Abstract
Background:
Breast cancer is the most common cancer form among women today. Depending on hormone receptor status, breast cancers are divided into different subtypes with vastly varying prognosis. S100A9 is a calcium-binding protein that is associated with inflammation and expressed not only in myeloid cells but also in some tumours. The role for S100A9 in the malignant cells is not well characterised; however, previous studies have shown that the protein could have important immune-modulating properties.
Methods:
Using a human breast cancer cohort consisting of 144 tumour samples and in vitro analysis of human breast cancer cell lines, we investigated the expression and function of S100A9 in human breast cancer.
Results:
We show that S100A9 expression in breast cancer correlated with the ER−PgR− breast tumour subtype (P<0.001) and with Ki67 (P=0.024) and was expressed both in the malignant cells and in the tumour-infiltrating anti-inflammatory CD163+ myeloid cells (P<0.001). Stromal expression of S100A9 also correlated to nodal stage, tumour size and Her2 positivity. Within the ER−PgR− subgroup, all Her2+ and EGFR+ tumours expressed S100A9 in the cytoplasm. Both cytoplasmic staining in the malignant cells as well as stromal S100A9 expression in myeloid cells correlated with a decreased overall survival in breast cancer patients. Furthermore, rS100A9 homodimers induced expression of pro-inflammatory cytokines (IL-6, IL-8 and IL-1β) in a TLR4- and EGFR-dependent manner in human breast cancer cells in vitro.
Conclusion:
We suggest that S100A9 could be viewed as a novel therapeutic target for patients with ER−PgR− breast cancers.
Similar content being viewed by others
Main
Breast cancer is the most common cancer form among women (Kamangar et al, 2006). Clinical breast cancer classification is partly based on hormone receptor status of oestrogen receptor (ER), progesterone receptor (PgR) and Her2 receptor (Sorlie et al, 2001; Allred et al, 2004; Schnitt, 2010). Depending on hormone receptor status, breast cancers are divided into different subtypes with vastly varying prognosis and treatment strategies. ER+ breast cancer patients have the best prognosis. In contrast, hormone receptor-negative breast cancers, that is, ER−, PgR− and Her2− (also denoted triple-negative (TN) subtype) have the worst prognosis (Schnitt, 2010). Although there are numerous treatment options for hormone receptor-positive patients, there are inadequate options for TN breast cancer patients (Schnitt, 2010). Therefore, novel treatment options are urgently needed for this breast cancer subgroup particularly.
Cancer cells are dependent on an uncontrolled and indefinite cell division. This can be mediated by different mutations and mechanisms supporting the cancer cell proliferation. Two typical growth factor receptors that are commonly upregulated in breast cancers are the epidermal growth factor receptor (EGFR/ERBB1) and human epidermal growth factor 2 receptor (Her2/ERBB2) (Slamon et al, 1987; Hanahan and Weinberg, 2000). These are also often mutated to generate a constitutive activation and thus enhanced cell proliferation. The expression of EGFR and Her2 in breast cancers is generally not beneficial for survival (Hanahan and Weinberg, 2000), although patients with Her2+ tumours have a slightly better prognosis owing to specialised treatment options (Schnitt, 2010).
S100A9 is a calcium-binding protein involved in inflammatory processes (Lagasse and Clerc, 1988; Zwadlo et al, 1988; Edgeworth et al, 1991; Hessian et al, 1993; Heizmann et al, 2002; Marenholz et al, 2004) and the protein can form homodimers or heterodimers with S100A8. S100A9 is mainly expressed in neutrophils and myeloid cells where it induces inflammatory cascades. Neutrophils primarily express S100A9/S100A8 heterodimers, thought to be involved in inflammatory diseases. In myeloid cells, it is believed that S100A9 signals as a Ca2+-and Zn2+-dependent damage-associated molecular pattern, via Toll-like receptor 4 (TLR4) and receptor for advanced glycation end products (RAGE) whereby activation of NFκB is induced (Roth et al, 2003; Sunahori et al, 2006; Benedyk et al, 2007; Sinha et al, 2008; Riva et al, 2012). S100A9 has a strong immunosuppressive function in tumour-infiltrating myeloid-derived suppressor cells (MDSCs) (Ostrand-Rosenberg and Sinha, 2009). This is in sharp contrast to the role for S100A9 in tumour-infiltrating neutrophils (Yui et al, 2003). Furthermore, S100A9 has been shown to act as a chemo-attractant for immunosuppressive cells such as MDSCs or anti-inflammatory myeloid cells in tumours (Srikrishna et al, 2001; Ostrand-Rosenberg and Sinha, 2009). The protein expression of S100A9 in breast cancer has previously been studied in smaller cohorts (Arai et al, 2004; Cross et al, 2005; Arai et al, 2008) and in a large protein profiling study performed on breast cancers (Goncalves et al, 2008) where only cytoplasmic S100A9 expression in the malignant cells was scored. The functional role of the protein in malignant breast epithelial cells is controversial (Arai et al, 2004, 2008; Moon et al, 2008; Yin et al, 2013; Cormier et al, 2014; Gumireddy et al, 2014; Bresnick et al, 2015).
We have previously shown that anti-inflammatory myeloid cells are preferentially expressed in the tumour stroma of TN breast tumours (Medrek et al, 2012). The reason to this preferential location is unknown and one factor could be expression of chemo-attractants, such as S100A9. In this study, we investigated the expression and localisation pattern of S100A9 in 144 breast tumours and found a strong correlation not only to ER−PgR− tumours (irrespective of S100A9 localisation) and to the proliferation marker Ki67 (cytoplasmic and stromal localisation) but also to the presence of anti-inflammatory myeloid cells. Stromal S100A9 localisation further correlated to parameters such as larger tumour size, Her2 positivity and nodal stage. Within the ER−PgR− subgroup, all Her2+ and EGFR+ tumours expressed S100A9. This was further supported by gene expression profile analyses where ER, PgR, Her2 or EGFR mRNA expression strongly correlated with S100A9 expression, respectively. Also, stimulation with rEGF induced S100A9 mRNA expression. When breast cancer cell lines were stimulated with homodimers of rS100A9, induction of pro-inflammatory cytokines (IL-6, IL-8 and IL-1β) was observed in a TLR4-dependent manner. This was not observed when stimulating with S100A9/A8 heterodimers. Importantly, cytoplasmic staining of S100A9 in the malignant cells, as well as in stromal myeloid cells, correlated with an impaired overall survival in breast cancer patients. We suggest that S100A9 should be viewed as a potential therapeutic target for patients with ER−PgR− breast cancers.
