Abstract
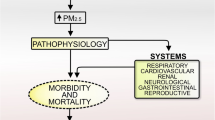
The harmful effects of fine particulate matter ≤2.5 µm in size (PM2.5) on human health have received considerable attention. However, while the impact of PM2.5 on the respiratory and cardiovascular systems has been well studied, less is known about the effects on stem cells in the bone marrow (BM). With an emphasis on the invasive characteristics of PM2.5, this review examines the current knowledge of the health effects of PM2.5 exposure on BM-residing stem cells. Recent studies have shown that PM2.5 enters the circulation and then travels to distant organs, including the BM, to induce oxidative stress, systemic inflammation and epigenetic changes, resulting in the reduction of BM-residing stem cell survival and function. Understanding the broader health effects of air pollution thus requires an understanding of the invasive characteristics of PM2.5 and its direct influence on stem cells in the BM. As noted in this review, further studies are needed to elucidate the underlying processes by which PM2.5 disturbs the BM microenvironment and inhibits stem cell functionality. Strategies to prevent or ameliorate the negative effects of PM2.5 exposure on BM-residing stem cells and to maintain the regenerative capacity of those cells must also be investigated. By focusing on the complex relationship between PM2.5 and BM-resident stem cells, this review highlights the importance of specific measures directed at safeguarding human health in the face of rising air pollution.
Similar content being viewed by others
Introduction
Particulate matter (PM) is a complex mixture of solid and liquid microscopic particles that enter the atmosphere as a result of natural environmental processes or human activities1. PM with aerodynamic diameters ≤10 µm cannot be filtered by the cilia and mucus of the nose and human respiratory tract, and instead infiltrate tracheobronchial and alveolar tissues, eventually entering the circulatory system and causing illness2. Fine particulate matter ≤2.5 µm in size (PM2.5) is especially harmful to human health3.
PM2.5 is generated from both natural environmental and human sources. The former include dust, soil particles, pollen, and sea salt aerosols, while the latter are primarily the products of combustion processes, such as fossil fuel combustion in power plants, industrial emissions, residential heating and cooking, and vehicle exhaust emissions4. The annual average concentration of PM2.5 measured in the Seoul Metropolitan area from November 2005 to March 2012 was 27 µg/m3, 5, and it was 25 µg/m3 in 20176, which is nearly three times the WHO standard. PM10-2.5 is less of a concern, although these particles can irritate the eyes, nose, and throat. The substantial health risk of PM2.5 is related to its ability to enter the circulation and infiltrate the lungs. PM deposition in the lungs triggers airway inflammation, compromising normal immune responses and rendering this vital organ vulnerable to infection7. Damage to the bronchial mucociliary system induced by PM2.5 can impede the effective clearance of bacteria8. Other adverse effects of PM2.5 include increased inflammatory cytokine release, which triggers lung fibroblast and epithelial cell death, interference with gap junctions and thus intercellular communication, and increased permeability of the epithelial barrier, impairing its function as a physical constraint and its role in innate pulmonary immunity9. Following the inhalation of PM2.5, the particles pass through the epithelial barrier and successively cross the basement membrane, subepithelial connective tissue layer, and endothelial cells to finally enter the bloodstream10,11. Ambient particles are toxic to cytochrome P450s, a ubiquitous superfamily of enzymes responsible for eliminating most hydrophobic compounds in tissues throughout the body12. PM2.5 and other foreign substances can also be transferred from the circulation through pores and fenestrate in the vascular endothelium to interact with the mononuclear phagocyte system in reticular connective tissues and thereby reach potentially sensitive target sites such as the bone marrow (BM)13,14, where they alter the activity of stem cells. Through its direct transfer via the blood circulation to major organs, PM2.5 has been implicated in cardiopulmonary15, respiratory16, and cardiovascular diseases17, lung cancer18, and an increased risk of death19,20. PM2.5 is also a risk factor for asthma21 and, in children, for cough variant asthma by reducing immune regulation and ventilatory function22. Exposure to PM2.5 and some of its constituents is associated with a reduced hemoglobin level during the third trimester in multiparous pregnancy23. The BM is the major source of adult stem cells, mainly hematopoietic stem cells (HSCs) and mesenchymal stem cells (MSCs). This review particularly focuses on the impact of exposure to PM2.5 on BM-residing stem cells, especially HSCs and MSCs, and the underlying mechanisms.
HSCs
HSCs are multipotent, self-renewing cells with the ability to differentiate into functional blood cells24. Disruption of HSC function can affect hematopoiesis, immune function, and overall health25. As multipotent primitive cells, HSCs can develop into all types of blood cells, including myeloid- and lymphoid-lineage cells26. Their maintenance is facilitated by a highly specialized niche microenvironment hosting mesenchymal stromal cells, endothelial cells, and megakaryocytes27. Blood cells have a high turnover rate and thus form a highly regenerative tissue. HSCs can be divided into long-term (LT)-HSCs, short-term (ST)-HSCs, and multipotent progenitor (MPP) cells. Under normal physiological conditions, LT-HSCs are in a resting state, but in response to stress, they are activated to develop into all lineage blood cells28,29, ensuring the long-term survival and lifelong hematopoietic function of HSCs.
HSCs undergo self-renewal, replication, and multilineage differentiation. Self-renewal is maintained by asymmetric mitosis30, in which one of the two daughter cells is an early progenitor cell and the other retains all of the stem cell characteristics. As a result, the number of HSCs remains constant regardless of the number of replication cycles, generating progenitor cells to meet an organism’s normal differentiation needs31,32. HSCs differentiate into two branches: lymphoid cells and myeloid progenitor cells. Lymphoid progenitor cells further differentiate into T cells, B cells, natural killer (NK) cells, and myeloid progenitor cells can further differentiate into red blood cells, platelets, granulocytes, macrophages, and dendritic cells33,34. Given the short lifespan of blood cells, HSCs must continuously differentiate to maintain their populations.
MSCs
MSCs have self-renewal potential and tri-lineage plasticity35. The regulation of HSCs is mediated by MSCs, which modulate the BM microenvironment in addition to providing an osteoblastic niche. MSCs express a particular set of markers on their surface, including cluster of differentiation (CD)73, CD90, and CD105, but not CD14, CD34, CD45, and human leukocyte antigen-DR (HLA-DR)35. The main sources of MSCs are the BM36, adipose tissue37,38, and umbilical cord tissue39. BM-MSCs undergo self-renewal and multilineage differentiation40 into osteocytes, chondrocytes, adipocytes37,41,42,43,44, hepatocytes45, cardiomyocytes46, pancreatic cells47,48,49, and neuronal cells50,51. This differentiation potential of BM-MSCs contributes to modulation of the BM microenvironment, which can affect HSCs in a noncell-autonomous manner. Maternal exposure to PM2.5 during pregnancy disrupts the BM microenvironment and causes MSC senescence, followed by the senescence of HSCs and the development of myeloproliferative disease52.
Mechanism of PM2.5 toxicity
The composition and characteristics of PM2.5 vary geographically and temporally depending on the source and atmospheric conditions. PM2.5 is mainly composed of black carbon53, polycyclic aromatic hydrocarbons54,55, aryl hydrocarbons56, volatile organic hydrocarbons57, heavy metals58, organic compounds59, minerals60, inorganic ions61, and biological materials62. Elements such as Al, As, Br, Ca, Cl, Cr, Fr, K, Mg, Mn, Na, Pb, Ti, and Zn, as well as sulfate, nitrate, and ammonium ions, are also commonly present in PM2.563.
There is a relationship between exposure to airborne pollutants and poor human health52. In humans, the initial phase of angiohematopoiesis occurs outside of the embryo, in the yolk sac, from approximately 16 days of development64. Hematopoiesis is next transiently relayed in the liver before it shifts to the BM. During maternal exposure to PM2.5 in pregnancy, the particles directly enter the alveoli of the lungs and penetrate the blood–gas and placental barriers65, potentially causing premature birth and increasing the risk of mortality of preterm infants66.
PM2.5 can interfere with normal hematopoiesis and has been implicated in leukemia and other hematopoiesis-associated diseases67. In mice receiving an intranasal instillation of PM2.5 over a period of months, the number of stem cells in the BM decreased68. Exposure to PM2.5 for 2 weeks was sufficient to impair hematopoiesis in the BM of mice69. A summary of investigational studies on the effects of PM2.5 is provided in Table 1.
The mechanisms by which PM2.5 affect HSPCs and HSCs
Hematopoietic stem and progenitor cells (HSPCs), the progenitors of HSCs in the BM, have the capacity for self-renewal and multilineage differentiation70. The regulation of survival, proliferation, differentiation, and migration of HSPCs is essential for hematopoietic hemostasis and therefore the maintenance of mature immune cell generation71,72,73.
