectly influencing intracellular signaling pathways to regulate cellular activities and metabolism [19,20,21]. The cytoplasmic structural domain contains palmitoylation sites on conserved intracellular cysteine residues. Specific functions are associated with the distinct structural domains of Tspans [22]. The transmembrane structural domains stabilize individual Tspans and facilitate binding to other Tspans on the cell membrane [22]. The variable region of EC2 mediates specific protein-protein interactions and the cytoplasmic region may provide a link to the cytoskeleton and signaling molecules [23]. Tspan5, Tspan10, Tspan14, Tspan15, Tspan17, and Tspan33 possess a unique property of containing 8 cysteines in their EC2. These proteins can undergo phosphorylation, methylation, and ubiquitination [18]. Palmitoylation of the EC2 of Tspan28 inhibits infection by the hepatitis C virus [24].
The role of the Tspan protein family in cancer progression
The progression of cancer involves a cascade reaction where cancer cells escape from the primary site, infiltrate blood vessels, circulate, and colonize distant sites. Accumulated evidence indicates that the Tspan protein family plays a regulatory role in various aspects of tumor biology, including cell proliferation, cell cycle progression, invasion, migration, autophagy, and apoptosis, mediated by signaling pathways [8, 14]. Furthermore, the members of the Tspan protein family exhibit distinct expression patterns across various types of cancers (Fig. 2). Despite the high structural conservation among members of the Tspan protein family and the absence of fundamental differences in the assembly of transmembrane and signal transduction molecules within TEMs, these proteins exert distinct regulatory roles in tumorigenesis. Specifically, they can either promote or inhibit the development of tumors (Table 1).
The role of the Tspan protein family in cell proliferation
The proliferation of cancer cells refers to the rapid division and multiplication of cancer cells under certain conditions, resulting in the formation of tumors [25, 26]. The proliferation of normal cells is regulated by the differentiation and death of organ cells as well as the regulation of signals, while the proliferation of cancer cells loses these self-regulatory abilities, which is considered to be the root cause of cancer [27]. According to the carcinogenic mechanism, the Tspan protein family is significantly associated with cancer proliferation in different cancers. Tspan further inhibits or promotes the ability of cancer cells to proliferate by inhibiting and activating signaling pathways related to cancer proliferation [28]. In other words, the Tspan protein family has two effects on cancer proliferation.
Tspan1 promotes the proliferation of cholangiocarcinoma and breast cancer by promoting the PI3K/AKT signaling pathway [28, 29]. Overexpression of Tspan15 in HepG2 significantly increased cell proliferation, which was achieved by activating the EGFR/MAPK/ERK axis [30]. In bladder cancer, the regulation of EGFR activity by Tspan27 is influenced by the ganglioside GD1a, which is important for the spatial organization of Tspan27-rich microstructural domains and interferes with Tspan27 recruitment of negatively regulated EGFR molecules such as tyrosine phosphatases, similar to the GD1a-Tspan27-EGFR complex, and the crosstalk between Tspan27, integrin α3 and HGFR intercrosstalk is also regulated by gangliosides. The ganglioside GM2-GM3-Tspan27 complex interferes with HGFR activation, accompanied by decreased GRB2 and HRA activity upstream of the MAPK pathway and GAB1 activity upstream of PI3K, thereby affecting cell motility and proliferation [31]. In HCC, Tspan31 promotes cell proliferation and motility by activating the AKT/GSK-3β/β-catenin pathway [32].
Conversely, in bladder cancer, overexpression of Tspan7 inhibits p-PI3K and p-AKT, thereby inhibiting the proliferation of bladder cancer [33]. In ovarian cancer and fibromasarco, Tspan29 inhibits cell proliferation and migration by affecting several β1 integrin subpopulations or forming EWI-2/EWI-F/β1 complexes, inactivating AKT, p38, and EGFR signaling pathways [34, 35]. As the research on Tspan proteins deepens, there is more and more evidence that the Tspan protein family is involved in the regulation of tumor proliferation. Based on the dual role of the Tspan protein family for tumors, the study of Tspan on cancer proliferation, prevention, treatment, and prognosis can provide new ideas (Fig. 3).
Mechanism summary
Tspan1 and Tspan15 activate the PI3K/AKT and EGFR/MAPK/ERK pathways, respectively, leading to enhanced proliferation in cholangiocarcinoma and breast cancer. Tspan27 facilitates EGFR activity and cell motility through interactions with gangliosides and other proteins. Tspan31 promotes proliferation and motility in hepatocellular carcinoma via the AKT/GSK-3β/β-catenin pathway. In contrast, Tspan7 suppresses proliferation in bladder cancer by inhibiting p-PI3K and p-AKT. Tspan29 exerts anti-proliferative and anti-migratory effects in ovarian cancer and fibrosarcoma by targeting specific β1 integrin subpopulations and signaling pathways.
The role of the Tspan protein family in the cell cycle
The intricate choreography of the cell cycle, encompassing Gap phases (G1, G2), DNA synthesis (S phase), and mitosis (M phase), is meticulously orchestrated by various factors, including the Tspan protein family. Their influence on this fundamental process is evident through their interactions with key regulatory components [36,37,38]. Malignant cells exhibit a distorted cell cycle, bypassing critical checkpoints that normally restrain excessive cell division. This deregulation enables cancer cells to evade normal growth constraints and acquire the ability to undergo limitless replication, forming tumors and metastasizing to distant sites [39]. The Tspan protein family plays an important role in the orderly operation of the cell cycle. In colon cancer, after Tspan1 is knocked down, the cell cycle is blocked in the G1/G0 phase [40]. Similarly, in gastric cancer, TSPAN1 knockdown induces G1/G0 phase block and inhibits invasion and migration [41]. In contrast, in gastric cancer, overexpression of Tspan9 significantly inhibited cell proliferation, with cells remaining in the G1 phase and a significant decrease in S-phase cells [42].
Mechanistically, Tspan proteins engage in a delicate tango with key orchestra conductors of the cell cycle. Tspan12 knockdown in ovarian cancer disrupts the rhythmic progression through G1-S-G2/M phases, implicating its involvement in the intricate interplay of cyclins and cyclin-dependent kinases (CDKs) like Cyclin A2, D1, E2, CDK2, and CDK4 [43]. This suggests that Tspan12 acts as a choreographer downstream of these critical cell cycle regulators. Likewise, in cervical cancer, Tspan31 directly silences CDK4 by targeting its 3′-untranslated region (3′-UTR), a master switch controlling cell cycle progression [44]. Furthermore, Silencing Tspan31 itself reverses this effect, highlighting its direct role in orchestrating the cell cycle tempo [44]. The downstream effects of the Tspan protein family usually serve to regulate the cell cycle. These findings paint a dynamic picture of Tspan proteins as context-dependent modulators of the cell cycle. While their downstream effects are evident, the precise mechanisms by which they interact with and manipulate specific signaling pathways remain shrouded in mystery. Future research endeavors that delve deeper into these intricate dances hold immense potential for unlocking the full therapeutic potential of Tspan proteins in the fight against cancer.
Mechanism summary
Knockdown of Tspan1 in colon and gastric cancer cells elicits G1/G0 phase arrest and inhibits cell proliferation, highlighting its tumor-suppressive potential. Tspan12 knockdown in ovarian cancer disrupts the rhythmic progression through G1-S-G2/M phases, suggesting its involvement in the intricate interplay of cyclins and CDKs, the orchestra conductors of the cell cycle. Tspan31 directly silences CDK4, a master switch controlling cell cycle progression, by targeting its 3′-untranslated region (3′-UTR) in cervical cancer, highlighting its ability to modulate cell cycle checkpoints.
The role of the Tspan protein family in invasion and metastasis
Tumor cell metastasis is a multi-stage process in which malignant tumor cells spread from the primary site, through the body cavity, and vascular and lymphatic pathways, to reach other sites for further growth, and the process requires the participation of cell motility [45, 46]. Therefore, regulating the motility of tumor cells to prevent tumor cells from spreading and improve patient survival has also become one of the focuses of cancer treatment. Tspan1 regulates MMP2 expression through PLCγ and inhibits migration and invasion of pancreatic cancer cells [20]. In lung cancer, Tspan2 enhances cell motility and invasiveness by assisting Tspan8 to scavenge intracellular reactive oxygen species [47]. Tspan8 expression is upregulated in breast cancer stem cells, and upregulation of Tspan8 expression leads to increased drug resistance and stemness in tumor cells [48]. Mechanistically, Tspan8 enhances breast cancer cell stemness by interacting with PTCH1, recruiting ATXN3 deubiquitinating enzymes to inhibit proteasome-mediated degradation of the SHH/PTCH1 complex, and activating the Hedgehog signaling pathway. In human epidermoid carcinoma, Tspan24 binds to integrin α6 and EGFR and regulates laminin adhesion and migration [49]. By binding to EWI-2, Tspan27 inhibits laminin and fibronectin activity and suppresses prostate cancer invasion and migration [50]. Tspan27 interferes with lung cancer growth factor receptor (HGFR) signaling and thus activates Rac and CDC42, among a series of other mechanisms to inhibit cell invasion [51].
Tspan28 regulates the development of colorectal cancers through epithelial-mesenchymal transition, and inhibition of Tspan28 expression leads to reduced migration ability of cancer cells and reduced lung metastasis [52]. In addition, increased expression of Tspan28 in melanoma was also reported to promote tumor progression and metastasis [53, 54]. Tspan29 inhibits tumor metastasis, and its function is related to the inhibition of integrin-mediated motility. In ovarian cancer, Tspan29 expression levels correlated with integrins β1, α2, α3, α5, and α6, and downregulation of Tspan29 expression resulted in diminished stromal adhesion and diffuse growth [35]. Tspan30 inhibition of metastasis formation may be dependent on integrin endocytosis, MMP14 lysosomal degradation, and recruitment of tissue inhibitor of metalloproteinases 1 (TIMP1) [55]. Numerous studies have shown that most members of the Tspan protein family exhibit significant aggressiveness and metastatic properties. Therefore, inhibition of Tspan-related targets may provide new avenues to mitigate cancer metastasis and improve patient survival.
