Abstract
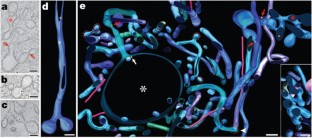
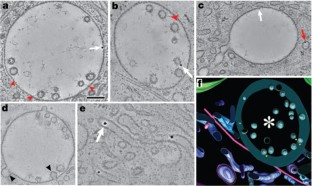
The neonatal Fc receptor (FcRn) transports maternal IgG across epithelial barriers1,2, thereby providing the fetus or newborn with humoral immunity before its immune system is fully functional. In newborn rats, FcRn transfers IgG from milk to blood by apical-to-basolateral transcytosis across intestinal epithelial cells. The pH difference between the apical (pH 6.0–6.5) and basolateral (pH 7.4) sides of intestinal epithelial cells facilitates the efficient unidirectional transport of IgG, because FcRn binds IgG at pH 6.0–6.5 but not at pH 7 or more1,2. As milk passes through the neonatal intestine, maternal IgG is removed by FcRn-expressing cells in the proximal small intestine (duodenum and jejunum); remaining proteins are absorbed and degraded by FcRn-negative cells in the distal small intestine (ileum)3,4,5,6. Here we use electron tomography to make jejunal transcytosis visible directly in space and time, developing new labelling and detection methods to map individual nanogold-labelled Fc within transport vesicles7 and simultaneously to characterize these vesicles by immunolabelling. Combining electron tomography with a non-perturbing endocytic label allowed us to conclusively identify receptor-bound ligands, resolve interconnecting vesicles, determine whether a vesicle was microtubule-associated, and accurately trace FcRn-mediated transport of IgG. Our results present a complex picture in which Fc moves through networks of entangled tubular and irregular vesicles, only some of which are microtubule-associated, as it migrates to the basolateral surface. New features of transcytosis are elucidated, including transport involving multivesicular body inner vesicles/tubules and exocytosis through clathrin-coated pits. Markers for early, late and recycling endosomes each labelled vesicles in different and overlapping morphological classes, revealing spatial complexity in endo-lysosomal trafficking.


Similar content being viewed by others
References
Rodewald, R. & Kraehenbuhl, J.-P. Receptor-mediated transport of IgG. J. Cell Biol. 99, S159–S164 (1984)
Simister, N. E. & Mostov, K. E. An Fc receptor structurally related to MHC class I antigens. Nature 337, 184–187 (1989)
Rodewald, R. Selective antibody transport in the proximal small intestine of the neonatal rat. J. Cell Biol. 45, 635–640 (1970)
Jones, E. A. & Waldmann, T. A. The mechanism of intestinal uptake and transcellular transport of IgG in the neonatal rat. J. Clin. Invest. 51, 2916–2927 (1972)
Rodewald, R. Intestinal transport of antibodies in the newborn rat. J. Cell Biol. 58, 189–211 (1973)
Rodewald, R. Distribution of immunoglobulin G receptors in the small intestine of the young rat. J. Cell Biol. 85, 18–32 (1980)
He, W. et al. A freeze substitution fixation-based gold enlarging technique for EM studies of endocytosed Nanogold-labeled molecules. J. Struct. Biol. 160, 103–111 (2007)
Benlounes, N. et al. Intestinal transport and processing of immunoglobulin G in the neonatal and adult rat. Biol. Neonate 67, 254–263 (1995)
McIntosh, J. R., Nicastro, D. & Mastronarde, D. N. New views of cells in 3D: an introduction to electron tomography. Trends Cell Biol. 15, 43–51 (2005)
Morphew, M., He, W., Bjorkman, P. J. & McIntosh, J. R. Silver enhancement of nanogold particles during freeze substitution fixation for electron microscopy. J. Microsc. 230, 263–267 (2008)
Hull, B. E. & Staehelin, L. A. The terminal web. A reevaluation of its structure and function. J. Cell Biol. 81, 67–82 (1979)
Schwartz, S. L., Cao, C., Pylypenko, O., Rak, A. & Wandinger-Ness, A. Rab GTPases at a glance. J. Cell Sci. 120, 3905–3910 (2007)