Material and methods
Cell culture
The human breast cancer cell lines MCF-7, T47-D, MDA-MB-231 and MDA-MB-468 were cultured in RPMI 1640 medium supplemented with 10% fetal bovine serum (Biosera, Boussens, France), 1% sodium pyruvate, 1% HEPES and penicillin/streptomycin (100 U ml−1 and 100 μg ml−1, respectively), CAMA-1 was cultured in MEM/EBSS supplemented with 10% FBS and penicillin/streptomycin, and SKBR3 was cultured in McCoys 5 A medium (without phenol red) supplemented with 10% FBS and penicillin/streptomycin. All cell lines were from ATCC (Wesel, Germany). Media and supplements were purchased from Thermo Scientific HyClone (South Logan, UT, USA) unless otherwise stated.
All stimulations were performed for a total of 20 h. Recombinant human S100A9 (rS100A9) and S100A9/S100A8 was a gift from Active Biotech AB (Lund, Sweden), and a detailed description on endotoxin-free S100A9 and S100A8/A9 generation, purification, bioactivity and binding has been published previously (Bjork et al, 2009; Riva et al, 2012; Bjork et al, 2013; Riva et al, 2013). Both S100A9 and S100A8/A9 were used at a concentration of 10 μg ml−1. As proper S100A9 homodimer activity and binding of S100A9 to its receptors requires the presence of calcium and zinc (Bjork et al, 2009; Markowitz and Carson, 2013), cells were pretreated with 10 μ M ZnCl2 ⩾15 min prior stimulation with rS100A9 at the indicated concentrations. All purchased media contained Ca2+ (⩾200 μ M).
Compounds and cytokine analysis
Antibodies used for S100A9 western blotting (WB), immunoprecipitation (IP) and IHC were Calgranulin B (clone MRP 1H9; Santa Cruz, Dallas, TX, USA), Lamin B (clone C-20; Santa Cruz) and Actin (clone C-4; MP Biomedicals, Solon, OH, USA). Supernatants from rS100A9-stimulated or siRNA-transfected cells were harvested and analysed using human inflammatory cytokine cytometric bead array (BD Biosciences, San Diego, CA, USA) or quantikine human CXCL8/IL-8 immunoassay (R&D Systems, Minneapolis, MN, USA) according to the manufacturers’ instructions. IL-10 and IL-12 were largely undetectable and hence excluded in this study.
Tissue microarray
The large breast cancer cohort analysed in this study consists of 144 patients diagnosed with invasive breast cancer at Skåne University Hospital, Malmö, Sweden between 2001 and 2002. The cohort and TMA has previously been described in detail (Elkabets et al, 2011; Gronberg et al, 2011; Svensson et al, 2011) and (Medrek et al, 2012).
Ethical considerations
Ethical permit was obtained from the local ethical committee at Lund University (Dnr 447/07). Signed informed consent was not collected but the patients were offered to opt out.
Immunohistochemistry
Four-μm-thick TMA sections were mounted onto glass slides and deparaffinised followed by antigen retrieval using the PT-link system (DAKO, Glostrup, Denmark) and stained in a Autostainer Plus (DAKO) with the EnVisionFlex High pH-kit (DAKO). Antibodies used were: anti-Calgranulin B (S100A9; clone MRP 1H9 dilution 1 : 200; Santa Cruz), anti-CD163 (10D6 dilution 1 : 250; Novocastra, GmbH, Nuβloch, Germany), and anti-CD68 (dilution 1 : 1500; DAKO).
Gene expression profile array
Correlations between S100A9 and ER, PgR, EGFR or Her2 in a separate data set (Tumour breast EXPO-351) was performed using the publicly available database R2: microarray analysis and visualisation platform (http://r2.amc.nl).
Quantitative real-time PCR (qRT–PCR)
Total RNA was extracted using the RNeasy Plus Kit according to the manufacturer’s instructions (Qiagen, Hilden, MD, USA). Equal amounts of RNA were used for cDNA synthesis using random hexamers and the M-MuLV reverse transcriptase enzyme (Thermo Scientific). qRT–PCR was performed in triplicates using Maxima SYBR Green/Rox (Thermo Scientific) according to the manufacturer’s instructions. qRT–PCR analysis was performed on the Mx3005P QPCR system (Agilent Technologies, Santa Clara, CA, USA) and the relative mRNA expression was normalised to YWHAZ, UBC and SDHA and calculated using the comparative Ct method (Vandesompele et al, 2002). For primer sequences, see Supplementary Table S1.
Transient transfections and luciferase assays
Transient siRNA transfections were performed using Lipofectamine 2000 according to the manufacturer’s instructions (Invitrogen, Carlsbad, CA, USA). In all, 10 nM of the following silencer select siRNA oligonucleotides were used: Silencer Select Negative Control #2, siS100A9 #1 (s12425), siS100A9 #2 (s12426), siTLR4 #1 (s14194) and siTLR4 #2 (s14195), all purchased from Ambion (Carlsbad, CA, USA). For proliferation and viability assays, the cells were re-seeded into 96-well plates after 24 h. All analyses were performed 72 h posttransfection.
For transient transfection and luciferase assays, breast cancer cells were co-transfected with a total of 0.5 μg pNFκB-luciferase (BD Biosciences) and 0.05 μg TK-renilla-luciferase (Promega, Madison, WI, USA) plasmids, w/wo 1 μg pDUO-MD2-hTLR4 (Invivogen, San Diego, CA, USA) plasmid or 10 nM of the respective siRNA, using Lipofectamine 2000. rS100A9 stimulation was performed 24 h posttransfection as described above. The NFκB activity was analysed using the Dual-Luciferase Reporter Assay System (Promega).