Mechanism summary
Tspan proteins interact with a wide range of signaling pathways, including integrin signaling, ECM remodeling, and angiogenesis, to orchestrate metastasis. Tspan1 regulates MMP2 expression and either promoting or inhibiting invasion. Tspan2 assists Tspan8 in scavenging intracellular reactive oxygen species. Tspan8 upregulation promotes breast cancer stemness and drug resistance.Tspan24 binds to integrin α6 and EGFR. Tspan27 binds to EWI-2, inhibiting laminin and fibronectin activity. It also interferes with HGFR signaling, activating Rac and CDC42.Tspan28 regulates through EMT. Tspan29 inhibits integrin-mediated motility. Tspan30 depends on integrin endocytosis, MMP14 lysosomal degradation, and recruitment of TIMP1.
The role of the Tspan protein family in cell apoptosis
Apoptosis is the autonomous, orderly death of cells that are genetically controlled, also known as programmed death [56]. The Tspan protein family has a dual function in cancer cell apoptosis, namely anti-apoptotic and pro-apoptotic activities [21, 57]. Depending on the tumor type and the specific member of the Tspan protein family, the effect of Tspan proteins on apoptosis can be either promoting or suppressing cancer. For example, in breast cancer, Tspan12 has a pro-apoptotic function, as it significantly reduced primary tumor xenograft growth, while increasing tumor apoptosis [57]. Tspan12 removal also altered the expression of several genes regulated by β-catenin (CCNA1, CCNE2, WISP1, ID4, SFN, ME1), inhibiting alterations in tumor growth and metastasis [57]. Similarly, in thyroid cancer, the downregulation of Tspan13 significantly inhibited the proliferation and promoted the apoptosis of TPC-1 cells [21]. On the other hand, in glioblastoma multiforme (GBM), Tspan17 has an anti-apoptotic function, as it is associated with poor prognosis in GBM patients. Tspan17 knockdown promotes cell apoptosis [58]. Likewise, in gastric cancer, Tspan31 has an anti-apoptotic function, as the level of apoptosis in gastric cancer cells was significantly increased after upregulation of Tspan31 expression and significantly decreased after downregulation of Tspan31 expression [59]. Tspan31 facilitated the proliferation and metastasis of gastric cancer through METTL1/CCT2 and PI3K/AKT [59, 60]. Elucidating the molecular mechanisms of the Tspan protein family in apoptosis can help identify new therapeutic targets and develop new treatment strategies.
Mechanism summary
Tspan proteins interact with various apoptosis signaling pathways, including β-catenin, METTL1/CCT2, and PI3K/AKT, to orchestrate apoptosis. Tspan12 removal reduces tumor growth and promotes apoptosis, altering the expression of β-catenin-regulated genes. Tspan13 inhibits proliferation and promotes apoptosis. Tspan17 promotes apoptosis, potentially improving prognosis. Tspan31 inhibits apoptosis and increases proliferation and metastasis through METTL1/CCT2 and PI3K/AKT signaling pathways.
The role of the Tspan protein family in cell autophagy
Autophagy is a process by which a cell engulfs its own cytoplasmic proteins or organelles and encapsulates them into vesicles and fuses with lysosomes to form autophagic lysosomes that degrade their encapsulated contents, thereby achieving the cell’s own metabolic needs and the renewal of certain organelles [61, 62]. Only Tspan1 and Tspan9 of the Tspan protein family have been reported to be associated with autophagy in cancer. Tspan1 expression was demonstrated to be upregulated in pancreatic cancer in vitro and in vivo, and Tspan1 deletion decreased the proliferation of pancreatic cancer cells. In addition, Tspan1 is a novel positive regulator of autophagy characterized by reduced expression of LC3-II and SQSTM1/p62 and inhibition of GFP-LC3 autophagic vacuoles [10]. In gastric cancer, Tspan9 knockdown was followed by a significant decrease in the LC3-II/I ratio and a significant increase in SQSTM1/p62 levels [63]. Tspan9 overexpression increased the LC3-II/I ratio and decreased SQSTM1/p62 levels [63]. The Tspan protein family has been less studied in cancer in relation to autophagy. Although it has been shown that Tspan proteins play an important role in cellular autophagy, an in-depth understanding of the effects of other Tspan protein family members in different cancers would make an essential contribution to the field of cancer therapy.
Mechanism summary
Tspan1 acts as a positive regulator of autophagy, characterized by reduced expression of LC3-II and SQSTM1/p62 and inhibition of GFP-LC3 autophagic vacuoles. Tspan9 knockdown decreases the LC3-II/I ratio and increases SQSTM1/p62 levels, while overexpression of Tspan9 increases the LC3-II/I ratio and decreases SQSTM1/p62 levels.
The role of the Tspan protein family in tumor immunity
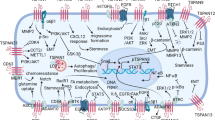
Antitumor immunity is formed by different types of immune cells present in the tumor microenvironment, and there is growing evidence demonstrating the involvement of the Tspan protein family in tumor-associated immune responses [64]. Tspans can promote or inhibit tumor invasion and metastasis by interacting with the tumor cell microenvironment, Tspans induce immune responses to tumors in a context-dependent manner, and they can directly influence immune-cell signaling by interacting with key immune receptors (e.g., MHC molecules, CD4, CD8, CD19), Tspan protein family directly impacts immune-cell signaling, influencing antigen presentation, immune-cell migration, cytokine production, as well as T cell proliferation, activation, and subpopulation differentiation (Fig. 4).
During tumor immunity, immune cells migrate from peripheral lymphoid organs to the tumor with the aim of destroying tumor cells, and the Tspan protein family is involved in this process; however, tumors can also undergo immune escape to evade immune surveillance, and tumor cells recruit different types of cells (e.g., Tregs and MDSCs) to create specific tumor microenvironments, and tumor microenvironments are composed of several different cells, including tumor cells and different immune cells, immune cells produce different soluble factors, including cytokines and antibodies, Tspan can produce immune-cell-mediated antitumor or pro-tumor immune effects in the tumor microenvironment.
The initial stage of the immune response involves the uptake, processing, and presentation of tumor antigens by APCs. Tspan25, Tspan26, Tspan27, and Tspan28 have demonstrated associations with the MHC class II complex [65]. Conversely, Tspan30 has been reported to inhibit antigen presentation [66]. Adequate immune responses require the migration of immune cells from the surrounding tissues to the tumor site. TSPAN proteins have been observed to influence dendritic cell migration [67]. Specifically, Tspan26 and Tspan28 have been shown to promote dendritic cell migration [68, 69], whereas Tspan27 inhibits dendritic cell migration68. Additionally, Tspan24 facilitates T cell migration [70]. The Tspan protein family can produce immune-cell-mediated antitumor or pro-tumor immune effects. Tspan28−/− mice are severely impaired in tumor growth and metastasis, and lacking Tspan28 are defective in the inhibitory capacity of both Tregs and MDSCs [54]. Tspan7 is highly upregulated in tertiary lymphoid structures-related vessels and Tspan7 plays a role in lymphocyte stasis and lymphocyte recruitment involved in tertiary lymphoid structures-related vessels [71]. Tspans were also found to reduce infiltration of tumor immune cells, enrich immune-related signaling pathways, and express immune checkpoint proteins in cancer patients [72]. Tspan9 expression was significantly negatively correlated with tumor immune-cell infiltration and the immune checkpoint CTLA4, Tspan9 can decrease tumor immune-cell infiltration, enrich immune-related signaling pathways, and express immune checkpoint proteins to exert its tumor suppressor effects [72]. Tspan11 is a tumor mesenchymal-associated biomarker that correlates with tumor immunity [73]. Tspan24 can regulate the interactions between tumor cells and immune cells, and Tspan24 has the function of regulating both innate immune cells and acquired immune cells [74]. Tspan24 peptide tumor vaccine prepared with this inspiration also has potential therapeutic value. Tspan24 peptide tumor vaccines also have potential therapeutic value, and antitumor active immunity initiated by Tspan24 peptide may be an effective and safe method to inhibit tumor progression. Using the synthetic Tspan24 peptide as a tumor vaccine, the vaccine triggered an active immune response in both primary and metastatic tumor models, promoted tumor CD8 lymphocyte invasion and reduced MDSCs, and effectively inhibited tumor growth and metastasis [75]. Tspans affect immune function through lateral interactions with their counterparts, GPR56 (Gprotein-coupled receptor56) was found to be an inhibitory receptor expressed on human NK cells, and cis interaction with Tspan28 attenuates the cytotoxic activity of NK cells [76].
The Tspan protein family has the ability to impact cytokine production. Tspan27 assumes a crucial role in shaping innate immune signaling, specifically in the transport and signaling of Toll-like receptor 9 (TLR9). TLR9 is responsible for recognizing unmethylated cytosine-phosphate-guanine (CpG) motifs found in viral, bacterial, and fungal DNA. Notably, Tspan27’s regulation of TLR9-dependent nuclear translocation of NF-κB is essential for the production of inflammatory cytokines [77]. The Tspan protein family is associated with T cell proliferation; activation and subpopulation differentiation, and the Tspan1 protein is strongly associated with the polarization ratio of CD4 T cells and Th17 cells [78]. Tspan25 is a chaperone of the T cell-surface tyrosine phosphatase CD45 and can regulate the CD45 activity, Tspan25 is required for T cell activation and proliferation [79]. In conclusion, different Tspans can play positive or negative regulatory roles in innate and acquired immunity during tumor immune surveillance. Further exploration of the Tspan protein family’s role in immune cells will deepen our understanding of these membrane proteins’ contributions to antitumor immunity and their potential as targets for immune-cell-based interventions [80].
