Abstract
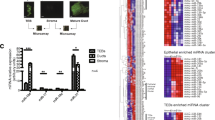
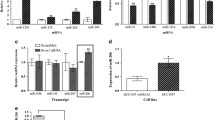
MicroRNAs are small noncoding RNAs that carry out post-transcriptional regulation of the expression of their target genes. However, their roles in mammalian organogenesis are only beginning to be understood. Here we show that the microRNA-212/132 family (which comprises miR-212 and miR-132) is indispensable during the development of the mammary glands in mice, particulary for the regulation of the outgrowth of the epithelial ducts. Mammary transplantation experiments revealed that the function of the miR-212/132 family is required in the stroma but not in the epithelia. Both miR-212 and miR-132 are expressed exclusively in mammary stroma and directly target the matrix metalloproteinase MMP-9. In glands that lack miR-212 and miR-132, MMP-9 expression increases and accumulates around the ducts. This may interfere with collagen deposition and lead to hyperactivation of the tumor growth factor-β signaling pathway, thereby impairing ductal outgrowth. Our results identify the miR-212/132 family as one of the main regulators of the epithelial-stromal interactions that are required for proper pubertal development of the mammary gland.







Similar content being viewed by others
Accession codes
References
Valencia-Sanchez, M.A., Liu, J., Hannon, G.J. & Parker, R. Control of translation and mRNA degradation by miRNAs and siRNAs. Genes Dev. 20, 515–524 (2006).
Bartel, D.P. MicroRNAs: Target recognition and regulatory functions. Cell 136, 215–233 (2009).
Zhao, Y. & Srivastava, D. A developmental view of microRNA function. Trends Biochem. Sci. 32, 189–197 (2007).
Smibert, P. & Lai, E.C. Lessons from microRNA mutants in worms, flies and mice. Cell Cycle 7, 2500–2508 (2008).
Vo, N. et al. A cAMP-response element binding protein-induced microRNA regulates neuronal morphogenesis. Proc. Natl. Acad. Sci. USA 102, 16426–16431 (2005).
Wayman, G.A. et al. An activity-regulated microRNA controls dendritic plasticity by down-regulating p250GAP. Proc. Natl. Acad. Sci. USA 105, 9093–9098 (2008).
Impey, S. et al. An activity-induced microRNA controls dendritic spine formation by regulating Rac1-PAK signaling. Mol. Cell. Neurosci. 43, 146–156 (2010).
Kawashima, H. et al. Glucocorticoid attenuates brain-derived neurotrophic factor-dependent upregulation of glutamate receptors via the suppression of microRNA-132 expression. Neuroscience 165, 1301–1311 (2010).
Cheng, H.Y. et al. MicroRNA modulation of circadian-clock period and entrainment. Neuron 54, 813–829 (2007).
Shaked, I. et al. MicroRNA-132 potentiates cholinergic anti-inflammatory signaling by targeting acetylcholinesterase. Immunity 31, 965–973 (2009).
Howlin, J., McBryan, J. & Martin, F. Pubertal mammary gland development: Insights from mouse models. J. Mammary Gland Biol. Neoplasia 11, 283–297 (2006).
Richert, M.M. et al. An atlas of mouse mammary gland development. J. Mammary Gland Biol. Neoplasia 5, 227–241 (2000).
Hennighausen, L. & Robinson, G.W. Signaling pathways in mammary gland development. Dev. Cell 1, 467–475 (2001).
Watson, C.J. & Khaled, W.T. Mammary development in the embryo and adult: a journey of morphogenesis and commitment. Development 135, 995–1003 (2008).
Hennighausen, L. & Robinson, G.W. Information networks in the mammary gland. Nat. Rev. Mol. Cell Biol. 6, 715–725 (2005).
Parmar, H. & Cunha, G.R. Epithelial-stromal interactions in the mouse and human mammary gland in vivo. Endocr. Relat. Cancer 11, 437–458 (2004).
Neville, M.C., Medina, D., Monks, J. & Hovey, R.C. The mammary fat pad. J. Mammary Gland Biol. Neoplasia 3, 109–116 (1998).
Wiseman, B.S. & Werb, Z. Stromal effects on mammary gland development and breast cancer. Science 296, 1046–1049 (2002).
Remenyi, J. et al. Regulation of the miR-212/132 locus by MSK1 and CREB in response to neurotrophins. Biochem. J. 428, 281–291 (2010).