Cell proliferation and viability assays
The effects of rS100A9 or S100A9 knockdown on cell proliferation and viability was assessed using thymidine incorporation, WST-1 assay and Annexin V-staining. In all, 1 μ Ci [methyl-3H] thymidine was added 18 h prior to analysis, and thymidine incorporation was determined in a Microbeta Counter (PerkinElmer, Waltham, MA, USA). The WST-1 assay was performed according to the manufacturer’s recommendations (Roche Applied Science, Indianapolis, IN, USA). For Annexin V staining, cells were detached using EDTA-free trypsin (Thermo Scientific), washed twice in PBS prior to Annexin V-APC and PI staining according to the manufacturer’s instructions (BD Biosciences). The amount of viable cells was analysed using a FACSVerse (BD).
Statistics and annotation
ANOVA or Student’s t-test were used for in vitro experiments as indicated, using the Graph Pad Prism software (La Jolla, CA, USA).
Immunohistochemical S100A9 expression in TMA breast cancer tumours was estimated in fraction (percent) and intensity (0–3) separately annotated for nuclear, cytoplasmic, membrane and stromal compartments. A multiplier of percent and intensity for each core was constructed, yielding a score of 0–3, and a mean value of the two cores was used in the analyses. Spearman’s Rho and χ2 test was used for comparison of CD163, CD68 and S100A9 expression. Classification and regression tree (CRT) was used for cutoff purposes, and Kaplan–Meier analysis with log rank tests were used to illustrate differences in survival. All statistical tests were two sided and P⩽0.05 were considered significant. Calculations were performed with IBM SPSS Statistics version 19.0 (IBM Corp, Armonk, NY, USA).
Results
S100A9 is expressed in ER−PgR− breast cancer cell lines expressing EGFR
To investigate the expression pattern of S100A9 in human breast cancers, we started by verifying a human specific anti-calgranulin B (S100A9) antibody (clone MRP 1H9). To this end, we first characterised the S100A9 expression pattern in six different breast cancer cell lines (MCF-7, T47D, CAMA-1 (all three ER+PgR+) and MDA-MB-231 (TN), MDA-MB-468 (TN EGFR++), SKBR3 (ER−PgR−Her2+)), by using Q-PCR, WB and IHC of paraffin-embedded cell pellets (Figure 1). The endogenous mRNA levels of S100A9 were compared using Q-PCR of the six cell lines (Figure 1A). Very low levels (<0.001 relative to control) of S100A9 were observed not only in all the ER+ cell lines but also in the TN MDA-MB-231 cell line. In contrast, a high level of S100A9 mRNA was found in MDA-MB-468 and SKBR3 cells (Figure 1A). A similar expression pattern could be seen at the protein level using IP with subsequent WB of cell lysates prepared from the cell lines (Figure 1B). IP was used as a weak background band and was present in all cell lysates at the size of S100A9 (14 kDa; data not shown). We then proceeded with an IHC staining of S100A9 on paraffin-embedded cell pellets from the cell lines used (Figure 1C). The cytoplasmic IHC staining was intense in the two cell lines expressing S100A9 (SKBR3 and MDA-MB-468; Figure 1C, right and center). A membranous as well as a nuclear staining was seen in these S100A9-expressing cell lines (Figure 1C). We next silenced S100A9 in MDA-MB-468 cells using siRNA and verified the knockdown of S100A9 and the specificity of the IHC stainings using Q-PCR (Supplementary Figure S1A) and IHC of paraffin-embedded cells (Supplementary Figure S1B), respectively. To confirm the nuclear staining in the S100A9-expressing cells, we also performed nuclear lysates from MDA-MB-468 cells showing that S100A9 was present in both the cytosol and nucleus (Figure 1D).
Analysis of S100A9 protein and mRNA expression in breast cancer cells lines. (A) The relative expression of S100A9 mRNA using Q-PCR on mRNA from the cell lines indicated. Error bars indicate s.e.m. ***P<0.001 using ANOVA test. (B) WB of immunoprecipitated (IP) cell lysates from the indicated cell lines. (C) IHC using an anti-human S100A9-specific antibody on paraffin-embedded cell pellets from the cell lines indicated. (D) WB of nuclear and cytoplasmic fractions. Nuclear extracts was controlled with the Lamin B-specific antibody. Actin shows the loading control for each lane. IHC, IP and WB antibody was the same in panels (A–C).
Cytokine release is induced upon rS100A9 stimulation
The functional role for S100A9 in human breast epithelial cells is not clear (Arai et al, 2004, 2008; Moon et al, 2008; Yin et al, 2013; Cormier et al, 2014; Gumireddy et al, 2014). As S100A9 is known to induce inflammatory cascades in certain cell types, we wanted to investigate whether rS100A9 could do this in malignant epithelial breast cancer cells as well. Using rS100A9 in Ca2+/Zn+ physiological conditions has previously been shown to trigger a conformational change of S100A9 (Bjork et al, 2009; Markowitz and Carson, 2013). We therefore added rS100A9 in Ca2+/Zn2+-high conditions and analysed whether inflammatory cytokines were released upon S100A9 stimulation of breast cancer cell lines. Indeed, in one of the cell lines we found a significantly enhanced expression of the cytokines IL-8, IL-6 and IL-1β at both the protein level (Figure 2A, C and E) and mRNA level (Figure 2B, D and F) upon stimulation with rS100A9 homodimers. This was preferentially seen in MDA-MB-231 cells that lack endogenous expression of S100A9 (TN cell line) and to some extent in the MDA-MB-468 cells (TN EGFR++). Stimulation with S100A9/A8 heterodimers did not induce this cytokine release (Supplementary Figure S1C). Interestingly, only very modest or no decrease in cytokine expression was seen when S100A9 was silenced in the S100A9-expressing cells (Supplementary Figure S1D).
S100A9 induced cytokine expression in human breast cancer cells in vitro. rS100A9 (10 μg ml−1) induced (A) IL-8, (C) IL-6 and (E) IL-1β levels were measured using cytometric bead array (CBA) in supernatants from stimulated breast cancer cell lines as indicated. The relative expression of corresponding mRNA was measured using Q-PCR (B) IL-8, (D) IL-6 and (F) IL-1β. Error bars indicate s.e.m. *P<0.05, **P<0.01, ***P<0.001 using ANOVA test.
