Abstract
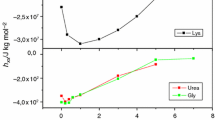
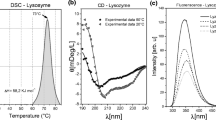
Procedures are described for the isolation of the individual components A1, A2, and A3 of native R-ovalbumin from freshly laid domestic hen eggs. Because heavy metal ion contaminants result in spurious irreproducible kinetics, particularly at high pH, considerable care is taken to avoid their presence. Kinetics studies are made of the behavior of whole R-ovalbumin and its individual components in urea solution over the pH range 3.7–9.6 following the reaction by determining absorbance differences at 233, 287, and 293 nm and ORD and CD changes at 350 and 221 nm, respectively. Reaction is rapid at low pH, slowing with increasing pH. Except under limited conditions, the reaction is not simple first order. Equations are presented for describing the reactions, and the nature of the reaction products is considered. Unfolding equilibrium profiles were also determined by ORD at several wavelengths and were not stigmoidal in shape and the normalized curves were not superimposed.
Similar content being viewed by others
REFERENCES
Ahmad, F., and Salahuddin, A. (1976). Biochemistry 15: 5168–5175.
Bates, R. (1973). Determination of pH: Theory and Practice (2nd Ed.), Wiley, New York, pp. 60–103.
Cho, Kon Ho (1969). The Conformation and Denaturation of Ovalbumin. Dissertation, Princeton University, Princeton, NJ.
Fasman, G. (ed.) (1996). Circular Dichroism and the Conformational Analysis of Biomolecules, Plenum, New York.
Glazer, A. N., McKenzie, H. A., and Wake, R. G. (1963). Biochim. Biophys. Acta 69: 240–248.
Hofmeister, F. (1889). Hoppe-Seyler's Z. Physiol. Chem. 14: 165–172.
Huntington, J. A., and Stein, P. E. (2001). J. Chromatogr. B 756: 189–198.
Kauzmann, W. (1959). Adv. Protein Chem. 14: 1–63.
McKenzie, H. A. (2003). J. Protein Chem. 22: (this issue).Author please supply missing information
McKenzie, H. A., and Frier, R. D. (2003). J. Protein Chem. 22: (this issue).Author please supply missing information
McKenzie, H. A., and Murphy, W. H. (1970). In Milk Proteins, Vol. 1 (McKenzie, H. A., ed.), Academic Press, New York, pp. 127–180.
McKenzie, H. A., and Ralston, G. B. (1971). Experientia 27: 617–624.
McKenzie, H. A., Smith, M. B., and Wake, R. G. (1963). Biochim. Biophys. Acta 69: 222–239.
Simpson, R. B., and Kauzmann, W. (1953). J. Am. Chem. Soc. 75: 5139–5152.
Smith, M. B. (1964). Aust. J. Biol. Sci. 17: 261–270.
Smith, M. B., and Back, J. F. (1965). Aust. J. Biol. Sci. 18: 365–377.
Stein, P. E., Leslie, A. G. W., Finch, J. T., and Carrell, R. W. (1991). J. Mol. Biol. 221: 941–959.
Tanford, C. (1968). Adv. Protein. Chem. 23: 121–282.
Tanford, C. (1970). Adv. Protein Chem. 24: 1–95.
Wright, H. T., Qian, H. X., and Huber, R. (1990). J. Mol. Biol. 213: 513–528.
Zemser, M., Friedman, M., Katzhendler, J., Greene, L. L., Minsky, A., and Gorinstein, S. (1994). J. Protein Chem. 13: 261–274.
Author information
Authors and Affiliations
Corresponding author
Additional information
Deceased December 8, 2001
Rights and permissions
About this article
Cite this article
McKenzie, H.A., Frier, R.D. The Behavior of R-Ovalbumin and Its Individual Components A1, A2, and A3 in Urea Solution: Kinetics and Equilibria. J Protein Chem 22, 207–214 (2003). https://doi.org/10.1023/A:1025076621515
Published:
Issue Date:
DOI: https://doi.org/10.1023/A:1025076621515