Mechanism summary
Tspan25, Tspan26, Tspan27, and Tspan28 interact with the MHC class II complex, influencing antigen presentation by antigen-presenting cells (APCs). Tspan26 and Tspan28 promote dendritic cell migration, while Tspan27 inhibits dendritic cell migration. Tspan24 facilitates T cell migration. Tspan27 plays a crucial role in innate immune signaling, specifically in the transport and signaling of Toll-like receptor 9 (TLR9), which is responsible for recognizing unmethylated cytosine-phosphate-guanine (CpG) motifs found in viral, bacterial, and fungal DNA. Tspan1 is strongly associated with the polarization ratio of CD4 T cells and Th17 cells. Tspan25 is a chaperone of the T cell-surface tyrosine phosphatase CD45 and regulates CD45 activity, which is required for T cell activation and proliferation.
The Tspan protein family in exosomes
The Tspan protein family is prominently present in the membranes of exosomes and extracellular vesicles [81]. Exosomes are small extracellular vesicles that cells secrete to facilitate intercellular communication by transferring cellular cargoes, including functional proteins, metabolites, and nucleic acids, to recipient cells [82]. Proteomics studies have revealed the widespread presence of the Tspan protein family in exosome membranes, where they can serve as targeting molecules for cell membrane binding or exert indirect control over cellular interactions through exosomes. Different tspan-associated proteins may be involved in the attachment process to specific proteins on target cells [83] These proteins play significant roles in physiological processes, including signal transduction, cell activation, motility, adhesion, and tissue differentiation [84]. The different roles that Tspan proteins can play in tumor progression may be the result of heterogeneous expression of Tspan proteins in exosomes [85]. An increase in Tspan29 and Prostate Specific Membrane Antigen (PSMA) double-positive plasma-derived circulating vesicles has been reported in patients with advanced metastatic prostate cancer, whereas double-positive Tspan29 and Tspan30 extracellular vesicles (EVs) are significantly increased in patients with limited prostate cancer [86]. Tspan proteins sorting and delivery to exosomes is tightly regulated. During advanced endosome stages, Tspan30 becomes enriched within luminal vesicles and is subsequently secreted as exosomes through the fusion of endosomes with the plasma membrane [55], The Alix and ESCRT-III pathways actively facilitate the sorting and delivery of Tspan proteins into exosomes. Additionally, Alix promotes the secretion of exosomes containing Tspan28, Tspan29, and Tspan30 [87].
The Tspan protein family exhibits diverse functions within exosomes. On one hand, it participates in morphogenesis, division, and fusion processes [88]. Tspan8 is able to modulate the content and function of EVs in breast cancer, and Tspan8 mediates a several-fold increase in the number of EVs in cell culture and the circulation of tumor-bearing animals [89]. Using breast cancer cells to obtain Tspan8-enriched small extracellular vesicles (SEVs), Tspan8 facilitated the binding of breast cancer cell-derived SEVs to target cells by enhancing the restriction of spreading and inducing restriction of fibroblasts. The binding or uptake of Tspan8-SEV promotes cellular motility and invasion and may be related to the FAK-SRC signaling pathway, EMT induction, and the upregulation of proteases in target cells [83]. On the other hand, multiple subpopulations of extracellular vesicles carrying distinct Tspan proteins are present in human blood, with varying levels of Tspan28, Tspan29, and Tspan30 across these subpopulations [90]. This variability enables Tspan proteins to serve as markers for exosome isolation and quantitative analysis [91]. Plasma EVs Tspan9 can be used as a marker for the detection of colorectal cancer [92]. Additionally, the heterogeneous expression of Tspan in exosomes allows them to determine the tumor type of a cancer patient with an unknown source of the primary tumor, supporting a diagnosis that leads to a more specific treatment plan for the cancer patient [93]. In addition, Tspan6 was found to be a negative regulator of exosome release, supporting lysosomal degradation of Syndecan4 and Syntenin, inhibiting the shedding of the extracellular structural domain of Syndecan4, and also having a positive effect on exosome formation or cargo composition [94]. The current study of exosome biology shows that Tspan6 is a negative regulator of exosome release, supporting lysosomal degradation of Syndecan4 and Syntenin, and inhibiting the shedding of the extracellular structural domain of Syndecan4. Exosomes may also provide some ideas for tumor therapy; hypoxic tumor cells release exosomes that induce tumorigenesis by promoting metastasis, angiogenesis, and modulation of the immune response, and targeting the biogenesis of these exosomes may be a therapeutic opportunity to reduce tumorigenesis, in addition, exosomes can act as drug delivery systems to transfer therapeutic compounds to cancer cells [95].
The main mechanisms of exosome biology have now been elucidated, but much remains unclear about the regulatory processes, and the heterogeneity of exosomes, their differential content, and their properties affected by donor and recipient cells add to the complexity of unraveling the regulatory processes [82] Despite the rapid development in the field of exosomes, the detailed understanding of the regulation and function of exosomes is still insufficient, the exact role of Tspan in exosome biogenesis is unclear, and there are still many challenges to be faced in the clinical application of tumor diagnosis and therapy.
Mechanism summary
The Tspan family plays a multifaceted role in regulating exosome biogenesis, function, and targeting, thereby shaping their impact on cellular processes. Tspan30, undergoes a complex sorting process, guided by the ALIX and ESCRT-III pathways, ensuring their efficient incorporation into exosomes.Tspan8, contributes to the unique composition of exosomes, influencing the cargo they carry and their functional properties.Tspan6, modulates exosome formation and dynamics, ensuring their timely release and regulating their interaction with recipient cells.Tspan8-enriched exosomes exert pro-invasive effects on target cells, potentially through the FAK-SRC signaling pathway, promoting their migration and invasion. Tspan29, Tspan30, and Tspan9, in exosomes, provide potential biomarkers for distinguishing tumor types, aiding in diagnosis and prognosis. Exosomes released from hypoxic tumor cells promote tumorigenesis, and targeting the biogenesis of these exosomes can reduce tumor progression. It can also be employed as drug delivery vehicles to cancer cells.
The role of the Tspan protein family as a biomarker in cancer diagnosis
The Tspan protein family exhibits variable expression levels across different cancer types, making it a potential biomarker for cancer (Table 2). Tspan1, Tspan11, Tspan13, Tspan28, Tspan29, and Tspan30 have been identified as biomarkers for tumor diagnosis and prognosis, while Tspan8, Tspan24, and Tspan27 show potential as biomarkers [96,97,98,99].
Tspan1 and CA2 serve as key genes for predicting prognosis and immunotherapy efficacy [100]. Tspan1, identified as a HUB gene closely associated with pancreatic cancer [101], exhibits significant correlations with tumor histological grade, T stage, clinical stage, and overall survival [102]. Tspan8 can promote the growth and migration of colorectal cancer cells [11]. Compared with normal adjacent tissues to cancer (NATs), the mRNA level of Tspan8 in pancreatic ductal adenocarcinoma, colon cancer, hepatocellular carcinoma, prostate cancer, rectal cancer, and gastric cancer is significantly increased [103]. Tspan24 serves as a favorable prognostic indicator in invasive lobular breast cancer and endometrial carcinoma. Conversely, its overexpression in gastric cancer, prostate cancer, and non-small-cell lung cancer indicates a poor prognosis. Circ_0020710, a CircRNA derived from Tspan24, promotes the proliferation, migration, and dissemination of melanoma cells, induces melanoma immune escape, and exhibits significant upregulation in melanoma. It is involved in various regulatory mechanisms, including ceRNAs, protein interactions, and the regulation of gene transcription and translation [104]. Tspan27 functions as a metastasis suppressor gene that is frequently downregulated in advanced stages of cancer [105]. Overexpression of Tspan27 can inhibit the invasion and migration of lung cancer cells. Tspan29 is sometimes employed as a biomarker for invasion and advanced stages of cancer, particularly in metastatic renal clear cell carcinoma, where it not only distinguishes tumor subtypes but also predicts the metastatic potential of renal cell carcinoma. In mesothelioma, the expression of Tspan29 is associated with survival and may serve as a favorable prognostic marker for patients [85]. Comparative proteomics analysis using a high-performance liquid chromatography-tandem mass spectrometry approach revealed TSPAN30, DDB1, TYMP, VDAC2, and DCXR as the top five candidate biomarkers for directly comparing samples of follicular adenoma and normal thyroid tissue [106].
The Tspan protein family can also be used as a biomarker of cancer-targeted therapy. The induction of autophagy gene ATG7 and the conversion of autophagy markers (LC3-I and LC3-II) and Tspan28, Tspan29, and Tspan30 in rotenone-treated prostate and breast cancer stem cells were detected by Western blotting. The results showed that rotenone could induce tumor stem cells to express Tspan28, Tspan29, Tspan30, and TSG101 [107, 108]. However, due to the limitation of experimental data and the presence of Tspan molecules on the surface of normal cells and tumor-related tissues, there are still some restrictions on the Tspan protein family as a biomarker.
The role of the Tspan protein family in cancer treatment
The significant role of the Tspan protein family in tumor development and therapy cannot be overlooked. Tspan proteins located in the biofilm of tumor cells can serve as target molecules for drugs and antibodies. Additionally, Tspan proteins secreted by exosomes, which detach from the cell membrane, play crucial roles in recognition and signaling. This suggests the potential for drug encapsulation in exosomes targeted specifically to tumor cells, enhancing therapy efficiency and precision. Moreover, microRNAs can directly target Tspan genes, altering their expression levels. The diverse range of effectors within the Tspan protein family offers numerous possibilities for clinical treatment (Table 3). Currently, specific antibodies or RNA interference strategies are being utilized to modulate Tspan protein family-associated signaling, with some agents demonstrating significant results in animal studies.