PM exposure induces the production of ROS and oxidative stress74,75. Both oxidative stress and inflammasome activation are enhanced in the BM of PM2.5-exposed mice (Fig. 1). In newborn mice, inflammasome activation can be detected in the BM for up to 12 months following PM2.5 inhalation, indicating that the particles enter not only the peripheral blood but also the BM76. Mice born after maternal PM2.5 exposure exhibited a similar number of lineage-Sca-1+c-Kit+ (LSK) cells and HSCs as control mice at 6 months of age, but at 12 months of age, the number of HSCs, but not LSK cells, was significantly increased76. LSK cells were highly enriched in HSPCs. In the BM, PM2.5 significantly increased the number of HSPCs (1.2-fold), GMPs (2.8-fold), MEPs (1.3-fold), and MkPs (1.7-fold) in wild-type mice but significantly decreased the number of HSPCs (0.8-fold) in Nrf2−/− mice, suggesting that NRF2 regulates HSPC differentiation to myeloid lineages and is involved in the proliferation or survival of HSPCs77. The numbers of GMPs, common myeloid progenitor cells (CMPs), MEPs, and common lymphoid progenitor cells (CLPs) in the BM did not differ significantly between newborn PM2.5-exposed mice and control mice at 6 months76. However, at 12 months, the proportion of myeloid lineage cells was increased, and the proportion of lymphoid lineage cells was reduced76. Similarly, tertiary transplanted PM2.5-exposed LSK cells in the peripheral blood were shown to consist of a higher proportion of myeloid lineage cells and a lower proportion of lymphoid lineage cells76. A similar hematopoietic skewing toward the myeloid lineage occurs in organs repopulated by myeloid-biased (My-bi) HSCs, while skewing toward a lymphoid lineage was observed in the blood, BM, and spleen after repopulation by lymphoid-biased (Ly-bi) HSCs78. In another study, mice with low- and high-dose PM2.5 exposure exhibited pathological changes in their BM79. PM2.5 exposure was shown to decrease the proliferation of BM-derived stromal cells by inhibiting the phosphorylation of Akt (without changing apoptosis) and increasing ROS production, both of which were reversed by treatment with N-acetyl-L-cysteine (NAC) (Fig. 1)80. However, offspring born from NAC-treated mice showed growth retardation and had a low body weight52. A model simulating respiratory tract lining fluid was used to demonstrate the depletion of antioxidants in response to PM exposure81. In fetal mice consecutively exposed to 50 µg/m3 PM2.5 during the first 12.5 days of gestation, an increase in the levels of 8-hydroxy-2’-deoxyguanosine (8-OHdG), a major biomarker of oxidative stress, cyclooxygenase-2 (COX-2), and TNF-α, a marker of inflammation, was observed in the lungs at E16.5d. In contrast, there was no increase in mitochondrial ROS levels in the liver-derived HSCs of PM2.5-exposed fetal mice because PM2.5 inhaled during pregnancy did not directly affect these cells in the fetus52. BM cellularity and the mitochondrial ROS levels of BM HSCs were not significantly altered between control and PM2.5-exposed offspring at 2 months, but at 6 months, the BM HSCs in PM2.5-exposed offspring had significantly increased levels of mitochondrial ROS and senescence-associated factors, such as senescence-associated beta-galactosidase (SA-β-gal), phospho-p38, and p16INK4a, and increased myeloid lineage-biased differentiation, leading to functional defects in clonogenic formation and donor cell-derived repopulation, engraftment, and self-renewal potentials in transplantation experiments. The fact that the senescent phenotype of HSCs induced by maternal PM2.5 exposure can be reversed by NAC treatment indicates that it is triggered mainly by oxidative stress52. NAC is an antioxidant that reduces the levels of ROS, produces cysteine by deacetylation, and increases glutathione levels, thereby promoting the scavenging of free radicals82. Treatment with NAC may reduce the telomere attrition rate associated with senescence, decrease lipid peroxidation, and activate the catalytic subunit of telomerase, thereby preventing the senescence of HSCs83. Similar to HSCs from maternal PM2.5-exposed offspring, those from newborn PM2.5-exposed middle-aged mice showed enhanced levels of mitochondrial ROS and senescence-related functional defects, as evidenced following serial transplantation and irradiation stress76. The mechanism is thought to involve a deficiency of telomerase activity, leading to telomere shortening and a reduction in the reconstitution potential of HSCs during transplantation84. In human lung epithelial cells, PM2.5 causes telomerase shortening and increases the levels of senescence markers85. While newborn control LSK cell-transplanted recipient-derived BM cells exhibited radioprotective effects and short-term repopulation of cells and thus almost rescued all of the lethally irradiated recipient mice, newborn PM2.5-exposed LSK cell-transplanted recipient-derived BM cells did not exert radioprotective effects on recipient mice, and all of these mice died within 9 weeks of transplantation76.
Inflammatory cytokines, including IL-1β, IL-6, IL-8, and TNF-α, are secreted by bronchial epithelial cells and alveolar macrophages in response to PM2.5 exposure. Systemic inflammatory responses are caused by the spread of these inflammatory factors from the lungs to the BM. The concentrations of PM2.5 and inflammatory cytokines are increased in the BM. PM2.5 inhibits the proliferation of HSCs, causing senescence and apoptosis. The proportion of quiescent HSCs in the BM is ~ 75%. The majority of cells are in G0 phase, and anchoring proteins on the surface of the bone, such as integrin and N-cadherin, as well as cytokines from myeloid stromal cells, such as CXCL12 and TGF-β, control the quiescence of HSCs. ROS, p53, p21, and p-p38 trigger the senescence of HSCs, whereas BAX, NOX, ROS, and p21 trigger their apoptosis. NRF2 inhibits the apoptosis of HSCs caused by PM2.5. In the differentiation of HSCs to macrophages, IL-6 promotes osteoclast formation in response to a low level of RANKL by controlling NF-κB, extracellular signal-regulated protein kinase/Jun N-terminal kinase, and mitogen-activated protein kinase signaling pathways, as well as transcription factors downstream of these pathways—nuclear factor-activated T-cell 1 (NFATc1), c-Fos, and tartrate resistance acid phosphatase (TRAP). TNF-α stimulates immunoreceptor tyrosine-based activation motif adaptor protein (ITAM), which promotes the transcription of NFATc1, c-Fos, and TRAP, thereby enhancing osteoclast formation and function in bone resorption. Sinusoidal endothelial (SER) cells supported by CXCL12-abundant reticular (CAR) cells in the BM regulate the proliferation, differentiation, homing, mobilization, and quiescence of HSCs. PM2.5 disrupts the function of sinusoid endothelial cells, resulting in a decrease in the self-renewal and proliferation abilities of HSCs and an increase in the number of quiescent HSCs. SER facilitates the differentiation of HSCs into lymphoid and myeloid progenitors. PM2.5 disrupts the differentiation of HSCs by decreasing the number of lymphoid progenitors and increasing the number of myeloid progenitors. Few lymphoid progenitors further differentiate into NK, T, and B cells, leading to the failure of immune functions. PM2.5 phosphorylates Akt and p38 in HSCs, stimulating the production of ROS and increasing oxidative stress. The increased ROS levels caused by PM2.5 exposure promote the excessive proliferation of myeloid progenitors, resulting in leukemia, anemia, and myelofibrosis. Superoxide ions produced by BMSCs cause mutations in HSCs, leading to the production of mutant progeny, which increases neutrophil extracellular trap (NET) formation and causes thromboses in blood vessels.
Exposure to PM2.5 activates the NRF2 pathway86, a regulator of HSPCs that induces their expansion in response to stress. NRF2 modulates HSPC retention and migration in the BM microenvironment; regulates the proliferation, differentiation, and survival of HSPCs and other progenitors87; and plays a role in the development of myeloid cells from HSCs88. The level of NRF2 is increased in the BM HSCs of PM2.5-exposed mice and in their offspring at 6 months52 but is significantly decreased in NRF2-knockout mice77. Intracellular ROS levels regulate the survival, proliferation, and differentiation of HSPCs and are critical for maintaining their homeostasis87,88. NRF2 increases the survival rate of HSPCs by inhibiting their apoptosis in the BM without changing the level of ROS in the hypoxic microenvironment of the BM88.