Mailleux, A.A., Overholtzer, M. & Brugge, J.S. Lumen formation during mammary epithelial morphogenesis: insights from in vitro and in vivo models. Cell Cycle 7, 57–62 (2008).
Fiedler, S.D., Carletti, M.Z., Hong, X. & Christenson, L.K. Hormonal regulation of microRNA expression in periovulatory mouse mural granulosa cells. Biol. Reprod. 79, 1030–1037 (2008).
Silberstein, G.B., Strickland, P., Coleman, S. & Daniel, C.W. Epithelium-dependent extracellular matrix synthesis in transforming growth factor-beta 1-growth-inhibited mouse mammary gland. J. Cell Biol. 110, 2209–2219 (1990).
Daniel, C.W., Robinson, S. & Silberstein, G.B. The role of TGF-beta in patterning and growth of the mammary ductal tree. J. Mammary Gland Biol. Neoplasia 1, 331–341 (1996).
Silberstein, G.B. Postnatal mammary gland morphogenesis. Microsc. Res. Tech. 52, 155–162 (2001).
Wiseman, B.S. et al. Site-specific inductive and inhibitory activities of MMP-2 and MMP-3 orchestrate mammary gland branching morphogenesis. J. Cell Biol. 162, 1123–1133 (2003).
Robinson, S.D. et al. Regulated expression and growth inhibitory effects of transforming growth factor-beta isoforms in mouse mammary gland development. Development 113, 867–878 (1991).
Kang, J.S., Liu, C. & Derynck, R. New regulatory mechanisms of TGF-beta receptor function. Trends Cell Biol. 19, 385–394 (2009).
Yu, Q. & Stamenkovic, I. Cell surface-localized matrix metalloproteinase-9 proteolytically activates TGF-beta and promotes tumor invasion and angiogenesis. Genes Dev. 14, 163–176 (2000).
Wilson, T.J., Nannuru, K.C. & Singh, R.K. Cathepsin G-mediated activation of pro-matrix metalloproteinase 9 at the tumor-bone interface promotes transforming growth factor-beta signaling and bone destruction. Mol. Cancer Res. 7, 1224–1233 (2009).
Okamoto, T. et al. Transforming growth factor-beta 1 induces matrix metalloproteinase-9 expression in human meningeal cells via ERK and Smad pathways. Biochem. Biophys. Res. Commun. 383, 475–479 (2009).
Sinpitaksakul, S.N., Pimkhaokham, A., Sanchavanakit, N. & Pavasant, P. TGF-beta 1 induced MMP-9 expression in HNSCC cell lines via Smad/MLCK pathway. Biochem. Biophys. Res. Commun. 371, 713–718 (2008).
Seomun, Y., Kim, J.T. & Joo, C.K. MMP-14 mediated MMP-9 expression is involved in TGF-beta 1-induced keratinocyte migration. J. Cell. Biochem. 104, 934–941 (2008).
Chou, Y.T., Wang, H., Chen, Y., Danielpour, D. & Yang, Y.C. Cited2 modulates TGF-beta-mediated upregulation of MMP9. Oncogene 25, 5547–5560 (2006).
Konrad, L., Scheiber, J.A., Schwarz, L., Schrader, A.J. & Hofmann, R. TGF-beta1 and TGF-beta2 strongly enhance the secretion of plasminogen activator inhibitor-1 and matrix metalloproteinase-9 of the human prostate cancer cell line PC-3. Regul. Pept. 155, 28–32 (2009).
Howlin, J. et al. CITED1 homozygous null mice display aberrant pubertal mammary ductal morphogenesis. Oncogene 25, 1532–1542 (2006).
Luetteke, N.C. et al. Targeted inactivation of the EGF and amphiregulin genes reveals distinct roles for EGF receptor ligands in mouse mammary gland development. Development 126, 2739–2750 (1999).
Ciarloni, L., Mallepell, S. & Brisken, C. Amphiregulin is an essential mediator of estrogen receptor alpha function in mammary gland development. Proc. Natl. Acad. Sci. USA 104, 5455–5460 (2007).
Wiesen, J.F., Young, P., Werb, Z. & Cunha, G.R. Signaling through the stromal epidermal growth factor receptor is necessary for mammary ductal development. Development 126, 335–344 (1999).
Shioda, T. et al. Transcriptional activating activity of Smad4: roles of SMAD hetero-oligomerization and enhancement by an associating transactivator. Proc. Natl. Acad. Sci. USA 95, 9785–9790 (1998).
Parsa, S. et al. Terminal end bud maintenance in mammary gland is dependent upon FGFR2b signaling. Dev. Biol. 317, 121–131 (2008).