The role of the Tspan protein family in drug therapy
The Tspan protein family plays a crucial role in the pharmacological treatment of tumors, serving as both targets for antitumor drugs and antibodies and as therapeutic targets to overcome drug resistance and enhance drug sensitivity. Clinically approved drugs often have limitations in target accumulation due to their lack of specificity, resulting in modest efficacy and numerous adverse effects. To enhance their potential, researchers utilize various targeting molecules, with Tspan proteins exhibiting significant promise in this regard [109]. Yue et al. were the first to demonstrate the binding of Andrographis paniculata water (APW), including its components Andrographis paniculata Lactone, Andrographis paniculata A, and Andrographis paniculata C, to Tspan28, resulting in the inhibition of its function. These findings suggest a potential relationship between these compounds and the anti-metastatic activity of APW in esophageal cancer [110].
The Tspan protein family is exposed on the surface of the cell membrane, which permits the immobilization of antibodies that can act as targeting molecules for related monoclonal antibodies. There is substantial evidence supporting the inhibition of tumor growth and potential induction of partial or complete remission through the targeting of Tspan proteins with antibodies. Overexpression of Tspan8 in rats promotes angiogenic activity and supports tumor growth, and an anti-rat Tspan8 monoclonal antibody effectively inhibits this process [111]. Four fully human antibodies with distinct complementarity-determining regions (CDRs) were isolated from a synthetic human antibody library using phage display technology. Among these antibodies, the one exhibiting the highest affinity for binding to Tspan8-LEL was selected. The antibody was found to specifically recognize amino acid residues 140–205 of Tspan8-LEL in a conformation-dependent manner, and these findings demonstrated that Tspan8-LEL may be a potential therapeutic target for cellular invasion of metastatic colorectal cancer cells [112]. The anti-Tspan24 antibodies inhibited metastatic spreading and primary tumor growth in a mouse model of human tumors, and they possessed the ability to destroy the ability to disrupt the complex between Tspan24 and α3β1 integrin [111]. Tspan26 has been suggested as a potential target for the treatment of B-cell lymphomas using radiolabeled anti-Tspan26 antibodies [111]. By introducing the E430G mutation into the humanized Tspan26 monoclonal antibody, IgG1, and utilizing intermolecular Fc-Fc interactions upon binding to cell-surface antigens, more efficient formation of IgG hexamers is achieved. These modified antibodies exhibit stronger complement-dependent cytotoxicity (CDC) activity compared to monoclonal antibodies lacking the E430G mutation, making them potential candidates for the treatment of B-cell malignancies [113]. Tspan28 is also an anticancer target, and Tspan28 signaling is involved in the development of solid tumors development and is associated with the aggressiveness of B-cell lymphomas, Tspan28 has been investigated as an antiviral and/or anticancer agent, and a number of anti-Tspan28 monoclonal antibodies have been developed, notably mAb5A6, which is anti-invasive and metastatic in triple-negative breast cancer cells [114]. Tspan28-EC2 can also be used as a natural product and as synthetic compounds as targets [114]. Intravenous injection of anti-Tspan29 monoclonal antibody ALB6 positively affected subcutaneous transplanted tumors in nude mice with human gastric carcinoma, with a reduction in tumor volume of 60–70% in the treated group of mice as compared to the control group, while at the same time, a significant reduction in cell proliferation and angiogenesis and an increase in apoptotic signals were observed [111].
Tspan proteins are capable of increasing drug resistance and can be inhibited to overcome resistance or increase drug sensitivity. Several studies have identified Tspan1 as a significant contributor to the acquired resistance of tumor cells to conventional chemotherapy [115]. miR-155 exerts its influence by targeting Tspan5, leading to increased stemness and resistance to Decitabine (DCA) in breast cancer cells [116]. Tspan8 is implicated as a drug-resistant protein in gastric cancer cells. However, silencing Tspan8 expression enhances the sensitivity of cancer cells to cisplatin, 5-fluorouracil (5-FU), and adriamycin. Tspan8 activates the Wnt/β-conjugated protein pathway by binding to NOTCH2, which increases the expression and accumulation of β-conjugated proteins in the nucleus and creates multidrug resistance. Tspan8 inhibitors may be developed as an adjuvant therapy for gastric cancer to reduce the cancer cells’ resistance [117]. Qi et al. discovered that 5-fluorouracil (5-FU)-resistant gastric cancer cells exhibit high expression of Tspan9 and P55, which binds to PIK3R3 and subsequently inhibits the activation of the PI3K/AKT/mTOR pathway. This inhibition promotes autophagy, contributing to 5-FU resistance. Sorafenib, a vascular endothelial growth factor (VEGF) inhibitor, is a first-line agent for treating hepatocellular carcinoma, and its monotherapy has become the standard systemic therapy for advanced cases. Strong correlations have been observed between the levels of Tspan24 in hepatocellular carcinoma cells and resistance to sorafenib [118]. Tspan28-positive AML (Acute myeloid leukemia) cells are 30–50% more resistant to conventional chemotherapy, and Tspan29 has also been shown to regulate the development of leukemogenesis and resistance to chemotherapy. Tspan29-positive AML cells are more likely than Tspan29-negative AML cells are more resistant and migratory to Ara-C chemotherapy [118]. Tspan3 has been shown to be upregulated in both adriamycin-resistant AML samples and cell lines [118]. In the tumor microenvironment, Tspan29 is responsible for the crosstalk between BMMSCs (bone marrow mesenchymal stem cells) and breast cancer cells, which leads to chemoresistance [119]. Consistent with this, studies in small-cell lung cancer cells have shown that ectopic overexpression of Tspan29 enhances β1 integrin-mediated cell adhesion to extracellular matrix fibronectin, a fibronectin that is involved in cell adhesion-mediated resistance to drugs. Koch et al. found that when B-cell lymphomas isolate Dox (doxorubicin) isolated within Tspan29-positive exosomes and then exported it outside the cell, chemoresistance developed [119].
Several studies have demonstrated the potential of cancer treatment through the upregulation of Tspan proteins. Tspan27, in particular, has been identified as a metastasis suppressor that could be employed to halt breast cancer metastasis. However, this strategy is only applicable to breast cancer patients with low levels of Tspan27 [120]. The Tspan protein family, as a specific surface molecule present on exosomes, can selectively bind to specific cells, enabling exosomes to exhibit natural cell targeting abilities. Intravenous administration of exosomes loaded with paclitaxel demonstrates potent antitumor efficacy [121]. In recent years, exosomes have emerged as a highly effective drug delivery system characterized by low immunogenicity, high biocompatibility, and potent therapeutic capabilities. These vesicles enable the organic loading of nucleic acids, peptides, lipids, or small molecules with therapeutic functions, facilitating their targeted delivery to specific cell types or tissues in the body, particularly tumor tissues [122].
Numerous studies have acknowledged and elucidated the targeting properties of Tspan proteins. However, several challenges persist in achieving effective clinical treatment, and the research for clinical applications is still in its early stages. There is still a considerable journey ahead before biologics targeting Tspan proteins can be implemented in clinical practice.
Mechanism summary
Antibodies targeting specific Tspan proteins can inhibit their function and tumor growth. For example, anti-Tspan8 antibody has been shown to inhibit tumor growth in mouse models.
Certain natural products, such as Andrographis paniculata water (APW), can bind to Tspan proteins and suppress their activity. Overexpression of Tspan1 contributes to the acquired resistance of tumor cells to conventional chemotherapy. Silencing Tspan5 expression enhances the sensitivity of cancer cells to Decitabine (DCA) in breast cancer cells. Tspan8 promotes drug resistance in gastric cancer cells, but silencing its expression enhances their sensitivity to cisplatin, 5-fluorouracil (5-FU), and adriamycin. Tspan9 promotes 5-fluorouracil (5-FU) resistance in gastric cancer cells. Sorafenib resistance is associated with high levels of Tspan24 in hepatocellular carcinoma (HCC) cells. Tspan28-positive acute myeloid leukemia (AML) cells are more resistant to chemotherapy.Tspan29 is involved in the development of leukemogenesis and resistance to chemotherapy. Tspan proteins present on exosomes can selectively bind to specific cells, enabling exosomes to exhibit natural cell targeting abilities.
The role of the Tspan protein family in microRNA
MicroRNAs (miRNAs) and the Tspan protein family form a tightly knit alliance, with miRNAs playing crucial roles in regulating key pathways associated with tumorigenesis [58]. Several miRNAs have been identified as direct targets of Tspan genes or mRNAs, often leading to the downregulation of Tspan gene expression and influencing tumor progression. This highlights the potential of miRNAs as therapeutic targets for cancer treatment [123].
