Abstract
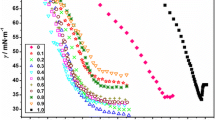
The surface tension of aqueous solutions of the cationic surfactant dodecylethylaminocarbonyldimethylbenzylammonium chloride is measured for the first time over a wide concentration range from 7.9×10- 7 to 3.9×10- 2 M at 20-35°C. The critical micelle concentration of DAEDMBAC as influenced by temperature is determined.
Similar content being viewed by others
REFERENCES
Rusanov, A.I., Mitselloobrazovanie v rastvorakh poverkhnostno-aktivnykh veshchestv (Micelle Formation in Surfactant Solutions), St. Petersburg: Khimiya, 1992.
Abdulin, N.G., Kochurova, N.N., and Rusanov, A.I., Kolloidn. Zh., 1997, vol. 59, no. 6, pp. 725–728.
Abramzon, A.A., Poverkhnostno-aktivnye veshchestva (Surfactants), Leningrad: Khimiya, 1962.
Burchfield, I.E. and Woolky, E.M., J. Phys. Chem., 1984, vol. 88, no. 10, pp. 2149–2155.
Samoilov, O.Ya., Struktura vodnykh rastvorov elektrolitov i gidratatsiya ionov (Structure of Aqueous Electrolyte Solutions and Ion Hydration), Moscow: Akad. Nauk SSSR, 1957.
Mehrian, T., Keizer, A., Korteweg, A.G. de, and Lyklema, J., Colloids Surf., Ser. A, 1993, vol. 71, pp. 255–267.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kochurova, N.N., Korotkikh, O.P. & Dmitrovskaya, M.V. Surface Tension in Aqueous Dodecylethylaminocarbonyldimethylbenzylammonium Chloride. Russian Journal of Applied Chemistry 77, 848–850 (2004). https://doi.org/10.1023/B:RJAC.0000038829.36267.ee
Issue Date:
DOI: https://doi.org/10.1023/B:RJAC.0000038829.36267.ee