NADPH oxidase (NOX)1, 2, and 4 are expressed in mouse BM LSK cells, whereas HPCs, Lin- cells, and mononuclear cells from mouse BM express NOX1 and 2, but not NOX4, suggesting that the expression of NOX4 is downregulated upon HSC differentiation and that NOX4 plays an important role in regulating HSC function89. The expression levels of NOX4 and the NOX-associated subunits p67phox, p47phox, and p22phox are increased in the heart tissue of PM2.5-exposed mice90. Elevated ROS levels modulate HSC-specific phosphorylation of p38, limiting the function of HSCs91. In maternal PM2.5-exposed offspring, p38 expression in HSCs is upregulated. Activation of the p38 pathway contributes to the induction of p16 and cellular senescence in response to other stimuli, including DNA damage resulting from exposure to genotoxic and oxidative stress and telomere shortening due to extensive replication92. Compared to the BM cells from control mice, BM cells from newborn PM2.5-exposed 12-month-old mice exhibit reduced clonogenicity, involving colony-forming unit (CFU)- induced granulocytes/monocytes, burst-forming-unit-erythrocytes, and CFU-induced erythrocytes/macrophages/megakaryocytes. Maternal exposure to PM2.5 during pregnancy selectively affects the BM microenvironment, causing premature aging, with BM HSCs gradually becoming senescent via a noncell autonomous pathway. The mechanisms by which PM2.5 affects HSCs are shown in Fig. 1.
The above studies indicate that the effects of PM2.5 on mice depend on the age at exposure, with more harmful effects during the embryonic stage than the adult stage. This finding was also shown in a mouse study in which 9 of 25 (36%) 1-year-old maternal PM2.5-exposed offspring had a higher propensity to develop a myeloproliferative illness, with enhanced SA-gal activity in MSCs and HSCs observed over time52.
The mechanisms by which PM2.5 affects MSCs
PM2.5 changes the BM microenvironment with subsequent effects on HSCs, including the induction of osteoclastogenesis and adipogenesis. The expression levels of runt-related transcription Factor 2 and osteopontin are lower in the BM cells of PM2.5-exposed mice than in those of control mice76. PM2.5-exposed BM cells have low bone mineralization ability and primarily differentiate into osteoclasts, both of which are closely associated with an increase in receptor activator of nuclear factor κB ligand (RANKL) levels and a decrease in osteoprotegerin (OPG) levels76. Exposure to cadmium significantly inhibits the differentiation of BMSCs and bone marrow macrophages (BMMs) into osteoblasts and osteoclasts and promotes apoptosis in vitro via P2X7/PI3K/AKT signaling and the RANKL/OPG system93. In humans, the serum level of RANKL was increased by 41% and that of OPG was reduced by 22% among 79 premenopausal women constantly exposed to PM10-2.5 biomass compared with control women, suggesting that chronic exposure to biomass smoke increases the risk of bone resorption and osteoporosis94.
BM-MSCs from PM2.5-exposed mice show increased adipogenesis (adiponectin and peroxisome proliferator-activated receptor γ) and senescence76. A low concentration of PM2.5 increases the proliferation of BM-MSCs in vitro, but an increase in the PM2.5 concentration decreases the number of BM-MSCs, induces cellular morphological changes, and increases cell death caused by stress-related cell shrinkage and membrane disruption95. In C10 alveolar epithelial cells exposed to 50 µg PM2.5/cm3, the sub-G0/G1 phase is prolonged, indicative of both apoptotic and necrotic cell death96. Receptor interacting protein (RIP) and the fas-associated death domain (FADD) complex act in concert with caspase-8 and have been implicated in PM2.5-induced apoptosis96. Inflammatory markers such as TNF-α and IL-6 are upregulated in response to PM2.595. TNF-α and FADD interact with RIP to induce apoptosis97. In BM-MSCs treated with 150 µg PM2.5/ml, antiapoptotic BCL2 is upregulated, while proapoptotic BAX and the tumor suppressor gene p53 are downregulated95. Exposure to PM2.5 promotes the secretion of inflammatory cytokines in the respiratory tract98,99 and promotes BM-MSC differentiation100 into endothelial-like cells and cancer-associated fibroblasts101. The levels of IL-1β, IL-6, and COX-2 mRNAs are increased in PM2.5-exposed 16HBE cells. IL-6 is important for inducing the expression of markers of differentiation, such as CD31, von Willebrand factor, α-smooth muscle actin (α-SMA), and fibroblast activation protein (FAP), whereas IL-1β and COX-2 induce the expression of α-SMA and FAP101. PM2.5 elevates ROS levels via NOX, a key factor in ROS generation102 that is also elevated in mitochondrial disorders103. The mechanisms by which PM2.5 affects MSCs are summarized in Fig. 2.
PM2.5 and inflammatory cytokines in bronchial epithelial cells enter the BM via blood vessels. PM2.5, inflammatory cytokines, and ROS inhibit the self-renewal and proliferation abilities of MSCs. Under normal conditions, TGF-β and Wnt facilitate the self-renewal of MSCs. ROS and oxidative stress cause the senescence of MSCs and increase their shrinkage and membrane disruption, resulting in MSC death. The production of IL-1β triggered by PM2.5 activates NF-κB, p38, and ERK, finally activating the Active 1 protein, which inhibits the transcription of bone morphogenic protein (BMP)-2, BMP-4, and BMP-9. These proteins are essential for the maturation of osteoblasts. TNF-α inhibits the osteogenic differentiation of MSCs by stimulating the production of Dickkopf-related protein 1 (DKK1), thereby inhibiting the transcription of osteocalcin, BMP-4, BMP-6, RUNX-2, and OPN. Furthermore, the ROS produced by MSCs after exposure to PM2.5 cause mitochondrial dysfunction via the Jun N-terminal kinase (JNK) and NOX pathways, leading to release of the apoptosis-promoting protein BAX and inhibition of BCL-2 production and the apoptosis of mature osteoblasts via PI3K/Akt/GSK3β signaling. TNF-α may activate FADD and bind to RIP, thereby activating caspase-8 and inducing the apoptosis of MSCs. Exposure of MSCs to PM2.5 stimulates the production of adiponectin and PPARγ, thereby increasing the differentiation of MSCs to adipocytes rather than osteocytes.
Hematopoiesis and blood disorders related to PM2.5
Mesodermal progenitor cells (MPCs) differentiate into CMPs, GMPs, and other oligo-potent or unipotent progenitor cells, which in turn give rise to mature blood cells104,105. The effects of PM2.5 that have reached the BM affect BM-conserved stem cells. Children in areas with high 24 h PM2.5 concentrations (mean 25−50 µg/m3) have a higher rate of mild to moderate anemia106, and those in areas with very high 24 h PM2.5 ( >50 µg/m3) have a higher rate of moderate to severe anemia95. In animal models, PM2.5 exposure causes more harmful effects, including BM microenvironment impairment, in young mice than in adolescent mice68,95. PM2.5 increases ROS production and thus the cellular levels of inflammatory cytokines (TNF-α, IL-1β, and IL-6), which in turn inhibit the differentiation and proliferation of erythroid precursor cells and promote an erythropoietin-resistant state52,68,95. Inflammatory cytokines also cause the breakdown of ferroportin by upregulating hepcidin synthesis, thereby reducing iron absorption in the gastrointestinal tract107,108. Murine erythrocytes are reportedly deformed in a mice after PM2.5 exposure109. As in humans, mice exposed to air pollution have an increased risk of anemia, depending on the PM2.5 dose and exposure time95. Mice exposed to PM2.5 for 100 s/day for eight consecutive days have increased erythrocyte distortion, leading to hemolytic anemia (Fig. 1)109. Leukemia is a malignant clonal disease characterized by abnormal proliferation and impaired differentiation of HSCs. Exposure to PM2.5 injures HSCs in the BM52 and has been implicated in adult leukemia and childhood hematological malignancy110,111. Among the effects of long-term exposure to PM2.5 is the alteration of DNA methylation112. Molecular epidemiologic studies indicate that DNA methylation of the genes associated with leukemia positively correlates with exposure to environmental toxins113,114. DNA methylation involves incorporation of a methyl group at the fifth carbon of cytosine to produce 5-methylcytosine, which is oxidized to 5-hydroxymethylcytosine, a suppressor of gene expression115,116, which prevents cellular differentiation and thus promotes the development of leukemia. PM2.5 is also linked to platelet function117,118, and deep venous thrombosis and hypercoagulability are associated with long-term ( ~ 8 years) exposure to PM2.5119. The mechanism of PM2.5-associated thrombosis involves increases in the levels of inflammatory cytokines, oxidative stress, and platelet activation as well as stimulation of coagulation pathways and reduced fibrinolysis120,121,122. The increased expression levels of inflammatory cytokines, such as IL-6 and IL-1β, in rats exposed to PM2.5 induces disseminated intravascular coagulation120. The effects of moderate to high doses of PM2.5 include the activation of tissue-factor-dependent extrinsic pathways, coagulation, and the upregulation of adhesion molecules, such as vascular cell adhesion molecule 1 and intracellular adhesion molecule 1118.