S100A9 affects NFκB activity in breast cancer cells
To investigate whether the S100A9-induced cytokine release was caused by an S100A9-induced activation of NFκB activity, we performed NFκB Dual-Luciferase reporter assays. As shown in Figure 3A and B, addition of rS100A9 induced a significant NFκB activity in the MDA-MB-231 cell line but not the MDA-MB-468 or SKBR3 cell lines. Addition of S100A8/A9 heterodimers induced a modest but significant NFκB activity in the MDA-MB-231 cell line (Figure 3B) but as described above absolutely no increase in cytokine expression (Supplementary Figure S1C). The most logical explanation to this is that S100A8/A9-induced NFκB activity is too low for pro-inflammatory cytokine induction. We next performed a Q-PCR analysis of the potential receptors for Ca2+-dependent S100A9 signalling: TLR4 and RAGE (Markowitz and Carson, 2013), and could show that only MDA-MB-231 cells expressed high levels of TLR4 (Figure 3C, left). RAGE was expressed at relatively similar levels (Figure 3C, right). As the S100A9-expressing cell lines SKBR3 and MDA-MB-468 cells did not express TLR4 (Figure 3C, left) but expressed RAGE at varying levels (Figure 3C, right), we next investigated the NFkB activity upon S100A9 silencing in SKBR3 and MDA-MB-468 cells (Figure 3D). Silencing of S100A9 did not promote a decreased NFkB activity in SKBR3 or MDA-MB-468 cells (Figure 3D). In line with this and as mentioned above, only very modest or no decrease in cytokine expression was seen when S100A9 was silenced in the S100A9-expressing cells that lack TLR4 or RAGE (Supplementary Figure S1D). We therefore performed a TLR4-silencing experiment and could show that the rS100A9-induced IL-8 release was decreased when TLR4 was silenced in MDA-MB-231 cells and subsequently treated with rS100A9 (Figure 3E). Also, introduction of the MD2-hTLR4 complex (pDUO-MD2-hTLR4) in otherwise MD2- and TLR4-negative MCF7 cells promoted a significant NFκB activity upon S100A9 stimulation (Figure 3F).
S100A9 induces NF κ B in breast cancer cells in vitro in a TLR4-dependent manner. (A) Dual luciferase reporter assays of breast cancer cells transfected with a NFκB reporter. TK-Renilla was co-transfected as control. rS100A9 was added to stimulate NFκB activity as described in Material and Methods. Error bars indicate s.e.m. *P<0.05 using Student’s t-test. (B) Dual luciferase reporter assays of MDA-MB-231 breast cancer cells transfected with a NFκB reporter. TK-Renilla was co-transfected as control. rS100A9 (10 μg ml−1) or rS100A8/A9 (10 μg ml−1) was added to stimulate NFκB activity in MDA-MB-231 cells as described in Material and Methods. Error bars indicate s.e.m. *P<0.05 using Student’s t-test. (C) TLR4 and RAGE mRNA expression levels in human breast cancer cell lines as measured by Q-PCR. Error bars indicate s.e.m. ***P<0.001 using ANOVA test. (D) Dual luciferase reporter assays of breast cancer cells transfected with siRNA directed against S100A9 mRNA together with a NFκB reporter and TK-Renilla as control. Error bars indicate s.e.m. **P<0.01 using ANOVA test. (E) IL-8 protein levels in supernatants from MDA-MB-231 breast cancer cells transfected with negative control (nc) siRNA or siRNA directed against TLR4 mRNA (si#1 and si#2) and subsequently stimulated or not with rS100A9 homodimers. Cytometric bead array (CBA) was used to measure IL-8 levels. Error bars indicate s.e.m. *P<0.05, **P<0.01, ***P<0.001 using ANOVA test. (F) Dual luciferase reporter assays of MCF-7 breast cancer cells transfected with the MD2-hTLR4 complex (pDUO-MD2-hTLR4) together with a NFκB reporter and TK-Renilla as control. Error bars indicate s.e.m. **P<0.01 using ANOVA test. (G) EGF induced S100A9 expression in human breast cancer cells in vitro. rEGF stimulation of indicated breast cancer cell lines induced S100A9 mRNA levels as measured using Q-PCR. Error bars indicate s.e.m. *P<0.05, **P<0.01, ***P<0.001 using ANOVA test.
Because of the inherent expression of EGFR in MDA-MB-468 (amplified; EGFR++) and SKBR3 (EGFR+) cells, we also wanted to analyse whether EGF could induce S100A9 per se. Indeed, we found that rEGF induced S100A9 at the mRNA level in SKBR3 but not in the EGFR-amplified MDA-MB-468 cells (Figure 3G). In an attempt to investigate further functions of S100A9, we performed an array of in vitro experiments, including cell cycle, proliferation, apoptosis, actin polymerisation, epithelial-to-mesenchymal transition (EMT) and migration analyses. Using the mentioned breast cancer cell lines rS100A9 homodimers or siRNA towards S100A9, we found no significant effects on cell cycle, apoptosis, EMT or migration (data not shown). We did, however, see a small but significant effect on breast cancer cell proliferation in siS100A9-transfected MDA-MB-468 cells (Figure 4A–C) and also on actin polymerisation in rS100A9-stimulated MCF-7 cells (Figure 4D) as previously shown (Yin et al, 2013; Cormier et al, 2014).
Effects of S100A9 on breast cancer cell proliferation, survival and F-actin polymerisation in vitro. (A) Effects of rS100A9 on breast cancer cell proliferation using thymidine 3H incorporation (left) or WST-1 assay (right) on the different breast cancer cell lines indicated. (B) Breast cancer cell proliferation using thymidine 3H incorporation on cell lines transfected with either negative control (nc) or silencing (si) S100A9 siRNAs as indicated. Error bars indicate s.e.m. *P<0.05 using ANOVA test. (C) Breast cancer cell proliferation/viability using WST-1 assay (right) on cell lines transfected with either negative control (nc) or silencing (si) S100A9 siRNAs as indicated. Error bars indicate s.e.m. NS=not significant using ANOVA test. (D) Effects of S100A9 on F-actin polymerisation in MCF-7 cells using phalloidin IF (green) staining. Blue staining=DAPI.