MiR454 and miR-573 affected the proliferation, autophagy, migration, and invasion of pancreatic cancer cells by targeting Tspan1 [10, 124]. In gastric cancer, overexpression of miR-573 similarly downregulated the expression of Tspan1, which inhibited the proliferation and invasion of cancer cells [41]. miR-361-3p can target Tspan1 and negatively regulate Tspan1 expression, and the overexpression of miR-361-3p inhibited cervical cancer cell survival, migration, and invasion [125]. In contrast, miR-570-3p has been found to inhibit the progression of aggressive AML by downregulating Tspan3. Moreover, CIRC_0004136 acts as a miR-570-3p sponge, regulating the expression of Tspan3 [126]. Additionally, miR-155 has been demonstrated to induce stemness and DCA resistance of breast cancer cells by inhibiting the direct target gene Tspan5 [116]. It was found that miR-324-5P reduced gastric cancer cell survival and induced apoptosis in vitro by downregulating the expression of Tspan8 in human gastric cancer, in addition, miR-324-5p overexpression significantly inhibited gastric cancer cell tumorigenesis in vivo, as evidenced by a smaller tumor volume than that of the control group [127]. Hsa-miR-9-5p has been found to be significantly upregulated in patients with hepatocellular carcinoma and is associated with poor prognosis. Additionally, hsa-miR-9-5p-mediated downregulation of Tspan9 expression is correlated with a poor prognosis of hepatocellular carcinoma, immunologically related signaling pathways, and immune infiltration of the tumor [72]. Hypomethylation of the promoter region of the miR-196b-5p gene in NSCLC promoted the expression of miR-196b-5p, resulting in a significant upregulation of miR-196b-5p expression in NSCLC tissues, and the upregulated miR-196b-5p promoted the migration of lung cancer cells by directly targeting the tumor suppressor genes GATA6 and Tspan12, proliferation; cell cycle and the expression of both GATA6 and Tspan12 was downregulated in NSCLC tissues and negatively correlated with that of miR-196b-5p [128]. Tspan13 is a direct target of miR-369-3p, and overexpression of miR-369-3p significantly inhibited the proliferation and promoted apoptosis of PTC (papillary thyroid carcinoma) cells, and silencing of Tspan13 inhibited the effect of miR-369-3p on PTC cells [21]. MiR-4732-5p can also directly target the 3′-UTR of Tspan13 and inhibit the expression of Tspan13 at both the mRNA and protein levels, it was also found that miR-4732-5p may function as a tumor suppressor in the initial stages of breast cancer, while plays a tumor-promoting role by targeting Tspan13 during breast cancer progression [129]. MiR-378a-3p inhibits the progression of GBM by decreasing the expression of Tspan17, promotes apoptosis, and reduces proliferation, migration, and invasion, and thus may be a potential target for the treatment of GBM [58]. MiR-124 plays an important role in inhibiting invasion and metastasis of breast cancer cells by directly targeting the 3′-UTR of Tspan24 mRNAs and inhibiting its mRNA expression and protein translation [130]. In gastric cancer, miR-197 targets the 3′-UTR of Tspan27 and thus plays an inhibitory role against Tspan27 [131]. HSA-miR-217 and its target gene sirtuin 1 act as metastasis suppressor and promoter genes, respectively, in NSCLC, and they form an hsa-miR-217/sirtuin1/p53/Tspan27 metastasis regulatory pathway that plays an important role in brain metastasis of NSCLC [123]. In H. pylori infection-associated gastritis and gastric cancer, Tspan29, MST1R, and ADAM10 are the most likely targets of miR-155-5p [132]. LSD1 (histone lysine-specific demethylase 1) expression was significantly higher in metastatic gastric cancer tissues than in normal tissues, and it was also found that LSD1 elevated intracellular miR-142-5p, leading to downregulation of the expression of Tspan29, a novel target of miR-142-5p, which in turn promotes gastric cancer migration [133]. MiR-518f-5p decreased the expression of Tspan29 in prostate and breast cancers and led to increased cell migration [134].
MicroRNAs can also serve as targets of Tspan genes or mRNAs. For instance, CircTspan24 targets miR-30d-5p to upregulate the expression of GLI2(Glioma cancer-related gene homologous protein 2) and promote the malignant proliferation of lung adenocarcinoma and promote the malignant proliferation of lung adenocarcinoma [135]. Increasing evidence suggests that there is aberrant expression of miR-203 in tumor stem cells, and it has been found that Tspan27 upregulates the expression of miR-203, which in turn regulates the expression of FZD2 [136]. The relationship between microRNAs and the Tspan protein family as well as their mechanism of action still requires more in-depth study, whether microRNAs are potential targets for cancer therapy, and how to translate this knowledge into clinical applications remains an open question.
Conclusions and future directions
Despite the growing number of studies revealing the importance of the Tspan protein family in tumor initiation and progression, the intricate nature of their biological functions necessitates further investigation into their specific molecular mechanisms and regulatory diversity. The Tspan protein family exerts a critical role in cancer by modulating the expression of oncogenes, influencing signaling pathways relevant to cancer development and progression, and regulating miRNA generation and signaling pathways. Moreover, preliminary data yield inconsistent findings regarding the role of Tspan proteins in either promoting or inhibiting tumorigenesis and progression. These discrepancies could potentially stem from variations in the composition of TEMs and the interacting protein chaperones.
Internalization of the Tspan protein family into intracellular vesicles and intercellular signaling via exosomal/extracellular vesicles represents a significant regulatory mechanism that can either impede or promote tumor metastasis. Tspan proteins, abundant in the membrane of exosomal/extracellular vesicles, not only engage in the signaling process by interacting with receptor proteins on the surface of recipient cells but also determine the sorting and selection of exosomal/extracellular vesicles upon detachment from the cell membrane. Both extracellular vesicles and intracellular multivesicular body vesicles encapsulate signaling molecules (proteins, microRNAs) during the sorting and selection process. With the increasing research on this family, the emergence of drug-targeted therapies against Tspan proteins has provided new opportunities for targeted cancer therapy. However, several challenges must be addressed. Firstly, as integral components of multiprotein complexes, Tspan proteins are influenced by various interacting elements and exhibit context-dependent functional roles. Secondly, the structural similarity among family members poses challenges in targeted therapy. These factors collectively impact the application of Tspan proteins as molecular markers and therapeutic targets. In the future, extensive and systematic investigations are warranted to elucidate the involvement of the Tspan protein family in cancer and its associated signaling pathways, establish interaction networks, uncover their precise mechanisms of action in cancer, and foster innovative approaches for targeted cancer therapy. Furthermore, efforts should be directed towards identifying natural bioactive compounds capable of replacing small molecule inhibitors to mitigate toxic side effects on normal cells. Simultaneously, microRNAs targeting the Tspan protein family offer a novel avenue for targeted cancer therapy, especially in conjunction with radiation therapy, chemotherapy, and other treatment modalities.
While current studies have only scratched the surface of Tspan’s involvement in tumorigenesis and progression, an increasing body of data is progressively unraveling its enigmatic nature. As our comprehension of Tspan’s functionality expands, it will pave the way for novel diagnostic and therapeutic approaches to precisely target interventions in the future.
Data availability
The authors confirm that the data supporting the findings of this study are available within the article.
References
Oren R, Takahashi S, Doss C, Levy R, Levy S. TAPA-1, the target of an antiproliferative antibody, defines a new family of transmembrane proteins. Mol Cell Biol. 1990;10:4007–15.
Yaseen IH, Monk PN, Partridge LJ. Tspan2: a tetraspanin protein involved in oligodendrogenesis and cancer metastasis. Biochem Soc Trans. 2017;45:465–75.
Zhou Z, Yang Z, Zhou L, Yang M, He S. The versatile roles of testrapanins in cancer from intracellular signaling to cell-cell communication: cell membrane proteins without ligands. Cell Biosci. 2023;13:59.
García-Frigola C, Burgaya F, de Lecea L, Soriano E. Pattern of expression of the tetraspanin Tspan-5 during brain development in the mouse. Mech Dev. 2001;106:207–12.
Perot BP, Ménager MM. Tetraspanin 7 and its closest paralog tetraspanin 6: membrane organizers with key functions in brain development, viral infection, innate immunity, diabetes and cancer. Med Microbiol Immunol. 2020;209:427–36.
Dickerson MT, Dadi PK, Butterworth RB, Nakhe AY, Graff SM, Zaborska KE, et al. Tetraspanin-7 regulation of L-type voltage-dependent calcium channels controls pancreatic β-cell insulin secretion. J Physiol. 2020;598:4887–905.
Xu-Monette ZY, Li L, Byrd JC, Jabbar KJ, Manyam GC, Maria de Winde C, et al. Assessment of CD37 B-cell antigen and cell of origin significantly improves risk prediction in diffuse large B-cell lymphoma. Blood. 2016;128:3083–100.
Huang R, Sun H, Lin R, Zhang J, Yin H, Xian S, et al. The role of tetraspanins pan-cancer. iScience. 2022;25:104777.
Huang Y, Zucker B, Zhang S, Elias S, Zhu Y, Chen H, et al. Migrasome formation is mediated by assembly of micron-scale tetraspanin macrodomains. Nat Cell Biol. 2019;21:991–1002.
Zhou C, Liang Y, Zhou L, Yan Y, Liu N, Zhang R, et al. TSPAN1 promotes autophagy flux and mediates cooperation between WNT-CTNNB1 signaling and autophagy via the MIR454-FAM83A-TSPAN1 axis in pancreatic cancer. Autophagy. 2021;17:3175–95.
Zhang HS, Liu HY, Zhou Z, Sun HL, Liu MY. TSPAN8 promotes colorectal cancer cell growth and migration in LSD1-dependent manner. Life Sci. 2020;241:117114.
Cai S, Deng Y, Peng H, Shen J. Role of tetraspanins in hepatocellular carcinoma. Front Oncol. 2021;11:723341.
Qiu Q, Sun Y, Yang L, Li Q, Feng Y, Li M, et al. TSPAN32 suppresses chronic myeloid leukemia pathogenesis and progression by stabilizing PTEN. Signal Transduct Target Ther. 2023;8:90.
Becic A, Leifeld J, Shaukat J, Hollmann M. Tetraspanins as potential modulators of glutamatergic synaptic function. Front Mol Neurosci. 2021;14:801882.
Todd SC, Doctor VS, Levy S. Sequences and expression of six new members of the tetraspanin/TM4SF family. Biochim Biophys Acta. 1998;1399:101–4.