Health implications and clinical outcomes
An increase in the incidence and mortality of certain illnesses has been linked to exposure to ambient PM2.5. Several studies have reported a high risk of PM2.5-related mortality in patients with lung cancer, ischemic heart disease, stroke, chronic obstructive pulmonary disease (COPD), and other disorders worldwide (Fig. 3)123,124,125,126,127,128. One of the main targets of PM2.5-induced toxicity is the lung, the first site of PM2.5 deposition in the airway. PM2.5 causes inflammation in the airways, impairs normal immunological responses in the lung, and increases their susceptibility to several respiratory diseases7. First, injury to the bronchial mucociliary system caused by PM2.5 suppresses the clearance of foreign particles8. Next, lung epithelial cells and fibroblasts undergo apoptosis due to PM2.5 exposure, resulting in the disruption of inflammatory cytokine networks, rendering them incapable of communicating through gap junctions. This impairment in cellular communication leads to increased permeability of the lung epithelial barrier, ultimately diminishing its efficacy as a physical component of pulmonary innate immunity9. PM2.5 reaches the lungs by inhalation and activates immune cells, such as macrophages and neutrophils, which release inflammatory mediators. Fine-tuned regulation of the survival, proliferation, differentiation, and migration of HSPCs is essential for hematopoietic homeostasis, including the normal generation of mature immune cells71,72,73. Excessive exposure to PM2.5 causes inflammation and the release of cytokines that act on HSPCs. Type I interferons (e.g., IFN-α) and type II interferons (e.g., IFN-γ) are important for the adaptation of HSPCs to inflammation and drive the proliferation of quiescent HSCs; however, their chronic administration impairs the self-renewal ability of HSCs129. This is a result of the induction of DNA damage in HSCs entering the cell cycle, which is linked to an increased mitochondrial membrane potential and mitochondrial production of ROS130. IL-1β is also a central mediator of innate immunity and acts directly on HSCs in vitro, promoting their proliferation and differentiation into myeloid lineage cells by activating the transcription factor PU.1. Chronic administration of IL-1β diminished the self-renewal ability of HSCs in a mouse model131. HSCs also respond to TNF, which induces their proliferation and directs myeloid lineage differentation in HSCs, thus compromising the repopulation of these cells132. The PM2.5-induced disruption of HSC maintenance limits the differentiation of HSCs to lymphoid-lineage progenitor cells, leading to deficiencies in NK cells and T and B lymphocytes and thus to an inability to combat infection133. PM2.5 also disrupts macrophage phagocytosis, increasing susceptibility to infection and inducing chronic lung injury117. Air pollution, including PM2.5, causes a T-cell imbalance, proinflammatory cytokine production, local pulmonary inflammation, oxidative stress, and methylation changes, all of which underlie the pathogenesis of autoimmune diseases56. For example, chronic exposure to PM2.5 causes several134,135 abnormalities linked to the development of type 2 diabetes mellitus (T2DM), adipose inflammation, insulin resistance (IR), and hepatic endoplasmic reticulum (ER) stress. PM2.5 has been suggested to modulate ER stress and inflammatory pathways, leading to IR and the development of T2DM136. In addition, exposure to PM2.5 not only causes subclinical alterations in cardiovascular function but also damages the heart’s autonomic nervous system (ANS), which decreases heart rate variability and is inextricably linked to cardiovascular morbidity and mortality137. Additionally, PM2.5 is linked to the prevalence and development of chronic kidney disease (CKD) and decreases the glomerular filtration rate (GFR)138. Maternal prenatal exposure to PM2.5 is linked to poor birth outcomes, such as preterm delivery, low birth weight, and neonatal infant death139,140,141. Furthermore, PM2.5 influences several other harmful health outcomes, including impaired antiviral immunity, bone loss, liver fibrosis, lung cancer, macrosomia, Alzheimer’s disease, and ovarian dysfunction (Fig. 3)142,143,144,145,146,147,148.
Epidemiological and experimental studies
PM2.5 has several implications for human health, mainly related to cardiovascular and respiratory diseases and cancer149. Long-term exposure to PM2.5 was associated with a decline in bone strength among adults in Southwest China150 and with a decrease in BMD and an increase in the likelihood of osteoporosis151. PM2.5 at 1 µg/m3 increased the risk of osteoporosis by 14.6% in 8033 rural residents of Henan Province, China, between July 2015 and September 2017152. In several cities in Brazil from 2010 to 2018, long-term exposure to PM2.5 was linked to an increase in the mortality rates of a variety of cancers, including bone cancer, and persons older than 60 years had a higher risk of succumbing to bone cancer153. Morales-Ancajima et al. evaluated the association between hemoglobin and air pollution in a residential area among 139,368 children (ages 6–35 months). Anemia was detected in 30.8% of the exposed children, and moderate to severe anemia was detected in 8.8%154. Honda et al. performed a study involving 4121 older persons with anemia (34.9%) who lived in areas with high annual mean PM2.5 levels (>11.1 ± 2.8 μg/m3). Individuals with 2–5 years of exposure to PM2.5 pollution had a mean decline in hemoglobin levels of 0.81 ± 0.06 g/dL155. The PM2.5 levels in blood samples from adult patients (age 20–71 years) were higher in those with acute myeloid leukemia than in healthy individuals156. Exposure of newborn mice to PM2.5 induced HSC senescence by causing chronic oxidative stress and inflammasome activation in the BM76. Exposure of pregnant mice to PM2.5 caused progressive senescence of HSCs as a result of the continuous impairment of the BM microenvironment52. PM2.5 increases the numbers of systematic inflammatory cells and the risks of associated diseases in adult mice by regulating NRF2-dependent myeloid-biased hematopoiesis77. The proliferation of BMSCs is inhibited by PM2.5 exposure, leading to their degeneration and death and increasing IL-6 and TNF-α production. The osteoblastic development of BMSCs is suppressed, as is the deposition of extracellular matrix, and TNF-α, IL-6, and IL-11 mRNA levels are elevated upon coculture of chronic lymphoblastic leukemia cells with BMSCs157. Continuous exposure to organic dust lowered the bone density and bone volume fraction in mice and increased bone trabecular spacing and the serum IL-6 concentration and number of osteoclasts. The lung-inflammatory-bone axis, which encompasses the systemic IL-6 effector pathway, is involved in inhaled organic dust extract-mediated bone damage in mice158. According to Calderon et al., 6-year-old children who lived in locations with high PM2.5 pollution levels had decreased bone density and increased IL-6 production compared with those who resided in areas with low PM2.5 and air pollution levels159. An analysis of 175,959 men and 186,437 women showed that for each 10 µg/m3 increase in PM2.5, men and women had 17% and 14%, respectively, greater likelihoods of an increased platelet count (≥90th percentile)117.
Future research
The effect of PM2.5 on the functional integrity of HSCs in the BM is a new avenue of investigation in research on immune disorders and blood cancers induced by PM2.5 exposure. However, such studies are complicated by the geographical and temporal variability of PM2.5 levels, which makes it difficult to determine individual exposure. Developing strategies for accurate exposure assessment, such as exposure modeling or personal monitoring approaches, would be beneficial. Among the methods through which PM2.5 impacts HSCs, inflammation, oxidative stress, and epigenetic alterations have been examined using cutting-edge experimental methods and molecular investigations. The identification of reliable biomarkers of PM2.5-induced adverse effects on HSCs is crucial for monitoring PM2.5 exposure in at-risk populations, analyzing PM2.5-derived health consequences, and evaluating therapeutic efficacy. The effects of PM2.5 on HSCs can be facilitated by animal models that appropriately represent human responses and by in vitro systems that accurately mimic the HSC milieu.
How long-term exposure to PM2.5 affects HSCs is another important area of research. Elucidating the impact of PM2.5 on the function, self-renewal, and differentiation potential of HSCs and thus on hematopoiesis can provide insights into the mechanism by which blood-related illnesses arise from PM2.5 exposure. The epigenetic changes induced by exposure to PM2.5 can be examined by studying DNA methylation patterns, histone modifications, and noncoding RNA expression. Epidemiological studies on the relationship between PM2.5 exposure and hematological diseases or anomalies in sizable populations are also essential to determine the potential hazards of PM2.5 and guide the development of preventative measures. All these areas of investigation should be accompanied by research on therapies and tactics that lessen the harmful effects of PM2.5 on HSCs, such as by determining the efficacy of protective agents, therapeutic intervention options, and air pollution reduction initiatives. Identifying susceptible groups, such as people with blood diseases or genetic predispositions, will be essential to develop focused preventative and intervention strategies.