S100A9 expression correlates with ER−, PgR−, Her2+ and EGFR+ expression in human breast tumours
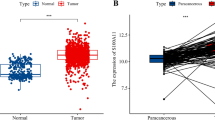
To investigate the relation between S100A9 expression and ER, PgR, Her2 (ERBB2) and EGFR expression in primary breast cancers in more detail, we next used a publicly available data site (R2: microarray analysis and visualisation platform http://r2.amc.nl; Tumour breast EXPO-351) with gene expression profiles of 351 primary breast cancers. We found a very strong negative correlation between expression of S100A9 mRNA with either ER (r-value −0.518; P=1.7e-25) and PgR (r-value −0.468; P=1.8e-20), and a positive correlation between expression of S100A9 mRNA with either ERBB2 (r-value 0.238; P=6.8e-06) or EGFR (r-value 0.319; P=9.6e-10) (Supplementary Figures S2A–D).
Following IHC treatment of our TMA comprising 144 breast cancers, cytoplasmic and nuclear S100A9 staining was successfully annotated in 129 (89.6%) tumours and 128 tumours (88.9%) for stromal compartment. In line with both the IHC data from breast cancer cell lines, as well as the gene expression profiling data, S100A9 expression (cytoplasmic, nuclear, membrane as well as stromal; Figure 5A–D) correlated to ER−PgR− hormone receptor status of the breast tumours (P<0.001; Tables 1a–c). Cytoplasmic S100A9 also correlated to expression of the Ki67 proliferation marker (Table 1a). Tumours presenting Her2 or EGFR positivity were relatively few (n=12 and n=11, respectively). Despite this, stromal S100A9 expression correlated to Her2 positivity (P=0.018; Table 1a) and presented a borderline correlation to EGFR positivity (P=0.064; Table 1a). All of the Her2+ (n=12) as well as all EGFR+ (n=11) showed cytoplasmic S100A9 expression (Table 1b).
S100A9 expression in breast tumours and survival curves. (A–D) Immunohistochemical S100A9 expression sample images from breast cancer TMA cores, from left to right: negative (A), cytoplasmic expression (B), cytoplasmic/nuclear (C), and stromal expression (D). (E) Kaplan–Meier curves illustrating differences in overall survival according to S100A9 cytoplasmic expression in breast cancer tumours. (F) Kaplan–Meier curves illustrating differences in overall survival according to S100A9 stromal expression in breast cancer tumours.
Stromal S100A9 expression correlates to the presence of anti-inflammatory CD163+ myeloid cells
Tumour stromal S100A9 expression has previously been described in different tumours (e.g., prostate cancer) (Tidehag et al, 2014). In this study, we found that stromal S100A9 expression (predominantly in cells with a leukocyte morphology) correlated not only with ER−PgR− tumours as mentioned above but also to parameters such as larger tumour size and Ki67 positivity and nodal stage (Table 1a; S100A9 stroma; right). We have previously shown that anti-inflammatory CD163+ myeloid cells are located preferentially in the tumour stroma of TN breast cancer and also that this correlated to higher grade, larger tumour size and Ki67 positivity (Medrek et al, 2012). As shown in Table 1c, breast cancer stromal S100A9 correlated with anti-inflammatory CD163+ myeloid cells but interestingly not to the macrophage marker CD68. This suggests that the myeloid cells expressing S100A9 are of immature myeloid origin (e.g., MDSCs). Supporting this notion, also anti-inflammatory CD163+ myeloid cells in the tumour nest actually expressed S100A9, but CD68+ macrophages did not (Table 1c).
S100A9 expression in breast cancer correlates with a poor survival
S100A9 expression in cytoplasm and in stroma was found to be significantly associated with an impaired survival (Figure 5). The same trend but non-significant was seen for membrane and nuclear expression (data not shown). For cytoplasmic expression, CRT analysis revealed a cutoff at 0.149/3.00 (Supplementary Figure S3A) where patients having positive (any) S100A9 expression denoted as moderate–high had significantly impaired survival (n=116/129, P=0.021, Figure 5E). For stromal expression, CRT analysis that revealed an optimal cutoff at 1.95/3.00 (Supplementary Figure S3B) also had significantly impaired survival (P<0.001, Figure 5F). This is in line with what we previously showed concerning correlation between impaired overall survival and the presence of macrophages and anti-inflammatory myeloid cells in the tumour stroma, with a preferential presence of anti-inflammatory myeloid cells in the tumour stroma of TN breast cancers particularly (Medrek et al, 2012).
Discussion
S100A9 expression in breast cancer has previously been explored in vitro and in small cohorts of patients (Arai et al, 2004; Cross et al, 2005; Arai et al, 2008; Moon et al, 2008; Markowitz and Carson, 2013; Yin et al, 2013; Cormier et al, 2014; Gumireddy et al, 2014). Also, a large protein profiling study was performed on breast cancers (Goncalves et al, 2008) where cytoplasmic S100A9 expression in the malignant breast epithelial cells was investigated. Studies concerning the role for S100A9 in breast cancer cells in vitro are indefinite (Markowitz and Carson, 2013) and one reason for this is the limited supply of reagents. As S100A9 acts as a chemo-attractant for certain immunosuppressive cells and is expressed in myeloid suppressor cells that are important in diseases such as cancer (Ostrand-Rosenberg and Sinha, 2009), we decided to perform a deeper analysis concerning the expression and localisation pattern of S100A9 and its correlation to the presence of different tumour-infiltrating myeloid cells. We stained TMA sections containing samples of 144 breast cancers using a carefully validated antibody towards human S100A9 and using a publicly available gene expression data set consisting of 351 primary breast tumours. The findings were endorsed by analysis regarding the functional role of S100A9 specifically using stimulation with rS100A9 or silencing of S100A9 in vitro.