Li H, Onbe K, Liu Q, Iijima M, Tatematsu K, Seno M, et al. Synthesis and assembly of hepatitis B virus envelope protein-derived particles in Escherichia coli. Biochem Biophys Res Commun. 2017;490:155–60.
Florin L, Lang T. Tetraspanin assemblies in virus infection. Front Immunol. 2018;9:1140.
Saint-Pol J, Eschenbrenner E, Dornier E, Boucheix C, Charrin S, Rubinstein E. Regulation of the trafficking and the function of the metalloprotease ADAM10 by tetraspanins. Biochem Soc Trans. 2017;45:937–44.
Chen L, Yuan D, Wang GL, Wang Y, Wu YY, Zhu J. Clinicopathological significance of expression of Tspan-1, Jab1 and p27 in human hepatocellular carcinoma. J Korean Med Sci. 2010;25:1438–42.
Zhang X, Shi G, Gao F, Liu P, Wang H, Tan X. TSPAN1 upregulates MMP2 to promote pancreatic cancer cell migration and invasion via PLCγ. Oncol Rep. 2019;41:2117–25.
Li P, Dong M, Wang Z. Downregulation of TSPAN13 by miR-369-3p inhibits cell proliferation in papillary thyroid cancer (PTC). Bosn J Basic Med Sci. 2019;19:146–54.
Veenbergen S, van Spriel AB. Tetraspanins in the immune response against cancer. Immunol Lett. 2011;138:129–36.
Dimitrijevic I, Axelsson L, Andersson T. The adhesion receptor CD-31 can be primed to rapidly adjust the neutrophil cytoskeleton. Biochem Biophys Res Commun. 2002;292:1092–7.
Montpellier C, Tews BA, Poitrimole J, Rocha-Perugini V, D’Arienzo V, Potel J, et al. Interacting regions of CD81 and two of its partners, EWI-2 and EWI-2wint, and their effect on hepatitis C virus infection. J Biol Chem. 2011;286:13954–65.
DeBerardinis RJ, Lum JJ, Hatzivassiliou G, Thompson CB. The biology of cancer: metabolic reprogramming fuels cell growth and proliferation. Cell Metab. 2008;7:11–20.
Chen G, Zhang H, Sun H, Ding X, Liu G, Yang F, et al. Bufalin targeting BFAR inhibits the occurrence and metastasis of gastric cancer through PI3K/AKT/mTOR signal pathway. Apoptosis. 2023;28:1390–405.
Wang Z, Wei D, Xiao H. Methods of cellular senescence induction using oxidative stress. Methods Mol Biol. 2013;1048:135–44.
Wang Y, Liang Y, Yang G, Lan Y, Han J, Wang J, et al. Tetraspanin 1 promotes epithelial-to-mesenchymal transition and metastasis of cholangiocarcinoma via PI3K/AKT signaling. J Exp Clin Cancer Res. 2018;37:300.
Wu Y, Chen W, Gong Y, Liu H, Zhang B. Tetraspanin 1 (TSPAN1) promotes growth and transferation of breast cancer cells via mediating PI3K/Akt pathway. Bioengineered. 2021;12:10761–70.
Sidahmed-Adrar N, Ottavi JF, Benzoubir N, Ait Saadi T, Bou Saleh M, Mauduit P, et al. Tspan15 is a new stemness-related marker in hepatocellular carcinoma. Proteomics. 2019;19:e1900025.
Todeschini AR, Dos Santos JN, Handa K, Hakomori SI. Ganglioside GM2/GM3 complex affixed on silica nanospheres strongly inhibits cell motility through CD82/cMet-mediated pathway. Proc Natl Acad Sci USA. 2008;105:1925–30.
Wang J, Zhou Y, Li D, Sun X, Deng Y, Zhao Q. TSPAN31 is a critical regulator on transduction of survival and apoptotic signals in hepatocellular carcinoma cells. FEBS Lett. 2017;591:2905–18.
Yu X, Li S, Pang M, Du Y, Xu T, Bai T, et al. TSPAN7 exerts anti-tumor effects in bladder cancer through the PTEN/PI3K/AKT pathway. Front Oncol. 2020;10:613869.
Chen S, Sun Y, Jin Z, Jing X. Functional and biochemical studies of CD9 in fibrosarcoma cell line. Mol Cell Biochem. 2011;350:89–99.
Furuya M, Kato H, Nishimura N, Ishiwata I, Ikeda H, Ito R, et al. Down-regulation of CD9 in human ovarian carcinoma cell might contribute to peritoneal dissemination: morphologic alteration and reduced expression of beta1 integrin subsets. Cancer Res. 2005;65:2617–25.
Sikora E, Bielak-Zmijewska A, Mosieniak G. A common signature of cellular senescence; does it exist? Ageing Res Rev. 2021;71:101458.
Hume S, Dianov GL, Ramadan K. A unified model for the G1/S cell cycle transition. Nucleic Acids Res. 2020;48:12483–501.
Stillman DJ. Dancing the cell cycle two-step: regulation of yeast G1-cell-cycle genes by chromatin structure. Trends Biochem Sci. 2013;38:467–75.
Barnaba N, LaRocque JR. Targeting cell cycle regulation via the G2-M checkpoint for synthetic lethality in melanoma. Cell Cycle. 2021;20:1041–51.
Chen L, Yuan D, Zhao R, Li H, Zhu J. Suppression of TSPAN1 by RNA interference inhibits proliferation and invasion of colon cancer cells in vitro. Tumori. 2010;96:744–50.
Lu Z, Luo T, Nie M, Pang T, Zhang X, Shen X, et al. TSPAN1 functions as an oncogene in gastric cancer and is downregulated by miR-573. FEBS Lett. 2015;589:1988–94.
Li PY, Lv J, Qi WW, Zhao SF, Sun LB, Liu N, et al. Tspan9 inhibits the proliferation, migration and invasion of human gastric cancer SGC7901 cells via the ERK1/2 pathway. Oncol Rep. 2016;36:448–54.
Ji G, Liang H, Wang F, Wang N, Fu S, Cui X. TSPAN12 precedes tumor proliferation by cell cycle control in ovarian cancer. Mol Cells. 2019;42:557–67.
Xia Y, Deng Y, Zhou Y, Li D, Sun X, Gu L, et al. TSPAN31 suppresses cell proliferation in human cervical cancer through down-regulation of its antisense pairing with CDK4. Cell Biochem Funct. 2020;38:660–8.
Githaka JM, Pirayeshfard L, Goping IS. Cancer invasion and metastasis: Insights from murine pubertal mammary gland morphogenesis. Biochim Biophys Acta Gen Subj. 2023;1867:130375.
Zhang H, Zhang Z, Guo T, Chen G, Liu G, Song Q, et al. Annexin A protein family: Focusing on the occurrence, progression and treatment of cancer. Front Cell Dev Biol. 2023;11:1141331.
Otsubo C, Otomo R, Miyazaki M, Matsushima-Hibiya Y, Kohno T, Iwakawa R, et al. TSPAN2 is involved in cell invasion and motility during lung cancer progression. Cell Rep. 2014;7:527–38.
Zhu R, Gires O, Zhu L, Liu J, Li J, Yang H, et al. TSPAN8 promotes cancer cell stemness via activation of sonic hedgehog signaling. Nat Commun. 2019;10:2863.
Hasegawa M, Furuya M, Kasuya Y, Nishiyama M, Sugiura T, Nikaido T, et al. CD151 dynamics in carcinoma-stroma interaction: integrin expression, adhesion strength and proteolytic activity. Lab Investig. 2007;87:882–92.
Zhang XA, Lane WS, Charrin S, Rubinstein E, Liu L. EWI2/PGRL associates with the metastasis suppressor KAI1/CD82 and inhibits the migration of prostate cancer cells. Cancer Res. 2003;63:2665–74.
Takahashi M, Sugiura T, Abe M, Ishii K, Shirasuna K. Regulation of c-Met signaling by the tetraspanin KAI-1/CD82 affects cancer cell migration. Int J Cancer. 2007;121:1919–29.
Yuan H, Zhao J, Yang Y, Wei R, Zhu L, Wang J, et al. SHP-2 interacts with CD81 and regulates the malignant evolution of colorectal cancer by inhibiting epithelial-mesenchymal transition. Cancer Manag Res. 2020;12:13273–84.
Hong IK, Byun HJ, Lee J, Jin YJ, Wang SJ, Jeoung DI, et al. The tetraspanin CD81 protein increases melanoma cell motility by up-regulating metalloproteinase MT1-MMP expression through the pro-oncogenic Akt-dependent Sp1 activation signaling pathways. J Biol Chem. 2014;289:15691–704.
Vences-Catalán F, Rajapaksa R, Srivastava MK, Marabelle A, Kuo CC, Levy R, et al. Tetraspanin CD81 promotes tumor growth and metastasis by modulating the functions of T regulatory and myeloid-derived suppressor cells. Cancer Res. 2015;75:4517–26.
Pols MS, Klumperman J. Trafficking and function of the tetraspanin CD63. Exp Cell Res. 2009;315:1584–92.
Kashyap D, Garg VK, Goel N. Intrinsic and extrinsic pathways of apoptosis: Role in cancer development and prognosis. Adv Protein Chem Struct Biol. 2021;125:73–120.
Knoblich K, Wang HX, Sharma C, Fletcher AL, Turley SJ, Hemler ME. Tetraspanin TSPAN12 regulates tumor growth and metastasis and inhibits β-catenin degradation. Cell Mol Life Sci. 2014;71:1305–14.
Guo XB, Zhang XC, Chen P, Ma LM, Shen ZQ. miR‑378a‑3p inhibits cellular proliferation and migration in glioblastoma multiforme by targeting tetraspanin 17. Oncol Rep. 2019;42:1957–71.