Conclusions
Studies on the effects of PM2.5 on BM-conserved MSCs and HSCs have revealed that exposure alters the BM microenvironment and causes oxidative stress, inflammation, and DNA damage in HSCs, reducing their ability to self-renew and differentiate, thereby leading to abnormal hematopoietic development. The PM2.5-mediated functional changes in HSCs impact the immune system and have been implicated in hematological diseases. Similar effects on BM-conserved MSCs, a cell population essential for BM microenvironment maintenance, immunological control, and tissue healing, have been linked to exposure to PM2.5. MSC equilibrium can be disrupted by PM2.5-induced oxidative stress and inflammation, resulting in decreased proliferation and poor differentiation of these cells together with weakened immunomodulatory capabilities. These results have important ramifications for MSC-mediated tissue regeneration and repair processes. The negative effects of PM2.5 on BM-conserved HSCs and MSCs are well established and include disturbances in hematopoiesis, the immune system, and tissue healing mechanisms. The findings summarized in this review highlight the urgent need for mitigation measures aimed at reducing PM2.5 pollution, the benefits of which will safeguard the structural and functional integrity of BM stem cells, including their regenerative capability. In addition to elucidating the molecular mechanisms by which PM2.5 exerts its detrimental effects on HSCs and MSCs, potential therapeutic interventions, such as antioxidant strategies or anti-inflammatory treatments, require increased research attention. The ability to determine the effects of PM2.5 on BM-conserved HSCs and MSCs in humans will encourage the development of strategies to mitigate PM2.5 pollution, protect human health, and advance well-being in the face of mounting environmental problems.
References
Dockery, D. W. et al. An association between air pollution and mortality in six US cities. N. Engl. J. Med. 329, 1753–1759 (1993).
Lippmann, M., Yeates, D., Albert, R. J. O. & Medicine, E. Deposition, retention, and clearance of inhaled particles. Br. J. Ind. Med. 37, 337–362 (1980).
Kappos, A. D. et al. Health effects of particles in ambient air. Int. J. Hyg. Environ. Health 207, 399–407 (2004).
Kelly, F. J. & Fussell, J. C. Air pollution and public health: emerging hazards and improved understanding of risk. Environ. Geochem. Health 37, 631–649 (2015).
Lee, M. An analysis on the concentration characteristics of PM2.5 in Seoul, Korea from 2005 to 2012. Asia Pac. J. Atmos. Sci. 50, 585–594 (2014).
Research, N.I.o.E. (National Institute of Environmental Research Incheon, Korea, (2017).
Jia, H. et al. PM2.5‐induced pulmonary inflammation via activating of the NLRP3/caspase‐1 signaling pathway. Environ. Toxicol. 36, 298–307 (2021).
Duan, Z. et al. Effects of PM2. 5 exposure on Klebsiella pneumoniae clearance in the lungs of rats. Zhonghua jie he he hu xi za zhi= Zhonghua jiehe he huxi zazhi= Chin. J. tuberculosis respiratory Dis. 36, 836–840 (2013).
Longhin, E. et al. Cell cycle alterations induced by urban PM2. 5 in bronchial epithelial cells: characterization of the process and possible mechanisms involved. Part. Fibre Toxicol. 10, 1–19 (2013).
Jin, X.-T. et al. Progression and inflammation of human myeloid leukemia induced by ambient PM 2.5 exposure. Arch. Toxicol. 90, 1929–1938 (2016).
Blank, F., von Garnier, C., Gehr, P. & Rothen-Rutishauser, B. Translocation across the air-blood tissue barrier. (CRC Press, 2014).
Reed, J. R., dela Cruz, A. L. N., Lomnicki, S. M. & Backes, W. L. Environmentally persistent free radical-containing particulate matter competitively inhibits metabolism by cytochrome P450 1A2. Toxicol. Appl. Pharmacol. 289, 223–230 (2015).
Oberdörster, G., Oberdörster, E. & Oberdörster, J. Nanotoxicology: an emerging discipline evolving from studies of ultrafine particles. Environ. Health Perspect. 113, 823–839 (2005).
Casals, E., Vázquez-Campos, S., Bastús, N. G. & Puntes, V. Distribution and potential toxicity of engineered inorganic nanoparticles and carbon nanostructures in biological systems. TrAC Trends Anal. Chem. 27, 672–683 (2008).
Xu, J.-J. et al. Relationship between PM 2. 5 exposure and pulmonary function in different working environments. J. Environ. Health 30, 1–4 (2013).
Xing, Y.-F., Xu, Y.-H., Shi, M.-H. & Lian, Y.-X. The impact of PM2. 5 on the human respiratory system. J. Thorac. Dis. 8, E69 (2016).
Cheng, Y. et al. Ambient PM2. 5 during pregnancy and risk on preterm birth. Zhonghua liu xing bing. xue za zhi= Zhonghua liuxingbingxue zazhi 37, 572–577 (2016).
Shu, Y. et al. Analysis of the relationship between PM2. 5 and lung cancer based on protein-protein interactions. Combinatorial Chem. high. throughput Screen. 19, 100–108 (2016).
Wu, J., Zhu, J., Li, W., Xu, D. & Liu, J. Estimation of the PM 2.5 health effects in China during 2000–2011. Environ. Sci. Pollut. Res. 24, 10695–10707 (2017).
Shi, T. et al. Association between PM2. 5 air pollution and daily resident mortality in Guangzhou urban area in winter. J. Environ. Health 32, 477–481 (2015).
Chen, Y., Wong, G. W. & Li, J. Environmental exposure and genetic predisposition as risk factors for asthma in China. Allergy, Asthma Immunol. Res. 8, 92–100 (2016).
Zhang, Y. et al. Correlational study on atmospheric concentrations of fine particulate matter and children cough variant asthma. Eur. Rev. Med. Pharm. Sci. 20, 2650–2654 (2016).
Xie, G. et al. Effects of PM2. 5 and its constituents on hemoglobin during the third trimester in pregnant women. Environ. Sci. Pollut. Res. 29, 35193–35203 (2022).
Seita, J. & Weissman, I. L. Hematopoietic stem cell: self‐renewal versus differentiation. Wiley Interdiscip. Rev.: Syst. Biol. Med. 2, 640–653 (2010).
Verovskaya, E. V., Dellorusso, P. V. & Passegué, E. Losing sense of self and surroundings: hematopoietic stem cell aging and leukemic transformation. Trends Mol. Med. 25, 494–515 (2019).
Till, J. E. & McCulloch, E. A. A direct measurement of the radiation sensitivity of normal mouse bone marrow cells. Radiat. Res. 178, AV3–AV7 (2012).
Wei, Q. & Frenette, P. S. Niches for hematopoietic stem cells and their progeny. Immunity 48, 632–648 (2018).
Yamada, T., Park, C. S. & Lacorazza, H. D. Genetic control of quiescence in hematopoietic stem cells. Cell Cycle 12, 2376–2383 (2013).
Cabezas-Wallscheid, N. et al. Vitamin A-retinoic acid signaling regulates hematopoietic stem cell dormancy. Cell 169, 807–823. (2017).
Shenghui, H., Nakada, D. & Morrison, S. J. Mechanisms of stem cell self-renewal. Annu. Rev. Cell Developmental 25, 377–406 (2009).
Hinge, A. et al. Asymmetrically segregated mitochondria provide cellular memory of hematopoietic stem cell replicative history and drive HSC attrition. Cell Stem Cell 26, 420–430. (2020).
Inaba, M. & Yamashita, Y. M. Asymmetric stem cell division: precision for robustness. Cell Stem Cell 11, 461–469 (2012).
Suda, T., Arai, F. & Hirao, A. Hematopoietic stem cells and their niche. Trends Immunol. 26, 426–433 (2005).
Majeti, R., Park, C. Y. & Weissman, I. L. Identification of a hierarchy of multipotent hematopoietic progenitors in human cord blood. Cell Stem Cell 1, 635–645 (2007).
Dominici, M. et al. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy 8, 315–317 (2006).
Pittenger, M. F. et al. Multilineage potential of adult human mesenchymal stem cells. Science 284, 143–147 (1999).
Wagner, W. et al. Comparative characteristics of mesenchymal stem cells from human bone marrow, adipose tissue, and umbilical cord blood. Exp. Hematol. 33, 1402–1416 (2005).
Zhang, X. et al. Runx2 overexpression enhances osteoblastic differentiation and mineralization in adipose-derived stem cells in vitro and in vivo. Calcif. tissue Int. 79, 169–178 (2006).
Kita, K., Gauglitz, G. G., Phan, T. T., Herndon, D. N. & Jeschke, M. G. Isolation and characterization of mesenchymal stem cells from the sub-amniotic human umbilical cord lining membrane. Stem Cells Dev. 19, 491–502 (2010).
Ullah, I., Subbarao, R. B. & Rho, G. J. Human mesenchymal stem cells-current trends and future prospective. Biosci. Rep. 35, 1–18 (2015).