Although S100A9 is expressed in some epithelial cell types and also in certain cancers, its functional role in these cell types is still obscure (Markowitz and Carson, 2013). Our in vitro experiments regarding cell cycle analysis, apoptosis, migration and EMT markers, using stimulation with rS100A9 or silencing of S100A9, did not yield consistent results. However, as published previously, both cell proliferation and f-actin polymerisation was affected slightly by S100A9 in some cell lines and should therefore be warranted more studies in the future (Yin et al, 2013; Cormier et al, 2014). Instead we found that inflammatory cytokines were released upon S100A9 stimulation of breast cancer cell lines but only in cells with a high expression of TLR4 (MDA-MB-231), similar to what has previously been published regarding myeloid cells (Riva et al, 2012). In line with this, only very modest or no decrease in cytokine expression was seen when S100A9 was silenced in the S100A9-expressing cells that lack TLR4 or RAGE. Only, S100A9 signalling, but not S100A9/S100A8, induced significant expression of inflammatory proteins. Indeed, S100A9 homodimers but not S100A9/S100A8 heterodimers have been proposed to signal via TLR4 in a Ca2+-dependent manner (Markowitz and Carson, 2013). As both membranous and vesicular TLR4 has been reported in cancer cells, the cytoplasmic S100A9 expression pattern could represent intracellular as well as extracellular signalling pathways (Hassan et al, 2006).
Breast tumours of the TN and ER−PgR− subtypes most often express pro-inflammatory proteins that activate STAT3 (Sasser et al, 2007). STAT3 has previously been shown to induce S100A9 (Li et al, 2004; Lee et al, 2012). This does, however, not reflect the expression pattern of S100A9 in breast cancer cell lines in vitro, as a high S100A9 expression is seen only in two out of three ER−PgR− breast cancer cell lines tested. Hence other explanations as to why S100A9 is expressed in malignant epithelial breast cells are likely to be found in the future. A link between S100A9 expression and EGFR has been suggested for other cancer types before (Kim et al, 2011, 2014). The fact that the two cell lines expressing S100A9 both expressed EGFR (MDA-MB-468, EGFR++ amplified; SKBR3, EGFR+) led us to the finding that also rEGF could induce S100A9 expression but only in EGFR-non-amplified SKBR3 cells. It would be interesting to investigate whether the tumour microenvironment as such could effect S100A9 expression in vivo.
After careful validation, we considered the S100A9 antibody used in this study to be specific using IHC on paraffin-embedded samples. We found that S100A9 was expressed primarily in tumours with an ER−PgR− phenotype. This correlation was found irrespective of the localisation of S100A9 (cytoplasmic, nuclear, membrane) in the malignant cells and was supported by a strong inverse correlation of the genes in a gene expression profile analysis made of 351 breast tumours (R2: microarray analysis and visualisation platform (http://r2.amc.nl; Tumour breast EXPO-351). Even stromal expression of S100A9 correlated not only with the ER−PgR− breast cancer phenotype but also to parameters such as larger tumour size, Her2 positivity and nodal stage. Stromal expression of S100A9 was mostly present in cells with a leukocyte morphology and correlated with the presence of anti-inflammatory myeloid cells (CD163+), but not with the macrophage marker CD68, in the same tumours. This support previous findings that S100A9 is expressed by MDSCs (Ostrand-Rosenberg and Sinha, 2009). In this context, it is interesting to note that we previously have shown that anti-inflammatory myeloid cells (CD163+) are preferentially enriched in the stromal areas of breast tumours of the TN subtype. Cytoplasmic S100A9 expression did not, however, correlate to Her2 in our breast cancer cohort. Instead we found that, out of the 11 Her2+ tumours, all (100%) expressed cytosolic S100A9 in the malignant cells. The same was true for EGFR+ tumours (n=12). The fact that no correlation can be found between S100A9 expression and Her2 or EGFR positivity is most likely due to the small amount of tumours with this particular subtype. This is supported by the gene expression profile analysis that showed a strong positive correlation between S100A9 and either ERBB2 (Her2) or EGFR at the gene expression level, as well as our in vitro analysis of breast cancer cell lines with this phenotype. A link between S100A9 expression and EGFR has previously been published regarding bladder cancer (Kim et al, 2011, 2014). Our findings that S100A9 expression correlates to ER−PgR− and Her2+ or EGFR+ breast tumours is also supported by a large protein profiling study performed on breast cancers (Goncalves et al, 2008).
The effect of S100A9 expression on primary breast cancer patient survival has not been studied in depth before, but the expression of S100A9 in metastatic lesions has been shown to correlate with a worse prognosis (Acharyya et al, 2012) and cytoplasmic S100A9 expression in malignant breast epithelial cells was correlated to worse overall survival, although it was concluded that this might be due to its correlation with other parameters such as grade, ER or ERBB2 status (Goncalves et al, 2008). In our study, patients having positive (any) cytoplasmic S100A9 expression were found to have a significant reduction in overall survival, where the main discrepancy was seen for patients having a minor increase in expression, and this was also seen for ER+PgR+ tumours. Even more accentuated, a similar trend was seen for stromal S100A9 expression, where a higher expression was progressively associated with a reduced overall survival. This result fits very well to what was found when we analysed the presence of stromal CD163+ anti-inflammatory myeloid cells previously, again indicating that stromal S100A9 expression is caused by myeloid cells primarily (Medrek et al, 2012). In summary, this suggests that S100A9 can be expressed both in malignant cells and in myeloid cells in breast tumours. It is most likely so that S100A9 might have different roles for these cell types, but according to this study also similar functions are expected. This would primarily concern induction of an inflammatory environment. When the chemo-attractive effects on MDSCs are added to these processes, it is not surprising that both a stromal as well as malignant S100A9 expression pattern will affect breast cancer patient’s overall survival. With these data, we propose that S100A9 would be a good future candidate drug target for patients with ER−PgR− breast cancer, especially in patients with tumours that express Her2 or EGFR.
References
Acharyya S, Oskarsson T, Vanharanta S, Malladi S, Kim J, Morris PG, Manova-Todorova K, Leversha M, Hogg N, Seshan VE, Norton L, Brogi E, Massague J (2012) A CXCL1 paracrine network links cancer chemoresistance and metastasis. Cell 150 (1): 165–178.
Allred DC, Brown P, Medina D (2004) The origins of estrogen receptor alpha-positive and estrogen receptor alpha-negative human breast cancer. Breast Cancer Res 6 (6): 240–245.
Arai K, Takano S, Teratani T, Ito Y, Yamada T, Nozawa R (2008) S100A8 and S100A9 overexpression is associated with poor pathological parameters in invasive ductal carcinoma of the breast. Curr Cancer Drug Targets 8 (4): 243–252.