Takashima Y, Komatsu S, Ohashi T, Kiuchi J, Kamiya H, Shimizu H, et al. Overexpression of Tetraspanin31 contributes to malignant potential and poor outcomes in gastric cancer. Cancer Sci. 2022;113:1984–98.
Ma X, Qiu S, Tang X, Song Q, Wang P, Wang J, et al. TSPAN31 regulates the proliferation, migration, and apoptosis of gastric cancer cells through the METTL1/CCT2 pathway. Transl Oncol. 2022;20:101423.
Sorice M. Crosstalk of autophagy and apoptosis. Cells. 2022;11:1479.
Rakesh R, PriyaDharshini LC, Sakthivel KM, Rasmi RR. Role and regulation of autophagy in cancer. Biochim Biophys Acta Mol Basis Dis. 2022;1868:166400.
Qi Y, Qi W, Liu S, Sun L, Ding A, Yu G, et al. TSPAN9 suppresses the chemosensitivity of gastric cancer to 5-fluorouracil by promoting autophagy. Cancer Cell Int. 2020;20:4.
Miyazaki T, Müller U, Campbell KS. Normal development but differentially altered proliferative responses of lymphocytes in mice lacking CD81. EMBO J. 1997;16:4217–25.
Rubinstein E, Le Naour F, Lagaudrière-Gesbert C, Billard M, Conjeaud H, Bouchei C. CD63, CD81, and CD82 are components of a surface tetraspan network connected to HLA-DR and VLA integrins. Eur J Immunol. 1996;26:2657–65.
Petersen SH, Odintsova E, Haigh TA, Rickinson AB, Taylor GS, Berditchevski F. The role of tetraspanin CD63 in antigen presentation via MHC class II. Eur J Immunol. 2011;41:2556–61.
de Winde CM, Munday C, Acton SE. Molecular mechanisms of dendritic cell migration in immunity and cancer. Med Microbiol Immunol. 2020;209:515–29.
Jones EL, Wee JL, Demaria MC, Blakeley J, Ho PK, Vega-Ramos J, et al. Dendritic cell migration and antigen presentation are coordinated by the opposing functions of the tetraspanins CD82 and CD37. J Immunol. 2016;196:978–87.
Quast T, Eppler F, Semmling V, Schild C, Homsi Y, Levy S, et al. CD81 is essential for the formation of membrane protrusions and regulates Rac1-activation in adhesion-dependent immune cell migration. Blood. 2011;118:1818–27.
Zelman-Toister E, Bakos E, Cohen S, Zigmond E, Shezen E, Grabovsky V, et al. CD151 regulates T-cell migration in health and inflammatory bowel disease. Inflamm Bowel Dis. 2016;22:257–67.
Sawada J, Hiraoka N, Qi R, Jiang L, Fournier-Goss AE, Yoshida M, et al. Molecular signature of tumor-associated high endothelial venules that can predict breast cancer survival. Cancer Immunol Res. 2022;10:468–81.
Tan S, Song X, Zhang C, Sun Y, Zhang J, Zhang Z, et al. hsa-miR-9-5p-mediated TSPAN9 downregulation is positively related to both poor hepatocellular carcinoma prognosis and the tumor immune infiltration. J Immunol Res. 2022;2022:9051229.
Uddin MN, Wang X. Identification of key tumor stroma-associated transcriptional signatures correlated with survival prognosis and tumor progression in breast cancer. Breast Cancer. 2022;29:541–61.
Malla R, Marni R, Chakraborty A. Exploring the role of CD151 in the tumor immune microenvironment: therapeutic and clinical perspectives. Biochim Biophys Acta Rev Cancer. 2023;1878:188898.
Lin W, Liu J, Chen J, Li J, Qiu S, Ma J, et al. Peptides of tetraspanin oncoprotein CD151 trigger active immunity against primary tumour and experimental lung metastasis. EBioMedicine. 2019;49:133–44.
Bilemjian V, Vlaming MR, Álvarez Freile J, Huls G, De Bruyn M, Bremer E. The novel immune checkpoint GPR56 is expressed on tumor-infiltrating lymphocytes and selectively upregulated upon TCR signaling. Cancers. 2022;14:3164.
Khan NS, Lukason DP, Feliu M, Ward RA, Lord AK, Reedy JL, et al. CD82 controls CpG-dependent TLR9 signaling. FASEB J. 2019;33:12500–14.
Lu Z, Pang T, Yin X, Cui H, Fang G, Xue X, et al. Delivery of TSPAN1 siRNA by novel Th17 targeted cationic liposomes for gastric cancer intervention. J Pharm Sci. 2020;109:2854–60.
Dunlock VE, Arp AB, Singh SP, Charrin S, Nguyen V, Jansen E, et al. Tetraspanin CD53 controls T cell immunity through regulation of CD45RO stability, mobility, and function. Cell Rep. 2022;39:111006.
Schaper F, van Spriel AB. Antitumor immunity is controlled by tetraspanin proteins. Front Immunol. 2018;9:1185.
Gao X, Ran N, Dong X, Zuo B, Yang R, Zhou Q, et al. Anchor peptide captures, targets, and loads exosomes of diverse origins for diagnostics and therapy. Sci Transl Med. 2018;10:eaat0195.
Gurung S, Perocheau D, Touramanidou L, Baruteau J. The exosome journey: from biogenesis to uptake and intracellular signalling. Cell Commun Signal. 2021;19:47.
Wang T, Wang X, Wang H, Li L, Zhang C, Xiang R, et al. High TSPAN8 expression in epithelial cancer cell-derived small extracellular vesicles promote confined diffusion and pronounced uptake. J Extracell Vesicles. 2021;10:e12167.
Salunkhe S, Dheeraj, Basak M, Chitkara D, Mittal A. Surface functionalization of exosomes for target-specific delivery and in vivo imaging & tracking: strategies and significance. J Control Release. 2020;326:599–614.
Brosseau C, Colas L, Magnan A, Brouard S. CD9 tetraspanin: a new pathway for the regulation of inflammation? Front Immunol. 2018;9:2316.
Martens-Uzunova ES, Kusuma GD, Crucitta S, Lim HK, Cooper C, Riches JE, et al. Androgens alter the heterogeneity of small extracellular vesicles and the small RNA cargo in prostate cancer. J Extracell Vesicles. 2021;10:e12136.
Larios J, Mercier V, Roux A, Gruenberg J. ALIX- and ESCRT-III-dependent sorting of tetraspanins to exosomes. J Cell Biol. 2020;219:e201904113.
Rana S, Zöller M. Exosome target cell selection and the importance of exosomal tetraspanins: a hypothesis. Biochem Soc Trans. 2011;39:559–62.
Voglstaetter M, Thomsen AR, Nouvel J, Koch A, Jank P, Navarro EG, et al. Tspan8 is expressed in breast cancer and regulates E-cadherin/catenin signalling and metastasis accompanied by increased circulating extracellular vesicles. J Pathol. 2019;248:421–37.
Karimi N, Dalirfardouei R, Dias T, Lötvall J, Lässer C. Tetraspanins distinguish separate extracellular vesicle subpopulations in human serum and plasma - contributions of platelet extracellular vesicles in plasma samples. J Extracell Vesicles. 2022;11:e12213.
Ter-Ovanesyan D, Norman M, Lazarovits R, Trieu W, Lee JH, Church GM, et al. Framework for rapid comparison of extracellular vesicle isolation methods. eLife. 2021;10:e70725.
Dash S, Wu CC, Wu CC, Chiang SF, Lu YT, Yeh CY, et al. Extracellular vesicle membrane protein profiling and targeted mass spectrometry unveil CD59 and tetraspanin 9 as novel plasma biomarkers for detection of colorectal cancer. Cancers. 2022;15:177.
Hoshino A, Kim HS, Bojmar L, Gyan KE, Cioffi M, Hernandez J, et al. Extracellular vesicle and particle biomarkers define multiple human cancers. Cell. 2020;182:1044–1061.e18.
Rodia MT, Solmi R, Pasini F, Nardi E, Mattei G, Ugolini G, et al. LGALS4, CEACAM6, TSPAN8, and COL1A2: blood markers for colorectal cancer-validation in a cohort of subjects with positive fecal immunochemical test result. Clin Colorectal Cancer. 2018;17:e217–e228.
Jafari R, Rahbarghazi R, Ahmadi M, Hassanpour M, Rezaie J. Hypoxic exosomes orchestrate tumorigenesis: molecular mechanisms and therapeutic implications. J Transl Med. 2020;18:474.
Ma C, Cui Z, Wang Y, Zhang L, Wen J, Guo H, et al. Bioinformatics analysis reveals TSPAN1 as a candidate biomarker of progression and prognosis in pancreatic cancer. Bosn J Basic Med Sci. 2021;21:47–60.
Yunusova N, Dzhugashvili E, Yalovaya A, Kolomiets L, Shefer A, Grigor’eva A, et al. Comparative analysis of tumor-associated microRNAs and tetraspanines from exosomes of plasma and ascitic fluids of ovarian cancer patients. Int J Mol Sci. 2022;24:464.
Jiang L, Zhang X, Geradts J, Wei Q, Hochwald S, Xu H, et al. Expression of tetraspanins NET-6 and CD151 in breast cancer as a potential tumor biomarker. Clin Exp Med. 2019;19:377–84.
Detchokul S, Newell B, Williams ED, Frauman AG. CD151 is associated with prostate cancer cell invasion and lymphangiogenesis in vivo. Oncol Rep. 2014;31:241–7.
Guo JN, Chen D, Deng SH, Huang JR, Song JX, Li XY, et al. Identification and quantification of immune infiltration landscape on therapy and prognosis in left- and right-sided colon cancer. Cancer Immunol Immunother. 2022;71:1313–30.