Lu, P., Ewald, A.J., Martin, G.R. & Werb, Z. Genetic mosaic analysis reveals FGF receptor 2 function in terminal end buds during mammary gland branching morphogenesis. Dev. Biol. 321, 77–87 (2008).
Roarty, K. & Serra, R. Wnt5a is required for proper mammary gland development and TGF-β-mediated inhibition of ductal outgrowth. Development 134, 3929–3939 (2007).
Ibarra, I., Erlich, Y., Muthuswamy, S.K., Sachidanandam, R. & Hannon, G.J. A role for microRNAs in maintenance of mouse mammary epithelial progenitor cells. Genes Dev. 21, 3238–3243 (2007).
Kong, W. et al. MicroRNA-155 is regulated by the transforming growth factor beta/Smad pathway and contributes to epithelial cell plasticity by targeting RhoA. Mol. Cell. Biol. 28, 6773–6784 (2008).
Tanaka, T., Haneda, S., Imakawa, K., Sakai, S. & Nagaoka, K. A microRNA, miR-101a, controls mammary gland development by regulating cyclooxygenase-2 expression. Differentiation 77, 181–187 (2009).
Rasmussen, S.B., Young, L.J.T. & Smith, G.H. in Methods in Mammary Gland Biology and Breast Cancer Research. (eds. Ip, M.M. and Asch, B.B.) 75–86 (Kluwer Academic/Plenum, New York, 2000).
Young, L.J.T. in Methods in Mammary Gland Biology and Breast Cancer Research (eds. Ip, M.M. and Asch, B.B.) 67–74 (Kluwer Academic/Plenum, New York, 2000).
Sangai, T. et al. Hormonal stimulation increases the recruitment of bone marrow-derived myoepithelial cells and periductal fibroblasts into the mammary gland. Biochem. Biophys. Res. Commun. 346, 1173–1180 (2006).
Acknowledgements
We thank S. Geisendorf for technical assistance, S. Hille for DNA sequencing, S. Mahsur and U. Franke for technical help in generation of knockout mouse line, A. Kurth, A. Driehorst, K. Kiel and U. Teichman for animal care work, M. Smalley for discussion about mammary epithelial transplantation method, N. Ba Tiep for technical help with collecting blood samples, A. Yalcin for creative contribution to the initial phase of the work, and P. Gruss and G. Eichele for encouragement and support. Funded by the Max Planck Society. Additional funding was provided by the Deutsche Forschungsgemeinschaft (DFG, GR 536/9-2 and GR 536/11-1 to B.G. and TH 903/7-2 to T.T.).
Author information
Authors and Affiliations
Contributions
A.U. and K.C. developed the concept of this study. A.U. performed all experiments except the mammary epithelial transplantation analyses, which were performed by V.V. and P.A.B.K. in the laboratory of B.G., serum hormone level analyses, which were done by H.J., and luciferase assays, which were done by J.F. in the laboratory of T.T. The manuscript was written by A.U. and K.C. Important suggestions were made by B.G., V.V. and T.T. which improved the quality of the manuscript. All authors contributed to the discussion of the data and commented on the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–10, Supplementary Table 1 and Supplementary Note (PDF 9611 kb)
Rights and permissions
About this article
Cite this article
Ucar, A., Vafaizadeh, V., Jarry, H. et al. miR-212 and miR-132 are required for epithelial stromal interactions necessary for mouse mammary gland development. Nat Genet 42, 1101–1108 (2010). https://doi.org/10.1038/ng.709
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng.709
- Springer Nature America, Inc.
This article is cited by
-
RAC1B function is essential for breast cancer stem cell maintenance and chemoresistance of breast tumor cells
Oncogene (2023)
-
Sfrp3 modulates stromal–epithelial crosstalk during mammary gland development by regulating Wnt levels
Nature Communications (2019)
-
Hypoxia-Induced MicroRNA-212/132 Alter Blood-Brain Barrier Integrity Through Inhibition of Tight Junction-Associated Proteins in Human and Mouse Brain Microvascular Endothelial Cells
Translational Stroke Research (2019)
-
Transcriptome sequencing to detect the potential role of long non-coding RNAs in bovine mammary gland during the dry and lactation period
BMC Genomics (2018)
-
Predictive value of microRNA-132 and its target gene NAG-1 in evaluating therapeutic efficacy of non-steroidal anti-inflammatory drugs treatment in patients with ankylosing spondylitis
Clinical Rheumatology (2018)