Ranera, B. et al. Expansion under hypoxic conditions enhances the chondrogenic potential of equine bone marrow-derived mesenchymal stem cells. Vet. J. 195, 248–251 (2013).
Zhang, X. et al. Isolation and characterization of mesenchymal stem cells from human umbilical cord blood: reevaluation of critical factors for successful isolation and high ability to proliferate and differentiate to chondrocytes as compared to mesenchymal stem cells from bone marrow and adipose tissue. J. Cell. Biochem. 112, 1206–1218 (2011).
Gronthos, S., Graves, S., Ohta, S. & Simmons, P. The STRO-1+ fraction of adult human bone marrow contains the osteogenic precursors. Blood 84, 4164–4173 (1994).
Muruganandan, S., Roman, A. & Sinal, C. Adipocyte differentiation of bone marrow-derived mesenchymal stem cells: cross talk with the osteoblastogenic program. Cell. Mol. life Sci. 66, 236–253 (2009).
Stock, P., Brückner, S., Winkler, S., Dollinger, M. M. & Christ, B. Human bone marrow mesenchymal stem cell-derived hepatocytes improve the mouse liver after acute acetaminophen intoxication by preventing progress of injury. Int. J. Mol. Sci. 15, 7004–7028 (2014).
Xu, W. et al. Mesenchymal stern cells from adult human bone marrow differentiate into a cardiomyocyte phenotype in vitro. Exp. Biol. Med. 229, 623–631 (2004).
Tang, D.-Q. et al. In vitro generation of functional insulin-producing cells from human bone marrow-derived stem cells, but long-term culture running risk of malignant transformation. Am. J. Stem Cells 1, 114 (2012).
Phadnis, S. M. et al. Human bone marrow-derived mesenchymal cells differentiate and mature into endocrine pancreatic lineage in vivo. Cytotherapy 13, 279–293 (2011).
Gabr, M. M. et al. Insulin-producing cells from adult human bone marrow mesenchymal stem cells control streptozotocin-induced diabetes in nude mice. Cell Transplant. 22, 133–145 (2013).
Barzilay, R., Ben-Zur, T., Bulvik, S., Melamed, E. & Offen, D. Lentiviral delivery of LMX1a enhances dopaminergic phenotype in differentiated human bone marrow mesenchymal stem cells. Stem Cells Dev. 18, 591–602 (2009).
Wilkins, A. et al. Human bone marrow-derived mesenchymal stem cells secrete brain-derived neurotrophic factor which promotes neuronal survival in vitro. Stem Cell Res. 3, 63–70 (2009).
Bhattarai, G. et al. Maternal exposure to fine particulate matter during pregnancy induces progressive senescence of hematopoietic stem cells under preferential impairment of the bone marrow microenvironment and aids development of myeloproliferative disease. Leukemia 34, 1481–1484 (2020).
Bové, H. et al. Ambient black carbon particles reach the fetal side of human placenta. Nat. Commun. 10, 3866 (2019).
Leachi, H. F. L. et al. Polycyclic aromatic hydrocarbons and development of respiratory and cardiovascular diseases in workers. Rev. Bras. Enferm. 73, e20180965 (2020).
Alegbeleye, O. O., Opeolu, B. O. & Jackson, V. A. Polycyclic aromatic hydrocarbons: a critical review of environmental occurrence and bioremediation. Environ. Manag. 60, 758–783 (2017).
Zhao, C.-N. et al. Emerging role of air pollution in autoimmune diseases. Autoimmun. Rev. 18, 607–614 (2019).
Puri, P., Nandar, S. K., Kathuria, S. & Ramesh, V. Effects of air pollution on the skin: A review. Indian J. Dermatol. Venereol. Leprol. 83, 415 (2017).
Ashmore, M.R. et al. World Atlas of Atmospheric Pollution, 77–94 (2008).
Grzywa-Celińska, A., Krusiński, A. & Milanowski, J. ‘Smoging kills’-effects of air pollution on human respiratory system. Ann. Agric. Environ. Med. 27, 1–5 (2020).
Kitamura, H. et al. Impact of secondary generated minerals on toxic element immobilization for air pollution control fly ash of a municipal solid waste incinerator. Environ. Sci. Pollut. Res. 25, 20700–20712 (2018).
Švédová, B. et al. Concentration variability of water-soluble ions during the acceptable and exceeded pollution in an industrial region. Int. J. Environ. Res. Public Health 17, 3447 (2020).
Shahrbaf, M. A., Akbarzadeh, M. A., Tabary, M. & Khaheshi, I. Air pollution and cardiac arrhythmias: A comprehensive review. Curr. Probl. Cardiol. 46, 100649 (2021).
Bangar, V., Mishra, A. K., Jangid, M. & Rajput, P. Elemental characteristics and source-apportionment of PM2. 5 during the post-monsoon season in Delhi, India. Front. Sustain. Cities 3, 648551 (2021).
Luckett, W. P. Origin and differentiation of the yolk sac and extraembryonic mesoderm in presomite human and rhesus monkey embryos. Am. J. Anat. 152, 59–97 (1978).
Onoda, A., Takeda, K. & Umezawa, M. Dose-dependent induction of astrocyte activation and reactive astrogliosis in mouse brain following maternal exposure to carbon black nanoparticle. Part. Fibre Toxicol. 14, 1–16 (2017).
Lelieveld, J., Evans, J. S., Fnais, M., Giannadaki, D. & Pozzer, A. The contribution of outdoor air pollution sources to premature mortality on a global scale. Nature 525, 367–371 (2015).
Puett, R. C. et al. Relationship of leukaemias with long-term ambient air pollution exposures in the adult Danish population. Br. J. Cancer 123, 1818–1824 (2020).
Cui, Y. et al. Ambient fine particulate matter induces apoptosis of endothelial progenitor cells through reactive oxygen species formation. Cell. Physiol. Biochem. 35, 353–363 (2015).
Ge, J. et al. Combined exposure to formaldehyde and PM2. 5: Hematopoietic toxicity and molecular mechanism in mice. Environ. Int. 144, 106050 (2020).
Kaushansky, K. Lineage-specific hematopoietic growth factors. N. Engl. J. Med. 354, 2034–2045 (2006).
Chavakis, T., Mitroulis, I. & Hajishengallis, G. Hematopoietic progenitor cells as integrative hubs for adaptation to and fine-tuning of inflammation. Nat. Immunol. 20, 802–811 (2019).
Ogawa, M. Differentiation and proliferation of hematopoietic stem cells. Blood 81, 2844–2853 (1993).
Wright, D. E., Wagers, A. J., Gulati, A. P., Johnson, F. L. & Weissman, I. L. Physiological migration of hematopoietic stem and progenitor cells. Science 294, 1933–1936 (2001).
Khurshid, S. S., Siegel, J. A. & Kinney, K. A. Indoor particulate reactive oxygen species concentrations. Environ. Res. 132, 46–53 (2014).
Daher, N. et al. Oxidative potential and chemical speciation of size-resolved particulate matter (PM) at near-freeway and urban background sites in the greater Beirut area. Sci. Total Environ. 470, 417–426 (2014).
Bhattarai, G., Sim, H.-J., So, H.-S., Lee, J.-C. & Kook, S.-H. Exposure of newborns to atmospherically relevant artificial particulate matter induces hematopoietic stem cell senescence. J. Hazard. Mater. 452, 131293 (2023).
Wang, Y. et al. PM2. 5 Increases Systemic Inflammatory Cells and Associated Disease Risks by Inducing NRF2-Dependent Myeloid-Biased Hematopoiesis in Adult Male Mice. Environ. Sci. Technol. 57, 7924–7937 (2023).
Muller-Sieburg, C. E., Cho, R. H., Karlsson, L., Huang, J.-F. & Sieburg, H. B. Myeloid-biased hematopoietic stem cells have extensive self-renewal capacity but generate diminished lymphoid progeny with impaired IL-7 responsiveness. Blood 103, 4111–4118 (2004).
Liu, L.-L. et al. Effects of Long-Term Exposure to PM2. 5 on Oxidative Stress Injury and Expression of Inflammatory Factors, NF-κB p65 and Cx43 in Bone Marrow of Mice. 10, 747286 (2022).
Cui, Y. et al. Ambient fine particulate matter suppresses in vivo proliferation of bone marrow stem cells through reactive oxygen species formation. PLoS One 10, e0127309 (2015).
Godri, K. J. et al. Particulate oxidative burden associated with firework activity. Environ. Sci. Technol. 44, 8295–8301 (2010).
Banaclocha, M. M., Hernandez, A. I., Martınez, N. & Ferrandiz, M. L. N-acetylcysteine protects against age-related increase in oxidized proteins in mouse synaptic mitochondria. Brain Res. 762, 256–258 (1997).