Stoorvogel, W., Oorschot, V. & Geuze, H. J. A novel class of clathrin-coated vesicles budding from endosomes. J. Cell Biol. 132, 21–33 (1996)
McCarthy, K. M., Yoong, Y. & Simister, N. E. Bidirectional transcytosis of IgG by the rat neonatal Fc receptor expressed in a rat kidney cell line: a system to study protein transport across epithelia. J. Cell Sci. 113, 1277–1285 (2000)
Murk, J. L. A. N. et al. Endosomal compartmentalization in three dimensions: Implications for membrane fusion. Proc. Natl Acad. Sci. USA 100, 13332–13337 (2003)
Kleijmeer, M. et al. Reorganization of multivesicular bodies regulates MHC class II antigen presentation by dendritic cells. J. Cell Biol. 155, 53–63 (2001)
Mari, M. et al. SNX1 defines an early endosomal recycling exit for sortilin and mannose 6-phosphate receptors. Traffic 9, 380–393 (2008)
Altstiel, L. & Branton, D. Fusion of coated vesicles with lysosomes: measurement with a fluorescence assay. Cell 32, 921–929 (1983)
Brodsky, F. M., Chen, C. Y., Knuehl, C., Towler, M. C. & Wakeham, D. E. Biological basket weaving: formation and function of clathrin-coated vesicles. Annu. Rev. Cell Dev. Biol. 17, 517–568 (2001)
Conner, S. D. & Schmid, S. L. Regulated portals of entry into the cell. Nature 422, 37–44 (2003)
Bonifacino, J. S. & Glick, B. S. The mechanisms of vesicle budding and fusion. Cell 116, 153–166 (2004)
Greene, L. E. & Eisenberg, E. Dissociation of clathrin from coated vesicles by the uncoating ATPase. J. Biol. Chem. 265, 6682–6687 (1990)
Fesce, R., Grohovaz, F., Valtorta, F. & Meldolesi, J. Neurotransmitter release: fusion or ‘kiss-and-run’? Trends Cell Biol. 4, 1–4 (1994)
Ober, R. J., Martinez, C., Lai, X., Zhou, J. & Ward, E. S. Exocytosis of IgG as mediated by the receptor, FcRn: an analysis at the single-molecule level. Proc. Natl Acad. Sci. USA 101, 11076–11081 (2004)
Sokac, A. M. & Bement, W. M. Kiss-and-coat and compartment mixing: coupling exocytosis to signal generation and local actin assembly. Mol. Biol. Cell 17, 1495–1502 (2006)
Granseth, B., Odermatt, B., Royle, S. J. & Lagnado, L. Clathrin-mediated endocytosis is the dominant mechanism of vesicle retrieval at hippocampal synapses. Neuron 51, 773–786 (2006)
Ghetie, V. & Ward, E. S. Multiple roles for the major histocompatibility complex class I-related FcRn. Annu. Rev. Immunol. 18, 739–766 (2000)
Mastronarde, D. N. Dual-axis tomography: an approach with alignment methods that preserve resolution. J. Struct. Biol. 120, 343–352 (1997)
Ladinsky, M. S. & Howell, K. E. Electron tomography of immunolabeled cryosections. Methods Cell Biol. 79, 543–548 (2007)
Huber, A. H., Kelley, R. F., Gastinel, L. N. & Bjorkman, P. J. Crystallization and stoichiometry of binding of a complex between a rat intestinal Fc receptor and Fc. J. Mol. Biol. 230, 1077–1083 (1993)
Halliday, R. The absorption of antibodies from immune sera by the gut of the young rat. Proc. R. Soc. Lond. B 143, 408–413 (1955)
Danscher, G. Localization of gold in biological tissue. A photochemical method for light and electron microscopy. Histochemistry 71, 1–16 (1981)
He, W., Cowin, P. & Stokes, D. L. Untangling desmosomal knots with electron tomography. Science 302, 109–113 (2003)
Mastronarde, D. N. Automated electron microscope tomography using robust prediction of specimen movements. J. Struct. Biol. 152, 36–51 (2005)
Kremer, J. R., Mastronarde, D. N. & McIntosh, J. R. Computer visualization of three-dimensional data using IMOD. J. Struct. Biol. 116, 71–76 (1996)
Ladinsky, M. S., Mastronarde, D. N., McIntosh, J. R., Howell, K. E. & Staehelin, L. A. Golgi structure in three dimensions: functional insights from the normal rat kidney cell. J. Cell Biol. 144, 1135–1149 (1999)