Arai K, Teratani T, Kuruto-Niwa R, Yamada T, Nozawa R (2004) S100A9 expression in invasive ductal carcinoma of the breast: S100A9 expression in adenocarcinoma is closely associated with poor tumour differentiation. Eur J Cancer 40 (8): 1179–1187.
Benedyk M, Sopalla C, Nacken W, Bode G, Melkonyan H, Banfi B, Kerkhoff C (2007) HaCaT keratinocytes overexpressing the S100 proteins S100A8 and S100A9 show increased NADPH oxidase and NF-kappaB activities. J Invest Dermatol 127 (8): 2001–2011.
Bjork P, Bjork A, Vogl T, Stenstrom M, Liberg D, Olsson A, Roth J, Ivars F, Leanderson T (2009) Identification of human S100A9 as a novel target for treatment of autoimmune disease via binding to quinoline-3-carboxamides. PLoS Biol 7 (4): e97.
Bjork P, Kallberg E, Wellmar U, Riva M, Olsson A, He Z, Torngren M, Liberg D, Ivars F, Leanderson T (2013) Common interactions between S100A4 and S100A9 defined by a novel chemical probe. PLoS One 8 (5): e63012.
Bresnick AR, Weber DJ, Zimmer DB (2015) S100 proteins in cancer. Nat Rev Cancer 15 (2): 96–109.
Cormier K, Harquail J, Ouellette RJ, Tessier PA, Guerrette R, Robichaud GA (2014) Intracellular expression of inflammatory proteins S100A8 and S100A9 leads to epithelial-mesenchymal transition and attenuated aggressivity of breast cancer cells. Anticancer Agents Med Chem 14 (1): 35–45.
Cross SS, Hamdy FC, Deloulme JC, Rehman I (2005) Expression of S100 proteins in normal human tissues and common cancers using tissue microarrays: S100A6, S100A8, S100A9 and S100A11 are all overexpressed in common cancers. Histopathology 46 (3): 256–269.
Edgeworth J, Gorman M, Bennett R, Freemont P, Hogg N (1991) Identification of p8,14 as a highly abundant heterodimeric calcium binding protein complex of myeloid cells. J Biol Chem 266 (12): 7706–7713.
Elkabets M, Gifford AM, Scheel C, Nilsson B, Reinhardt F, Bray MA, Carpenter AE, Jirstrom K, Magnusson K, Ebert BL, Ponten F, Weinberg RA, McAllister SS (2011) Human tumors instigate granulin-expressing hematopoietic cells that promote malignancy by activating stromal fibroblasts in mice. J Clin Invest 121 (2): 784–799.
Goncalves A, Charafe-Jauffret E, Bertucci F, Audebert S, Toiron Y, Esterni B, Monville F, Tarpin C, Jacquemier J, Houvenaeghel G, Chabannon C, Extra JM, Viens P, Borg JP, Birnbaum D (2008) Protein profiling of human breast tumor cells identifies novel biomarkers associated with molecular subtypes. Mol Cell Proteomics 7 (8): 1420–1433.
Gronberg M, Fjallskog ML, Jirstrom K, Janson ET (2011) Expression of ghrelin is correlated to a favorable outcome in invasive breast cancer. Acta Oncol 51 (3): 386–393.
Gumireddy K, Li A, Kossenkov AV, Cai KQ, Liu Q, Yan J, Xu H, Showe L, Zhang L, Huang Q (2014) ID1 promotes breast cancer metastasis by S100A9 regulation. Mol Cancer Res 12 (9): 1334–1343.
Hanahan D, Weinberg RA (2000) The hallmarks of cancer. Cell 100 (1): 57–70.
Hassan F, Islam S, Tumurkhuu G, Naiki Y, Koide N, Mori I, Yoshida T, Yokochi T (2006) Intracellular expression of toll-like receptor 4 in neuroblastoma cells and their unresponsiveness to lipopolysaccharide. BMC Cancer 6: 281.
Heizmann CW, Fritz G, Schafer BW (2002) S100 proteins: structure, functions and pathology. Front Biosci 7: d1356–d1368.
Hessian PA, Edgeworth J, Hogg N (1993) MRP-8 and MRP-14, two abundant Ca(2+)-binding proteins of neutrophils and monocytes. J Leukoc Biol 53 (2): 197–204.
Kamangar F, Dores GM, Anderson WF (2006) Patterns of cancer incidence, mortality, and prevalence across five continents: defining priorities to reduce cancer disparities in different geographic regions of the world. J Clin Oncol 24 (14): 2137–2150.
Kim WJ, Kim SK, Jeong P, Yun SJ, Cho IC, Kim IY, Moon SK, Um HD, Choi YH (2011) A four-gene signature predicts disease progression in muscle invasive bladder cancer. Mol Med 17 (5-6): 478–485.
Kim WT, Kim J, Yan C, Jeong P, Choi SY, Lee OJ, Chae YB, Yun SJ, Lee SC, Kim WJ (2014) S100A9 and EGFR gene signatures predict disease progression in muscle invasive bladder cancer patients after chemotherapy. Ann Oncol 25 (5): 974–979.
Lagasse E, Clerc RG (1988) Cloning and expression of two human genes encoding calcium-binding proteins that are regulated during myeloid differentiation. Mol Cell Biol 8 (6): 2402–2410.
Lee MJ, Lee JK, Choi JW, Lee CS, Sim JH, Cho CH, Lee KH, Cho IH, Chung MH, Kim HR, Ye SK (2012) Interleukin-6 induces S100A9 expression in colonic epithelial cells through STAT3 activation in experimental ulcerative colitis. PLoS One 7 (9): e38801.
Li C, Zhang F, Lin M, Liu J (2004) Induction of S100A9 gene expression by cytokine oncostatin M in breast cancer cells through the STAT3 signaling cascade. Breast Cancer Res Treat 87 (2): 123–134.
Marenholz I, Heizmann CW, Fritz G (2004) S100 proteins in mouse and man: from evolution to function and pathology (including an update of the nomenclature). Biochem Biophys Res Commun 322 (4): 1111–1122.
Markowitz J, Carson WE 3rd (2013) Review of S100A9 biology and its role in cancer. Biochim Biophys Acta 1835 (1): 100–109.
