Abstract
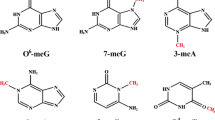
Repair of DNA damage is essential for maintaining genome integrity, and repair deficiencies in mammals are associated with cancer, neurological disease and developmental defects1. Alkylation damage in DNA is repaired by at least three different mechanisms, including damage reversal by oxidative demethylation of 1-methyladenine and 3-methylcytosine by Escherichia coli AlkB2,3. By contrast, little is known about consequences and cellular handling of alkylation damage to RNA4. Here we show that two human AlkB homologues, hABH2 and hABH3, also are oxidative DNA demethylases and that AlkB and hABH3, but not hABH2, also repair RNA. Whereas AlkB and hABH3 prefer single-stranded nucleic acids, hABH2 acts more efficiently on double-stranded DNA. In addition, AlkB and hABH3 expressed in E. coli reactivate methylated RNA bacteriophage MS2 in vivo, illustrating the biological relevance of this repair activity and establishing RNA repair as a potentially important defence mechanism in living cells. The different catalytic properties and the different subnuclear localization patterns shown by the human homologues indicate that hABH2 and hABH3 have distinct roles in the cellular response to alkylation damage.




Similar content being viewed by others
References
Friedberg, E. C., Walker, G. C. & Siede, W. DNA Repair and Mutagenesis (ASM, Washington DC, 1995)
Falnes, P. Ø., Johansen, R. F. & Seeberg, E. AlkB-mediated oxidative demethylation reverses DNA damage in Escherichia coli. Nature 419, 178–182 (2002)
Trewick, S. C., Henshaw, T. F., Hausinger, R. P., Lindahl, T. & Sedgwick, B. Oxidative demethylation by Escherichia coli AlkB directly reverts DNA base damage. Nature 419, 174–178 (2002)
Singer, B. & Grunberger, D. Molecular Biology of Mutagens and Carcinogens (Plenum, New York, 1983)
Boiteux, S. & Laval, J. Mutagenesis by alkylating agents: coding properties for DNA polymerase of poly(dC) template containing 3-methylcytosine. Biochimie 64, 637–641 (1982)
Saffhill, R. Differences in the promutagenic nature of 3-methylcytosine as revealed by DNA and RNA polymerising enzymes. Carcinogenesis 5, 691–693 (1984)
Rozenski, J., Crain, P. F. & McCloskey, J. A. The RNA Modification Database: 1999 update. Nucleic Acids Res. 27, 196–197 (1999)
Agris, P. F. The importance of being modified: roles of modified nucleosides and Mg2+ in RNA structure and function. Prog. Nucleic Acid Res. Mol. Biol. 53, 79–129 (1996)
Yoshizawa, S., Fourmy, D. & Puglisi, J. D. Recognition of the codon–anticodon helix by ribosomal RNA. Science 285, 1722–1725 (1999)
Matsugi, J. & Murao, K. Study on construction of a cDNA library corresponding to an amino acid-specific tRNA and influence of the modified nucleotide upon nucleotide misincorporations in reverse transcription. Biochim. Biophys. Acta 1521, 81–88 (2001)
Haug, T. et al. Human uracil-DNA glycosylase gene: sequence organization, methylation pattern, and mapping to chromosome 12q22–q24.1. Genomics 36, 408–416 (1996)
Wei, Y. F., Carter, K. C., Wang, R. P. & Shell, B. K. Molecular cloning and functional analysis of a human cDNA encoding an Escherichia coli AlkB homolog, a protein involved in DNA alkylation damage repair. Nucleic Acids Res. 24, 931–937 (1996)
Aravind, L. & Koonin, E. V. The DNA-repair protein AlkB, EGL-9, and leprecan define new families of 2-oxoglutarate- and iron-dependent dioxygenases. Genome Biol. 2, Research0007 [online] 〈http://genomebiology.com/2001/2/3/research/0007〉 (2001)