Tokuyasu, K. T. Application of cryoultramicrotomy to immunocytochemistry. J. Microsc. 143, 139–149 (1986)
Acknowledgements
We thank D. Mastronarde for advice about setting up SerialEM on the Caltech microscopes and three-dimensional modelling; C. Kivork for preparing Au-Fc and Au-dextran; B. Tivol for assistance with electron microscopes; M. Murphy for help with schematic figures; M. Morphew for contributions to silver enhancement methods; Y. Nie, A. Feuerabendt, A. Kules, C. Luna, K. McKenzie, R. Sander and L. Zinn-Bjorkman for segmenting tomograms; J. Vielmetter for SPR studies of Au-Fc; and F. Brodsky, F. Maxfield, S. Schmid and D. Tesar for discussions. This work was supported by the National Institutes of Health (2 R37 AI041239-06A1 to P.J.B. and RR000592 to J.R.M.), a Max Planck Research Award (P.J.B.), gifts from the Gordon and Betty Moore Foundation and the Agouron Institute to support electron microscopy at Caltech, and National University of Singapore AcRF start-up funds (R-154-000-339-133 to W.H.).
Author Contributions W.H. and P.J.B. conceived the experiments. Electron microscopy data were collected by W.H. in collaboration with and in the microscopy laboratory established by G.J.J. at Caltech. W.H. configured SerialEM on the Caltech electron microscopes, prepared intestinal samples by chemical fixation or HPF/FSF, conceived and developed the gold enhancement procedures, and collected, processed, interpreted and modelled tomograms. W.H. and K.E.H.T conducted kinetic experiments. Immunolabelling and associated imaging was done by M.S.L. in Boulder with enhanced samples provided by W.H. and K.E.H.T. Initial phases of the project using transfected cells were conducted by P.J.B. in Boulder in collaboration with J.R.M. and the Boulder Laboratory for 3D Microscopy of Cells. All authors discussed and interpreted the results and edited the manuscript.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Information
This file contains Supplementary Figures S1-S11 with Legends, Supplementary Tables S1-S3, Supplementary Results and Discussion, Supplementary Movie Legends 1-8 and Supplementary References (PDF 30861 kb)
Supplementary Movie 1
This file contains Supplementary Movie 1 (MOV 8125 kb)
Supplementary Movie 2
This file contains Supplementary Movie 2 (MOV 8275 kb)
Supplementary Movie 3
This file contains Supplementary Movie 3 (MOV 10088 kb)
Supplementary Movie 4
This file contains Supplementary Movie 4 (MOV 10489 kb)
Supplementary Movie 5
This file contains Supplementary Movie 5 (MOV 10410 kb)
Supplementary Movie 6
This file contains Supplementary Movie 6 (MOV 5110 kb)
Supplementary Movie 7
This file contains Supplementary Movie 7 (MOV 9878 kb)
Supplementary Movie 8
This file contains Supplementary Movie 8 (MOV 9591 kb)
Rights and permissions
About this article
Cite this article
He, W., Ladinsky, M., Huey-Tubman, K. et al. FcRn-mediated antibody transport across epithelial cells revealed by electron tomography. Nature 455, 542–546 (2008). https://doi.org/10.1038/nature07255
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature07255
- Springer Nature Limited
This article is cited by
-
The application and development of electron microscopy for three-dimensional reconstruction in life science: a review
Cell and Tissue Research (2024)
-
Maternal brain reactive antibodies profile in autism spectrum disorder: an update
Translational Psychiatry (2023)
-
Gut–liver axis: barriers and functional circuits
Nature Reviews Gastroenterology & Hepatology (2023)
-
An FcRn-targeted mucosal vaccine against SARS-CoV-2 infection and transmission
Nature Communications (2023)
-
Transport of Designed Ankyrin Repeat Proteins through reconstituted human bronchial epithelia and protection against SARS-CoV-2
Scientific Reports (2023)