Medrek C, Ponten F, Jirstrom K, Leandersson K (2012) The presence of tumor associated macrophages in tumor stroma as a prognostic marker for breast cancer patients. BMC Cancer 12: 306.
Moon A, Yong HY, Song JI, Cukovic D, Salagrama S, Kaplan D, Putt D, Kim H, Dombkowski A, Kim HR (2008) Global gene expression profiling unveils S100A8/A9 as candidate markers in H-ras-mediated human breast epithelial cell invasion. Mol Cancer Res 6 (10): 1544–1553.
Ostrand-Rosenberg S, Sinha P (2009) Myeloid-derived suppressor cells: linking inflammation and cancer. J Immunol 182 (8): 4499–4506.
Riva M, He Z, Kallberg E, Ivars F, Leanderson T (2013) Human S100A9 protein is stabilized by inflammatory stimuli via the formation of proteolytically-resistant homodimers. PLoS One 8 (4): e61832.
Riva M, Kallberg E, Bjork P, Hancz D, Vogl T, Roth J, Ivars F, Leanderson T (2012) Induction of nuclear factor-kappaB responses by the S100A9 protein is Toll-like receptor-4-dependent. Immunology 137 (2): 172–182.
Roth J, Vogl T, Sorg C, Sunderkotter C (2003) Phagocyte-specific S100 proteins: a novel group of proinflammatory molecules. Trends Immunol 24 (4): 155–158.
Sasser AK, Sullivan NJ, Studebaker AW, Hendey LF, Axel AE, Hall BM (2007) Interleukin-6 is a potent growth factor for ER-alpha-positive human breast cancer. FASEB J 21 (13): 3763–3770.
Schnitt SJ (2010) Classification and prognosis of invasive breast cancer: from morphology to molecular taxonomy. Mod Pathol 23 (Suppl 2): S60–S64.
Sinha P, Okoro C, Foell D, Freeze HH, Ostrand-Rosenberg S, Srikrishna G (2008) Proinflammatory S100 proteins regulate the accumulation of myeloid-derived suppressor cells. J Immunol 181 (7): 4666–4675.
Slamon DJ, Clark GM, Wong SG, Levin WJ, Ullrich A, McGuire WL (1987) Human breast cancer: correlation of relapse and survival with amplification of the HER-2/neu oncogene. Science 235 (4785): 177–182.
Sorlie T, Perou CM, Tibshirani R, Aas T, Geisler S, Johnsen H, Hastie T, Eisen MB, van de Rijn M, Jeffrey SS, Thorsen T, Quist H, Matese JC, Brown PO, Botstein D, Lonning PE, Borresen-Dale AL (2001) Gene expression patterns of breast carcinomas distinguish tumor subclasses with clinical implications. Proc Natl Acad Sci USA 98 (19): 10869–10874.
Srikrishna G, Panneerselvam K, Westphal V, Abraham V, Varki A, Freeze HH (2001) Two proteins modulating transendothelial migration of leukocytes recognize novel carboxylated glycans on endothelial cells. J Immunol 166 (7): 4678–4688.
Sunahori K, Yamamura M, Yamana J, Takasugi K, Kawashima M, Yamamoto H, Chazin WJ, Nakatani Y, Yui S, Makino H (2006) The S100A8/A9 heterodimer amplifies proinflammatory cytokine production by macrophages via activation of nuclear factor kappa B and p38 mitogen-activated protein kinase in rheumatoid arthritis. Arthritis Res Ther 8 (3): R69.
Svensson KJ, Christianson HC, Kucharzewska P, Fagerstrom V, Lundstedt L, Borgquist S, Jirstrom K, Belting M (2011) Chondroitin sulfate expression predicts poor outcome in breast cancer. Int J Oncol 39 (6): 1421–1428.
Tidehag V, Hammarsten P, Egevad L, Granfors T, Stattin P, Leanderson T, Wikstrom P, Josefsson A, Hagglof C, Bergh A (2014) High density of S100A9 positive inflammatory cells in prostate cancer stroma is associated with poor outcome. Eur J Cancer 50 (10): 1829–1835.
Vandesompele J, De Preter K, Pattyn F, Poppe B, Van Roy N, De Paepe A, Speleman F (2002) Accurate normalization of real-time quantitative RT-PCR data by geometric averaging of multiple internal control genes. Genome Biol 3 (7): RESEARCH0034.
Yin C, Li H, Zhang B, Liu Y, Lu G, Lu S, Sun L, Qi Y, Li X, Chen W (2013) RAGE-binding S100A8/A9 promotes the migration and invasion of human breast cancer cells through actin polymerization and epithelial-mesenchymal transition. Breast Cancer Res Treat 142 (2): 297–309.
Yui S, Nakatani Y, Mikami M (2003) Calprotectin (S100A8/S100A9), an inflammatory protein complex from neutrophils with a broad apoptosis-inducing activity. Biol Pharm Bull 26 (6): 753–760.
Zwadlo G, Bruggen J, Gerhards G, Schlegel R, Sorg C (1988) Two calcium-binding proteins associated with specific stages of myeloid cell differentiation are expressed by subsets of macrophages in inflammatory tissues. Clin Exp Immunol 72 (3): 510–515.
Acknowledgements
We thank Ms. Elise Nilsson for professional technical skills in preparation of TMAs and IHC. This work was supported by grants from the Swedish Research Council, The Swedish Cancer Foundation, Kocks Stiftelser, Österlunds Stiftelse, Gunnar Nilsson Cancer Foundation, MAS Cancer Foundation and Åke Winbergs Foundation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
TL is a part-time employee at Active Biotech that develops S100A9 inhibitors for the treatment of autoimmune diseases and cancer.
Additional information
Supplementary Information accompanies this paper on British Journal of Cancer website
Rights and permissions
This work is licensed under the Creative Commons Attribution-Non-Commercial-Share Alike 4.0 International License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/4.0/
About this article
Cite this article
Bergenfelz, C., Gaber, A., Allaoui, R. et al. S100A9 expressed in ER−PgR− breast cancers induces inflammatory cytokines and is associated with an impaired overall survival. Br J Cancer 113, 1234–1243 (2015). https://doi.org/10.1038/bjc.2015.346
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bjc.2015.346
- Springer Nature Limited