Voghel, G. et al. Chronic treatment with N-acetyl-cystein delays cellular senescence in endothelial cells isolated from a subgroup of atherosclerotic patients. Mech. Ageing Dev. 129, 261–270 (2008).
Samper, E. et al. Long-term repopulating ability of telomerase-deficient murine hematopoietic stem cells. Blood, J. Am. Soc. Hematol. 99, 2767–2775 (2002).
Chang-Chien, J. et al. Particulate matter causes telomere shortening and increase in cellular senescence markers in human lung epithelial cells. Ecotoxicol. Environ. Saf. 222, 112484 (2021).
Park, B. et al. In International Forum of Allergy & Rhinology, Vol. 12 1424 (Wiley-Blackwell, 2022).
Tsai, J. J. et al. Nrf2 regulates haematopoietic stem cell function. Nat. Cell Biol. 15, 309–316 (2013).
Dai, X. et al. Nrf2: redox and metabolic regulator of stem cell state and function. Trends Mol. Med 26, 185–200 (2020).
Wang, Y. et al. Total body irradiation causes residual bone marrow injury by induction of persistent oxidative stress in murine hematopoietic stem cells. Free Radic. Biol. Med 48, 348–356 (2010).
Wu, F. & Zhang, J. The involvement of Nox4 in fine particulate matter exposure-induced cardiac injury in mice. J. Toxicological Sci. 43, 171–181 (2018).
Ito, K. et al. Reactive oxygen species act through p38 MAPK to limit the lifespan of hematopoietic stem cells. Nat. Med. 12, 446–451 (2006).
Iwasa, H., Han, J. & Ishikawa, F. Mitogen‐activated protein kinase p38 defines the common senescence‐signalling pathway. Genes Cells 8, 131–144 (2003).
Ma, Y. et al. Cadmium exposure triggers osteoporosis in duck via P2X7/PI3K/AKT-mediated osteoblast and osteoclast differentiation. Sci. Total Environ. 750, 141638 (2021).
Saha, H., Mukherjee, B., Bindhani, B. & Ray, M. R. Changes in RANKL and osteoprotegerin expression after chronic exposure to indoor air pollution as a result of cooking with biomass fuel. J. Appl Toxicol. 36, 969–976 (2016).
Abu-Elmagd, M. et al. Evaluation of the effects of airborne particulate matter on bone marrow-mesenchymal stem cells (BM-MSCs): cellular, molecular and systems biological approaches. Int. J. Environ. Res. Public Health 14, 440 (2017).
Altekruse, S. SEER cancer statistics review, 1975–2007. http://seer.cancer.gov/csr/1975_2007/results_merged/sect_13_leukemia.pdf (2009).
Humphries, F., Yang, S., Wang, B. & Moynagh, P. N. RIP kinases: key decision makers in cell death and innate immunity. Cell Death Differ. 22, 225–236 (2015).
Lee, M.-S. et al. Oxidative stress and systemic inflammation as modifiers of cardiac autonomic responses to particulate air pollution. Int. J. Cardiol. 176, 166–170 (2014).
Corsini, E. et al. Comparison of wood smoke PM2. 5 obtained from the combustion of FIR and beech pellets on inflammation and DNA damage in A549 and THP-1 human cell lines. Arch. Toxicol. 87, 2187–2199 (2013).
Ostro, B. et al. Chronic PM2. 5 exposure and inflammation: determining sensitive subgroups in mid-life women. Environ. Res. 132, 168–175 (2014).
Jin, X., Su, R., Li, R., Cheng, L. & Li, Z. Crucial role of pro-inflammatory cytokines from respiratory tract upon PM2. 5 exposure in causing the BMSCs differentiation in cells and animals. Oncotarget 9, 1745 (2018).
Lambeth, J. D. NOX enzymes and the biology of reactive oxygen. Nat. Rev. Immunol. 4, 181–189 (2004).
Jin, X. et al. Amelioration of particulate matter-induced oxidative damage by vitamin C and quercetin in human bronchial epithelial cells. Chemosphere 144, 459–466 (2016).
Manz, M. G. & Boettcher, S. Emergency granulopoiesis. Nat. Rev. Immunol. 14, 302–314 (2014).
Jacobsen, S. E. W. & Nerlov, C. Haematopoiesis in the era of advanced single-cell technologies. Nat. Cell Biol. 21, 2–8 (2019).
Organization, W. H. Ambient air pollution: A global assessment of exposure and burden of disease. (2016).
Ferrucci, L. & Balducci, L. In Seminars in hematology, Vol. 45 242–249 (Elsevier, 2008).
Kido, T. et al. Particulate matter induces translocation of IL-6 from the lung to the systemic circulation. Am. J. Respir. Cell Mol. Biol. 44, 197–204 (2011).
Wardoyo, A. Y., Juswono, U. P. & Noor, J. A. How exposure to ultrafine and fine particles of car smoke can alter erythrocyte forms of male mice. Pol. J. Environ. Stud. 28, 2901 (2019).
Lavigne, É. et al. Maternal exposure to ambient air pollution and risk of early childhood cancers: a population-based study in Ontario, Canada. Environ. Int. 100, 139–147 (2017).
Hvidtfeldt, U. A. et al. Residential exposure to PM2. 5 components and risk of childhood non-hodgkin lymphoma in Denmark: a nationwide register-based case-control Study. Int. J. Environ. Res. Public Health 17, 8949 (2020).
Sanchez-Guerra, M. et al. Effects of particulate matter exposure on blood 5-hydroxymethylation: results from the Beijing truck driver air pollution study. Epigenetics 10, 633–642 (2015).
Cortessis, V. K. et al. Environmental epigenetics: prospects for studying epigenetic mediation of exposure–response relationships. Hum. Genet. 131, 1565–1589 (2012).
Hou, L., Zhang, X., Wang, D. & Baccarelli, A. Environmental chemical exposures and human epigenetics. Int. J. Epidemiol. 41, 79–105 (2012).
Madakashira, B. P. & Sadler, K. C. DNA methylation, nuclear organization, and cancer. Front. Genet. 8, 76 (2017).
Koch, A. et al. Analysis of DNA methylation in cancer: location revisited. Nat. Rev. Clin. Oncol. 15, 459–466 (2018).
Zhang, Z. et al. Long-term exposure to ambient particulate matter (PM2. 5) is associated with platelet counts in adults. Environ. Pollut. 240, 432–439 (2018).
Robertson, S. & Miller, M. R. Ambient air pollution and thrombosis. Part. Fibre Toxicol. 15, 1–16 (2018).
Brook, R. D. et al. Particulate matter air pollution and cardiovascular disease: an update to the scientific statement from the American Heart Association. Circulation 121, 2331–2378 (2010).
Liang, S. et al. Repeat dose exposure of PM2. 5 triggers the disseminated intravascular coagulation (DIC) in SD rats. Sci. Total Environ. 663, 245–253 (2019).
Rückerl, R. et al. Associations between ambient air pollution and blood markers of inflammation and coagulation/fibrinolysis in susceptible populations. Environ. Int. 70, 32–49 (2014).
Hajat, A. et al. Long-term exposure to air pollution and markers of inflammation, coagulation, and endothelial activation: a repeat-measures analysis in the Multi-Ethnic Study of Atherosclerosis (MESA). Epidemiol. (Camb., Mass.) 26, 310 (2015).
Hystad, P. et al. Associations of outdoor fine particulate air pollution and cardiovascular disease in 157 436 individuals from 21 high-income, middle-income, and low-income countries (PURE): a prospective cohort study. Lancet Planet Health 4, e235–e245 (2020).
Gao, X. et al. Short-term exposure to PM2. 5 components and renal health: Findings from the Veterans Affairs Normative Aging Study. J. Hazard Mater. 420, 126557 (2021).
Liu, L., Zhang, Y., Yang, Z., Luo, S. & Zhang, Y. Long-term exposure to fine particulate constituents and cardiovascular diseases in Chinese adults. J. Hazard Mater. 416, 126051 (2021).
Lao, X. Q. et al. Long-term exposure to ambient fine particulate matter (PM 2.5) and incident type 2 diabetes: a longitudinal cohort study. Diabetologia 62, 759–769 (2019).
Gilcrease, G. W. et al. Is air pollution affecting the disease activity in patients with systemic lupus erythematosus? State of the art and a systematic literature review. Eur. J. Rheumatol. 7, 31 (2020).
Pun, V. C., Kazemiparkouhi, F., Manjourides, J. & Suh, H. H. Long-term PM2. 5 exposure and respiratory, cancer, and cardiovascular mortality in older US adults. Am. J. Epidemiol. 186, 961–969 (2017).
Essers, M. A. et al. IFNα activates dormant haematopoietic stem cells in vivo. Nature 458, 904–908 (2009).