Ye H, Li T, Wang H, Wu J, Yi C, Shi J, et al. TSPAN1, TMPRSS4, SDR16C5, and CTSE as novel panel for pancreatic cancer: a bioinformatics analysis and experiments validation. Front Immunol. 2021;12:649551.
Liu S, Cai Y, Changyong E, Sheng J, Zhang X. Screening and validation of independent predictors of poor survival in pancreatic cancer. Pathol Oncol Res. 2021;27:1609868.
Li J, Chen X, Zhu L, Lao Z, Zhou T, Zang L, et al. SOX9 is a critical regulator of TSPAN8-mediated metastasis in pancreatic cancer. Oncogene. 2021;40:4884–93.
Wei CY, Zhu MX, Lu NH, Liu JQ, Yang YW, Zhang Y, et al. Circular RNA circ_0020710 drives tumor progression and immune evasion by regulating the miR-370-3p/CXCL12 axis in melanoma. Mol Cancer. 2020;19:84.
Al-Khater KM, Almofty S, Ravinayagam V, Alrushaid N, Rehman S. Role of a metastatic suppressor gene KAI1/CD82 in the diagnosis and prognosis of breast cancer. Saudi J Biol Sci. 2021;28:3391–8.
Lai X, Chen S. Identification of novel biomarker and therapeutic target candidates for diagnosis and treatment of follicular adenoma. Cancer Genom Proteom. 2015;12:271–81.
Kumar D, Gupta D, Shankar S, Srivastava RK. Biomolecular characterization of exosomes released from cancer stem cells: possible implications for biomarker and treatment of cancer. Oncotarget. 2015;6:3280–91.
Gao Y, Li X, Zeng C, Liu C, Hao Q, Li W, et al. CD63(+) cancer-associated fibroblasts confer tamoxifen resistance to breast cancer cells through exosomal miR-22. Adv Sci. 2020;7:2002518.
Titu S, Grapa CM, Mocan T, Balacescu O, Irimie A. Tetraspanins: physiology, colorectal cancer development, and nanomediated applications. Cancers. 2021;13:5662.
Yue GG, Gomes AJ, Saeed MEM, Tsui KY, Dawood M, Drif AI, et al. Identification of active components in Andrographis paniculata targeting on CD81 in esophageal cancer in vitro and in vivo. Phytomedicine. 2022;102:154183.
Bonnet M, Maisonial-Besset A, Zhu Y, Witkowski T, Roche G, Boucheix C, et al. Targeting the tetraspanins with monoclonal antibodies in oncology: focus on Tspan8/Co-029. Cancers. 2019;11:179.
Heo K, Lee S. TSPAN8 as a novel emerging therapeutic target in cancer for monoclonal antibody therapy. Biomolecules. 2020;10:388.
Oostindie SC, van der Horst HJ, Kil LP, Strumane K, Overdijk MB, van den Brink EN, et al. DuoHexaBody-CD37(®), a novel biparatopic CD37 antibody with enhanced Fc-mediated hexamerization as a potential therapy for B-cell malignancies. Blood Cancer J. 2020;10:30.
Vences-Catalán F, Rajapaksa R, Kuo CC, Miller CL, Lee A, Ramani VC, et al. Targeting the tetraspanin CD81 reduces cancer invasion and metastasis. Proc Natl Acad Sci USA. 2021;118:e2018961118.
Garcia-Mayea Y, Mir C, Carballo L, Sánchez-García A, Bataller M, ME LL. TSPAN1, a novel tetraspanin member highly involved in carcinogenesis and chemoresistance. Biochim Biophys Acta Rev Cancer. 2022;1877:188674.
Yang LW, Wu XJ, Liang Y, Ye GQ, Che YC, Wu XZ, et al. miR-155 increases stemness and decitabine resistance in triple-negative breast cancer cells by inhibiting TSPAN5. Mol Carcinog. 2020;59:447–61.
Deng Y, Cai S, Shen J, Peng H. Tetraspanins: novel molecular regulators of gastric cancer. Front Oncol. 2021;11:702510.
Floren M, Gillette JM. Acute myeloid leukemia: therapy resistance and a potential role for tetraspanin membrane scaffolds. Int J Biochem Cell Biol. 2021;137:106029.
Ullah M, Akbar A, Ng NN, Concepcion W, Thakor AS. Mesenchymal stem cells confer chemoresistance in breast cancer via a CD9 dependent mechanism. Oncotarget. 2019;10:3435–50.
Viera M, Yip GWC, Shen HM, Baeg GH, Bay BH. Targeting CD82/KAI1 for precision therapeutics in surmounting metastatic potential in breast cancer. Cancers. 2021;13:4486.
Melzer C, Ohe JV, Hass R. Anti-tumor effects of exosomes derived from drug-incubated permanently growing human MSC. Int J Mol Sci. 2020;21:7311.
He J, Ren W, Wang W, Han W, Jiang L, Zhang D, et al. Exosomal targeting and its potential clinical application. Drug Deliv Transl Res. 2022;12:2385–402.
Jiang W, Hou L, Wei J, Du Y, Zhao Y, Deng X, et al. Hsa-miR-217 inhibits the proliferation, migration, and invasion in non-small cell lung cancer cells via targeting SIRT1 and P53/KAI1 signaling. Balk Med J. 2020;37:208–14.
Wang L, Gao P, Yuan P, Zhou P, Fan H, Lin X, et al. miR-573 suppresses pancreatic cancer cell proliferation, migration, and invasion through targeting TSPAN1. Strahlenther Onkol. 2021;197:438–48.
Li C, Li Y, Zhang Y, Yan H, Huang C. Knockdown of LINC01123 inhibits cell viability, migration and invasion via miR-361-3p/TSPAN1 targeting in cervical cancer. Exp Ther Med. 2021;22:1184.
Bi J, Pu Y, Yu X. Exosomal circ_0004136 enhances the progression of pediatric acute myeloid leukemia depending on the regulation of miR-570-3p/TSPAN3 axis. Anticancer Drugs. 2021;32:802–11.
Cui J, Smolinski SE, Liu F, Xu D, Dulaimy K, Irani Z. Incrementally expandable transjugular intrahepatic portosystemic shunts: single-center experience. Am J Roentgenol. 2018;210:438–46.
Liang G, Meng W, Huang X, Zhu W, Yin C, Wang C, et al. miR-196b-5p-mediated downregulation of TSPAN12 and GATA6 promotes tumor progression in non-small cell lung cancer. Proc Natl Acad Sci USA. 2020;117:4347–57.
Wang YW, Zhao S, Yuan XY, Liu Y, Zhang K, Wang J, et al. miR-4732-5p promotes breast cancer progression by targeting TSPAN13. J Cell Mol Med. 2019;23:2549–57.
Han ZB, Yang Z, Chi Y, Zhang L, Wang Y, Ji Y, et al. MicroRNA-124 suppresses breast cancer cell growth and motility by targeting CD151. Cell Physiol Biochem. 2013;31:823–32.
Xu L, Hou Y, Tu G, Chen Y, Du YE, Zhang H, et al. Nuclear Drosha enhances cell invasion via an EGFR-ERK1/2-MMP7 signaling pathway induced by dysregulated miRNA-622/197 and their targets LAMC2 and CD82 in gastric cancer. Cell Death Dis. 2017;8:e2642.
Mahbobi R, Fallah F, Behmanesh A, Yadegar A, Hakemi-Vala M, Ehsanzadeh SJ, et al. Helicobacter pylori infection mediates inflammation and tumorigenesis-associated genes through miR-155-5p: an integrative omics and bioinformatics-based investigation. Curr Microbiol. 2022;79:192.
Zhao LJ, Fan QQ, Li YY, Ren HM, Zhang T, Liu S, et al. LSD1 deletion represses gastric cancer migration by upregulating a novel miR-142-5p target protein CD9. Pharmacol Res. 2020;159:104991.
Bond DR, Kahl R, Brzozowski JS, Jankowski H, Naudin C, Pariyar M, et al. Tetraspanin CD9 is regulated by miR-518f-5p and functions in breast cell migration and in vivo tumor growth. Cancers. 2020;12:795.
Zhao L, Zheng H, Jiang P. circCD151 promotes GLI2 expression by regulating miR‑30d‑5p and enhancing proliferation, invasion and stemness of lung cancer. Mol Med Rep. 2021;24:699.
Mine M, Yamaguchi K, Sugiura T, Chigita S, Yoshihama N, Yoshihama R, et al. miR-203 inhibits frizzled-2 expression via CD82/KAI1 expression in human lung carcinoma cells. PLoS ONE. 2015;10:e0131350.
Lin H, Zhou AJ, Zhang JY, Liu SF, Gu JX. MiR-324-5p reduces viability and induces apoptosis in gastric cancer cells through modulating TSPAN8. J Pharm Pharmacol. 2018;70:1513–20.
Acknowledgements
Figures 2–4 were drawn by Figdraw (www.figdraw.com).
Funding
This work was supported by the National Natural Science Foundation of China (No.81803895), Shandong Province Natural Science Foundation (ZR2021YQ57), Medical and Health Science and Technology Development Program of Shandong Province(2019WS123), and China Postdoctoral Science Foundation (No.2020M682131, No.2021T140357).
Author information
Authors and Affiliations
Contributions
Huhu Zhang, Qinghang Song, and Kaiwen Shang wrote the article and drew the pictures. Ya Li and Liangqian Jiang draw the tables. Lina Yang supervised and corrected the writing process. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zhang, H., Song, Q., Shang, K. et al. Tspan protein family: focusing on the occurrence, progression, and treatment of cancer. Cell Death Discov. 10, 187 (2024). https://doi.org/10.1038/s41420-024-01961-0
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41420-024-01961-0
- Springer Nature Limited