Myllyharju, J. & Kivirikko, K. I. Characterization of the iron- and 2-oxoglutarate-binding sites of human prolyl 4-hydroxylase. EMBO J. 17, 1173–1180 (1997)
Valegård, K. et al. Structure of a cephalosporin synthase. Nature 394, 805–809 (1998)
Nash, T. The colorimetric estimation of formaldehyde by means of the Hantzsch reaction. Biochem. J. 55, 416–421 (1953)
Kaasen, I., Evensen, G. & Seeberg, E. Amplified expression of the tag+ and alkA+ genes in Escherichia coli: identification of gene products and effects on alkylation resistance. J. Bacteriol. 168, 642–647 (1986)
Leonhardt, H. et al. Dynamics of DNA replication factories in living cells. J. Cell Biol. 149, 271–280 (2000)
Warner, J. R. Nascent ribosomes. Cell 107, 133–136 (2001)
Hartl, F. U. & Hayer-Hartl, M. Molecular chaperones in the cytosol: from nascent chain to folded protein. Science 295, 1852–1858 (2002)
Sambrook, J., Fritsch, E. F. & Maniatis, T. Molecular Cloning. A Laboratory Manual, 2nd edn (Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York, 1989)
Yanisch-Perron, C., Vieira, J. & Messing, J. Improved M13 phage cloning vectors and host strains: nucleotide sequences of the M13mp18 and pUC19 vectors. Gene 33, 103–119 (1985)
Bjelland, S., Bjørås, M. & Seeberg, E. Excision of 3-methylguanine from alkylated DNA by 3-methyladenine DNA glycosylase I of Escherichia coli. Nucleic Acids Res. 21, 2045–2049 (1993)
Huang, X., Powell, J., Mooney, L. A., Li, C. & Frenkel, K. Importance of complete DNA digestion in minimizing variability of 8-oxo-dG analyses. Free Radicals Biol. Med. 31, 1341–1351 (2001)
Crain, P. F. Preparation and enzymatic hydrolysis of DNA and RNA for mass spectrometry. Methods Enzymol. 193, 782–790 (1990)
Blatny, J. M., Brautaset, T., Winther-Larsen, H. C., Haugan, K. & Valla, S. Construction and use of a versatile set of broad-host-range cloning and expression vectors based on the RK2 replicon. Appl. Environ. Microbiol. 63, 370–379 (1997)
Dinglay, S., Trewick, S. C., Lindahl, T. & Sedgwick, B. Defective processing of methylated single-stranded DNA by E. coli AlkB mutants. Genes Dev. 14, 2097–2105 (2000)
Acknowledgements
We thank E. Feyzi, K. Baynton and B. Kavli for comments; and A. Nilsen, M. Westbye, R. F. Johansen and L. Hagen for technical support. This work was supported by The Research Council of Norway and the Norwegian Cancer Society. H.E.K. acknowledges support from The Cancer Research Fund at the Regional Hospital in Trondheim, and Svanhild and Arne Must's Fund for Medical Research. E.S. acknowledges support from the European Commission and Simon Fougner Hartmann's Family Fund.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Rights and permissions
About this article
Cite this article
Aas, P., Otterlei, M., Falnes, P. et al. Human and bacterial oxidative demethylases repair alkylation damage in both RNA and DNA. Nature 421, 859–863 (2003). https://doi.org/10.1038/nature01363
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature01363
- Springer Nature Limited
This article is cited by
-
The genome of a vestimentiferan tubeworm (Ridgeia piscesae) provides insights into its adaptation to a deep-sea environment
BMC Genomics (2023)
-
Signaling pathways in brain tumors and therapeutic interventions
Signal Transduction and Targeted Therapy (2023)
-
m1A in CAG repeat RNA binds to TDP-43 and induces neurodegeneration
Nature (2023)
-
Interactions between HIV protease inhibitor ritonavir and human DNA repair enzyme ALKBH2: a molecular dynamics simulation study
Molecular Diversity (2023)
-
DNA damage in IDH-mutant gliomas: mechanisms and clinical implications
Journal of Neuro-Oncology (2023)