Walter, D. et al. Exit from dormancy provokes DNA-damage-induced attrition in haematopoietic stem cells. Nature 520, 549–552 (2015).
Pietras, E. M. et al. Chronic interleukin-1 exposure drives haematopoietic stem cells towards precocious myeloid differentiation at the expense of self-renewal. Nat. Cell Biol. 18, 607–618 (2016).
Pronk, C. J., Veiby, O. P., Bryder, D. & Jacobsen, S. E. W. Tumor necrosis factor restricts hematopoietic stem cell activity in mice: involvement of two distinct receptors. J. Exp. Med. 208, 1563–1570 (2011).
Makris, S. et al. Immune function and dysfunction are determined by lymphoid tissue efficacy. Dis. Model. Mech. 15, dmm049256 (2022).
Jung, E. M. et al. Association between prenatal exposure to PM2. 5 and the increased risk of specified infant mortality in South Korea. Environ. Int 144, 105997 (2020).
Ortigoza, A. et al. Association between ambient PM2· 5 and under-5, infant, and child mortality in Latin America, 2010–15: a longitudinal analysis. 5, S16 (2021).
Bo, Y. et al. Associations of reduced ambient PM2. 5 level with lower plasma glucose concentration and decreased risk of type 2 diabetes in adults: a longitudinal cohort study. Am. J. Epidemiol. 190, 2148–2157 (2021).
Hayes, R. B. et al. PM2. 5 air pollution and cause-specific cardiovascular disease mortality. Int J. Epidemiol. 49, 25–35 (2020).
Bowe, B. et al. Particulate matter air pollution and the risk of incident CKD and progression to ESRD. J. Am. Soc. Nephrol. 29, 218 (2018).
Lee, J.-T. J. C. & Pediatrics, E. Review of epidemiological studies on air pollution and health effects in children. Clin. Exp. Pediatr. 64, 3 (2021).
Li, G., Li, L., Liu, D., Qin, J. & Zhu, H. J. S. R. Effect of PM2. 5 pollution on perinatal mortality in China. Sci. Rep. 11, 7596 (2021).
Alman, B. L. et al. Associations between PM2. 5 and risk of preterm birth among liveborn infants. Ann. Epidemiol. 39, 46–53 (2019).
Tao, R.-j et al. PM2. 5 compromises antiviral immunity in influenza infection by inhibiting activation of NLRP3 inflammasome and expression of interferon-β. Mol. Immunol. 125, 178–186 (2020).
Prada, D., López, G., Solleiro-Villavicencio, H., Garcia-Cuellar, C. & Baccarelli, A. A. Molecular and cellular mechanisms linking air pollution and bone damage. Environ. Res 185, 109465 (2020).
Qiu, Y.-N. et al. PM2. 5 induces liver fibrosis via triggering ROS-mediated mitophagy. Ecotoxicol. Environ. Saf. 167, 178–187 (2019).
Li, R., Zhou, R. & Zhang, J. Function of PM2. 5 in the pathogenesis of lung cancer and chronic airway inflammatory diseases. Oncol. Lett. 15, 7506–7514 (2018).
Chen, S. et al. Effect of PM2. 5 on macrosomia in China: A nationwide prospective cohort study. Pediatr. Obes. 15, e12584 (2020).
Nääv, Å. et al. Urban PM2. 5 induces cellular toxicity, hormone dysregulation, oxidative damage, inflammation, and mitochondrial interference in the HRT8 trophoblast cell line. Front Endocrinol. (Lausanne) 11, 75 (2020).
Zhou, S. et al. Ovarian dysfunction induced by chronic whole‐body PM2. 5 exposure. Small 16, 2000845 (2020).
int/mediacentre/factsheets/fs313/en/, W.H.O.J.R.f.W.H.O.A.f.h.w.w. Ambient (outdoor) air quality and health. 2018 (2016).
Wu, J. et al. The association between long-term exposure to ambient air pollution and bone strength in China. J. Clin. Endocrinol. Metab. 106, e5097–e5108 (2021).
Yang, Y. et al. Ambient air pollution, bone mineral density and osteoporosis: results from a national population-based cohort study. Chemosphere 310, 136871 (2023).
Qiao, D. et al. Long-term exposure to air pollution might increase prevalence of osteoporosis in Chinese rural population. Environ. Res 183, 109264 (2020).
Yu, P. et al. Associations between long-term exposure to PM2. 5 and site-specific cancer mortality: A nationwide study in Brazil between 2010 and 2018. Environ. Pollut. 302, 119070 (2022).
Morales-Ancajima, V. C. et al. Increased outdoor PM2. 5 concentration is associated with moderate/severe anemia in children aged 6–59 months in Lima, Peru. J. Environ. Public Health 2019, 6127845 (2019).
Honda, T., Pun, V. C., Manjourides, J. & Suh, H. Anemia prevalence and hemoglobin levels are associated with long-term exposure to air pollution in an older population. Environ. Int. 101, 125–132 (2017).
Visani, G. et al. Environmental nanoparticles are significantly over-expressed in acute myeloid leukemia. Leuk. Res. 50, 50–56 (2016).
Giannoni, P. et al. Chronic lymphocytic leukemia cells impair osteoblastogenesis and promote osteoclastogenesis: role of TNFα, IL-6 and IL-11 cytokines. Haematologica 106, 2598 (2021).
Wells, A. et al. Systemic IL-6 effector response in mediating systemic bone loss following inhalation of organic dust. J. Interferon Cytokine Res. 37, 9–19 (2017).
Calderón-Garcidueñas, L. et al. Exposure to urban air pollution and bone health in clinically healthy six-year-old children. Arh. Hig. Rada Toksikol. 64, 23–23 (2013).
Chen, Y. et al. Probucol protects circulating endothelial progenitor cells from ambient PM2. 5 damage via inhibition of reactive oxygen species and inflammatory cytokine production in vivo. Exp. Ther. Med. 16, 4322–4328 (2018).
Liberda, E. N. et al. Exposure to inhaled nickel nanoparticles causes a reduction in number and function of bone marrow endothelial progenitor cells. Inhal. Toxicol. 22, 95–99 (2010).
Haberzettl, P. et al. Exposure to ambient air fine particulate matter prevents VEGF-induced mobilization of endothelial progenitor cells from the bone marrow. Environ. Health Perspect. 120, 848–856 (2012).
O’Toole, T. E. et al. Episodic exposure to fine particulate air pollution decreases circulating levels of endothelial progenitor cells. Circ. Res 107, 200–203 (2010).
Yamaguchi, M. & Kashiwakura, I. Role of reactive oxygen species in the radiation response of human hematopoietic stem/progenitor cells. PLoS One 8, e70503 (2013).
Rodrigues-Moreira, S. et al. Low-dose irradiation promotes persistent oxidative stress and decreases self-renewal in hematopoietic stem cells. Cell Rep. 20, 3199–3211 (2017).
Eliasson, P. et al. Hypoxia mediates low cell-cycle activity and increases the proportion of long-term–reconstituting hematopoietic stem cells during in vitro culture. Exp. Hematol. 38, 301–310 (2010).
Ito, K. et al. Regulation of oxidative stress by ATM is required for self-renewal of haematopoietic stem cells. Nature 431, 997–1002 (2004).
Renzi, M. et al. Short-term exposure to PM2. 5 and risk of venous thromboembolism: A case-crossover study. Thromb. Res 190, 52–57 (2020).
Cliff, R. et al. Effect of diesel exhaust inhalation on blood markers of inflammation and neurotoxicity: a controlled, blinded crossover study. Inhal. Toxicol. 28, 145–153 (2016).
Orona, N. S. et al. Acute exposure to Buenos Aires air particles (UAP-BA) induces local and systemic inflammatory response in middle-aged mice: A time course study. Environ. Pollut. 208, 261–270 (2016).
Park, S.-R. et al. The impact of fine particulate matter (PM) on various beneficial functions of human endometrial stem cells through its key regulator SERPINB2. Exp. Mol. Med 53, 1850–1865 (2021).
Acknowledgements
This work was supported by the Basic Science Research Program of the National Research Foundation of Korea funded by the Ministry of Science, Information and Communications Technology and Future Planning, Republic of Korea (2019R1A2C2084453, 2020R1C1C1004968, 2021R1A2C2006032, and 2021R1I1A1A01044453).
Author information
Authors and Affiliations
Contributions
G.B., J.C.L., and S.H.K. conceptualized and supervised manuscript preparation. All the authors contributed to the drafting of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Bhattarai, G., Shrestha, S.K., Sim, HJ. et al. Effects of fine particulate matter on bone marrow-conserved hematopoietic and mesenchymal stem cells: a systematic review. Exp Mol Med 56, 118–128 (2024). https://doi.org/10.1038/s12276-023-01149-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s12276-023-01149-z
- Springer Nature Limited